Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Access video content for this chapter online at Elsevier eBooks+ ![]()
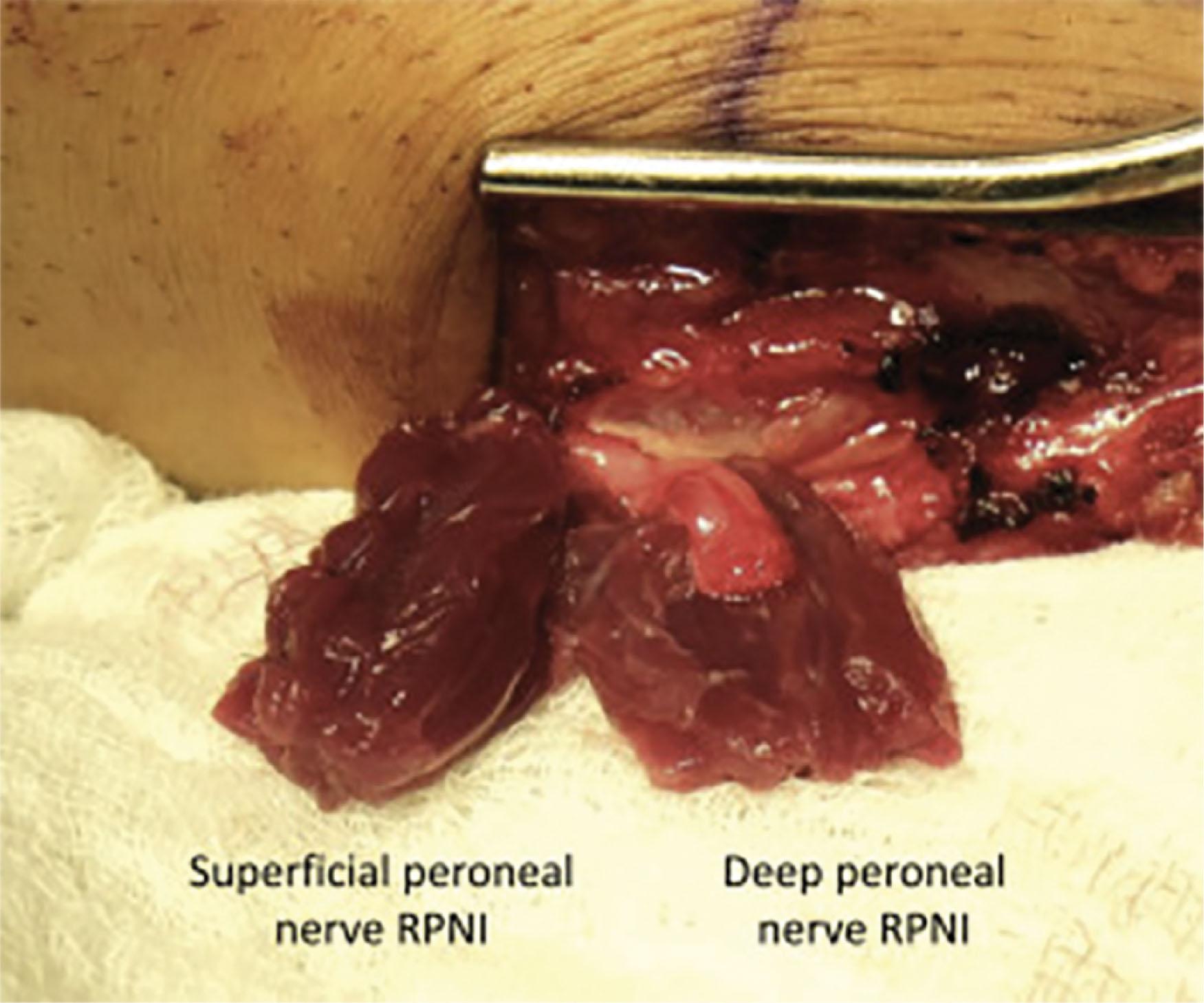
Lower extremity pain can have a variety of sources including bone, joint, tendon, ligament, muscle, and nerve. Neuropathic pain can be particularly severe and adversely impacts a patient’s quality of life, ability to work, and even the ability to perform simple activities of daily living. Adequate treatment of neuropathic pain may require a series of interventions including medical management or surgical interventions. Due to the complexity of neuropathic pain, and the various potential etiologies for this pain including compression, traction, inflammation, or injury, successful treatment requires a clear delineation of the cause and a personalized intervention to address it. In this chapter, we discuss neuropathic pain secondary to symptomatic neuromas, the pathophysiology behind this pain, and its maladaptive sequelae (i.e., peripheral and central sensitization). In addition, we will highlight the development and use of the regenerative peripheral nerve interface (RPNI) as a surgical treatment strategy for lower extremity neuropathic pain. The RPNI is a surgical technique that represents a physiologic solution to treat and prevent neuropathic pain from symptomatic neuromas. The surgery involves securing a free autologous skeletal muscle graft around the terminal end of a peripheral nerve or nerve fascicle ( Fig. 6.3.1 ). RPNI surgery has been performed successfully on all major peripheral nerves of the lower and upper extremity and has been effective in mitigating and preventing neuropathic pain, improving psychosocial wellbeing, and reducing central sources of pain including phantom limb pain.
Traumatic injury to the axons of a peripheral nerve will result in Wallerian degeneration. At the distal stump of the injured peripheral nerve, cytoskeletal disintegration results in the inflow of calcium and sodium ions which initiates a cascade of events, similar to apoptosis, to recruit inflammatory cells (i.e., macrophages) into the zone of injury. Macrophages aid in the clearance of debris surrounding the injured peripheral nerve to create an environment conducive to axonal regeneration. The Schwann cells then mediate a process where the injured axons develop a growth cone and begin to regenerate towards potential peripheral targets for reinnervation. This entire process proceeds in a logical and stepwise fashion to facilitate successful axonal sprouting, elongation, and end organ reinnervation to complete neural recovery.
This entire process may be unsuccessful for a number of reasons including: (1) no distal targets available for reinnervation secondary to amputation; (2) diminished regenerative capacity of the axons in the injured nerve; and (3) limited support of the Schwann cells. In these cases, axonal regeneration can occur in a disorganized fashion and potentially lead to neuroma formation. In addition, concurrent collagen remodeling, myofibroblast proliferation, and scar formation occur at the site of neural injury leading to compression of the injured peripheral nerve in the poorly vascularized, dense fibrotic tissue. This fibrotic reaction in combination with the disorganized axonal outgrowth create the characteristic findings seen in peripheral nerve neuromas. After injury, phenotypic changes occur in the peripheral nerve that are characterized by upregulation of sodium channels, adrenergic receptors, and nicotinic cholinergic receptors. Typically, nociceptors in a peripheral nerve transduce increasing amounts of pain based on the severity of external stimulation of the nerve. However, with the phenotypic changes that occur in the peripheral nerve after injury, nociceptors can repetitively depolarize and transmit the sensation of pain, even in the absence of external stimuli, leading to abnormal sensitivity and spontaneous activity of the injured axons. Hence, simply protecting the end of a divided nerve from external stimulation will not necessarily prevent the sensation of pain in an injured nerve or a peripheral neuroma.
The physiologic changes that occur in peripheral nerves following injury also increase the excitability and firing of uninjured pain-transmitting neurons in the spinal cord in a process termed central sensitization. Central sensitization of pain is a complex series of events following peripheral nerve injury that is defined by changes in the properties of central nervous system neurons whereby pain is no longer coupled to a particular peripheral noxious stimulus. In the dorsal horn of the spinal cord, a series of changes occur following peripheral nerve injury that result in increased membrane excitability, synaptic efficacy, and reduced inhibition of firing that results in a pain response to a peripheral noxious stimulation in situations which would normally not produce pain.
The processes that influence the development of central sensitization occur both peripherally and centrally. Periodic peripheral painful inputs during the acute and subacute phase of an injury can begin a cascade of peripheral changes that influence the central somatosensory changes associated with central sensitization. Furthermore, once centralization of pain occurs, it can continue without sustained peripheral inputs, and can contribute to states of chronic pain. For instance, centralization has been implicated in the development of phantom limb pain and chronic back pain. Since centralization involves neuronal plasticity, additional peripheral injuries can further increase preexisting central pain. Therefore, in clinically managing lower extremity pain, considerations should be given to the peripheral and central processes of pain, since addressing only the peripheral component may not sufficiently improve all of the pain experienced by a patient.
There are over 1.7 million people living with limb loss in the United States alone, with over 185,000 new amputations each year. Some 75%–85% of these people suffer from post-amputation pain (PAP) including residual limb pain (i.e., pain in the residual limb that includes neuroma pain) and phantom limb pain (i.e., pain in the distribution of the amputated limb). The PAP associated with limb loss may contribute to depression, anxiety, and long-term opioid use, which can severely impact a patient’s quality of life. PAP affects the ability of patients to ambulate, conduct work, attend to personal hygiene, and maintain interpersonal relationships. Keeping this context in mind is critical in approaching the patient with PAP as, ultimately, successful management of the physiologic causes of pain is contingent on addressing the psychosocial and emotional factors at play.
Before diagnosing pain from a symptomatic neuroma, the surgeon should consider alternative causes of residual limb pain in patients with amputation ( Algorithm 6.3.1 ). These include inflammatory, infectious, and pathologic processes such as bursitis, osteomyelitis, and heterotopic ossification. Additionally, residual bone spurs, bony malunion or non-union, inadequate soft-tissue bulk at the site of amputation, and microvascular and macrovascular pathologies (such as diabetes and peripheral vascular disease) can contribute to residual limb pain. If diagnosed, these sources of pain should be incorporated into the treatment plan when managing neuropathic lower extremity pain.
Classically, patients with neuropathic lower extremity pain from symptomatic neuromas will complain of cold sensitivity, hypersensitivity, sharp pain, shooting sensations, and paresthesias. In many instances, patients will have pain and symptoms that have persisted for longer than three months, which is generally considered the time point for a diagnosis of chronic pain. The diagnosis of a symptomatic neuroma in the lower extremity can be made during physical examination by eliciting tenderness from point percussion at the site of the suspected neuroma (Tinel’s sign). Clinical examination can be supplemented by the injection of a local anesthetic (e.g., 1% lidocaine) at this site of maximal tenderness and subsequent relief of pain on percussion.
When the clinical diagnosis of neuroma is unclear, imaging can be considered to assist in making the diagnosis and potentially guiding the surgical planning. CT scans have been used in the past to identify symptomatic neuromas, but are now used predominantly to assess bony pathology. To assess any peripheral nerve pathology, high-resolution ultrasound and MRI are highly effective. Most importantly, all of these forms of imaging can be used to define the sources of pain in the lower extremity including bone spurs, heterotopic ossification, osteomyelitis, and vascular insufficiency, to ensure that all factors contributing to pain are adequately addressed.
It is important to note that although imaging of the lower extremity is very helpful to identify symptomatic neuromas, it may also reveal asymptomatic neuromas. Pain from these asymptomatic neuromas may be masked by more symptomatic neuromas that are clinically bothersome to the patient. If surgical treatment of a symptomatic neuroma is planned, consideration should be given to surgically treating any asymptomatic neuroma identified. Once pain from a symptomatic neuroma has been successfully treated surgically, previously “asymptomatic” neuromas may become more obvious to the patient and necessitate surgical intervention. Furthermore, as discussed earlier, these asymptomatic neuromas might be a source for the maladaptive process of central sensitization of pain. Therefore, when obtaining imaging to characterize symptomatic neuromas, it is important to consider imaging of additional suspected nerves, with corresponding neuromas, that may require surgical treatment.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here