Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
We are in a technological era where advancements in image-guided systems, robotics, machine learning/neuromodulation, and state-of-the-art image-guided operating suites are creating new possibilities in operative neurosurgery. This is resulting in numerous changes and improvements with respect to patient experience, care, and outcomes. Spine surgery is uniquely affected given its heavy reliance on intraoperative devices, biologics, and imaging. Novel tools are allowing us to perform surgeries with smaller incisions, as is the case with video-assisted endoscopy, with others fully relying on computer assisted navigation (CAN), as in cases of minimally invasive spine surgery (MISS). The integration of these, and similar technologies, is pushing the boundaries of spine tumor surgery, degenerative spine surgery, deformity surgery, MISS, and spine trauma surgery.
While traditional intraoperative imaging tools (i.e., radiography, C-arm fluoroscopy, biplanar fluoroscopy) remain a mainstay of most spine practices, newer technologies like 3D fluoroscopy, cone-beam CT, real-time flash registration, and intraoperative CT/MRI are allowing a new generation of spine surgeons safer, more accurate placement of hardware and better localization of difficult anatomy in real time. These modalities offer the added benefit of less radiation exposure to the surgeon, operative staff, and patient. Naturally, there are limitations to these technologies that have kept them from replacing the aforementioned mainstays. Among the challenges posed by novel technology is a steep learning curve for the appropriate implementation and effective use of such tools and techniques. As with all new developments, the upfront costs associated with the technology demands there be critical analysis of patient outcomes and long-term cost-effectiveness, prior to embracing them fully. Spine trauma surgery specifically has benefited from these advances as we continue to learn from its limitations to help make these technologies better for surgeon use.
The management of spine trauma patients has evolved rapidly over the last decade, from the imaging modalities used to diagnose them to the treatment of spinal cord injury. CAN is among the most revolutionary additions to spine surgery, allowing real-time feedback based on intraoperative radiography (3D x-ray, CT, and/or MRI) or registration points. It has been well established in the literature that, for pedicle screw placement, CAN demonstrates superior accuracy over that of a free-hand technique in the placement of pedicle screws. A case report by Kakarla et al. showed the use of navigated percutaneous thoracic pedicle screw fixation in six spine trauma patients with unstable spine fractures. In this study, a total of 37 pedicle screws were placed, with 16% of screws having a breach less than 2 mm and 3% with a breach beyond 2 mm. Additionally, the study noted no neurological complications detected and no revisions. Studies in both elective and trauma spine surgery repeatedly demonstrate the accuracy of pedicle screw placement with CAN, as well as the safety and decreased need for revision surgery for malpositioned screws.
Other modalities for spinal trauma, like percutaneous MISS techniques, are believed to improve outcomes by limiting soft tissue disruption and avoiding the risks of conventional open approaches. These result in shorter recovery periods and hospital stays. The application of this technique additionally limits blood loss, which, importantly, can be substantial in open trauma cases, and is not infrequently complicated by patients with multisystem issues. Percutaneous CAN options offer an important alternative in such cases. Furthermore, for trauma cases where the anatomy is completely distorted from injury and/or prior intervention, navigation can be useful for real-time feedback in instrumentation and assessing the corrected deformity. Although the literature is starting to support the application of MISS in degenerative surgery, the level of evidence supporting its use in the trauma population is lacking compared to its use in those with degenerative disease.
While intraoperative time was originally felt to be a likely disadvantage of the technology, the concern has been dispelled. Certainly, there is an upfront learning curve for both surgeons and operating room staff; however, the literature suggests that as the utilization of CAN has increased at certain centers and institutions, their operative time has concurrently either remained unchanged or decreased. As an example, a study by Lang et al. using intraoperative 3D fluoroscopy-based navigation for treating Hangman fractures through a C2–C3 pedicle screw fixation construct, showed that CAN did not lengthen the operative time (134.2 ± 8.0 min in MISS vs. 139.3 ± 25.8 in open, P >.01). In an effort to help address some of these concerns regarding intraoperative times, groups like Navarro-Ramirez et al., who have switched to a total navigation system for MISS and completely eliminated fluoroscopy in most surgeries, have developed algorithms to help with workflow. Their eight-step surgical workflow for total navigation is just one example of how a systems approach can help make certain CAN procedures faster than open techniques, even in spine trauma cases.
Beyond the benefits of reducing operative time, there is a significant reduction in radiation exposure to both the patient and staff when using CAN. The increased awareness of the hazardous effects of radiation exposure is changing the way surgeons, OR staff, and hospital administrators overseeing operating rooms perform surgeries in order to make the environment safer to all involved. A spine surgeon often experiences as much as 50 times the lifetime radiation dose of a surgeon in other orthopedic subspecialties, such as a hip surgeon. More complex spine procedures like MISS, tumor, and trauma that rely on fluoroscopy, given the complicated anatomy or lack of visible anatomical landmarks, subject all parties to higher doses of radiation. CAN is uniquely positioned to help in reducing this effect as it does not rely on the same radiation doses. It has the added benefit of enhanced 3D visual experience in real time.
•The patient is an 80-year-old man with a history of hypertension and osteopenia who sustained a fall from standing a month prior to his current hospitalization. From his original fall, a CT revealed a mild compression fracture at T12 that was treated conservatively with bracing, given its imaging features as well as the minimal subjective back pain ( Fig. 3.1A ). However, over the course of a month, his back pain worsened significantly and he was readmitted. The neurologic examination revealed normal strength, reflexes, and sphincter tone. However, he had substantial back pain with midline tenderness to palpation.
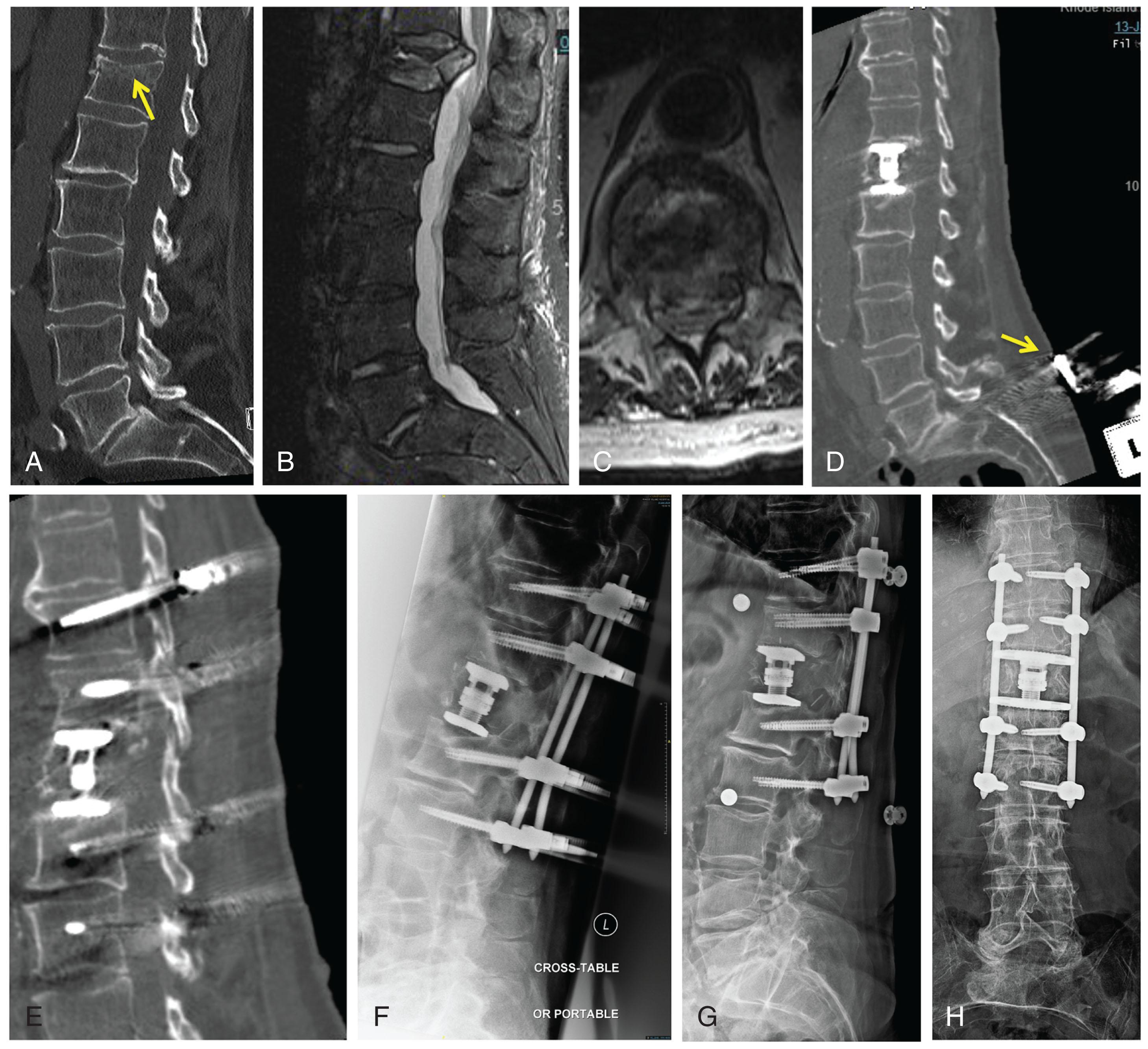
•His original CT scan from his fall a month prior revealed only a mild superior endplate fracture at T12 with no other bony fractures or associated focal kyphosis ( Fig. 3.1A ). There was also no fracture to the posterior elements or splaying of the posterior elements. An MRI obtained upon readmission a month later revealed progression of his T12 fracture, now with significant height loss, and a burst component into the canal with focal kyphosis (see Fig. 3.1B ). The posterior ligamentous complex was without injury on MRI. On the axial view of the MRI, the T12/L1 space was moderately stenotic from the burst fragment abutting the conus (see Fig. 3.1C ).
•In order to decompress the burst fragment and restore height and alignment, a decision was made for a right lateral decubitus transthoracic MISS approach for corpectomy of T12 followed by posterior percutaneous stabilization. The posterior percutaneous stabilization was done with CT-guided intraoperative navigation (BrainLab). Iliac pins with an array were placed for intraoperative navigation (see Fig. 3.1D ). An intraoperative CT scan was obtained following the placement of the array. Four small incisions were made for the posterior percutaneous surgery, two on each side below and above the cage reconstruction at T12. These incisions were mapped out using our navigation to ensure they would encompass our screw trajectories.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here