Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Surgical pathologists encounter a wide variety of nodules and tumors of hepatic origin, including both neoplastic and non-neoplastic lesions. Distinguishing between them can be challenging, but the distinction is critically important given the differing prognostic and therapeutic implications. Therefore, pathologists must understand the diagnostic criteria of these entities as well as the pitfalls inherent in interpreting small samples, such as core needle biopsies or fine needle aspirate (FNA) specimens. Furthermore, it is necessary to be aware of how the differential diagnosis varies in a cirrhotic versus a noncirrhotic liver, and to be aware of the context of the background liver, ideally from the tissue but at least from radiographic studies.
Nodular regenerative hyperplasia (NRH) is a rare condition characterized by transformation of the liver parenchyma into regenerative, non-neoplastic nodules that are not delimited by fibrous septa. It is an important cause of noncirrhotic portal hypertension (see also Chapter 19 ) and is associated with a wide range of conditions, including autoimmune diseases, common variable immunodeficiency, hematological disorders, and numerous drugs, including azathioprine, didanosine, and oxaliplatin. NRH may also occur in livers containing metastatic or primary tumor. Wanless originally postulated that the pathogenesis appears to be related to abnormalities of portal hepatic blood flow (particularly portal venous thrombosis), leading to localized areas of hypo- and hyper-perfusion. Patients may have elevated alkaline phosphatase, GGT, and/or transaminases.
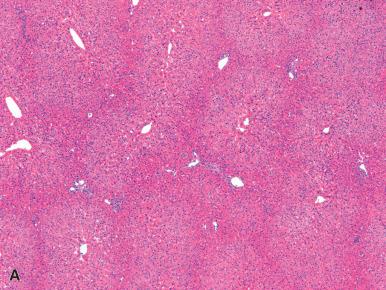
Macroscopically, the surface of the liver may be nodular and resemble micronodular cirrhosis. The histopathologic diagnosis of NRH is easier on wedge biopsies or surgical specimens than on needle biopsies, as the features can be difficult to appreciate in small specimens. Microscopically, NRH is characterized by nodules of hyperplastic hepatocytes arranged in plates more than one cell thick, whereas the parenchymal cells adjacent to the nodules (usually centrilobular areas, but not always) are compressed and atrophic ( Fig. 20.1A ). Fibrous septa must be absent, which differentiates the lesion from cirrhosis ; however, in some cases, the compressed parenchyma between the nodules shows perisinusoidal fibrosis. In general, there is no or only minimal inflammation and no evidence of liver cell damage, although sinusoidal lymphocytosis has been reported in association with NRH. The nodularity is better revealed on a reticulin stain (see Fig. 20.1B ). Vascular changes may be subtle and inconspicuous in needle biopsies and may include periportal angiomatosis as well as obliteration of portal vein branches; the latter is strongly associated with clinically evident portal hypertension.
Partial nodular transformation is a rare entity in which the nodules may be larger than in NRH and are often restricted to the perihilar region, mimicking a neoplasm. Again, portal venous thrombosis is invoked in the pathogenesis of the lesions but in this instance situated near the porta hepatis in the larger portal vein branches.
Focal nodular hyperplasia (FNH) can occur in any age group, including childhood; most cases, however, are seen during the third to fifth decades of life. The female to male ratio has ranged from 2 : 1 to 8 : 1 in the various adult and pediatric series. In approximately 80% of cases, the lesion is asymptomatic. In contrast to hepatocellular adenoma, hemoperitoneum as a complication is extremely rare, although it has been described. FNH is usually solitary; multicentricity has been described in 20% of adult cases, however, and in an even higher percentage of pediatric cases. Multicentric cases may be associated with other lesions (particularly vascular), including hemangiomas of the liver, telangiectasias of the brain, berry aneurysm, and portal vein atresia, as well as neoplasms of the brain including astrocytoma and meningioma.
Overall, the pathogenesis of FNH remains controversial. A possible relationship to oral contraceptives has been postulated, but it is not as clear-cut as for liver cell adenoma. There is no question that this disease occurred prior to the widespread use of oral contraceptives and that it can occur in males, particularly in association with chronic alcohol abuse. It has also been associated (together with hepatocellular adenomas and large regenerative nodules) in patients with hepatic venous outflow obstruction and following orthotopic liver transplantation, suggesting that vascular anomalies or a hyperplastic/regenerative response to a localized insult to the liver parenchyma may also play a role in pathogenesis. This hypothesis is also supported by the fact that lesions indistinguishable from nodular hyperplasia have been reported in cirrhotic livers. Clonality studies of FNH have shown mixed results.
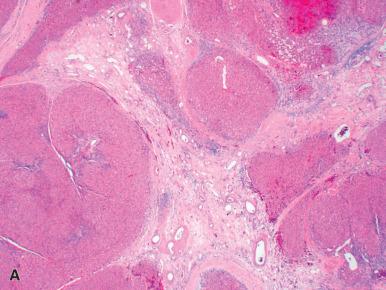
Grossly, FNH usually presents as a subcapsular, gray-white, solid mass that is sometimes pedunculated. On cut section, a white depressed area of fibrosis is often seen in the center, with broad strands radiating from it to the periphery in a stellate configuration ( Fig. 20.2A and B ). This can be detected in more than half of cases by MRI techniques. Microscopically, FNH has a nodular appearance with a central stellate scar ( Fig. 20.3A ), although it is important to note that the scar (at least a well-formed one) is not invariably present. Large, often thick-walled vessels are present in the fibrous bands (see Fig. 20.3B ), but they do not have accompanying bile ducts. Patchy areas of ductular reaction are characteristically present at the junction of the fibrous septa with the hepatocellular component (see Fig. 20.3C ), and this may be an important clue to the diagnosis, particularly in needle biopsies. Acute or chronic inflammation is common. The hepatocytes may contain increased amounts of glycogen and fat.

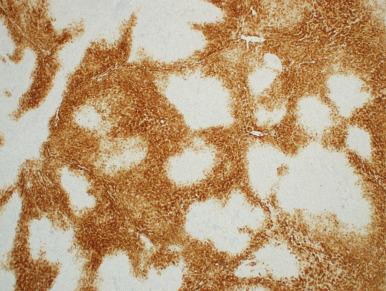
FNH typically shows a map-like staining pattern with glutamine synthetase (GS) ( Fig. 20.4 ), which may help to distinguish them from hepatocellular adenoma. CK7, CK19, and neuronal cell adhesion molecule may be used to highlight the ductular reaction. Immunostains for estrogen hormone receptors have yielded mixed results.
The differential diagnosis of FNH includes—at both the clinical, radiographic, and histologic level—hepatocellular adenoma, well-differentiated hepatocellular carcinoma (HCC), and cirrhosis; the latter is a particular problem on needle biopsy, and knowledge of the state of the background liver (cirrhotic vs. noncirrhotic) is invaluable in this instance. The lack of a fibrous scar may also be challenging, although often the presence of nodularity, ductular reaction, dysmorphic vessels, and/or a poorly formed fibrous scar may aid in diagnosis. Reticulin stain is useful in distinguishing between FNH and HCC, in that FNH has no loss of reticulin, and immunohistochemistry may aid in distinguishing FNH from hepatocellular adenomas. A summary of the histologic and immunohistochemical differences between FNH and liver cell adenoma is given in Table 20.1 . This distinction is clinically important, as asymptomatic FNH may be followed, but hepatocellular adenomas (particularly those with β-catenin mutations, as discussed later) are more likely to be resected or ablated.
| FEATURE | FOCAL NODULAR HYPERPLASIA | LIVER CELL ADENOMA |
|---|---|---|
| Capsule | No capsule | Well demarcated but unencapsulated |
| Stellate scar | Typically present | Absent |
| Parenchyma | Nodular | Homogeneous |
| Hemorrhage, necrosis | Rare | Common |
| Ductular reaction | Present | Absent |
| Vascularity | Large thick-walled vessels | Thin-walled sinusoids |
| Telangiectasia, dysmorphic vessels (inflammatory HA) | ||
| Background liver | Noncirrhotic | Noncirrhotic |
| Cytologic atypia | Rare | Can be present |
| Acinar change | Typically absent | May be focally present |
| Reticulin framework | Intact | Intact overall |
| Glutamine synthetase | Positive, map-like pattern | Positive in β-catenin activated HA only |
| β-Catenin | Negative | Positive in β-catenin activated HA |
| LFABP | Positive | Negative in HNF1α inactivated HA |
| Glypican 3 | Negative | Negative |
| SAA/CRP | Negative | Positive in inflammatory HA |
True adenomas of the liver are rare. They have a striking predilection for females and are pathogenetically related to the use of oral contraceptives, a fact supported by their occasional total regression following discontinuation of the hormones. These neoplasms have also been reported following anabolic–androgenic steroid therapy, following long-term carbamazepine therapy, in association with glycogen storage diseases and other genetically determined diseases, and in children with sex hormone disturbances. When compared with FNH, the adenomas are more often symptomatic and can lead to severe and even fatal intraperitoneal hemorrhage. Most of the patients are in the third to fifth decades of life, but cases have also been reported in children.
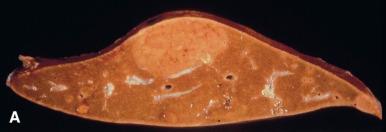
Hepatocellular adenomas (HA) arise in noncirrhotic livers, and approximately 70% of the lesions are solitary. They can occur anywhere in the liver but are usually located in the right lobe. Adenomas are usually visualized as defects on 99m technetium–sulfur colloid liver scans and may also have characteristic CT and MRI findings; however, many adenomas do exhibit typical radiographic features, and thus biopsy may be required for diagnosis. Grossly, they are well demarcated but usually unencapsulated and are more pale and yellow than the surrounding liver. The typical central scar of FNH is absent ( Fig. 20.5A and B ).
Advances in molecular biology have resulted in the classification of hepatic adenomas into four major groups, based on both genotypic and phenotypic characteristics. Adenomas with hepatocyte nuclear factor 1α (HNF1 α ) mutations account for 30%–50% of cases and are characterized by biallelic inactivating mutations of the HNF1α gene (both somatic, or one germline and one somatic). These tumors are associated with steatosis, insulin resistance, and absence of liver fatty acid–binding protein (LFABP) expression by immunohistochemistry (as compared with positive staining in normal liver); they may be single or multiple. Microscopically, adenomas with HNF1α mutations are diffusely steatotic, with numerous thin-walled vessels ( Fig. 20.6A ). The reticulin framework should be intact, and inflammation and nuclear atypia are absent.
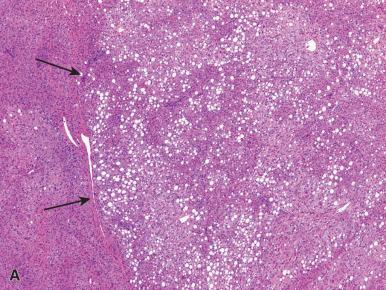
The second group are those with CTNNB1 mutations, accounting for 10%–15% of cases. These are the most highly correlated with malignant transformation, and they are associated with male gender, androgen use, and glycogenesis. Immunohistochemically, they overexpress GS, and show nuclear positivity with β-catenin staining (see Fig. 20.6B ), although it may be very focal. Microscopically, the features are nonspecific (see Fig. 20.6C ), but mild cellular atypia and focal rosette formation can be seen. They typically do not have steatosis.
The third group is known as inflammatory or telangiectatic adenomas, which were previously thought to represent a variant of FNH. They are the most common, comprising 40%–50% of adenomas. These tumors are associated with increased BMI and occasionally present with fever and clinical signs of inflammation; these are also the type most commonly seen in men. Histologically, they have characteristic inflammation, sinusoidal dilatation, and dystrophic vessels (see Fig. 20.6D and E ), and they express serum amyloid A and C-reactive protein by immunohistochemistry. LFABP is normally expressed. From a molecular standpoint, they lack HNF1α mutations, but 10% harbor CTNNB1 mutations, and thus are at risk for malignant transformation. The fourth group (<10%), known as unclassified adenomas, lacks specific molecular and phenotypic features.
Other features occasionally seen in hepatocellular adenomas include oncocytic changes, presence of Mallory hyaline, and pigmentation. An associated granulomatous reaction can also be seen with hepatic adenoma, and the granulomas may be within or outside of the tumor. In addition to the immunohistochemical features discussed previously, estrogen and progesterone receptors are present in about 75% of hepatic adenomas, whereas androgen receptors are expressed in only 20% of cases.
The risk of malignant transformation in adenomas is approximately 5%, and risk factors include male gender, size, and adenoma subtype (see earlier). Multiple sections and reticulin stains should be carefully evaluated in these tumors in order to rule out a well-differentiated HCC. The distinction between hepatocellular adenoma and well-differentiated carcinoma may be extremely difficult, however. Recent studies have suggested that GS and heat-shock protein 70 (HSP70) may be useful in this scenario, as these markers are more likely to be positive in cases of well-differentiated malignancy, but the usefulness of this remains controversial. Occasionally, an unequivocal and distinct focus of hepatocellular carcinoma is seen arising within an hepatic adenoma, indicating that these tumors do have a potential for malignant transformation. In rare instances, coexistence of adenoma and carcinoma in the same liver have been described.
Reportedly one third of patients with hepatic adenomas have multiple tumors, most commonly associated with the HNF1α subtype. When 10 or more tumors are present, this condition is referred to as liver cell adenomatosis . A proportion of cases are familial, due to germline mutations in the (TCF1/HNF1α) gene, which also causes maturity-onset diabetes of the young type 3 (MODY3).
Advances in imaging have made it possible to detect increasingly smaller lesions in cirrhotic patients, which are often biopsied to exclude HCC. The terminology used for these lesions has historically been inconsistent and confusing and has included terms such as ordinary and atypical adenomatous hyperplasia, minimal deviation hepatoma, paraplasia, large regenerative nodule, types I and II macroregenerative nodule, adenomatous hyperplasia, and low-grade dysplastic nodule. In a laudable attempt to standardize the diagnostic criteria and terminology for these lesions, a multinational panel of liver pathologists has proposed the following classification, which has been generally accepted by experienced hepatopathologists.
Regenerative nodules (also known as large regenerative nodules, macroregenerative nodules, adenomatous hyperplasia, and low-grade dysplastic nodules) are greater than 1.0 cm, with normal architecture and intact reticulin framework ( Fig. 20.7 ). The majority of regenerative nodules are probably not premalignant, and, like the rest of a cirrhotic liver, they may contain iron, fat, bile, Mallory hyaline, or large cell change but should not feature small cell change (see later). There are no histologic criteria to distinguish a non-clonal regenerative nodule from a nodule with low-grade dysplasia, as clonality is assessed via molecular means and the likelihood of progression to malignancy is unclear. For these reasons, most pathologists avoid this term.
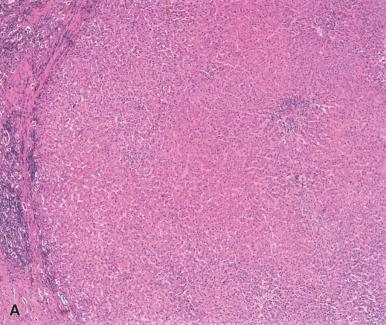
High-grade dysplastic nodules (HGDNs, also known as borderline nodules or atypical adenomatous hyperplasia) have both architectural and cytologic atypia, and are believed to be precursors of HCC. They may contain a number of abnormalities, including thickening of cell plates up to three cells thick, focal acinar formation, and occasional unpaired arterioles ( Fig. 20.8 ). Small cell change is often present as well (see later). HGDN may contain fat, bile, and Mallory hyaline, as in regenerative nodules, but iron is often absent. Reticulin should be intact or only focally decreased. Both regenerative nodules and HGDNs may differ in color from the surrounding liver and may protrude out from the cut surface.
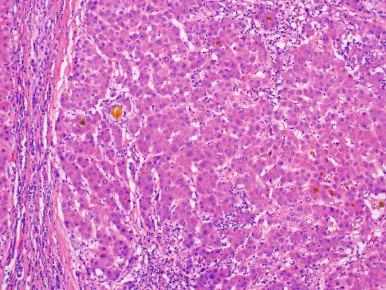
There are two types of liver cell dysplasia. Large cell change is defined by both nuclear and cytoplasmic enlargement (i.e. preserved N:C ratio), nuclear pleomorphism, nuclear hyperchromasia, and occasional multinucleated hepatocytes ( Fig. 20.9 ). Large cell change can be seen throughout a cirrhotic liver, including within regenerative nodules and HGDNs, and the biologic significance remains unelucidated. However, the presence of large cell change in cirrhotic livers due to hepatitis B has been reported as an independent risk factor for the development of HCC. Small cell change is characterized by a decreased volume of hepatocytic cytoplasm associated with moderately enlarged nuclear size, resulting in an increased nucleocytoplasmic ratio and low-power appearance of increased nuclear density ( Fig. 20.10 ). Small cell change is considered to be a premalignant lesion, based on its proliferative activity, molecular characteristics, and presence within both HGDN and HCCs. Both large cell change and small cell change should be noted in the pathology report.
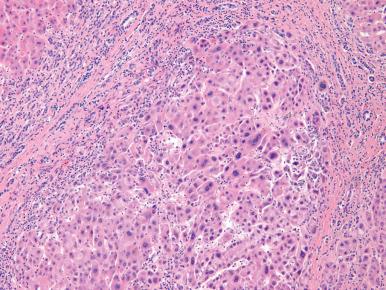
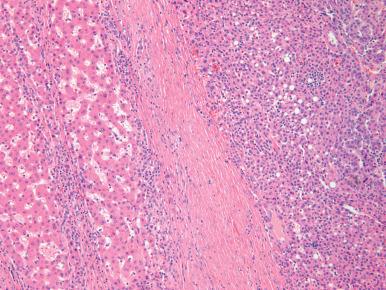
HCC (also known as liver cell carcinoma, and previously termed hepatocarcinoma or hepatoma) is the sixth most common malignancy worldwide and the second most common cause of cancer-associated death. Historically, it has been relatively rare in the United States, but during the past two decades the incidence has tripled secondary to the hepatitis C epidemic. The highest incidence is still in regions with endemic hepatitis B infection, such as Southeast Asia and Subsaharan Africa ; China alone accounts for over half of the world's cases of HCC. Most cases are seen in patients older than 40 years, with a peak at 70 years of age, but this tumor can also occur in younger individuals and even in children. Some cases have even occurred as second malignancies in successfully treated Wilms tumor patients. Currently, the greatest increase in HCC is among Caucasians and Hispanics between 45 and 60 years of age.
HCC is 2–4 times more common in males than in females. It usually presents with anorexia, malaise, abdominal pain, ascites, and liver enlargement. Obstructive jaundice may result from tumor invasion into the common bile duct. HCC has also been associated with numerous systemic manifestations such as hypoglycemia, hypercholesterolemia, erythrocytosis, hypercalcemia, carcinoid syndrome, ectopic production of chorionic gonadotropin and prostaglandin, and dysfibrinogenemia, and several others.
Serum elevation of alpha-fetoprotein (AFP) occurs in a majority of patients with HCC, but up to 40% of HCC patients have normal AFP levels, particularly when tumors are small. Elevated serum AFP can also be seen with malignant germ cell tumors, liver metastases, liver trauma, cirrhosis, chronic hepatitis, and cholangiocarcinoma. For these reasons, recent studies have argued that AFP is not an effective screening or surveillance tool for HCC.
The major risk factors for HCC include the hepatotropic viruses hepatitis B virus (HBV) and hepatitis C virus (HCV), alcoholic liver disease, and probably non-alcoholic fatty liver disease (NAFLD).
The majority of the risk factors for HCC result in cirrhosis, which is present in over 80% of patients with HCC. Patients with cirrhosis have an estimated 5-year cumulative risk of developing HCC of 5%–30%, depending on the underlying risk factor, geographic area, and ethnicity of the patient.
Hepatitis B infection is associated with approximately half of the cases of HCC in the world and virtually all childhood cases. Although both hepatitis B and hepatitis C can cause HCC in the absence of cirrhosis, the vast majority of patients with hepatotropic virus-related HCC have cirrhosis. Patients with chronic HBV have a 5–15-fold increased risk of developing HCC, whereas the increased risk for patients with HCV is 15–20 times that of the general population. The risk of developing HCC in patients with chronic HBV is further increased in men, elderly patients, and those of Asian or African race; with exposure to aflatoxin; with alcohol and tobacco use; if there is coinfection with HCV or the delta virus; with high levels of HBV DNA, positivity for hepatitis B e antigen (HBeAg), or infection with HBV genotype C; and with increasing length of time of infection. Similar factors are associated with increased risk of HCC in patients with chronic HCV infection, including older age, male gender, metabolic syndrome, and alcohol use.
Integration of hepatitis virus DNA into host genomic DNA is believed to be an early event in the development of HBV-associated HCC. HBV DNA integration induces both genomic instability and mutagenesis of several cancer-related genes. A major role is also played by the HBV-associated protein HBx, which stimulates virus gene expression and replication, and also activates a number of cell cycle pathways that are critical to hepatocyte growth and survival. HBV DNA is detectable in patients who are HBsAg negative, but the risk of progression to HCC is unclear in these individuals. The role of the hepatitis C virus in the development of HCC remains less well elucidated, but animal studies have indicated that the core protein of HCV appears to have oncogenic potential.
Heavy alcohol intake is a well-established risk factor for the development of HCC, although the direct carcinogenic effect of alcohol remains unproven and there is also evidence of a synergistic effect of heavy alcohol use and HBV or HCV infection. The overall risk of developing HCC in the context of alcoholic cirrhosis is estimated at 15%.
There is a growing body of evidence that NAFLD is associated with the development of HCC, even in the absence of cirrhosis, and that the annual incidence of NAFLD-associated HCC in the United States increased by almost 10% between 2004 and 2009. The prevalence of HCC in the context of NAFLD is expected to rise as more and more adults develop this disease worldwide, given the increasing rates of obesity, diabetes, and dyslipidemia. HCC in this context is also associated with a higher mortality rate.
A number of drugs and toxins are associated with the development of HCC. Thorium dioxide suspension (Thorotrast), a radiographic contrast medium, has resulted in the development of many liver cell carcinomas, the average latent period being 20 years. The geographic distribution of HCC has also suggested a relationship with the ingestion of aflatoxins, which are metabolic products of the ubiquitous fungus Aspergillus flavus ; contamination of food with aflatoxins remains a serious problem in parts of the world where the incidence of HCC is highest. Although the exact mechanism remains to be elucidated, it is known that aflatoxin B1 induces p53 mutations and may act synergistically with HBV to contribute to carcinogenesis. Rare cases of HCC have been reported in males in association with long-term administration of androgenic–anabolic steroids. Data on a potential association between oral contraceptive pills and HCC remain controversial. Although some studies have shown an increased risk of HCC in women with long-term oral contraceptive use (>5 years), the studies are generally small, and more recent studies have shown no definite evidence of an increased risk. There is also little information regarding the risk in patients who are taking newer generation, low-dose contraceptive pills.
A number of metabolic disorders are risk factors for HCC as well. Tyrosinemia type 1 is associated with a high incidence of liver cell carcinoma, usually in a background of cirrhosis. In one series, 37% of patients surviving beyond 2 years of age developed this malignancy in childhood. Cases of HCC associated with α1-antitrypsin deficiency have also been reported. Patients with the PiZ variant of antitrypsin deficiency seem to be particularly prone to this complication and also to the development of cholangiocarcinoma.
Other hereditary disorders associated with HCC include ataxia–telangiectasia ; glycogen storage disease , particularly type 1a; hereditary hemochromatosis , in which the risk of HCC is approximately 20 times higher than in the general population ; and porphyria. Cases of HCC have also been reported following the Kasai procedure in patients with biliary atresia, in association with familial cholestatic cirrhosis of childhood, and with congenital hepatic fibrosis associated with adult polycystic kidneys.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here