Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Vascular lesions are among the most common indications for laser treatment.
Treatment relies on the theory of selective photothermolysis and aims to confine thermal injury to targeted vessels.
Pulsed dye laser (PDL) is the treatment of choice for port-wine birthmarks (PWBs), but multiple vascular targeting devices can be used. Early treatment is thought to enhance response. Resistant or hypertrophic lesions can be treated with an alexandrite laser.
β-blockers are standard of care for treatment of infantile hemangiomas.
Combined treatment with β-blockers and PDL can be considered for infantile hemangiomas, particularly those which are ulcerated or in cosmetically sensitive areas. Low energies should be used for proliferative lesions treated with PDL. Involuted lesions with residual telangiectasias and/or textural change can be treated with PDL and fractional lasers.
Erythema and telangiectasias associated with rosacea may be treated with vascular targeting light sources, including PDL or intense pulsed light.
The treatment of vascular lesions was among the first applications of lasers in dermatology. Laser surgery has since become the treatment of choice for many vascular lesions, including port-wine birthmarks (PWBs), rosacea, and poikiloderma. Laser can also be used for infantile hemangiomas (IH).
Vascular-specific lasers have evolved considerably. The 1960s saw the emergence of ruby and argon lasers, which improved the color of PWBs and IH, but relatively nonspecific heating of the skin resulted in scarring. In 1983, the theory of selective photothermolysis proposed a way to confine thermal injury to the target of interest while minimizing damage to surrounding tissue.
Three components are necessary for selective photothermolysis: a laser wavelength with preferential absorption of the target chromophore, appropriate pulse duration matched to the target size, and a fluence that both treats the target and minimizes nonspecific thermal injury. Thermal relaxation time is roughly defined as time needed for the heated target to cool approximately halfway to its initial temperature. The ideal pulse duration is shorter than or equal to the thermal relaxation time of the target. A pulse duration that is too short may not be effective, whereas one that is too long may cause heat to dissipate to surrounding structures and cause unwanted thermal injury. The target chromophore for vascular lesions is oxyhemoglobin which has greatest absorption peaks at 418, 542, and 577 nm ( Fig. 2.1 ). Laser light is absorbed by oxyhemoglobin and converted to heat, which is transferred to the vessel wall causing coagulation and vessel closure. Other hemoglobin species can also be targeted. For example, venous lesions may benefit from wavelengths that target deoxyhemoglobin. The alexandrite laser at 755 nm is close to a deoxyhemoglobin absorption peak and has been used for refractory or hypertrophic PWBs, venocapillary malformations, and venous lakes. Methemoglobin absorption (which forms after 585 or 595 nm treatment at or above 5 J/cm 2 ) has been recognized as another potential target chromophore.
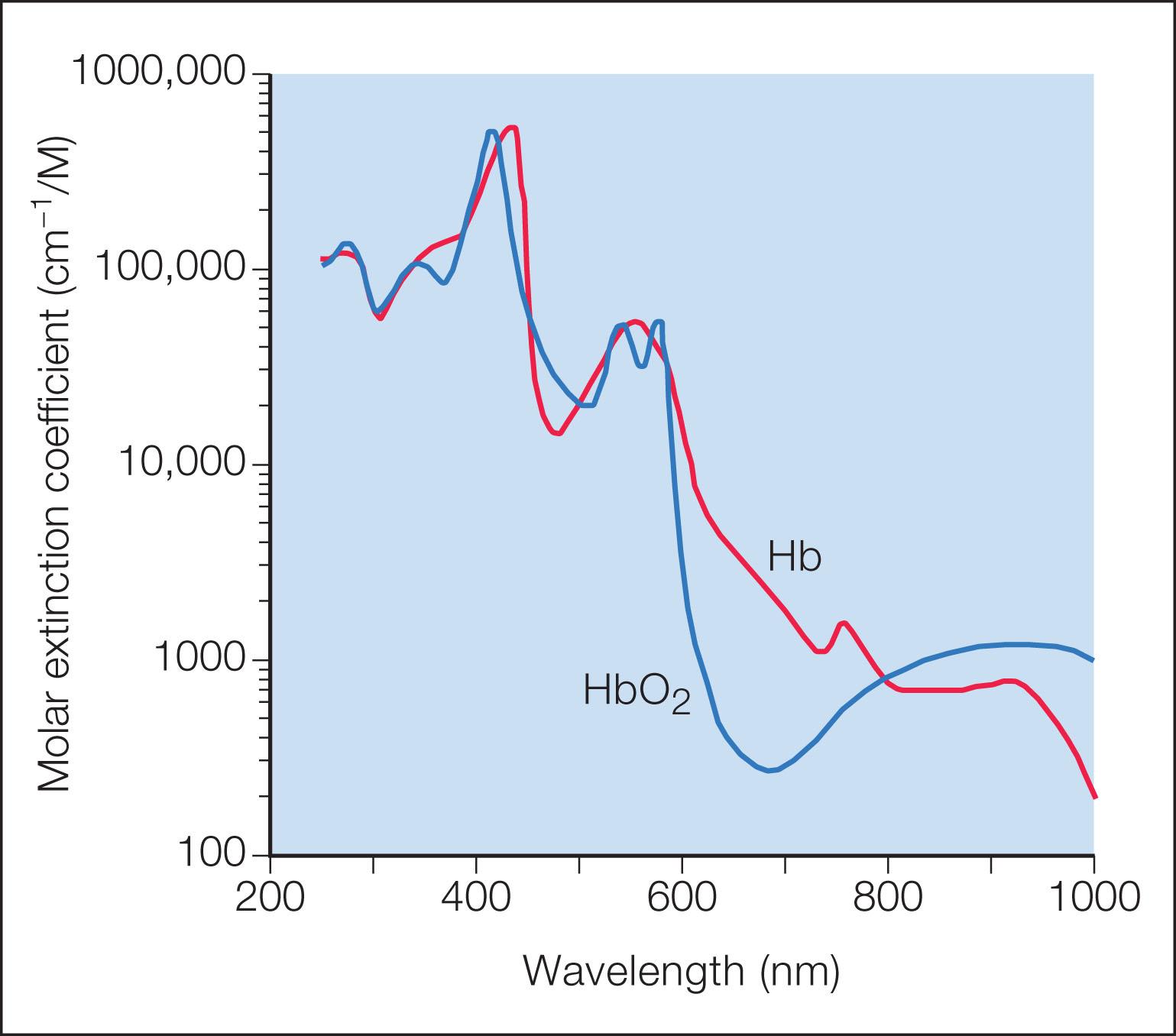
The pulsed dye laser (PDL) became available in 1986 and was initially developed at 577 nm to target oxyhemoglobin’s yellow absorption peak. It was later realized that selective photothermolysis could be achieved with wavelengths near absorption peaks, if preferential absorption still occurred. PDLs shifted to 585 nm, allowing for a depth of penetration of approximately 1.16 mm; 595 nm PDLs later became available to achieve greater depth of penetration. PDLs also evolved to incorporate longer pulse durations. Early PDLs had a fixed pulse duration of 0.45 ms, whereas currently available PDLs have pulse durations from 0.45 to 40 ms. Longer pulse durations can minimize posttreatment purpura.
Epidermal cooling was introduced in the 1990s to protect the epidermis, minimizing pigmentary changes and scarring. Cooling also permits use of higher fluences and provides greater treatment efficacy while minimizing discomfort. Modern cooling methods include cryogen spray, contact, and forced cold air.
Because PDL reaches a depth of only 1–2 mm, other lasers can be used to treat vascular lesions with greater depth. For example, the 755 nm alexandrite laser and 1064 nm neodymium:yttrium-aluminum-garnet (Nd:YAG) laser penetrate up to 50%–75% deeper into the skin. Given that the absolute absorption of hemoglobin is lower at these wavelengths, higher fluences are required and risk of injury is greater.
Intense pulsed light (IPL) devices emit polychromatic noncoherent broadband light from 420 to 1400 nm with varying pulse durations. Filters are implemented to remove unwanted wavelengths.
Other potential vascular targeting lasers include potassium titanyl phosphate (KTP) (532 nm), other near-infrared, long-pulsed lasers such as diode (including 800–810 nm, 940 nm), and dual wavelength lasers such as PDL and Nd:YAG (595 nm and 1064 nm).
The International Society for the Study of Vascular Anomalies (ISSVA) publishes a widely accepted standard for classification of vascular anomalies that was updated in 2018. Lesions are broadly grouped as either vascular tumors characterized by a proliferation of blood vessels, or vascular malformations characterized by vessels with abnormal structure. IH, a type of vascular tumor, and PWB, a type of vascular malformation are common vascular anomalies that present to the dermatologist ( Table 2.1 ). Of note, certain rare lesions seem to defy precise classification under these terms and have been listed in a provisional category. The classification also provides a list of known causal genes related to each vascular anomaly, which is an exciting area of evolving research.
| Infantile Hemangioma | Port-Wine Birthmark | |
|---|---|---|
| Onset |
|
|
| Course |
|
|
| Tissue marker |
|
|
PWBs are vascular malformations that are composed of ectatic capillaries and postcapillary venules in the superficial vascular plexus. PWB vessels vary in size from 7 to 300 µm, with older patients, particularly those naive to laser treatment, tending to demonstrate larger vessels. In most cases, PWBs are congenital, although acquired cases are reported. PWBs occur in approximately 0.3%–0.5% of newborns. They often involve cosmetically sensitive areas including the head and neck but may appear anywhere on the body. Over time, some PWBs darken in color with acquisition of secondary changes including soft tissue overgrowth and development of vascular nodules ( Fig. 2.2 ). In untreated patients, the mean age of hypertrophy has been reported as 37 years, and by the fifth decade approximately 65% of lesions are hypertrophic or nodular. Over time, significant soft tissue overgrowth, if it occurs, can lead to functional impairment in areas such as the lip or eyelid. Vascular nodules or blebs may develop. Some are pyogenic granulomas, which are proliferative and associated with second genetic hits, but others defy classification and may bleed with minimal trauma. Early treatment of PWBs can improve outcomes and is restorative in nature, minimizing disfigurement and psychosocial morbidity.
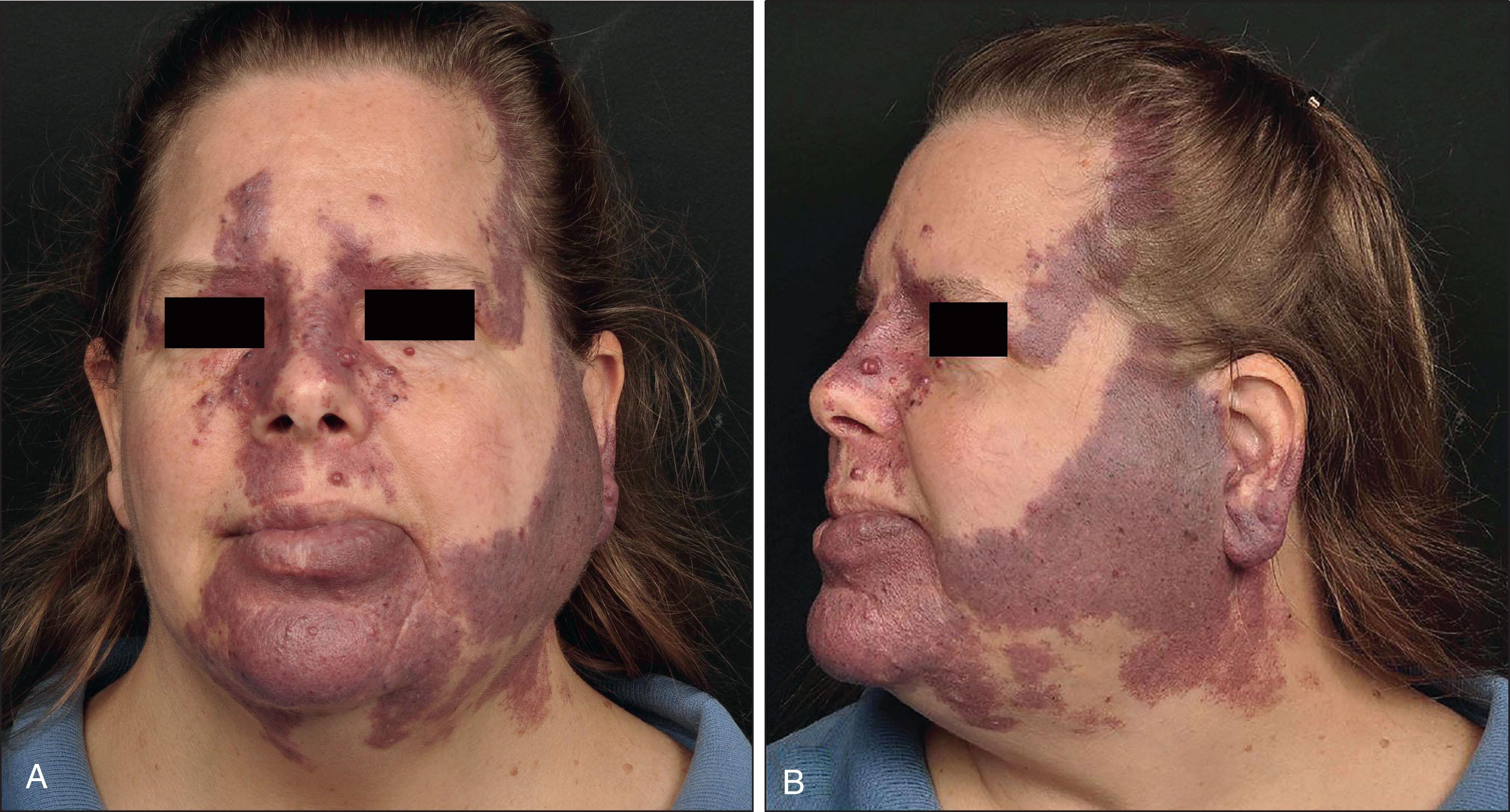
The natural history of PWB is explained by the recent discovery of mosaic mutations in highly conserved genes including GNAQ, GNA11, PIK3CA , and others that control cell-cycle proliferation and survival. Their mosaic origin accounts for low familial recurrence risk. Gain-of-function mutations in PWBs activate oncogenic pathways that result in synchronous, tightly regulated cellular proliferation and growth. These genes are known to be critical regulators of fetal vascular development, and very low allele frequency is required for alterations in embryonic vasculature.
Genotype-phenotype correlations exist but are not yet well characterized due to varied phenotype expression. Contributing factors to varied phenotype expression are hypothesized to include low variant allele frequency, anatomical location, age, prior treatment, and ancestry. In general, GNAQ mutations yield PWBs which are often red and moderately demarcated. PWBs resulting from PIK3CA hotspot mutations are generally deep red, sharply demarcated, and often associated with overgrowth, particularly leg length discrepancy. Lighter pink and blotchy or reticulated lesions are often associated with GNA11 mutations.
PWBs can be isolated or associated with various genetic syndromes. Sturge-Weber syndrome involves a facial PWB with associated eye and/or cerebral nervous system involvement, including glaucoma, seizures, and developmental delay. Numerous studies have indicated that facial PWBs follow embryologic vascular development of the face and not dermatomal nerve distribution, which was previously suspected. The most common mutation in both isolated PWBs and Sturge-Weber syndrome is a somatic activating mutation in GNAQ , the gene encoding guanine nucleotide-binding protein G(q) subunit alpha, as detailed in a seminal paper by Shirley, et al. The highest risk area for CNS involvement in Sturge-Weber syndrome is PWB of the forehead region (including from the lateral canthus to the top of the helices of the ear), with hemi-segmental or segmental lesions at highest risk. All patients with eyelid involvement should be referred to ophthalmology for glaucoma surveillance. More than 75% of patients with Sturge-Weber syndrome develop seizures before one year of age, and early referral to neurology are critical components of management. Mutations in PiK3CA are known to cause another PWB-associated syndrome, Klippel-Trénaunay syndrome (KTS), which is characterized by PWBs on an extremity associated with limb hypertrophy and underlying lymphatic and/or venous malformations. PWBs can also occur in association with arteriovenous malformations in capillary malformation–arteriovenous malformation syndrome, which can be caused by mutations in RASA-1 or EPHB4 , both involved in regulation of the RAS/MAPK signaling pathway.
Goals of PWB treatment include decreasing erythema, reducing psychosocial distress, and preventing development of blebs that may bleed or become secondarily infected. In addition, it has been theorized that treating PWBs early may minimize some PWB progression including nodularity later in life. PDL is considered the gold standard for PWB treatment.
Despite the overall effectiveness of PDL, individual patient response is variable. Approximately 80% of treated patients see a successful decrease in redness or thickness, but only approximately 20% will clear completely. One study found predictors of improved response include small size (less than 20 cm 2 ), location over bony areas (in particular the central forehead), and early treatment. Another study in 49 infants who started laser treatment before 6 months of age demonstrated an impressive average clearance of 88.6% after 1 year. Early treatment may be more beneficial due to thinner and overall smaller lesions, as well as the presence of hemoglobin F (a type of hemoglobin present during gestation and the first year of life) and less dilated vessels in infants. PWBs may darken again after laser therapy, a phenomenon dubbed "re-darkening,” hypothesized to occur due to progressive genetic etiology, lack of complete vessel eradication, and suboptimal laser treatment. Still, re-darkened areas remain lighter than their original untreated color.
Treatments are typically performed at 3- to 6-week intervals for 10 or more treatments until a plateau is reached or the lesion clears ( Fig. 2.3 ). When choosing settings, it is advisable to determine the fluence threshold on the darkest portion of the PWB with one or two test pulses. The fluence is adjusted to achieve the desired end point, which in the case of PDL using short pulse durations (e.g., 1.5 ms) is immediate purpura. If instead a gray color appears, the fluence is too high. Careful monitoring for tissue response is essential.
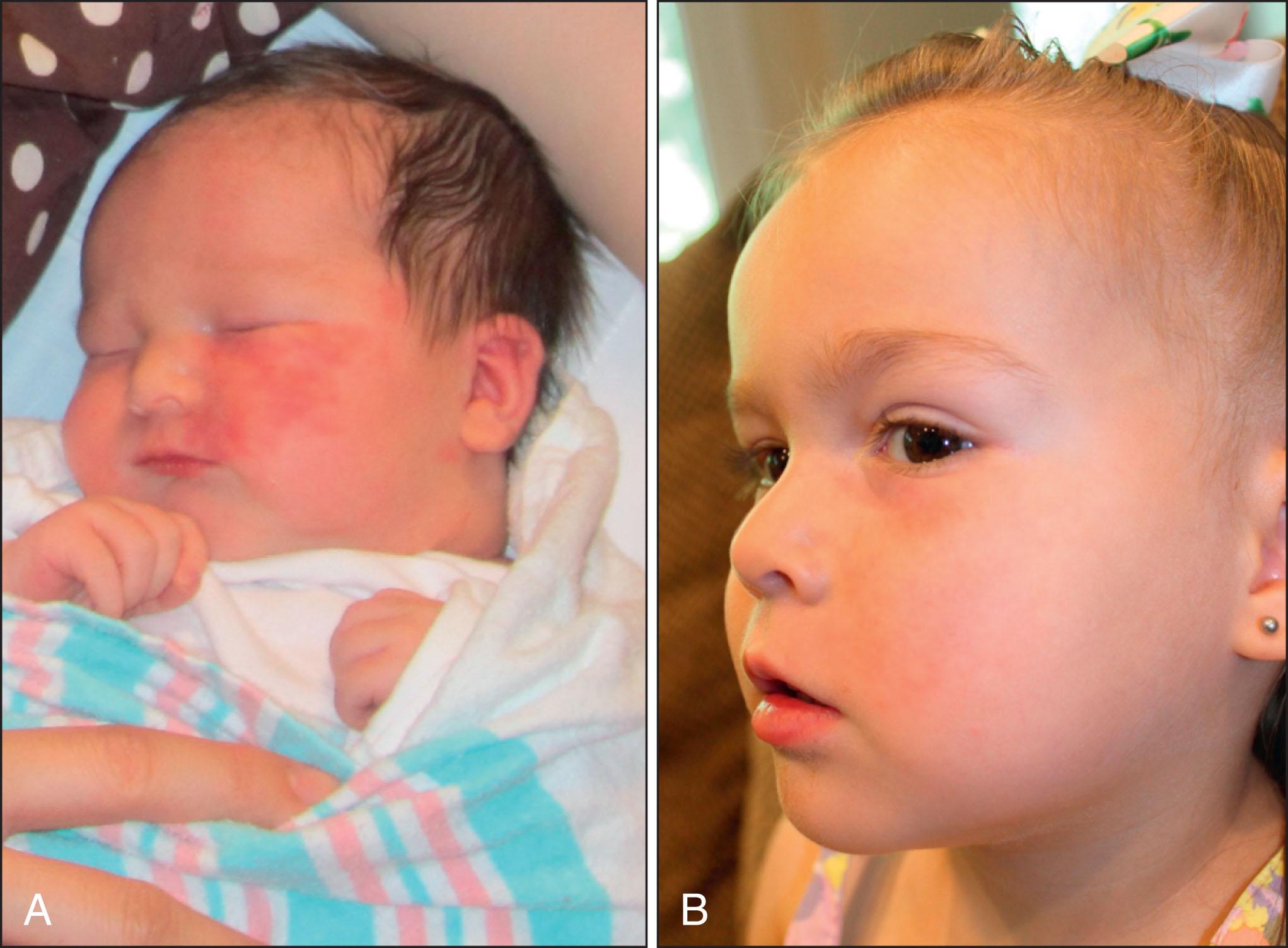
Changing the pulse duration may allow targeting of different sizes of vessels. The ideal pulse duration for PWB treatment has been proposed at 1–10 ms. In practice, treatment often begins at 1.5 ms, or in young children at 0.45 ms. Parameters to consider include 7- to 10-mm spot size, pulse duration of 0.45–6 ms, with appropriate epidermal cooling, such as cryogen spray cooling of 30 ms with a 20- to 30-ms delay. Longer pulse durations may be considered in darker skin types. Treatment should start at a lower energy that can be increased as tolerated. Parameters will vary by device.
As with any laser treatment, complications of PDL may include pigmentary change. When treating darker skin types, the risk of hypopigmentation and hyperpigmentation can be minimized by using appropriate cooling and longer pulse durations. Treatment intervals may need to be longer to allow for any pigmentation changes to resolve before proceeding with additional treatment. Care must be taken with leg lesions, which are prone to hyperpigmentation.
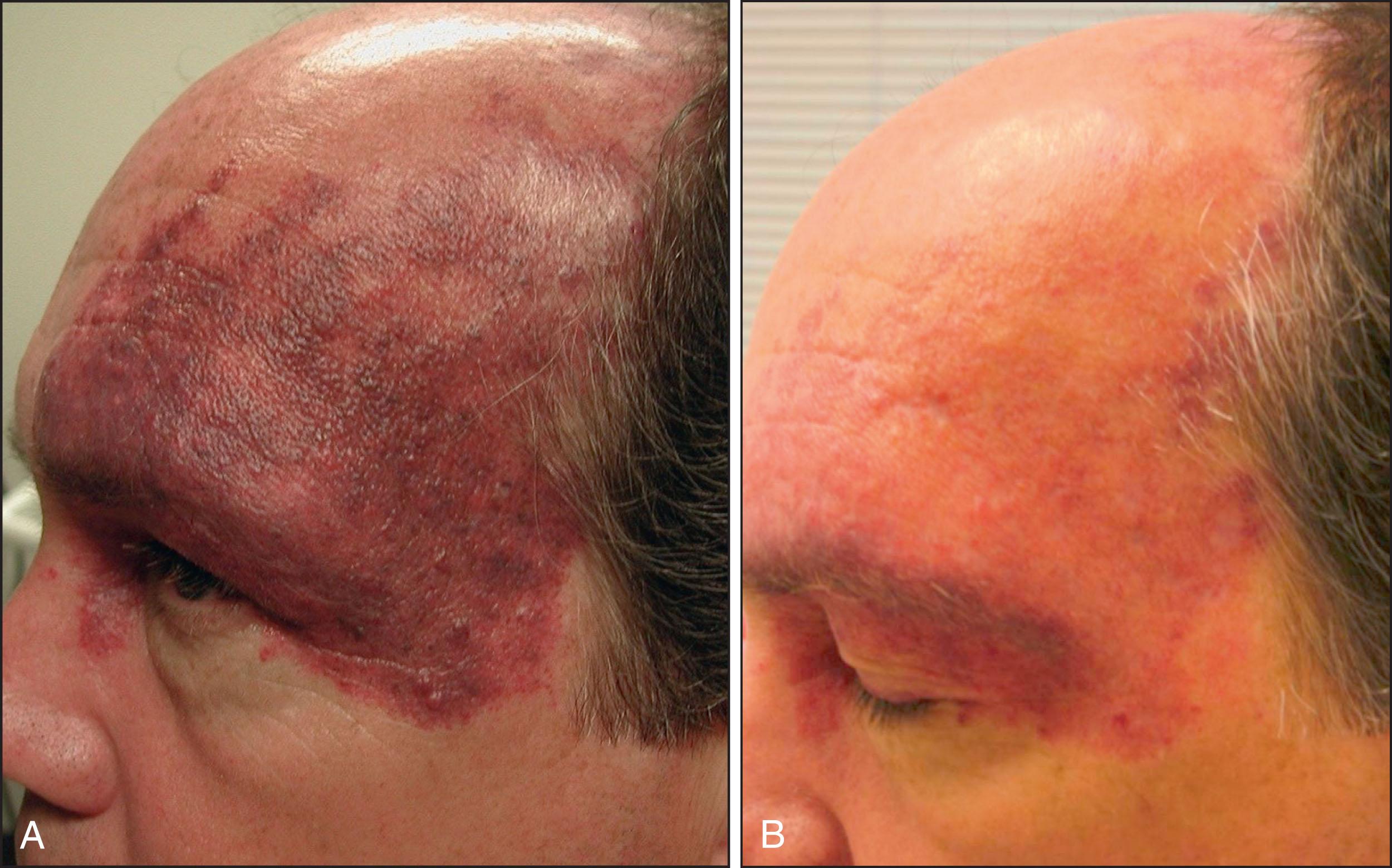
The alexandrite laser is typically used for PDL-resistant lesions, although it may be implemented as a first-line treatment for more hypertrophic, violaceous PWB in adults ( Fig. 2.4 ). The desired end point in this case is a transient gray color that evolves into a deeper persistent purpura over several minutes, as described by Izikson and Anderson in 2009. Care must be taken not to overlap or “stack” pulses because scarring can occur. Notably, the range of appropriate fluences for alexandrite laser is quite broad.
Nd:YAG laser and combined 595 nm and 1064 nm lasers can also be used for PWBs. Although depth of penetration can be increased, there is a narrow therapeutic window with these devices due to reduced oxyhemoglobin and caution is advised to avoid scarring. Scarring can occur even at low fluences. These devices should only be used by experienced laser surgeons and we prefer to utilize mostly for nodules that develop within PWB.
IPL with appropriate vascular filters can also be used, and some IPL devices have filters to optimize the wavelength range for PWB. As with many lasers, treatment in hair-bearing areas may lead to permanent hair loss. The risk is greater on the eyelashes, given the proximity of follicles to the surface, and on the eyebrows and scalp of young children, particularly those of darker skin types. Hair loss can also occur with any long-pulsed laser treatment of vascular lesions.
Nodules or blebs within PWB can be treated with excision or laser. In the case of PDL, several pulses or intentionally stacked pulses may be required. Given the limited depth of penetration of PDL, deeper penetrating lasers such as alexandrite or Nd:YAG can be used or nodules can be ablated with carbon dioxide (CO 2 ), or erbium-doped yttrium-aluminum-garnet (Er:YAG) lasers.
Photodynamic therapy (PDT) has been used with some success but is limited by the administration of systemic hematoporphyrin photosensitizers, which result in prolonged photosensitivity over weeks. There is also an increased risk of scarring or pigmentary change when compared to PDL.
Recent research has sought to improve treatment efficacy by combining light-based removal of PWBs with posttreatment antiangiogenic agents, especially rapamycin, which inhibits the mTOR enzyme that regulates cell growth and metabolism. There is no direct mechanistic data to suggest that mutant GNAQ or GNA11 directly impacts mTOR signaling, so rapamycin may not exert a pathway-specific effect. There was initial enthusiasm for combined PDL plus rapamycin but in clinical practice, most patients do not achieve significantly enhanced lightening. Other agents are being evaluated for use as adjuvants with laser and this is a promising area of research.
Optical coherence tomography (OCT) is a high-resolution bedside imaging tool that can rapidly characterize the diameter and depth of blood vessels in PWBs to a depth of 1 mm. Dynamic-OCT with matching of laser settings to vessel depth and diameter at each treatment session, may be promising in future clinical practice for more precise laser dosimetry.
Outline Borders of Pwbs Prior to Treatment
Laser pulses can induce erythema that blurs the border, making it difficult to appreciate the target. Prior to treatment with yellow light lasers such as PDL, lesion edges may be outlined with a yellow highlighter or white pencil to facilitate consistent visualization. This is particularly important prior to general anesthesia as vasodilation of blood vessels often obscure borders of the vascular birthmark.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here