Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pain management in the critically ill patient is complex. Each patient brings a unique set of sociodemographic, pharmacokinetic, and pharmacogenomic variables that are coupled with underlying psychosocial and medical comorbidities. These not only influence a patient’s response to painful stimuli but also to treatment. We know that critically ill patients experience pain at rest, during routine intensive care unit (ICU) care, and during procedures. , Pain can originate from multiple sources, including somatic, visceral, and neuropathic. Pain may be acute, chronic, or acute on chronic in nature. Comorbidities such as depression and anxiety can exacerbate pain. , Patients that are younger, female, and of nonwhite ethnicity are more likely to experience more intense pain. Aging patients experience many physiologic changes that may decrease their perception of pain and increase their vulnerability to the adverse effects of pain medications.
It is imperative to adequately treat pain in the ICU patient. Pain during a procedure is not only influenced by the type of procedure (including routine care such as turning) but also by preprocedural pain intensity. , , , , Adequate assessment of pain and preemptive analgesia are essential. Inadequate pain control contributes to the development of delirium, and severe pain may lead to cardiac instability (tachycardia, bradycardia, hypertension, and/or hypotension), respiratory distress (desaturation, bradypnea, and/or ventilator distress), and immunosuppression. ,
To optimize an analgesic regimen, it is important to obtain a thorough past medical history that includes a complete list of medications and whether alcohol, tobacco, or opioid dependence is present. Managing drug withdrawal in addition to achieving satisfactory analgesia requires a multimodal approach.
Multimodal analgesia combines two or more drug classes or techniques, employing different mechanisms of action that may target multiple pain pathways in order to achieve a synergistic or additive effect. This results in lower opioid consumption with the same or improved level of comfort. Multimodal analgesia may include opioids, nonopioid analgesics such as nonsteroidal antiinflammatory drugs (NSAIDs) or gabapentinoids, regional or neuraxial blocks, and nonpharmacologic therapies. Each patient’s medical history, allergies, age, injuries, and comorbidities will dictate the optimum regimen. Pain management protocols that mandate the use of validated pain and sedation scales consistently decrease the consumption of opioids and sedatives in ICU patients. ,
An assessment-driven and standardized pain management protocol improves ICU patient outcomes, but assessment of pain in the critically ill patient can be challenging. Many ICU patients are ventilated, cognitively impaired, and/or unable to self-report pain. Consequently, these patients are at risk for undertreatment of their pain. Valid assessment tools help guide analgesia while avoiding excess medication administration in those patients with adequate pain control. , A number of assessment tools are available for use in the ICU patient.
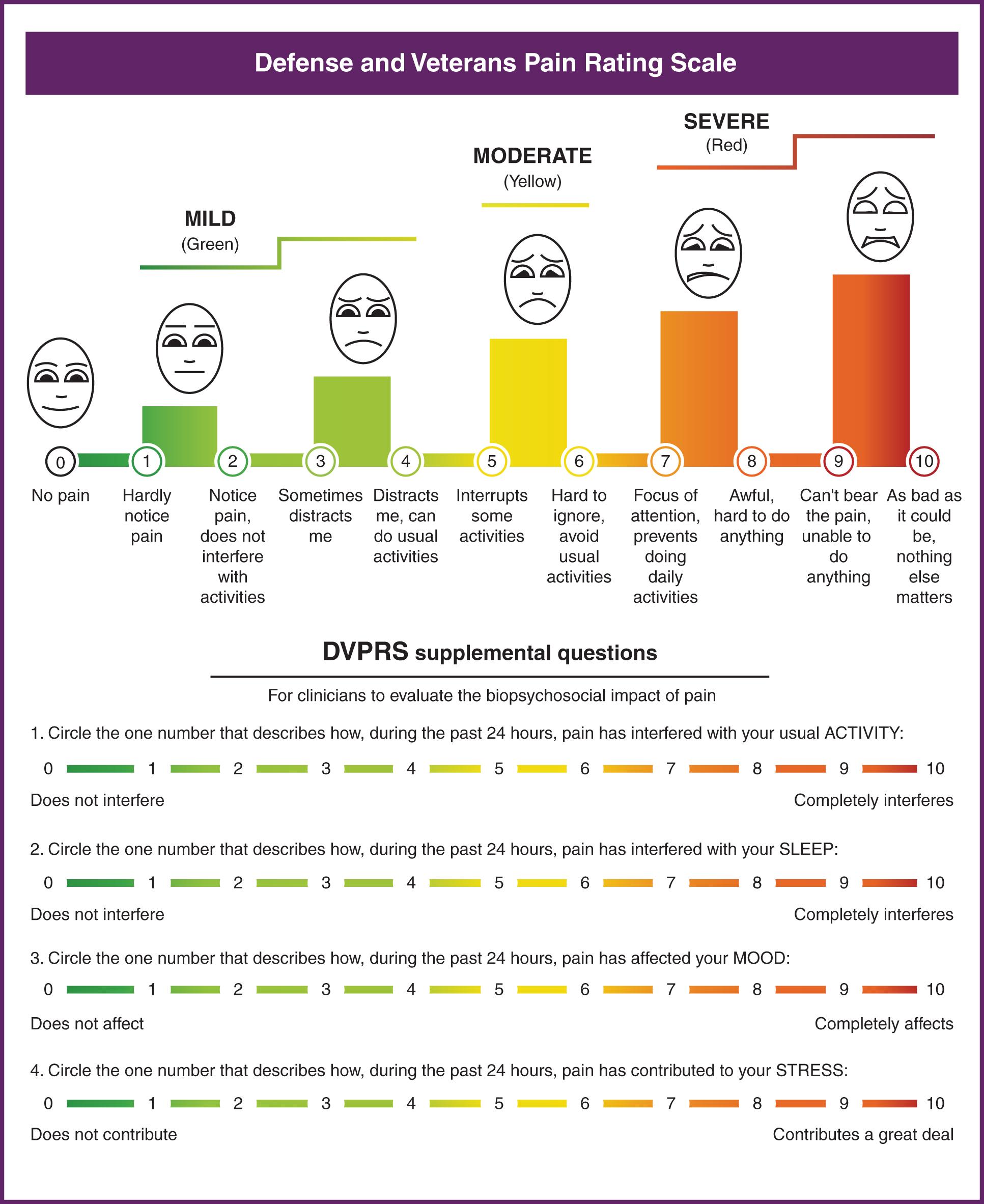
For those patients able to self-report pain, the Numeric Rating Scale (NRS) in a visual format had the best sensitivity, negative predictive value, and accuracy in ICU patients. More recently, the Defense and Veterans Pain Rating Scale (DVPRS) has become increasingly popular. It combines a 1–10 pain scale with facial expressions and colors to express pain intensity. The DVPRS also includes supplementary questions to measure the degree to which pain interferes with a patient’s usual activity, sleep, mood, and stress ( Fig. 3.1 ).
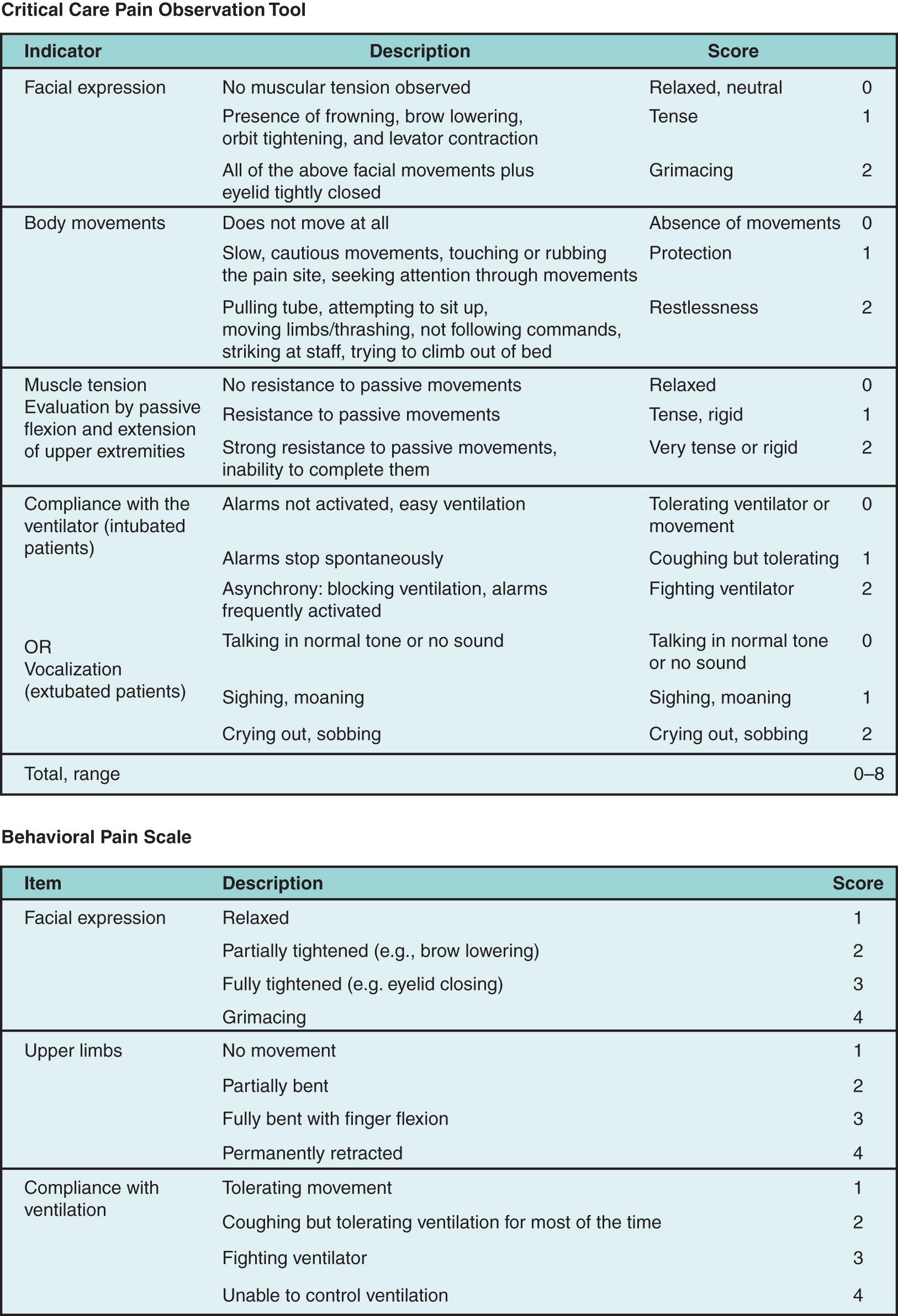
The Behavioral Pain Scale (BPS) and the Critical Care Pain Observation Tool (CPOT) ( Fig. 3.2 ) have the greatest validity and reliability for monitoring pain in those unable to self-report. BPS is a three-domain assessment tool with four possible scores in each domain. Scores range from 3 to 12, with scores above 6 indicating an unacceptable level of pain. , CPOT is similar, with four domains and scores ranging from 0 to 2 in each domain. The possible score ranges from 0 to 8, with 3 or above indicating the presence of pain.
Vital signs (heart rate, blood pressure, respiratory rate, oxygen saturation, and end tidal carbon dioxide) are not considered valid indicators for pain in critically ill patients and should only be used as cues to initiate further evaluation using one of the validated assessment measures. In unresponsive or paralyzed patients in which the validated scales are impossible to use, no current assessment methods are available. Promising technology under development for this patient population includes measuring heart rate variability (Analgesia Nociception Index), incorporating several physiologic parameters (Nociception Level Index), and examining pupillary reflex dilation using video pupillometry.
Historically, opioids were considered the mainstay of treatment for non-neuropathic pain in the critically ill patient population because of their high potency and efficacy. , , Opioids act at specific G-protein–coupled receptors designated delta, kappa, and mu. These exist predominantly in the central nervous system, but also peripherally and in certain organs (notably the gastrointestinal tract and heart). By acting at these receptors, opioids decrease the perception of pain without inducing loss of consciousness. All clinically relevant opioids are active at the mu receptor, and some have additional activity at other receptors.
The decision regarding which opioid to prescribe is highly dependent on specific patient comorbidities and circumstances. When titrated appropriately, all intravenous opioids are considered equally effective with regard to analgesic efficacy and clinical outcomes. However, studies have shown patients may demonstrate variability in opioid pharmacodynamics and pharmacokinetics, resulting in more favorable reactions to one opioid over another. Thus providers must be comfortable with many different opioids, depending on the patient’s reaction and clinical course. Common parenteral and oral opioids are summarized in Table 3.1 .
| Opioid | Dosage Forms | Onset | Pharmacokinetics | Considerations |
|---|---|---|---|---|
| Fentanyl | IV, IM, intranasal, transdermal, transmucosal | 1–2 min |
|
|
| Hydromorphone | PO, IV, IM, SQ |
|
|
|
| Morphine | PO, IV, IM, SQ, transdermal, transmucosal |
|
|
|
| Oxycodone | PO | 10–30 min |
|
|
| Hydrocodone | PO | 15–30 min |
|
|
| Methadone | PO | 30–60 min |
|
|
| Tramadol * | PO | 60 min |
|
|
* Although not a typical opioid, tramadol is structurally related to morphine and has effects at opioid receptors
Though opioids remain a cornerstone of pain management, considerable attention is now focused on limiting the opioids required through the use of multimodal therapy. This is primarily because of the many side effects of opioid-centered analgesia. Short term, these can include physical dependence, ileus, constipation, nausea and vomiting, respiratory depression, sedation, pruritus, and urinary retention. Opioids may increase ICU length of stay and worsen post-ICU patient outcomes. Long-term sequelae of opioid use include addiction, immunosuppression, and opioid-induced hyperalgesia. These complications are prevalent, regardless of the specific opioid used or the route of administration.
Particular attention should be paid to bowel dysfunction associated with opioids secondary to binding of mu receptors in the enteric nervous system. Opioids often induce constipation or worsen preexisting constipation; prevalence of these conditions with opioid use vary in the literature from 22% to 81% of patients. Thus prevention via a robust bowel regimen that includes laxatives and/or bulking agents is an essential consideration when prescribing opioids.
Despite being a commonly used analgesic and antipyretic, acetaminophen’s exact mechanism of action remains unknown. It has consistently been shown to reduce opioid requirements when used as an adjunct; furthermore, acetaminophen has minimal side effects. For the critically ill patient, acetaminophen offers an advantage in that it can be administered via multiple routes of administration (intravenous [IV], orally [PO], nasogastric [NG], per rectum [PR]). Although the IV formulation has a slightly faster onset when used as a first dose, there is minimal difference between the IV and enteral/rectal formulations when administered in a scheduled manner. Acetaminophen carries a risk of hepatotoxicity, though this is more commonly noted when high doses are used for chronic pain rather than short-term acute pain. In 2011, the Food and Drug Administration (FDA) released a recommendation to reduce the maximum daily dose of acetaminophen from 4000 mg/day to 3000 mg/day; however, it is important to note that this recommendation was aimed at consumers exposed to numerous over-the-counter acetaminophen-containing combination products. In the inpatient setting, the maximum daily dose should remain 4000 mg/day.
NSAIDs work as analgesics and antipyretics by inhibiting cyclooxygenase (COX) enzymes, thereby reducing the formation of prostaglandins. A wide variety of NSAIDs are available with numerous routes of administration (IV, PO, NG, PR, topical) and varying degrees of selectivity to COX-1 and COX-2.
Historically, there has been a reluctance to use NSAIDs in the acute pain setting because of their adverse effects, including increased risk of gastrointestinal bleeding, nephrotoxicity, impaired platelet function, and impaired wound healing. NSAIDs have long been avoided in the setting of fractures because of the risk of nonunion; however, more recent literature reviews have shown that this does not appear to be a long-term issue, particularly with early postinjury or postoperative administration. Thus the most recent guidelines support their use in this setting.
Platelet inhibition is primarily associated with inhibition of COX-1 and can be avoided with the use of COX-2 selective agents such as celecoxib. Unfortunately, COX-2 selective agents are not without risk—they are contraindicated after coronary artery bypass graft surgery and carry a risk of cardiovascular thrombotic events, such as myocardial infarction and stroke. Despite their known risks, NSAIDs can be beneficial in mitigating opioid exposure in select patient populations.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here