Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The major salivary glands are the paired parotid, submandibular (submaxillary), and sublingual glands. The parotid gland weighs 14–28 g, the submandibular gland weighs 7–8 g, and the sublingual gland weighs 3 g. The main duct of the parotid gland (Stensen duct) empties into the oral cavity opposite the crown of the second maxillary molar. The main ducts of both the submandibular glands (Wharton duct) and sublingual glands (Bartholin duct) open in the floor of the mouth, on each side of the frenulum of the tongue. Several minor sublingual gland ducts (Rivinus ducts) drain into the main duct or empty into the oral cavity along the plica sub lingualis on either side of the frenulum.
The parotid gland is composed of a broad superficial lobe and a smaller deep lobe, with the facial nerve running between, and artificially separating, the two lobes. Variations of this anatomy and the distribution of the facial nerve occur.
Minor salivary gland tissue is present in many other locations in the head and neck region, where it may give rise to inflammatory conditions, benign tumors, and malignant tumors. They are found in the lips (more in the upper than the lower lip), gingiva, floor of mouth, cheek, hard and soft palates, tongue, tonsillar areas, oropharynx, and upper respiratory tract.
Microscopically, salivary glands are compound exocrine glands composed of a ductal and an acinar portion, the latter of either serous or mucinous type. The parotid gland is exclusively serous, the submandibular gland is mixed with serous predominance, and the sublingual gland is mixed with mucinous predominance. Minor salivary glands can be pure mucinous (anterior tongue, palate), pure serous (posterior tongue), or mixed (lip, buccal, sinonasal tract, nasopharynx). The intercalated ducts and acini represent the terminal portion of the system (ductoacinar unit). Under normal conditions, rare sebaceous glands are annexed to the duct system in the parotid and, less commonly, the submandibular glands. The reserve cells of the intercalated ducts are the source of regeneration of the acinar tissue and the terminal duct system and are thought to be the progenitors (together with the closely apposed myoepithelial cells) of most salivary gland tumors. However, it has been pointed out that basal and luminal cells at all levels of the duct system and even acinar cells are capable of DNA synthesis and mitosis, and therefore they all have the potential to give rise to neoplasms.
Immunohistochemically, the luminal cells stain for low-molecular-weight keratin (including CK7), CD117, carcinoembryonic antigen (CEA), and epithelial membrane antigen (EMA). The intercalated duct luminal cells are also positive for lysozyme but not for S-100 protein. Serous acinar cells are positive for low-molecular-weight keratin and DOG-1. The myoepithelial cell component can show some minor differences in its immunohistochemical profile depending on its location, but this is an inconsistent finding: in the parotid gland the myoepithelial cells express mainly calponin and h-caldesmon; in the submaxillary glands they also express smooth muscle actin; and in the minor salivary glands there is an additional expression of smooth muscle myosin heavy chain. Myoepithelial cells also stain for p63, p40, CD10, and maspin, as do the basal cells.
The lymphoid tissue of the parotid region is represented by small nodes located near or within the parotid gland and by scattered lymphoid cells located in the connective tissue around the acini and ducts. The latter are part of the mucosa-associated lymphoid tissue (MALT). Periparotid lymph nodes often contain benign salivary gland inclusions. No lymph nodes are present within the submandibular or sublingual glands.
Heterotopia of salivary gland tissue has been divided into intranodal and extranodal types. The intranodal variety is more frequent. Almost all lymph nodes located within or near the parotid gland in infants contain salivary gland tissue. The finding is not as prevalent in adults, but it is still very frequent. The salivary gland tissue is usually located in the medullary portion of the node and is predominantly composed of intercalated and intralobular ducts; it may also contain acini (mainly of serous type) and small ducts of immature type.
Extranodal heterotopia has been divided into high and low forms, depending on its location in the head and neck region. Sites of high heterotopia include the mandible, ear, palatine tonsil, mylohyoid muscle, pituitary gland, and cerebellopontine angle; these are probably all the result of abnormalities in the embryonic migration of the salivary glands. Low heterotopia is related to the branchial pouches and is found in association with cysts and sinuses in the lower neck and in the thyroid gland. The most common location is along the medial border of the right sternomastoid muscle near the sternoclavicular joint. In this location, the ectopic glands can secrete saliva onto the skin during feeding.
Heterotopic salivary tissue is subject to the same pathologic changes as its orthotopic counterpart, including cystic formation, oncocytic metaplasia, ductal hyperplasia, and neoplasms. Among the latter, Warthin tumor is the most frequent, but several other benign and malignant types have been described.
Calculi may form in the major ducts of the submandibular, sublingual, and parotid glands, sometimes in a multicentric and bilateral fashion. They are more common in the submandibular gland than in the parotid gland, presumably because in the former the saliva is more saturated with calcium salts and the duct is longer with a sharp upward angle prior to its termination onto the floor of the mouth ( Fig. 6.1 ). Rarely, they affect minor salivary glands. Some of the stones have a foreign body or bacterial nidus. Others do not have an identifiable nidus, are laminated, and are composed of the crystalline compound carbonate apatite. The formation of calculi blocks secretion and produces swelling of the distal salivary gland tissue. If ductal obstruction persists, the gland becomes inflamed and atrophic as acinar tissue is destroyed. With obstructed ducts of the submandibular and sublingual glands, marked induration can occur in the floor of the mouth that may be mistaken for neoplasm by palpation. The duct orifices become erythematous and swollen. Radiographic examination may demonstrate a radiopaque mass, and sialography will show partial or total blockage of the duct. The stones can also be demonstrated by ultrasound. Microscopic examination of glands that have been affected by stones shows dilation of ducts with intraluminal neutrophils, at times squamous metaplasia of the epithelium, moderate to prominent chronic inflammation, and a variable destruction of acinar tissue ( Fig. 6.2 ). Degenerative changes in the secretory and myoepithelial cells are also apparent at the immunohistochemical and ultrastructural levels.
The treatment of symptomatic sialolithiasis consists of surgical removal or disintegration of the calculus. The latter is achieved with techniques such as intracorporeal or extracorporeal shock-wave lithotripsy. In most cases the gland can be preserved, but surgical resection is occasionally necessary.
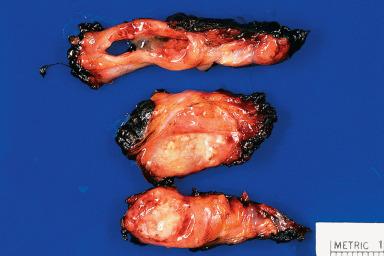
Acute sialadenitis can be localized to one salivary gland (usually parotid or submandibular) or be the expression of a systemic infection. Viral sialadenitis (rarely seen as a surgical specimen or biopsy) can be caused by paramyxovirus (mumps), cytomegalovirus, Epstein–Barr virus (EBV), coxsackievirus, and influenza A and parainfluenza viruses. Acute suppurative sialadenitis is generally caused by Staphylococcus aureus , Streptococcus species, and gram-negative bacteria. Dehydration, malnutrition, immunosuppression, and sialolithiasis are predisposing factors. Once an abscess has formed, surgical drainage may be necessary ( Fig. 6.3 ).
Chronic sialadenitis in the form of mild lymphocytic infiltration of the major salivary gland unaccompanied by clinical symptoms is relatively common. Some cases are of focal obstructive nature and are accompanied by various degrees of parenchymal atrophy, fibrosis, and microliths ; others, more common in females, are age related, have a high statistical association with rheumatoid arthritis and Sjögren syndrome, and are probably immune mediated. In clinically apparent cases, sialolithiasis is the most common cause.
Kuttner tumor is a chronic sclerosing sialadenitis of the submandibular gland. This disorder, which is usually unilateral, is characterized by a plasmacytic and lymphocytic periductal infiltrate eventually leading to encasement of ducts in thick fibrous tissue. The lobular architecture is preserved and obliterative phlebitis is frequently seen. The lymphocytes that infiltrate the epithelial component are mainly B cells. The plasma cells contain abundant immunoglobulin G4 (IgG4), suggesting that Kuttner tumor is part of the widening spectrum of IgG4-associated diseases. Cases in which the lymphoid infiltrate is very florid can simulate marginal zone B-cell lymphoma. Surgical excision may be necessary in some cases.
Sclerosing polycystic adenosis is characterized by the presence of a discrete mass (usually in the parotid gland) formed by fibrohyaline stroma enclosing dilated and hyperplastic ductal and acinar structures ( Fig. 6.4 ). Apocrine-like metaplasia and transluminal bridges with a cribriform pattern of growth are present. The appearance resembles that of sclerosing adenosis and fibrocystic disease of the breast. Some of the epithelial cells contain large, brightly eosinophilic cytoplasmic granules. The behavior is characteristically indolent. Although initially reported as a reactive inflammatory process, most authors now believe this is a low-grade neoplasm, and a study with the HUMARA technique has shown that the disease is of clonal nature.
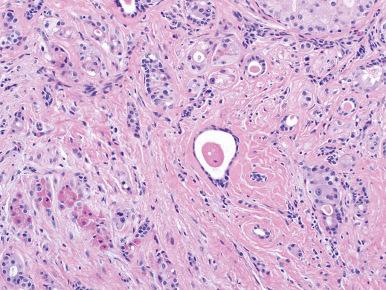
Granulomatous sialadenitis is uncommon and can result from tuberculosis, fungal infection, sarcoidosis, or duct obstruction from calculi or malignant tumors. In the latter instance, the granulomas result from rupture of ducts and may contain small pools of mucin. A xanthogranulomatous variant of sialadenitis has also been described.
Kimura disease can involve the salivary glands (especially the parotid) by spread from adjacent lymph nodes.
Benign lymphoepithelial cysts are lesions of the parotid or upper cervical lymph nodes characterized by unilocular or multilocular cystic formations lined by glandular or squamous epithelium surrounded by a florid lymphoid hyperplasia with prominent germinal centers. The amount of the lymphoid component is variable, and the cyst lining is often infiltrated by lymphocytes. This is likely an acquired process resulting from the proliferation of branchial pouch-derived or analogous epithelium (e.g. intranodal salivary gland inclusions) induced by the lymphoid hyperplasia, probably through a specific interaction between the epithelial cells and a subset of lymphocytes. Other examples of this phenomenon in the head and neck area are branchial cleft cyst, the branchial cleft cyst-like formations sometimes seen in Hashimoto thyroiditis, and multilocular thymic cyst.
Another salivary gland lesion that merits mention in this context is human immunodeficiency virus (HIV)-associated lymphoepithelial cyst ( Fig. 6.5 ). The morphologic changes in this condition include multiloculated lymphoepithelial cysts similar to those previously described, solid lymphoepithelial lesions (so-called epimyoepithelial islands) similar to those seen in Mikulicz disease (see next section), and a combination of both. These lesions can be unilateral but are often bilateral. The more prominent the combination of these changes, the more likely that HIV infection is the causative factor. Three-dimensional reconstructions have shown that the cysts arise from the intralobular duct system rather than from intraparotid lymph nodes.
The lymphoid follicles of HIV-induced salivary gland disease show a prominent network of follicular dendritic cells and numerous intrafollicular CD8+ lymphocytes. The follicular dendritic cells show a strong expression of HIV-1 major core protein and HIV-1 RNA, indicating that there is active replication of the virus within them.
There are two other polycystic diseases of the salivary glands that need to be mentioned here for differential diagnosis purposes. The first is polycystic (dysgenetic) disease , a developmental disorder so far reported only in females and characterized by bilateral parotid gland enlargement. The lesion is characterized by small epithelial cysts replacing most of the parotid gland parenchyma with small foci of residual acini interspersed throughout. The other is the already mentioned sclerosing polycystic adenosis . One should also keep in mind that some neoplasms of salivary gland may undergo prominent cystic changes. This is particularly true of mucoepidermoid carcinoma, acinic cell carcinoma, basal cell adenoma, sebaceous lymphadenoma, and, occasionally, benign mixed tumor.
Lymphoepithelial sialadenitis (Mikulicz disease, benign lymphoepithelial lesion) most often presents as a slowly increasing and eventually striking enlargement of the salivary and/or lacrimal glands. This enlargement is usually bilateral and symmetric, but it can also be unilateral and localized, at least at the clinical level. If an infection develops, the process may subside only to recur when the infection is gone.
Grossly, the gland is enlarged and whitish, sometimes admixed with occasional cysts ( Fig. 6.6A ). Microscopically, the two cardinal changes are marked lymphoid infiltration and so-called epimyoepithelial islands. This combination of findings has been used to create a synonym for the disease associated with a catchy acronym, that is, myoepithelial sialadenitis ( MESA ), recently modified to lymphoepithelial sialadenitis (LESA) . The lymphoid tissue contains numerous well-formed germinal centers and is composed of a mixed population of B and T lymphocytes, accompanied by scattered histiocytes and dendritic cells. The so-called epimyoepithelial islands appear as solid epithelial nests surrounded and infiltrated by lymphoid cells, which are mainly of monocytoid B type (see Fig. 6.6B ). A hyaline substance is deposited between the cells, which is shown ultrastructurally to represent basement membrane material, including type IV collagen. The nature of the cells within the islands, that is, whether ductal, basal or myoepithelial, remains a controversial subject. Current evidence suggests a lack of myoepithelial cell participation, which—if true—would make the term “epimyoepithelial islands” inaccurate and the alternative “lymphoepithelial lesions” more appropriate.
LESA can remain localized to the salivary gland but more often is a manifestation of a generalized symptom complex known as Sjögren syndrome , the other components of which are keratoconjunctivitis, xerostomia, rheumatoid arthritis, and hypergammaglobulinemia. In this condition, lymphoid infiltrates similar to those present in the major salivary glands are also seen in the lacrimal glands and minor salivary glands of the oral cavity. This fact is of diagnostic utility, in the sense that biopsy of the labial glands is often used to document the diagnosis of this condition. It should be noted, however, that so-called epimyoepithelial islands are usually scanty or absent at these sites. Occasionally, the lymphoid infiltrates extend to other organ systems, such as lymph nodes, lung, kidney, bone marrow, skeletal muscle, skin, or liver.
The etiology of LESA, with or without Sjögren syndrome, remains enigmatic. The incidence of the disease is markedly increased in HIV-infected patients. As a matter of fact, there is a marked morphologic similarity (and probably a close pathogenetic relationship) between Sjögren syndrome as seen in this population and the HIV-related changes described in the previous section. Interestingly, a chronic lymphocytic sialadenitis morphologically very similar to Mikulicz disease occurs in hepatitis C virus (HCV)-related chronic liver disease.
The prevalent view is that LESA is a systemic autoimmune process. In the typical case the lymphocyte population is polytypic, in keeping with a reactive process. However, this population may undergo small clonal expansions, detectable on Southern blots. In some cases, these evolve into full-blown lymphomas, whether in the salivary gland or in extraglandular locations. Some of these are large B-cell lymphomas (often with an immunoblastic appearance), whereas others are small lymphocytic lymphomas with or without plasmacytoid features (see later), and exceptional cases of Hodgkin lymphoma and peripheral T-cell lymphoma have been reported. When one of these lymphoma types develops, there is usually not much difficulty in identifying it. The vexing problem is how to interpret the usual case of LESA that is composed of a morphologic mature (“benign-looking”) lymphocytic population, especially if a monotypic component has been detected with molecular techniques. Is it a reactive, possibly autoimmune process with a tendency to evolve into a recognizable malignant lymphoma, or is it a malignant lymphoma already? Some authors have taken the latter view and have placed LESA in the ever-expanding MALT lymphoma category, mainly on the basis of the molecular findings. It seems to us that to label a process as malignant at a stage at which it shows neither clinical nor morphologic signs of malignancy is neither conceptually nor medically sound. At the present state of knowledge, it is probably wiser to restrict the diagnosis of lymphoma to those cases showing some morphologic evidence of such. The most important feature in this regard is the number and distribution of the monocytoid B cells. The presence of these cells outside of the lymphoepithelial lesions, and especially as broad anastomosing strands, is in favor of lymphoma; the same is true for sheets of plasma cells, with or without Dutcher bodies. In fact, probably the earliest morphologic feature of MALT lymphoma arising in LESA is the presence of a “halo” of pale-staining sheets of monocytoid B cells surrounding the “epimyoepithelial” islands.
The submandibular glands, which are often included in the field of irradiation for tumors of the oral cavity, swell and become firm as a result of the therapy. These changes may be mistaken clinically for metastatic carcinoma in submandibular lymph nodes and sometimes have led to unnecessary radical surgery. Not surprisingly, these changes are frequently seen in cervical lymph node dissection specimens in patients previously treated with radiation therapy. Microscopic examination shows atrophy of acinar elements with associated fibrosis and chronic inflammatory cells in the stroma. The lining of the duct epithelium may show prominent squamous metaplasia and degenerative-type atypia.
Lymphoid disorders of reactive nature can involve the intraparotid lymph nodes and be confused clinically with a primary salivary gland tumor. These include nonspecific follicular hyperplasia, healed abscesses, and granulomatous inflammations, such as those produced by cat-scratch disease.
Amyloidosis may involve the salivary gland as part of a generalized process or as a localized pseudotumoral mass (“amyloid tumor”), and it may result in the sicca syndrome.
Nodular fasciitis can present as a primary intraparotid or periparotid lesion; its microscopic appearance is identical to that of its more common soft tissue counterpart.
Inflammatory pseudotumor is composed of a predominant population of myofibroblast-like cells in an edematous and inflammatory background. As in other sites where this entity occurs, there is a question as to whether it may represent a neoplastic condition, hence the alternative designation of inflammatory myofibroblastic tumor .
Rosai–Dorfman disease (sinus histiocytosis with massive lymphadenopathy) can involve the major salivary glands, with or without accompanying lymph node enlargement.
Intercalated duct hyperplasia of major salivary glands may coexist with salivary gland tumors of various types (especially epithelial–myoepithelial carcinomas) and with chronic parotitis. It is composed of intercalated duct epithelium and therefore different from the adenomatoid acinar hyperplasia found predominantly in intraoral salivary glands. It has been suggested that intercalated duct hyperplasia may be a precursor lesion of some salivary gland neoplasms (see next section).
The classification of salivary gland tumors has proved a most difficult and frustrating exercise. If anything, it is even more complex than that of the somewhat related breast and sweat gland tumors. One of the major stumbling blocks is that most of the salivary gland tumors arise from—or differentiate toward—the same cell lines: epithelial (ductal and/or acinar) and abluminal (myoepithelial and/or basal cell). This results in a considerable overlap at all levels, compounded by the fact that each of these cells can undergo a variety of metaplastic changes (e.g. oncocytic, clear cell, sebaceous, squamous, chondroid). In addition, with some exceptions, these lesions are uncommon. The World Health Organization (WHO) classification stresses the distinction between benign and malignant tumors (i.e. adenomas and carcinomas). In this chapter, a somewhat different approach has been used. The tumors have been grouped, whenever possible, according to their predominant line of differentiation; this has been followed by a discussion of the benign and malignant members within each group.
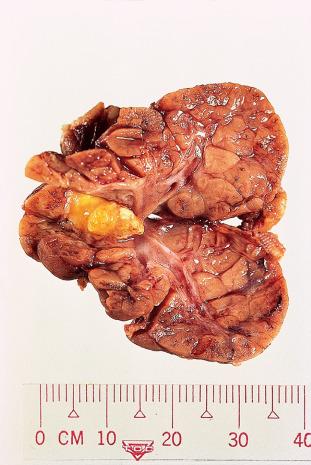
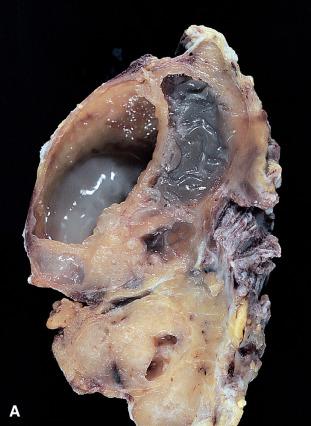
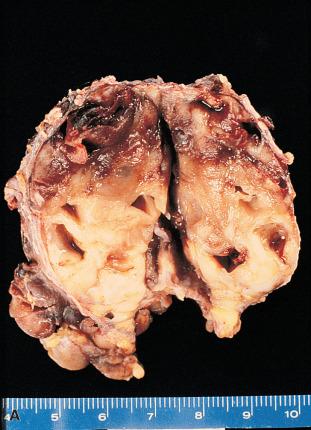
Pleomorphic adenoma is the most common neoplasm of the salivary glands. It is most frequent in women in the fourth decade of life, but it can be seen in children and in elderly persons of either gender. It is approximately 10 times more common in the parotid than in the submandibular gland and is very rare in the sublingual gland. In the parotid gland, most tumors arise within the superficial lobe, from either the tail (50%) or the anterior portion (25%). The remaining 25% arise from the deep lobe and often present as a parapharyngeal mass. They are also seen in various minor salivary gland sites (e.g. palate).
Grossly, the tumor forms a rubbery, tan-white, firm mass with a bosselated surface and may grow to a large size but are usually between 2 and 5 cm ( Fig. 6.7 ). The consistency depends on the relative amount of epithelial cells and stroma and the type of the latter. Although the tumor tends to be well circumscribed, small extensions (so-called pseudopodia) can often be seen protruding into the adjacent normal tissue. Islands of cartilage can be recognized by their glistening, translucent appearance. In rare cases, foci of mature bone are identified.
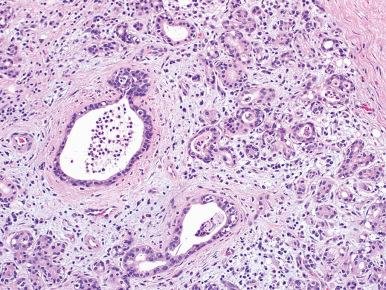
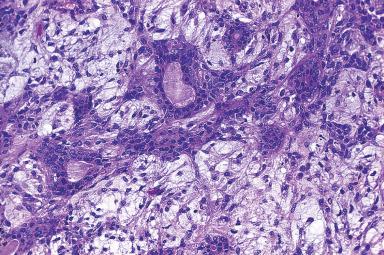
The typical pleomorphic adenoma has a biphasic appearance resulting from the intimate admixture of epithelium and stroma ( Figs. 6.8 and 6.9 ). Most of the epithelial component is of a glandular nature, but foci of squamous metaplasia are common, sometimes accompanied by keratinized epithelial plugs in the lumen. The neoplastic glands have a lining composed of two cell types, the basally located cells displaying morphologic features of myoepithelial and basal cells. They may be cuboidal, oncocytic, clear, spindled, or “hyaline” (see Fig. 6.8 ). The latter cells are also referred to as plasmacytoid myoepithelial cells and are frequently seen in pleomorphic adenomas. The stroma may have a nonspecific myxoid appearance, sometimes containing abundant elastic tissue or extensive adipose tissue ; however, areas of clear-cut cartilaginous differentiation are usually found. There is convincing morphologic, ultrastructural, immunohistochemical, and molecular evidence to suggest that these mesenchymal elements share the same origin as the epithelial cells and that they represent modified myoepithelial cells. Ultrastructurally, a continuum of cytoplasmic features from epithelial to mesenchymal cells is also present.
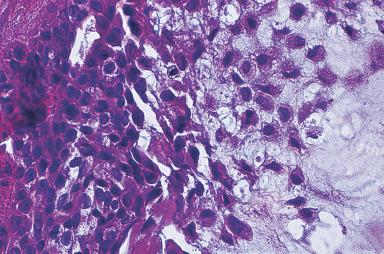
Additional stromal changes seen in pleomorphic adenoma include osseous metaplasia and areas of fibrosis. Cutaneous adnexal differentiation can be seen in pleomorphic adenomas, especially those arising in the palate and lip. This is most commonly manifest as trichilemmal differentiation, infundibulocystic structures, and trichohyalin granules. Other substances sometimes found in pleomorphic adenomas include tyrosine-rich crystalloids in the myxoid areas, collagenous spherules, calcium oxalate crystals, intraductal birefringent crystalloids of unknown chemical composition, and melanin.
Some benign pleomorphic adenomas are extremely cellular, the tumor cells being either round or spindle shaped ( Fig. 6.10 ). Others may show cytologic atypia in the form of scattered large hyperchromatic nuclei (which have been shown to be polyploid). Follow-up studies have indicated that these tumors do not behave differently from the ordinary variety. The rarity of mitotic figures and absence of necrosis aid in the differential diagnosis with true malignant neoplasms. Something similar can be said of pleomorphic adenomas with foci that superficially resemble the adenoid cystic carcinoma pattern. The presence of these foci does not influence the prognosis and therefore should be disregarded.
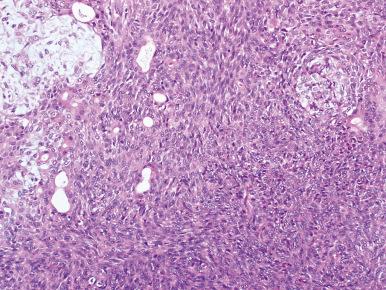
Immunohistochemically, the ductal epithelial component is positive for keratin, EMA, and CEA. The keratins most frequently expressed are CK7, CK8/18, and CK19 (with or without CK14) in the luminal cells of the tubular structures, and CK14 in the abluminal cells. The myoepithelial component is immunoreactive for low-molecular-weight keratins (CK7, CK8/18, and CK14), actin, myosin, other smooth muscle-specific proteins, p63, p40, and sometimes S-100 protein. The latter marker is also strongly expressed in the cartilaginous areas and in subpopulations of the epithelial ductal cells. The demonstration in some of the tumors of glial fibrillary acidic protein and astroprotein, two glial markers, is more difficult to explain ; it has been suggested that it is the result of cross-reactivity with other intermediate filaments of similar structure and chemical composition. Regardless of the reason for this occurrence, it seems to be related to the capacity of this tumor for cartilaginous differentiation, and it therefore may be useful in the differential diagnosis with adenoid cystic carcinoma and basal cell adenoma, in which such a differentiation does not occur.
Cytogenetically, pleomorphic adenoma shows distinctive chromosomal translocations involving 8q12 or 12q13–15. The PLAG1 gene at 8q12 can be fused with CTNNB1 (β-catenin), LIFR (leukemia inhibitory factor receptor), or SII (transcription elongation factor SII), or is overexpressed due to intrachromosomal 8q rearrangements. The HMGA2 gene at 12q13–15 can be fused with FHIT (fragile histidine triad) or NFIB (nuclear protein involved in transcriptional regulation). Overexpression of p53 or mutation in the TP53 gene is rare in pleomorphic adenoma, in contrast to its malignant counterpart.
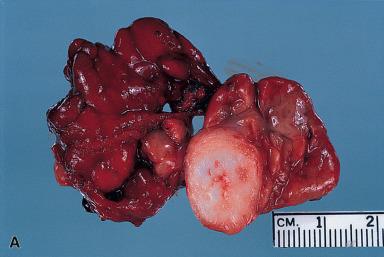
The recurrence rate of pleomorphic adenoma depends almost entirely on the adequacy of the primary excision, as several classic studies have conclusively demonstrated. Recurrence is very high if the tumor is removed by a simple enucleation ( Fig. 6.11 ). This is because small inconspicuous nodules (pseudopodia) attached by threadlike filaments of neoplastic tissue may be present surrounding the main mass. They may have the shape and appearance of lymph nodes and may be mistaken for nodal metastases by both the surgeon and the pathologist. If the tumor is enucleated, these small remnants may be left behind and will provide the nidus for recurrence. Most of these recurrences will appear during the first 18 months after surgery, but others supervene over an exceedingly long period (50 years or more). Because of this, long-term follow-up is essential. It has been proposed that patients with tumor spillage at operation or residual tumor left behind should have radiation therapy immediately to minimize the possibility of recurrence. Usually the microscopic pattern of the recurrent tumor exactly mimics that of the original neoplasm ( Fig. 6.12 ). Surgery for recurrent tumor often fails. In approximately one-fourth of cases, further recurrences develop, often in the form of multiple foci. The proper therapy for pleomorphic adenoma is its total surgical removal, along with a margin of normal salivary tissue that surrounds it. For the tumors located in the superficial lobe of the parotid gland, which represent the majority, the standard surgery is a superficial parotidectomy with preservation of the facial nerve. The incidence of recurrence after this procedure is almost zero, and the long-term prognosis for properly treated pleomorphic adenoma is excellent.
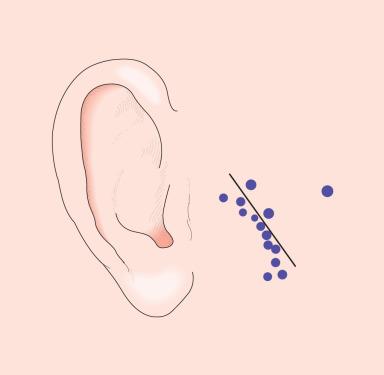
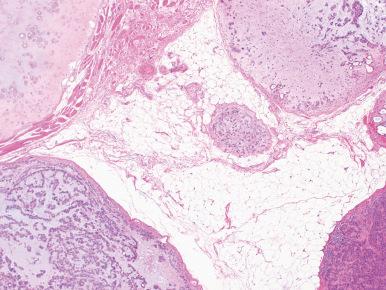
Under rare circumstances, a pleomorphic adenoma of ordinary microscopic appearance will metastasize to the lymph nodes, lungs, bone, or other organs, with the metastases appearing as benign as the original tumor (“benign” metastasizing pleomorphic adenoma). Some of these cases have occurred in immunologically compromised patients and have followed a rapidly aggressive clinical course. Usually these metastases are preceded by one or more local recurrences. Curiously, a good number of these metastases have presented as isolated masses in the kidney.
Two major categories of malignant mixed tumor exist. The first and more common can be viewed as a malignant transformation of a preexisting pleomorphic adenoma (carcinoma ex pleomorphic adenoma). This complication occurs in approximately 5%–10% of these neoplasms. Clinical features that suggest this event in a long-standing tumor are sudden increase in growth, pain, and facial paralysis. Documentation that a malignant salivary gland tumor arose from a preexistent pleomorphic adenoma may be difficult to obtain. The clinical history, although suggestive of the process, is not by itself diagnostic. It is necessary to have microscopic evidence of a previously existing benign tumor or to have benign and malignant tumor in the same neoplasm. This may require thorough sampling of the tumor. Sometimes the preexisting benign lesion is represented only by a totally hyalinized round nodule surrounded by carcinoma. Features said to indicate a greater likelihood of malignant transformation of a pleomorphic adenoma are submandibular (as opposed to parotid) location, long duration, older patient age, large tumor size, and—at the microscopic level—prominent zones of hyalinization and a moderate degree of mitotic activity.
The malignant component of this tumor typically has an epithelial appearance usually comparable to that of the luminal cells, less commonly that of the myoepithelial cells, or both ( Fig. 6.13 ). It has been stated that these malignant areas often assume the appearance of one of the well-recognized variants of salivary gland carcinoma (such as mucoepidermoid carcinoma or adenoid cystic carcinoma), but salivary duct carcinoma and adenocarcinoma, not otherwise specified (N.O.S.) type, appear to be the most common. For example, in the series of Tortoledo et al., the malignant component was classified as salivary ductal carcinoma in 13 cases, undifferentiated carcinoma in 10, terminal duct carcinoma in 9, myoepithelial carcinoma in 3, and unclassified in 2. Most of the tumors have a poorly differentiated appearance. As a matter of fact, whenever a high-grade adenocarcinoma that is difficult to classify is found in the salivary gland, the possibility of it having arisen from a pleomorphic adenoma should be considered. In a high percentage of early cases the malignant change is restricted to foci of intraductal carcinoma, with an intact layer of neoplastic myoepithelial cells on the outside.
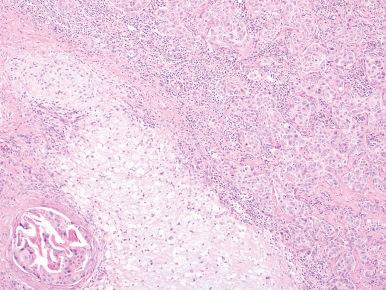
In most cases, the immunohistochemical profile of the malignant cells is comparable to that of the ductal luminal cells of the benign component. In the malignant areas there is often overexpression of TP53 (as a result of mutation) and of HER2 . However, this is also true in a small subset of pleomorphic adenoma lacking malignant change. Genetic alterations that mediate malignant transformation may include amplification of genes at 12q13–15 (including CDK4 , HMGA2 , MDM2 ), TP53 mutation, or HER2 amplification.
LiVolsi and Perzin have pointed out that if the cytologically malignant foci are found entirely within a pleomorphic adenoma (i.e. intracapsular or in situ) they are not associated with clinical malignancy if the tumor is completely excised. Only when invasion occurs beyond the capsule of the original neoplasm will the lesion potentially behave clinically in a malignant fashion. For invasive tumors, the extent of invasion beyond the capsule is of importance: minimally invasive (≤1.5 mm from the tumor capsule) versus widely or frankly invasive (<1.5 mm), as defined by the WHO. In one series, all patients whose malignant neoplasms extended for more than 8 mm beyond the capsule died of tumor. Therefore, the importance of thorough sampling cannot be overemphasized. The prognosis also depends on the histologic type of carcinoma, the microscopic grade (which is related to the former), and the proliferative index.
The most common sites of metastases are regional lymph nodes, lungs, bone (especially the vertebral column), and abdominal organs.
The second type of malignant mixed tumor has a biphasic composition similar to that of pleomorphic adenoma, but both the epithelial and the mesenchyme-like elements have a malignant appearance, the former often in the form of a ductal carcinoma and the latter in the form of chondrosarcoma or undifferentiated pleomorphic sarcoma. Because no preexisting benign tumor is found, the designation of carcinoma ex pleomorphic adenoma , which has been suggested as an alternative term for malignant mixed tumor, is inappropriate. Names such as true malignant mixed tumor and carcinosarcoma have been proposed instead. This is an aggressive, often rapidly lethal neoplasm.
Oncocytes or oxyphilic cells are large ductal epithelial cells with a granular, deeply eosinophilic cytoplasm crowded with mitochondria. Their number in the normal salivary gland increases with age, and their secretory activity is minimal, suggesting that they are the expression of a degenerative change. Sometimes one finds multiple parotid cysts lined by oncocytic epithelium, associated with tyrosine crystals in the lumen. In other instances, oncocytes form well-defined clusters scattered throughout the gland. It is not clear whether these clusters represent an exaggeration of the age-related hyperplastic process or the emergence of a neoplasm, an uncertainty reflected by the terms that have been used to designate this process: oncocytosis , multinodular oncocytoma , multinodular oncocytic hyperplasia, and multifocal adenomatous oncocytic hyperplasia .
The two major tumor types composed of oncocytes are oncocytic adenoma and Warthin tumor. There are no transitional forms between these two tumors, suggesting that their pathogenesis is different.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here