Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Nematode infections are highly prevalent, affecting millions of people worldwide. Nematodes are complex, nonsegmented roundworms with specialized organs that include a protective outer coating or cuticle, a complete and functional gastrointestinal tract, and muscular, nervous, and reproductive systems. Most species are free-living in soil or water, but a few parasitize humans.
Nematodes of medical importance can be categorized into those that primarily affect the gastrointestinal tract, where adult worms become established and cause disease, and those that affect other tissues and organ systems. The former includes the roundworm Ascaris lumbricoides , the hookworms Ancylostoma duodenale and Necator americanus , the pinworm Enterobius vermicularis , the whipworm Trichuris trichiura , and the threadworm Strongyloides stercoralis . Zoonotic intestinal nematodes such as Trichostrongylus and Anisakis also can incidentally infect and cause disease in humans. Nematodes that invade and cause disease primarily outside the gastrointestinal tract include those that cause lymphatic filariasis ( Wuchereria bancrofti , Brugia malayi , and Brugia timori ), Onchocerca volvulus , Loa loa , and the guinea worm Dracunculus medinensis , as well as Trichinella and Angiostrongylus species.
Nematodes that infect humans measure from several millimeters to more than a meter in length and often survive for months to years within their host. Except for S. stercoralis and Capillaria philippinensis , adult worms cannot complete their life cycle within a human host. Instead, sexually mature adult worms mate and produce eggs or larvae that must have at least one stage of development outside the host, either in the environment or in an intermediate host.
Nematode infections are rarely fatal; they more commonly result in chronic morbidity such as iron deficiency anemia caused by hookworm or blindness due to onchocerciasis. For most nematodes, the severity of the clinical manifestations of infection is proportional to the number of worms harbored by a given host; although light infections with only a few worms are usually asymptomatic, heavier worm burdens often cause symptoms and clinical sequelae.
Nematode infections are prevalent in the temperate and tropical regions of Africa, Asia, and Latin America. They are transmitted by the oral ingestion of embryonated eggs or by penetration of infective larvae through the skin, either by direct contact with contaminated soil or by the bite of an arthropod vector. Nematode infections are most common in areas with poor sanitation, where the environment is contaminated by human feces, and in climates that support survival of the insect vector if one is involved in the life cycle.
A. lumbricoides , commonly known as roundworm, is acquired by oral ingestion of embryonated eggs that have been expelled in the feces of humans, who are the only host. Adult worms ( Fig. 327-1 ) range in length between 15 and 50 cm and survive in the host for approximately 18 months.
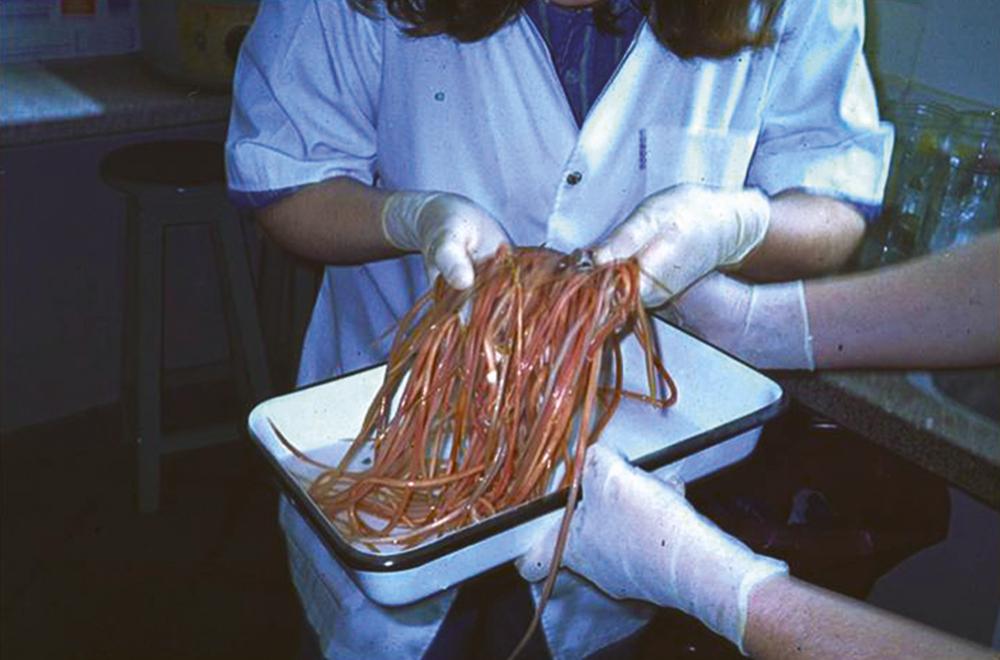
A. lumbricoides is the most prevalent nematode infection worldwide, affecting approximately 730 million people in sub-Saharan Africa, South and Southeast Asia, and Latin America, primarily in rural areas of high population density that lack adequate sanitation or treatment of sewage or where untreated human feces are used as fertilizer. Climate is an important determinant of disease since warm temperatures and adequate moisture are needed for embryonation of eggs in soil. In endemic areas, the prevalence and intensity of infection increase significantly during the first 2 to 3 years of life, remain high between the ages of 4 and 15 years, and then decline during adulthood.
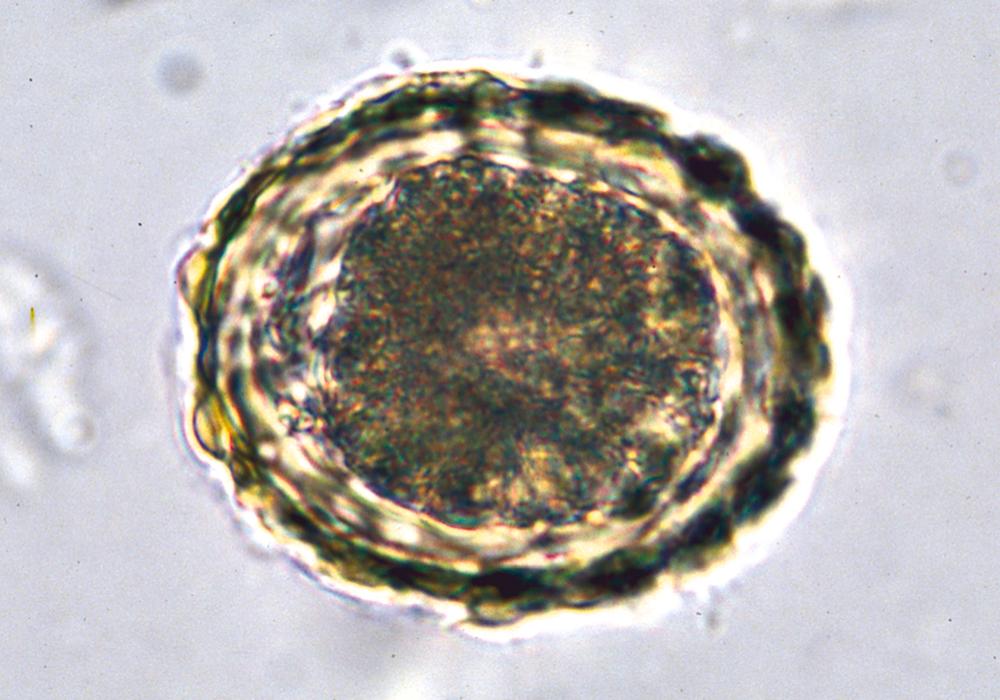
In the stomach, the egg’s outer shell is dissolved by gastric acid, thereby releasing larvae into the small intestine, where they penetrate the intestinal wall and enter the portal circulation. The larvae migrate to the pulmonary vasculature, where they penetrate the alveoli, ascend the bronchial tree, and are swallowed back into the intestinal tract, where they develop into adult worms 9 to 11 weeks after egg ingestion. Each day, a female adult Ascaris worm releases over 200,000 eggs that are expelled in feces. Fertilized eggs ( Fig. 327-2 ) become infectious after embryonating in warm, moist, shady soil. Eggs are resistant to extreme temperatures and to desiccation, and they can survive up to 15 years in the environment.
Clinical manifestations are associated with heavy worm burdens and result from larval migration through the lungs and from parasitism of the gastrointestinal tract by adult worms. During the pulmonary phase of the life cycle, A. lumbricoides larvae can induce an intense reaction that is due to both physical disruption and a hypersensitivity response to secreted antigens. This phenomenon is more common in areas in which transmission is seasonal, such as on the Arabian Peninsula, where outbreaks of pneumonitis typically follow the rainy season because of resumption of transmission.
Low-intensity A. lumbricoides infections are usually asymptomatic, but heavy worm burdens cause symptoms including sudden onset of wheezing, dyspnea, paroxysmal nonproductive cough, and high fever. Symptoms may persist for 2 to 3 weeks before resolving spontaneously. Respiratory symptoms may coincide with or be preceded by urticarial rash, angioedema, abdominal pain, and vomiting.
With moderate or heavy infections, a mass of worms can cause obstruction in the small intestine or migration of worms to the biliary tree, pancreatic duct, or appendix. Intestinal obstruction, which is more common in young children because of their smaller lumen size, is characterized by colicky abdominal pain and vomiting that may progress to signs of intestinal perforation and be confused with acute appendicitis ( Chapter 128 ). Hepatobiliary and pancreatic ascariasis are more common in adults. Chronic intestinal infection can manifest as abdominal pain and distention, diarrhea, and nausea. More insidious effects, especially in children, include decreased protein and fat absorption, development of vitamin A and C deficiencies, and lactose intolerance, which together lead to asthenia, stunted growth, and impaired cognitive development.
Diagnosis of ascariasis is usually made by microscopic examination of a sample of feces for characteristic thick-shelled eggs. Polymerase chain reaction (PCR) testing, although currently available only in specialized laboratories as a research tool, is also highly sensitive and specific. However, during the pulmonary phase of infection, ova will not be detectable in feces because adult worms have not yet matured and begun producing eggs; instead, larvae, as well as eosinophils or Charcot-Leyden crystals (formed from the breakdown of eosinophils), may be visualized on microscopic examination of sputum. Pulmonary disease is also usually characterized by peripheral eosinophilia and transient infiltrates on chest radiographs. The diagnosis of intestinal or biliary obstruction caused by A. lumbricoides is usually made by ultrasound examination or endoscopic retrograde cholangiopancreatography (ERCP).
Intestinal ascariasis is usually cured with a single oral dose of albendazole ( Table 327-1 ). Alternatives include mebendazole, ivermectin, moxidectin, tribendimidine, or pyrantel pamoate. No specific treatment is recommended for symptoms of pulmonary ascariasis since the condition is self-limited. In severe cases of biliary obstruction, including cholangitis, ERCP with or without resection of the ampulla of Vater is highly successful and may preclude the need for surgery.
| NEMATODE | TREATMENT |
|---|---|
| Ascaris lumbricoides | Albendazole 400 mg once. Alternatives: mebendazole 500 mg once or 100 mg bid for 3 days, ivermectin 150-200 µg/kg once, moxidectin 8 mg once, or pyrantel pamoate 11 mg/kg once with the maximum dose not to exceed 1 g |
| Hookworm ( Necator americanus and Ancylostoma duodenale ) |
Albendazole 400 mg daily for 3 days. Alternatives: tribendimidine 400 mg once; mebendazole 500 mg daily or 100 mg bid for 3 days; or pyrantel pamoate 11 mg/kg for 3 days with the maximum daily dose not to exceed 1 g |
| Cutaneous larva migrans ( Ancylostoma caninum and Ancylostoma braziliensis ) |
Ivermectin 200 µg/kg daily for 1-2 days. Alternative: albendazole 400 mg daily for 3 days |
| Trichuris trichiura | Albendazole 400 mg daily for 3 days. Alternative: mebendazole 100 mg bid for 3 days. The addition of ivermectin 200 µg/kg or moxidectin 8 mg daily for 3 days improves efficacy |
| Enterobius vermicularis | Pyrantel pamoate 11 mg/kg once, with a second dose 2 wk later; maximum dose of 1 g. Alternatives: mebendazole 100 mg once or albendazole 400 mg once, repeated in 2 wk |
| Strongyloides stercoralis | Uncomplicated infection: ivermectin 200 μg/kg daily for 2 days. ∗ Alternative: albendazole 400 mg bid for 7 days |
| Trichostrongylus spp | Pyrantel pamoate 11 mg/kg once; maximum dose of 1 g. Alternatives: albendazole 400 mg daily for 10 days or mebendazole 500 mg daily for 10 days |
| Capillaria philippinensis | Albendazole 400 mg bid for 10 days. Alternative: mebendazole 500 mg daily for 20 days |
∗ Treatment may need to be extended in immunocompromised patients with disseminated disease.
The definitive means of preventing Ascaris infection is improvement of hygiene and proper disposal of human waste. In endemic communities where this is not feasible, morbidity control consists of regular (usually annual) mass administration of an anthelminthic medication such as albendazole or mebendazole to preschool and school-aged children.
Untreated, ascariasis may lead to chronic infection that persists for years and can result in clinical complications ranging from rarely fatal intestinal or biliary obstruction to more insidious sequelae such as malnutrition. Although treatment with albendazole is highly effective, children living in endemic communities often become reinfected within months owing to continued exposure from the contaminated environment.
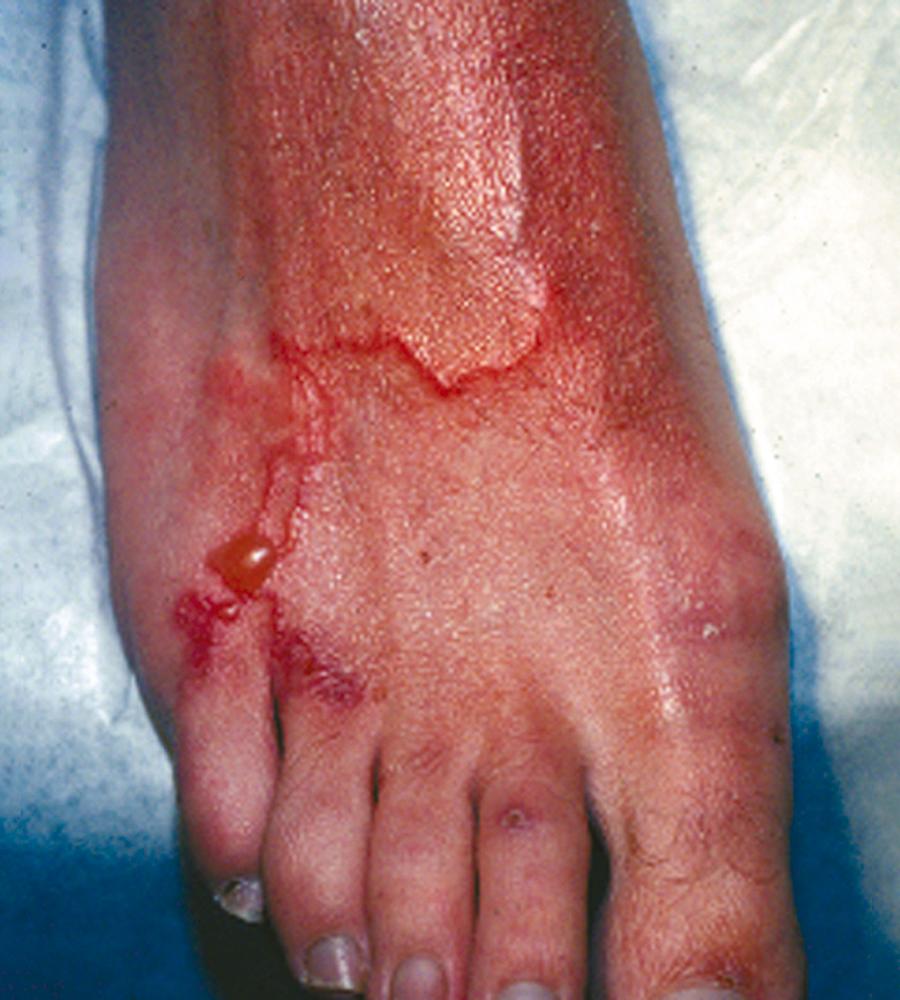
Hookworm infection in humans is due almost exclusively to two species: N. americanus and A. duodenale . However, incidental infection with the zoonotic hookworms Ancylostoma caninum , Ancylostoma braziliensis , Bunostomum phlebotomum , and Uncinaria stenocephala can cause self-limited dermatologic lesions known as cutaneous larva migrans ( Fig. 327-3 ). Additionally, Ancylostoma ceylanicum , normally a hookworm infecting cats, has been reported to cause intestinal disease in humans, especially in Asia, whereas A. caninum has been implicated as a cause of eosinophilic enteritis.
Over 400 million people are infected with hookworms worldwide. N. americanus is widespread, whereas infection with A. duodenale is more geographically restricted. The highest prevalence of infection occurs in rural areas of tropical and less developed countries, where environmental and socioeconomic conditions favor transmission. Like A. lumbricoides , hookworm eggs require soil of adequate moisture and warm temperature to incubate and develop into larvae. Equally important determinants of infection are poverty and lack of access to adequate sanitation and clean water. The prevalence of infection increases with age in children living in endemic areas until it plateaus by about age 10 years. By comparison, the intensity of infection rises at a slower rate during childhood, reaching a plateau by about age 20 years and then increasing again in the elderly.
Although cutaneous larva migrans is found throughout the tropics, in the United States it is diagnosed primarily in travelers who have recently returned from a tropical beach vacation, especially in the Caribbean, South and Central America, and Southeast Asia. Occasionally, autochthonous cases (originating where found) have been reported in the United States, usually from southeastern coastal states such as Florida and Texas. Cutaneous larva migrans occurs when larvae of the dog or cat hookworms ( A. caninum or A. braziliense , respectively) are expelled in their feces, contaminate moist soil or sand, and then come into contact with exposed skin. Other animal hookworms such as U. stenocephala and B. phlebotomum are less common causes.
Infection occurs when exposed skin comes in contact with infective filariform larvae in fecally contaminated soil or grass. Larvae penetrate the skin and enter the venous circulation. After reaching the pulmonary vasculature, larvae penetrate into alveolae, ascend the bronchial tree to the larynx, and are swallowed into the gastrointestinal tract. Larvae develop into sexually mature adult worms 5 to 9 weeks after skin penetration. Adult hookworms reside in the lumen of the small intestine, where they attach to the mucosa by means of cutting teeth ( A. duodenale ) or a rounded cutting plate ( N. americanus ). After mating, female worms produce eggs that are expelled from the body in feces; A. duodenale female worms lay approximately 28,000 eggs daily, whereas the output from N. americanus worms averages around 10,000 a day. Hookworm eggs hatch in warm, moist, sandy soil and release larvae that can infect another host. Humans are the only natural definitive hosts for these two parasites, and there are no intermediate or reservoir hosts. A. duodenale worms survive on average for 1 year in the human intestine, whereas N. americanus lives for 3 to 5 years.
The major pathology of hookworm infection is the associated gastrointestinal blood loss and resulting iron deficiency anemia. Hookworms attach to the intestinal mucosa and secrete enzymes that enable them to invade submucosal tissues and ingest villous tissue and blood. Hemoglobinases within the hookworm’s digestive canal degrade host hemoglobin for use as an essential nutrient source. The amount of blood loss is directly related to the total worm burden. A. duodenale causes more blood loss than N. americanus : each N. americanus worm results in a daily blood loss of 0.03 to 0.1 mL, whereas the corresponding figure for A. duodenale is between 0.15 and 0.26 mL.
The clinical features of hookworm infection are related to the initial larval migration through the skin and other tissues and the acute and chronic manifestations resulting from parasitism of the gastrointestinal tract by adult worms. Repeated skin exposure to hookworm larvae can result in a hypersensitivity reaction known as “ground itch,” a pruritic erythematous and papular rash that appears most commonly on the hands and feet. In contrast, when zoonotic hookworm larvae penetrate the skin to produce cutaneous larva migrans, most commonly on the feet, legs, and buttocks, they are unable to complete their life cycle in the human host and eventually die after causing a typical rash consisting of intensely pruritic, erythematous serpiginous tracks (see Fig. 327-3 ). Tracks appear after an incubation period of a few days, can be single or multiple, and advance by millimeters to a few centimeters each day. Vesiculobullous or papular lesions may develop along the tracks, as can secondary bacterial infection because of scratching. Untreated, lesions usually heal spontaneously within weeks to months following death of the larvae in the skin.
Migration of hookworm larvae through the lungs may induce mild and transient pulmonary symptoms consisting of dry cough, sore throat, wheezing, and low-grade fever. Uncommonly, acute symptomatic disease may follow the oral ingestion of A. duodenale larvae, known as Wakana syndrome, which is characterized by pharyngeal irritation, cough, dyspnea, hoarseness, nausea, and vomiting.
Gastrointestinal symptoms and signs caused by hookworm infection are uncommon. Instead, the manifestations of hookworm disease occur when intestinal blood loss exceeds the nutritional reserves of the host and results in iron deficiency anemia. Usually only moderate- and high-intensity (≥2000 eggs per gram of feces) hookworm infections produce clinical disease, which resembles that of iron deficiency anemia secondary to other causes ( Chapter 145 ). In addition, the protein losses associated with heavy hookworm infection can result in hypoproteinemia and anasarca. Clinical manifestations of iron deficiency anemia include weakness, palpitations, fainting, dizziness, dyspnea, lassitude, and headache. Uncommonly, patients may have constipation or diarrhea with occult blood in the stool or even frank melena, especially in children; there may also be an urge to eat soil (pica). Overwhelming hookworm infection may cause listlessness, coma, and even death, especially in infants. Because children and women of reproductive age have lower iron reserves, they are at increased risk of symptomatic disease.
The diagnosis of hookworm infection is made by microscopic identification of characteristic eggs in the stool. The eggs of N. americanus and A. duodenale cannot be distinguished because both are colorless and have a single thin hyaline shell with blunted ends; they range in size from 55 to 75 μm by 36 to 40 μm. Egg concentration techniques, such as the formalin–ethyl acetate sedimentation method, can be used to detect even light infections, although a direct wet mount examination is adequate for detecting moderate to heavy infections. As with A. lumbricoides , PCR testing that has been reported to be highly sensitive and specific is performed by a few specialized laboratories but is not commercially available or approved as a diagnostic test. In addition, eosinophilia is a common finding in chronic infection and during larval migration through the lungs.
For intestinal hookworm infection, three daily oral doses of albendazole 400 mg is the recommended treatment (see Table 327-1 ). Tribendimidine, which is not FDA approved and is not available or approved in most countries, has a similar efficacy profile as albendazole as a single oral dose of 400 mg, alone or in combination with other anthelminthics, such as single-dose ivermectin or oxantel pamoate. Less effective alternatives include mebendazole, pyrantel pamoate, and single-dose albendazole. Iron supplementation is recommended in patients with significant or symptomatic anemia.
For cutaneous larva migrans, although the disease is self-limited and will resolve spontaneously within weeks to a few months, treatment with one to two daily doses of ivermectin (200 µg/kg) leads to faster resolution of symptoms and skin manifestations. Albendazole is an alternative treatment of cutaneous larva migrans.
The ideal method for preventing hookworm infection is improvement in hygiene and proper disposal of human waste. In endemic communities where these public health measures have not been accomplished, control of disease consists of regular (at least annual) mass administration of single doses of an anthelminthic medication such as albendazole or mebendazole. For cutaneous larva migrans, tourists should be advised to wear shoes or sandals when walking on beaches and to avoid areas frequented by stray cats and dogs.
Severe iron deficiency anemia caused by hookworm during pregnancy can result in adverse consequences for the mother, the fetus (spontaneous abortion, intrauterine growth restriction), and the neonate (anemia, failure to thrive). In children, the anemia and protein malnutrition ( Chapter 197 ) associated with chronic infection cause long-term deficits in physical and cognitive development.
T. trichiura nematodes, or whipworms, which grow to as long as 50 mm, spend their entire life cycle in the host’s gastrointestinal tract.
The estimated worldwide prevalence of trichuriasis is 465 million, most commonly in poor, rural areas of the tropics and subtropics where disposal and treatment of human waste are inadequate. Children are more frequently infected than adults and more likely to have higher worm burdens. Humans are the only host.
T. trichiura does not have a tissue migratory phase like A. lumbricoides and hookworm. After embryonated eggs are ingested orally, they hatch and release larvae into the proximal large intestine, where they undergo a series of molts before being carried passively to the transverse and descending colon. The adult worm’s narrow anterior end embeds in the columnar epithelium, while the posterior portion protrudes into the lumen. After mating, female worms expel unembryonated eggs into the feces, which are then passed into the environment, where they must embryonate in warm, moist soil to complete the life cycle. Adult worms can survive in the host for approximately 1.5 to 2 years. The period between ingestion of eggs and detection of eggs in feces is about 90 days.
Most T. trichiura infections are asymptomatic. Symptomatic disease occurs primarily in children because most heavy infections (>10,000 eggs per gram of feces) occur in this age group. Heavy infections can manifest as an acute dysentery or as a chronic colitis resembling inflammatory bowel disease with recurrent abdominal pain and diarrhea.
Infection is diagnosed by microscopic identification of the typical barrel-shaped eggs with bipolar plugs in direct or concentrated smears of fecal specimens. PCR testing has been reported to be highly sensitive and specific by a few specialized laboratories but is not commercially available or approved as a diagnostic test.
T. trichiura responds less effectively than A. lumbricoides or hookworm to treatment with single-dose albendazole or mebendazole, so a 3-day course of either benzimidazole drug is recommended (see Table 327-1 ). The addition of ivermectin (200 μg/kg) to either drug can increase the response rate significantly. Some data also indicate that the combination of moxidectin (8 mg) plus albendazole may be somewhat more effective than albendazole alone, but the complete response rate is still below 25%.
As for A. lumbricoides and hookworm, control of trichuriasis in endemic areas consists of regular mass anthelminthic drug administration, primarily to preschool and school-aged children. Although the single doses of albendazole or mebendazole that are used in most mass drug administration programs worldwide are poorly effective for this intestinal nematode, their use is still recommended by the World Health Organization.
Chronic mucosal inflammation and edema of the colon and rectum can lead to protracted tenesmus that can result in rectal prolapse. Chronic Trichuris colitis can also lead to malnutrition, impaired growth, and anemia.
E. vermicularis , or pinworm, is transmitted by the fecal-oral route. Adult worms measure from 2 to 5 mm.
E. vermicularis is found worldwide and is the most prevalent nematode infection in temperate climes. Transmission is frequent in primary schools and daycare centers.
After ingestion, embryonated eggs hatch in the small intestine, where larvae develop into adult worms that migrate to the large intestine, where they mate. Gravid female worms emerge nightly from the anus to deposit large numbers of eggs (11,000 per worm) on the perianal and perineal skin, where they embryonate within 6 hours. If they are still on the skin, infective larvae are released and can migrate back through the anus into the rectum (retroinfection). Alternatively, autoinfection occurs when eggs are transferred to the mouth via scratching skin on which eggs have been deposited. Larvae may also migrate into the female genital tract and establish ectopic infection.
Although pinworm infections may be asymptomatic, perianal pruritus is the most common symptom and is due to an allergic response to worm proteins. The pruritus can be intense and result in chronic sleep deprivation in children. Rarely, adult E. vermicularis may precipitate appendicitis. When hatched larvae migrate into the female genital tract, vulvovaginitis, salpingitis, or peritonitis may develop.
Pinworm infection is diagnosed by identifying the characteristic eggs through microscopic examination of a piece of cellophane tape applied to the perianal region immediately after waking in the morning. E. vermicularis eggs are oval and slightly flattened on one side. Eggs are not usually found in fecal samples. Repeated examination may be necessary.
Pinworm infection is treated with a single dose of pyrantel pamoate, mebendazole, or albendazole, which must be repeated 2 weeks later because the drugs do not kill eggs or developing larvae (see Table 327-1 ). Given the high rate of transmission, all household members and individuals in close contact with the patient (e.g., other children attending the same daycare center) should also be treated. Bedding and underclothes should be thoroughly laundered in hot water followed by a hot dryer to kill remaining eggs.
Enterobiasis usually does not result in severe manifestations, particularly in children, and it responds rapidly to appropriate treatment.
S. stercoralis , commonly known as threadworm, is endemic in warm climates worldwide, including parts of the United States. Adult males are about 0.75 mm long, and adult females are about 1.0 mm long.
S. stercoralis infection is endemic in the tropical and subtropical regions of sub-Saharan Africa, Asia, Latin America, and areas of Eastern and Southern Europe, with a worldwide prevalence of up to 100 million. In the United States, Strongyloides -related hospitalizations are rare, and infection is diagnosed most frequently in immigrants; however, strongyloidiasis is still endemic in parts of rural Appalachia. S. stercoralis can also be transmitted sexually through oral-anal contact, most often among men who have sex with men, or via transplantation of solid organs.
Infection occurs when exposed skin comes in contact with free-living filariform larvae in soil contaminated with human feces. Like hookworm, larvae penetrate the skin, enter the vasculature, and migrate to the pulmonary capillaries, where they penetrate into the alveoli, ascend the bronchial tree to the pharynx, and are swallowed into the gastrointestinal tract. Further development into adult worms occurs in the upper small intestine, where parasites live embedded in the mucosa. Unlike most nematodes, S. stercoralis reproduces by parthenogenesis, with no apparent parasitic male worm present in the human host. Within 25 to 30 days after infection, female worms begin laying eggs, which rapidly hatch in the intestinal lumen and release noninfectious rhabditiform larvae that migrate to the colon and are excreted in feces. Alternatively, larvae may penetrate the colonic mucosa or perianal skin after migrating out of the anus and enter the circulation directly, a mechanism known as autoinfection that can lead to maintenance of parasitism in the host for decades.
In the soil, infectious filariform larvae can be produced either directly via transformation from rhabditiform larvae or indirectly from eggs released by free-living adult worms that have developed from rhabditiform larvae in warm, moist, sandy soil.
In immunologically competent individuals, infection does not usually result in symptomatic disease. However, serious complications may occur in individuals with cell-mediated immunodeficiency such as patients chronically taking corticosteroids, solid organ transplant recipients, patients with hematologic malignancies, or patients infected with human T-cell lymphotropic virus type 1. In these patients, the S. stercoralis autoinfection cycle can become amplified and lead to a hyperinfection syndrome with a large increase in the total worm burden in the infected person. Hyperinfection can lead to life-threatening dissemination of larvae and adult worms to aberrant sites such as the brain, pancreas, and kidneys. For unknown reasons, acquired immunodeficiency syndrome (AIDS) has not been associated with hyperinfection syndrome or with disseminated strongyloidiasis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here