Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The oropharynx is a common site for the presentation of upper aerodigestive tract malignancies, more than 90% of which are histopathologically squamous cell carcinoma.
A steadily progressive epidemic of human papillomavirus–associated oropharynx squamous cell carcinoma marks current head and neck epidemiology; these tumors exhibit a unique molecular biology characterized by p16 tumor suppressor protein overexpression and favorable oncologic outcomes compared with chemical carcinogen–associated tumors.
Diagnostic evaluation includes a thorough history, physical examination, imaging, endoscopy, and biopsy/cytology; p16 immunohistochemistry is performed for prognostication.
Therapeutic management of oropharyngeal cancer depends on staging, tumor location, projected posttreatment function, and the patient's preference after thorough informed consent.
Transoral approaches, including transoral laser microsurgery and robotic surgery, are being increasingly used in the surgical treatment of oropharyngeal cancers to achieve good functional and oncologic outcomes.
Most oropharyngeal cancers can be approached transorally. However, lateral and transhyoid pharyngotomy may be necessary for selected tumors with difficult transoral access.
Early-stage lesions can be treated with single-modality therapy including transoral surgery or radiation. Advanced-stage lesions often need multimodality therapy, including definitive surgery and postoperative radiation therapy with or without chemotherapy or definitive chemoradiation.
As for other malignancies of the head and neck, thorough follow-up enables early detection of recurrences and timely intervention for effective disease control in oropharyngeal cancers.
In addition to oncologic control and patient survival, quality of life, functional outcomes, and treatment morbidity are key factors in the judgment of therapeutic success.
The oropharynx is a common site for presentation of upper aerodigestive tract malignancies, the majority of which are squamous cell carcinoma (SCC) histopathologically. The most recent GLOBOCAN report (2012) from the International Agency for Research on Cancer estimates that there are over 142,387 new cases of pharyngeal cancer (excluding nasopharynx) worldwide annually as well as an age-standardized incidence of 1.9 per 100,000. In the United States in 2017—based on Surveillance, Epidemiology, and End Results (SEER) estimates—the number of new cases was about 17,000. Accurate population-level incidence estimation for oropharyngeal cancer is made difficult by the practice of often classifying these neoplasms in combination with the oral cavity and/or hypopharyngeal primaries. Tobacco and/or alcohol consumption have been the traditional risk factors worldwide for oropharyngeal SCC (OPSCC). However, the epidemiology has drastically changed—mainly in North America, Europe, and Australia—toward a more common etiologic association with the human papillomavirus (HPV). A meta-analysis of 102 studies, including 5396 OPSCC patients across the world, reported changes in the overall prevalence of HPV-associated OPSCC over time from 40.5% before 2000 to 64.3% between 2000 and 2004 and 72.2% between 2005 and 2009. The population-level incidence of HPV-associated OPSCC in the United States increased by 225% (0.8 per 100,000 to 2.6 per 100,000) from 1988 to 2004, with a decline in the non-HPV OPSCC by 50% (2.0 per 100,000 to 1.0 per 100,000). Given the current rates of increase in the prevalence of HPV-associated OPSCC, these cancers are projected to comprise about 47% of all head and neck cancers in the United States by 2030. Survival and prognosis for patients with oropharyngeal malignancies depend on early detection, initial staging of the disease, and the treatment modality. Therapeutic options as well as the understanding of the etiopathology for these tumors have greatly evolved in recent years. Therefore intensive knowledge of the existing treatment modalities is required, and new treatment strategies based on the latest studies and research must be aimed for in the future.
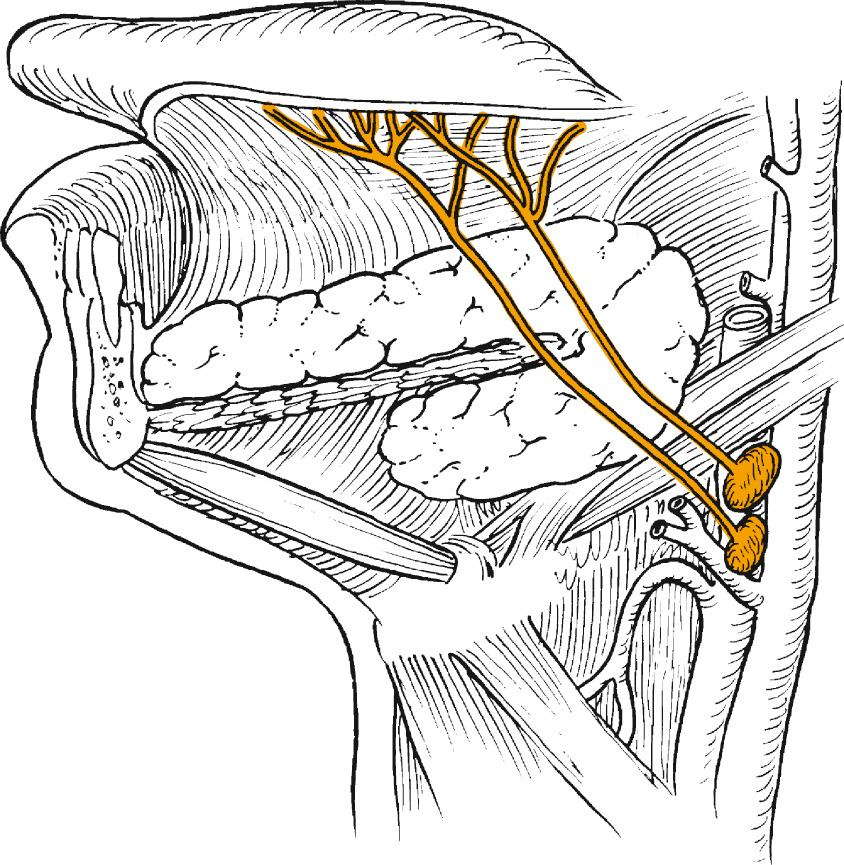
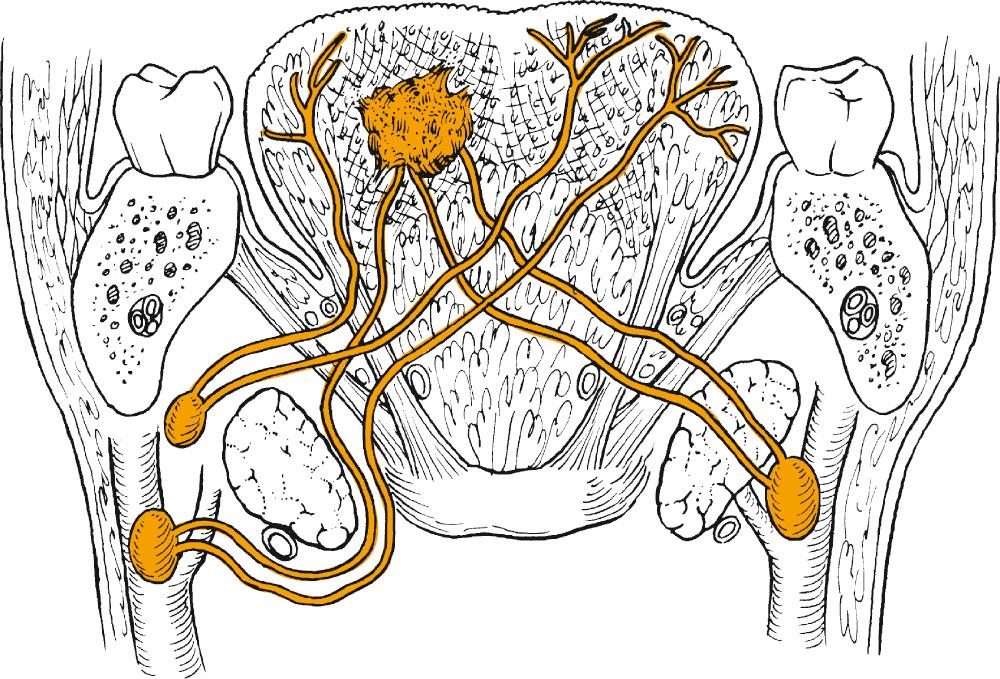
The structure of the oropharynx can be subdivided into the soft palate, tonsils, base of the tongue, and posterior oropharyngeal wall. The oropharyngeal borders are the soft palate superiorly and the hyoid bone and vallecula inferiorly; the ventral border is the base of the tongue, which ends at the circumvallate papillae. The palatine tonsillar fossae form the lateral borders. In the following sections, the anatomic sites are described separately, accounting for their specific characteristics ( Figs. 96.1 and 96.2 ).
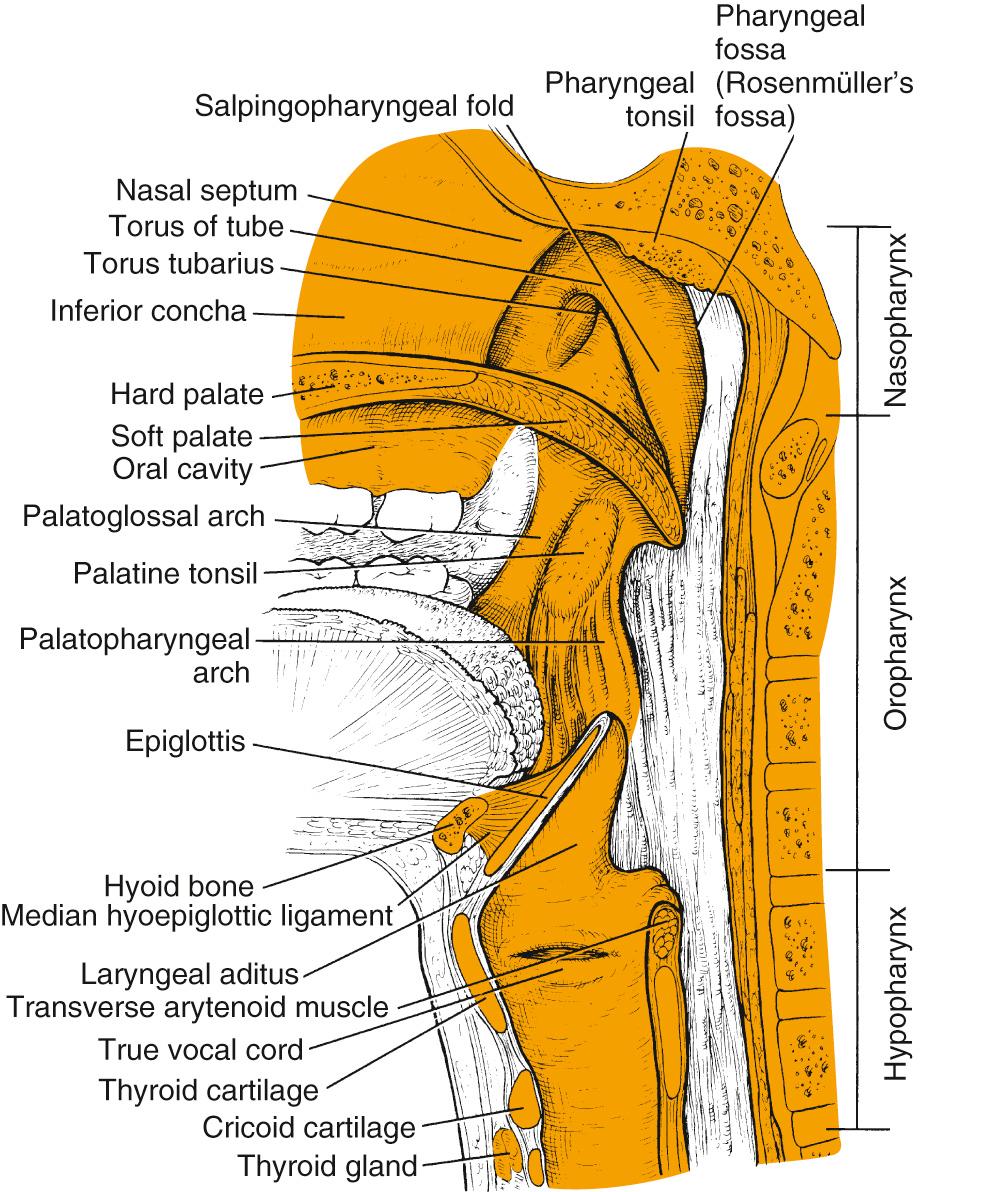
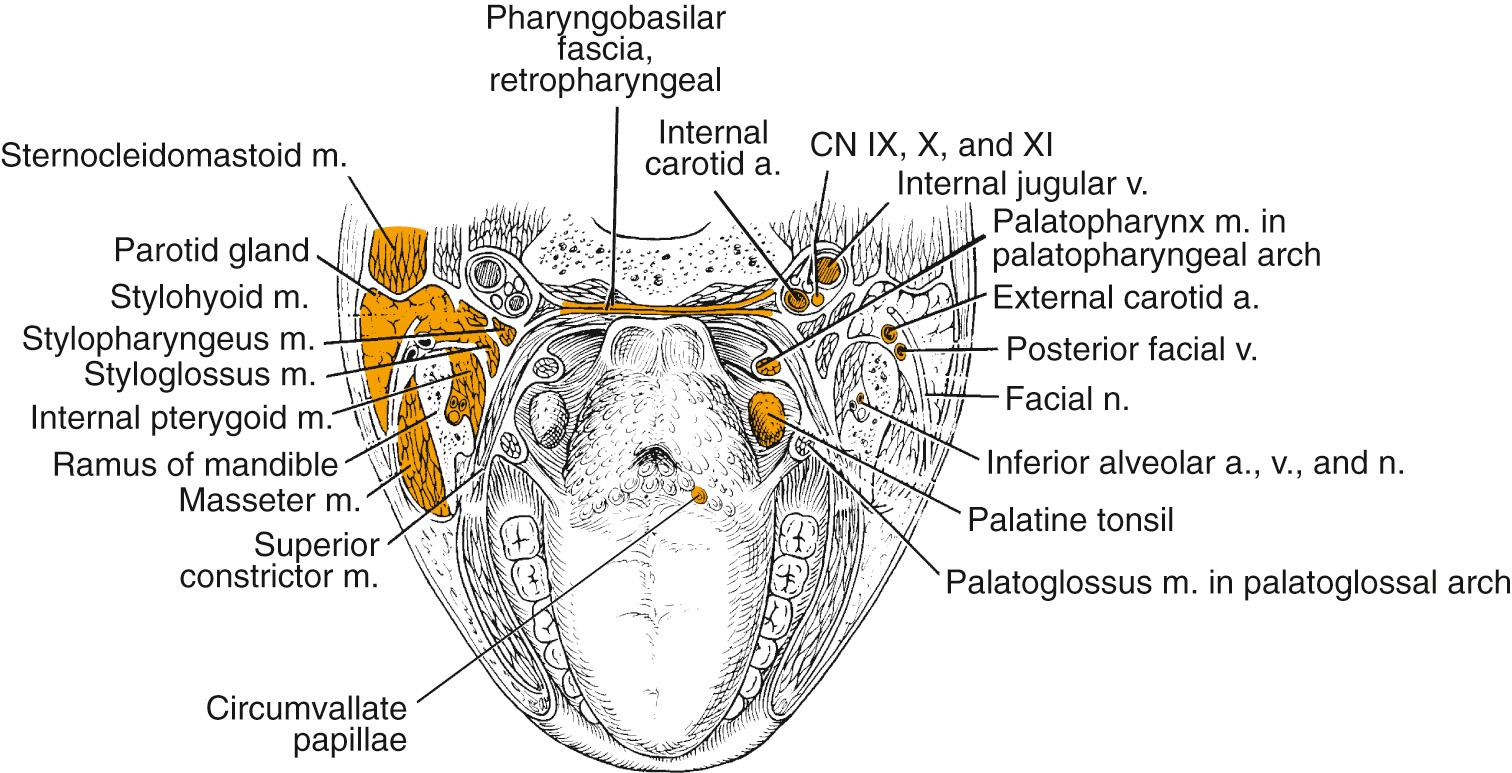
The soft palate incompletely separates the nasopharynx and oral cavity from the oropharynx. It is formed by the palatopharyngeal arch and the uvula and contains the levator and tensor muscle of the palate, the uvular muscle, and the palatopharyngeal and constrictor muscles laterally. Minor salivary glands exist within the soft palate tissue, and the blood supply is based on the ascending palatine branch of the facial artery. The cranial fibers of the accessory nerve provide motor innervation to the soft palate musculature via the pharyngeal branch of the vagus nerve except for the tensor veli palatini, which is supplied by the mandibular nerve. The sensory supply to the soft palate is via the glossopharyngeal nerve and lesser palatine nerve.
The tonsillar region contains lymphoid tissue of the palatine tonsil, located on the lateral pharyngeal wall. The palatine tonsils are embedded in the fossae between the anterior and posterior faucial pillars, formed by the palatoglossus and the palatopharyngeus muscle, respectively. The tonsils receive their vascular supply by the ascending pharyngeal artery, branches of the facial artery, posterior branches of the lingual artery, and palatine branches of the internal maxillary artery. Lesser palatine branches of the maxillary nerve and glossopharyngeal nerve branches provide neural innervation.
The base of tongue extends from the circumvallate papillae to the vallecula and to the glossopalatine sulci laterally. The tongue base is rich in lymphatics, and lingual tonsils along with the palatine tonsils form part of the Waldeyer ring . The blood supply is provided by the lingual arteries, mainly the dorsal lingual branch. The motor innervation of the tongue musculature is provided by the hypoglossal nerve. The glossopharyngeal nerve provides sensory and taste innervation except for the most posteroinferior part, which receives innervation from the internal laryngeal branch of the vagus nerve.
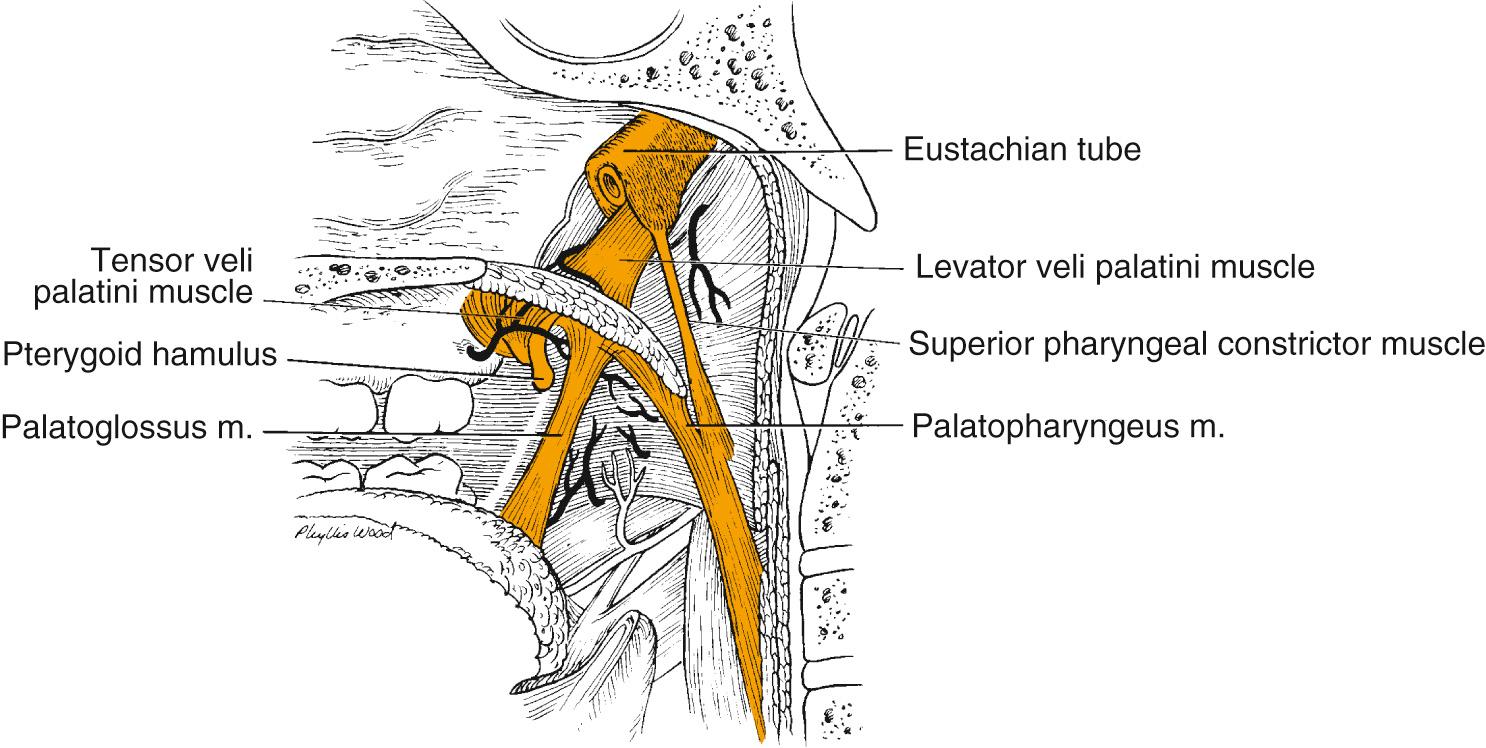
The posterior pharyngeal wall of the oropharynx extends from the region of the soft palate to the epiglottis and borders the tonsillar fossae and the lateral aspect of the piriform sinuses laterally. The wall is composed of mucosa, submucosa, pharyngobasilar fascia, pharyngeal muscles including the superior constrictor and upper fibers of the middle constrictor, and buccopharyngeal fascia ( Fig. 96.3 ). Motor supply is derived from the cranial accessory nerve through the pharyngeal branches of the vagus, whereas sensory efferents travel mainly via the pharyngeal branches of the glossopharyngeal nerve and partly through branches of the vagus.
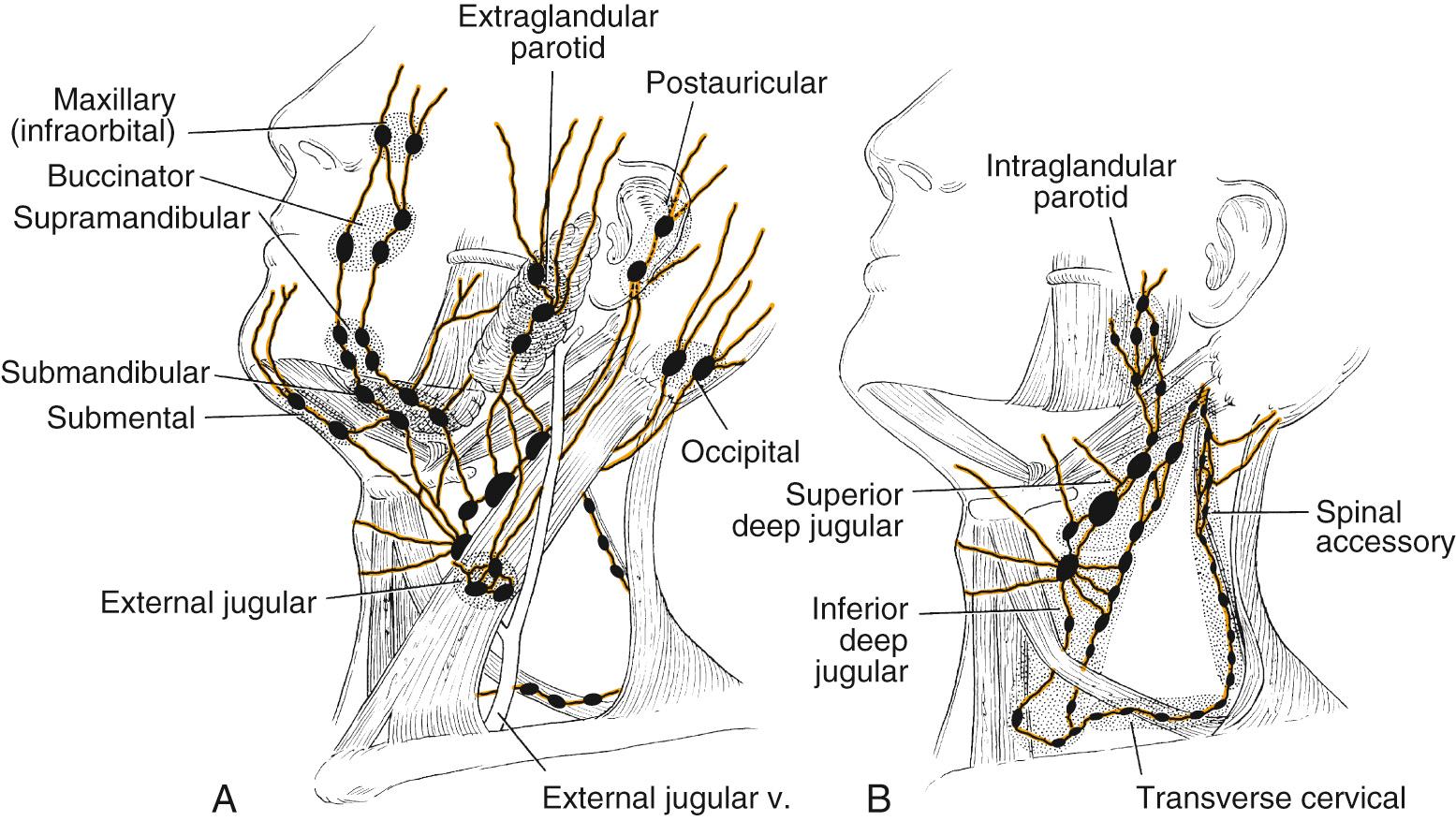
The lymphatic spread from the oropharynx depends on the location and size of the primary tumor. A solid understanding of the lymphatics in this region is of great importance for the outcome of surgical and/or radiotherapy (RT) treatment of oropharyngeal malignancies. A number of previous studies have defined the routes of lymphatic spread in the head and neck. The clinical neck levels are defined by levels I through VI with subdivisions A and B for levels I, II, and V ( Fig. 96.4 ). The jugulodigastric node in the upper deep jugular chain (level II) is the primary echelon of lymphatic drainage from the oropharynx. Level IIB lymph node metastases are rarely noted in isolation and are usually associated with the presence of level IIA metastasis, advanced T and N stage, and tumor location in the tonsil. In addition to level II, the retropharyngeal and parapharyngeal nodes can also serve as the primary drainage for the tonsil, soft palate, and posterior pharyngeal wall. Lymphatic drainage can advance to levels I, midcervical (III), and lower cervical nodes (IV) in extensive disease. In a retrospective study of 1119 radical neck dissection specimens, Shah described tumors of the oropharynx that characteristically metastasize to lymph node levels II through IV. No positive node involvement of level V was found when levels II through IV were negative.
The location of the primary malignancy in relation to the midline is an important consideration for guiding treatment of the neck. Tumors of the base of the tongue, soft palate, and posterior pharyngeal wall are associated with a higher incidence of bilateral lymphadenopathy. In a retrospective study of 352 patients with OPSCC who had a bilateral neck dissection, T2 or greater lesions of the tonsillar fossa and any stage lesion of the soft palate, base of tongue, and pharyngeal wall showed a high frequency of bilateral metastases. These considerations should influence planning to optimize therapy and minimize patient morbidity ( Figs. 96.5 and 96.6 ). Presentation with an N0 neck is infrequent in HPV-associated primaries, and higher N-stage disease is commonly seen even in early T stage at all oropharyngeal subsites.
SCC is the most common malignancy of the oropharynx and comprises more than 90% of all malignant oropharyngeal tumors. The 2017 edition of the Classification of Tumors of the Head and Neck , from the World Health Organization (WHO), classifies OPSCC as HPV-positive or HPV-negative ( Box 96.1 ). It also highlights some of the salivary gland and hematolymphoid tumors, but additional histologic types of epithelial, minor salivary gland, and soft tissue tumors can also occur, along with rare malignancies such as malignant melanoma (MM) and metastatic lesions, some of which are discussed in detail in later text.
Squamous cell carcinoma, HPV-positive
Squamous cell carcinoma, HPV-negative
Pleomorphic adenoma
Adenoid cystic carcinoma
Polymorphous adenocarcinoma
Hodgkin lymphoma, nodular lymphocyte–predominant
Classical Hodgkin lymphoma
Nodular sclerosis
Mixed cellularity
Lymphocyte-rich
Lymphocyte-depleted
Burkitt lymphoma
Follicular lymphoma
Mantle cell lymphoma
T-lymphoblastic leukemia/lymphoma
Follicular dendritic cell sarcoma/tumor
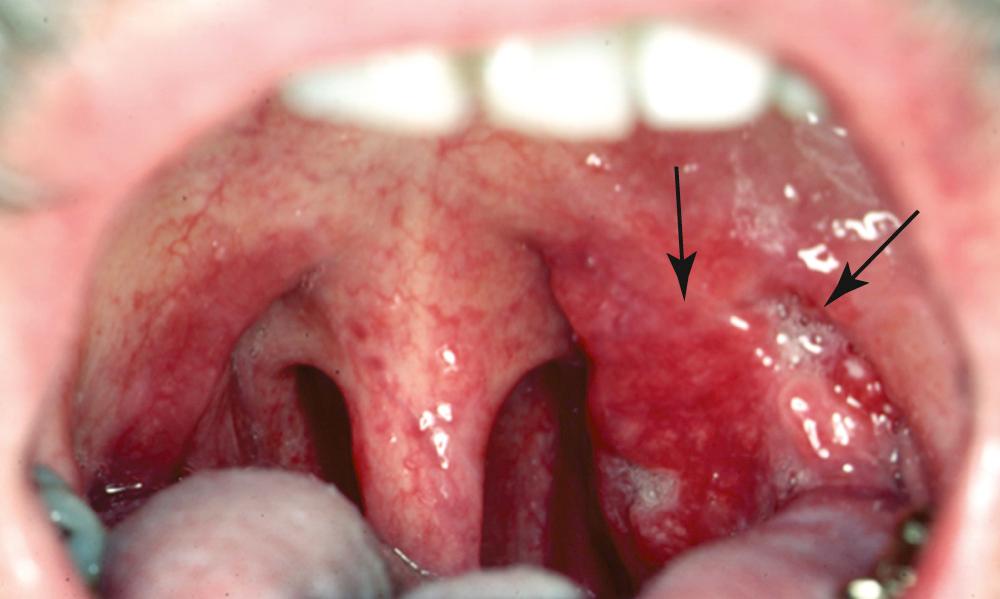
In general, precursor lesions of the oropharynx present clinically as white patches (leukoplakia) or red patches (erythroplakia). Some lesions appear as mixed variants with white and red components. Most white lesions do not show dysplastic cells and relate to hyperplasia. However, erythroplakia or mixed lesions frequently display dysplasia. Whereas leukoplakia rarely undergoes malignant transformation and may even regress after underlying etiologic factors have been eliminated, erythroplakia can often lead to malignancy.
Hyperplasia denotes increased cell counts in the spinous layer (acanthosis) or in the basal/parabasal layers. The cells are free of atypia, and tissue architecture is regular. In the presence of changes in architecture and cellular atypia, the histology is classified as dysplasia, which is characterized by a wide range of microscopic features; it is somewhat challenging to divide the dysplastic spectrum into mild, moderate, and severe. Mild dysplasia is defined by cytologic atypia limited to the basal third of the epithelium, moderate dysplasia by extension into the middle third, and s evere dysplasia by extension into the upper third. Again, no definite division of dysplastic variants has been made, and—depending on the degree of architecture and cytologic atypia—upgrading of dysplasia categories might have to be considered. Carcinoma in situ (CIS) is described as malignant transformation without invasion, and is considered by the WHO as synonymous with severe dysplasia.
OPSCC is an invasive epithelial neoplasm with varying degrees of squamous differentiation and a propensity to early and extensive lymph node metastases. The clinical appearance varies; it presents as a neoplasm that may be exophytic, endophytic, ulcerated, verrucoid, or papillary in growth ( Fig. 96.7 ). Independent from the gross appearance, histopathologic patterns of invasion demonstrate multiple variations as well. Severe dysplasia and CIS are common elements found in association with invasive SCC but are mainly seen in patients with a history of tobacco and alcohol consumption; these are rare in the presence of HPV-associated pathology. Invasive carcinoma eliminates the regular cellular architecture and can include invasion of the lymphovascular space, neurotropism, and infiltration of other tissue components such as muscle or cartilage, demonstrating the aggressive behavior of the tumor. SCC can vary from keratinizing (KSCC) to nonkeratinizing (NKSCC) and well-differentiated to poorly differentiated.
HPV-associated OPSCC displays a histomorphology that is distinct from the traditional tobacco-/alcohol-associated keratinizing SCC. These tumors frequently lack keratinization and mature squamous differentiation. A histologic typing system for OPSCC by Chernock et al. classifies these tumors mainly in three categories: type 1 is KSCC; type 2 is a hybrid, an intermediate, or mixed category with features of both KSCC and NKSCC; and type 3 is NKSCC, as shown in Fig. 96.8 . Type 1 is associated with a strong smoking or drinking history, whereas type 3 is common in HPV-associated tumors (∼70% are NKSCC). Type 2 is mostly seen in patients with HPV-associated tumors who have also been exposed to chemical carcinogens. A subset of HPV-associated OPSCC may have variant forms, such as basaloid, papillary, adenosquamous, sarcomatoid (spindle cell) and lymphoepithelial carcinomas. The available data, though limited, suggest that prognostic outcomes for these variants remain favorable as HPV-associated OPSCCs of typical morphology.
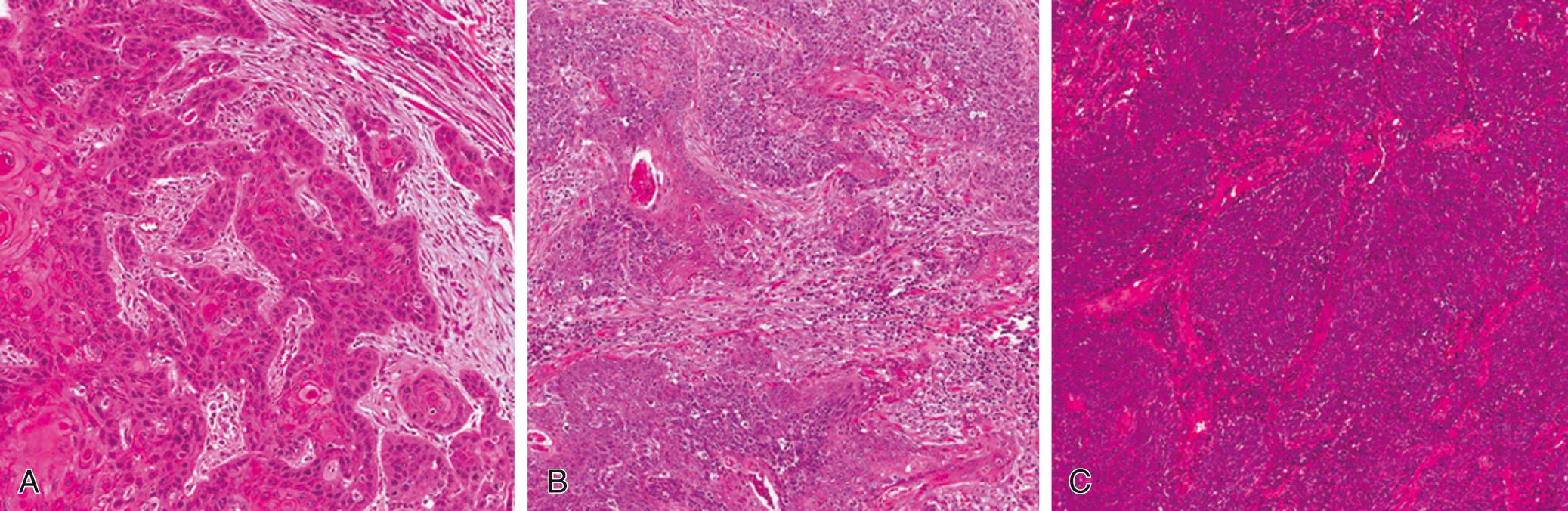
Most invasive OPSCCs can easily be identified by experienced pathologists. However, early clinical stages might significantly complicate the diagnosis of invasive patterns. Inflammation can make detailed analysis of the deep epithelial layers and the lamina propria difficult. However, the presence of cytologic atypia and mitotic features will lead to the correct diagnosis. The grading system for histopathologic typing is based on Broders’ system and includes three grades: well, moderately, and poorly differentiated. The utility of this system, however, is limited mainly to non–HPV-associated OPSCCs, because most of the HPV-associated tumors are poorly differentiated and have a nonkeratinizing morphology.
Lymphoepithelial carcinoma is defined as a poorly differentiated SCC or undifferentiated carcinoma accompanied by a prominent reactive lymphoplasmacytic infiltrate. Lymphoepithelial carcinoma is rare and is diagnosed in 0.8% to 2% of all oral and oropharyngeal malignancies. More than 90% of all lymphoepithelial carcinomas in the oral and oropharyngeal region are found in the tonsils and the base of tongue, although some are found in the buccal mucosa and the palate. The histopathologic pattern is described as invasive and comprises syncytial sheets and clusters of carcinoma cells with prominent nucleoli, ill-defined cell borders, and an often intact surface. The tumor sites are accompanied by a rich lymphoplasmacytic infiltrate. These tumors are radiosensitive, and local control can be achieved in a high percentage of cases.
Among malignant salivary gland tumors, 9% to 23% are found in the oral cavity and oropharynx. Minor salivary gland tumors account for 15% to 20% of all malignant salivary gland tumors. Most minor salivary gland neoplasms arise in the oral cavity; the oropharyngeal tumors arising in the soft palate, tonsillar fossae, or tongue base comprise only 1.1% to 3.3% of all head and neck minor salivary gland tumors. Among these, adenoid cystic carcinoma (ACC) and mucoepidermoid carcinoma (MEC) are the most common.
Adenoid cystic carcinoma is a relatively common minor salivary gland malignancy. A large series found that 42.5% of these lesions arise from the minor salivary glands and 20.5% of all minor salivary gland ACCs are located in oral or oropharyngeal sites. They usually present as slow-growing masses and have a propensity for perineural invasion. Symptoms such as pain signal nerve invasion and are suggestive of progressive disease. The histopathologic pattern is cylindromatous or cribriform, although some can show tubular areas or may appear solid. Cribriform lesions have a more favorable prognosis than solid tumors. Surgical resection is the treatment of choice. Adjuvant RT may be considered for positive or close margins. The spread of adenoid cystic lesions is commonly hematogenous, typically in the lung and bones. Lymph node involvement is somewhat rare; therefore neck dissection is generally reserved for patients with palpable disease. However, ACC of the tongue base has a higher rate of cervical metastasis compared with other sites of the head and neck. In RT for these rather moderately radiosensitive tumors, neutron beam radiation is recommended. Variables affecting survival outcome of ACC include advanced age (>70 years), histologic subtype, grade, osseous invasion, tumor size, clinical stage, positive margins, local recurrence, and distant metastasis.
Mucoepidermoid carcinoma is the most common salivary gland malignancy accounting for 9.5% to 23% of all minor salivary gland tumors. Half of these often asymptomatic lesions arise in the palate, whereas the base of the tongue and other oropharyngeal locations are rare. They are commonly well differentiated. Such a tumor appears as a bluish swelling and some show granulation or a papillary surface. Histopathologically, a mixture of epidermal epithelium and mucous membrane–producing cells is found. Treatment usually includes wide surgical excision, and neck dissection appears to be beneficial in patients with suspected or clinically obvious metastases. In a National Cancer Database study of 3005 patients with MEC of the oral cavity and oropharynx, Ellis et al. observed that increasing age, male sex, a Charlson-Deyo comorbidity score of 2+, clinical T3 to T4 tumors, nodal disease, high-grade tumors, and positive margins to be independently associated with a decreased 5-year overall survival (OS). The rate of occult nodal disease was below 6%, but higher rates of 14.1% and 17.3% were found in the presence of high-grade and clinical T3 to T4 tumors, respectively.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here