Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This work was supported by the National Institutes of Health (U01-HL141074).
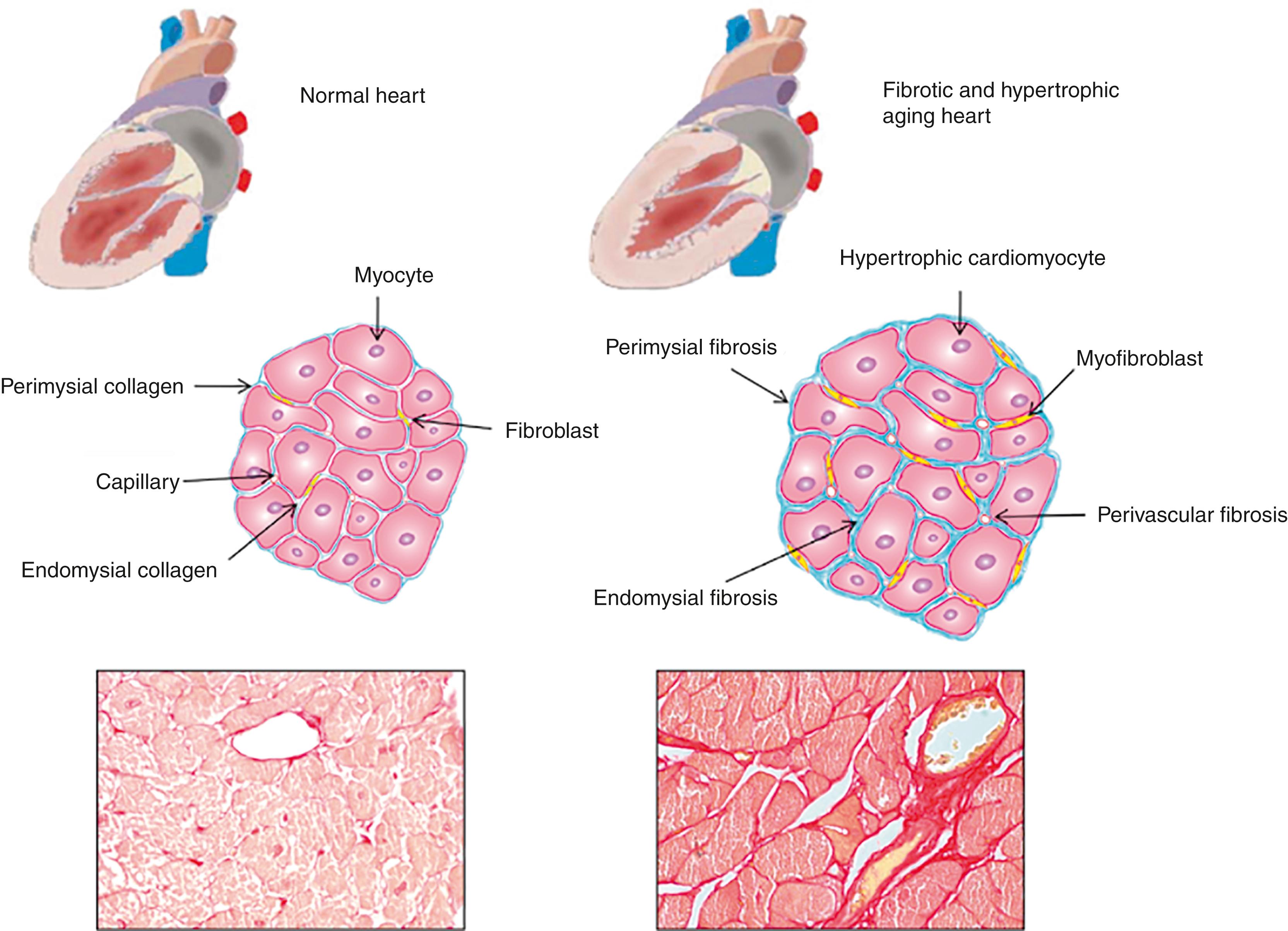
The aging heart is characterized by a number of morphologic and structural changes manifested at the different levels of biologic hierarchy. These changes lead to the functional decline of the aging heart and, particularly, to its diminished ability to meet the increases in demand. The progressive changes in cardiac anatomy and physiology occur even in apparently healthy individuals. Age-dependent remodeling of the heart in healthy populations is predominantly associated with cardiomyocyte hypertrophy and fibrotic remodeling, accompanied by a decline in diastolic function, as demonstrated by the Framingham Heart Study and the Baltimore Longitudinal Study on Aging.
As reviewed extensively by Biernacka et al., animal model studies provide consistent evidence of aging-related cardiomyocyte hypertrophy accompanied by an increase in myocardial collagen content. Additionally, histologic analysis of nonhypertensive aging hearts reveals progressive loss of cardiomyocytes because of necrotic and apoptotic cell death. , Animal experiments have also revealed an increased collagen deposition in the aging heart. The findings are similar in human hearts. The aging human heart exhibits a progressive increase in left ventricular (LV) mass despite a reduction in the total myocyte number. Collagen content increases with age in the normal human heart ; studies involving human subjects have documented an age-related increase in cardiac fibrosis. , Furthermore, myocardium from senescent individuals exhibits increased collagen deposition and thicker endomysial and perimysial collagen fibers. Autopsy studies of cardiac tissue from human subjects free of pathologic conditions have shown that collagen content increases by almost 50% between the third and seventh decades of life.
The differences between the normal and senescent heart are illustrated in Fig. 36.1 . In the normal heart, thin layers of perimysium and endomysium surround myocardial bundles and individual myocytes, respectively. In the senescent heart, there is increased deposition of perimysial and endomysial collagen, referred to as interstitial fibrosis . Interstitial fibrosis initially progresses without myocyte loss and is accompanied by cardiomyocyte hypertrophy as part of an adaptive response aimed at preserving cardiac output and normalizing wall stress. Eventually, however, replacement fibrosis takes place as cardiomyocytes undergo necrosis and apoptosis. Finally, in the aging heart there is transition of fibroblasts to myofibroblasts. Fibroblasts primarily regulate collagen turnover in tissues; however, under certain conditions that remain controversial, fibroblasts are activated and undergo phenotypic transition into myofibroblasts, the key effector cells in fibrotic remodeling.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here