Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Intra-axial tumours: usually of low attenuation on NECT ▸ high attenuation areas within a tumour indicate tumour calcification or recent intratumoural haemorrhage
Extra-axial tumours: associated with bone erosion or hyperostosis
Improved visualization of an enhancing mass lesion (e.g. a meningioma or metastases)
This can assess tumour relative cerebral blood volume (rCBV) and permeability changes ▸ it provides a limited area of coverage (compared with MRI) ▸ unlike MRI it can provide a direct relationship between the CT attenuation value and tissue contrast material concentration
T1WI: low SI ▸ T2WI/FLAIR: high SI
FLAIR: this provides particularly good contrast between normal brain tissue and glial tumours ▸ signal loss is seen within any cystic tumour components
Highly cellular tumours (e.g. lymphoma): a corresponding decreased water content (relatively low SI on T2WI)
Vascular extra-axial tumours (e.g. meningioma)
Following disruption of the blood–brain barrier (generally high-grade tumours) ▸ it may also be seen with certain low-grade tumours (e.g. pilocytic astrocytomas)
Haemorrhage/calcification becomes more conspicuous
T2WI: low SI ▸ this is more conspicuous on T2*WI (stronger magnetic susceptibility effects)
If there is haemorrhage, proteinaceous fluid, melanin (e.g. metastatic melanomas) or fat
This can assess tumour blood vessel density (an indirect measure of tumour neovascularity malignancy)
rCBV measurements correlate closely with markers of tumour vascularity and angiogenesis
indirect measure of tumour neovascularity
Higher rCBV values with high-grade tumours
rCBV maps can aid stereotactic tumour biopsies
In radiation necrosis the residual enhancing lesion has a low rCBV (higher with tumour recurrence due to new vessel formation)
DSC imaging differs from contrast enhancement, which is an indicator of vascular endothelial (blood–brain barrier) integrity
T1WI + Gad ▸ DCE imaging can generate time–signal intensity curves, and analysed with mathematical models (e.g. K TRANS )
This quantifies tumour microvascular permeability and correlates with tumour grade
Measured using T1W steady-state or first-pass T2*W gradient-echo imaging
Labelled endogenous hydrogen measures cerebral blood flow (rCBF)
Useful in identifying acute infarcts or abscesses (which can mimic brain tumours)
ADC measurements correlate inversely with the histological glioma cell count
ADC measurements of any enhancing components in radiation necrosis are significantly higher than with recurrent tumour (mirroring the higher cellular density with a recurrent neoplasm)
Diffusion tensor imaging (DTI) provides additional information about the direction of water diffusion ▸ the normally high anisotropy within white matter tracts can be lost if infiltrated by tumour
MRS is a sensitive but not specific technique
The common pattern seen with brain tumours:
Decreased:
N-acetylaspartate (NAA): a neuron-specific marker
Creatine (Cr)
Increased:
Lipids (L)
Lactate (Lac): a marker of tumour tissue hypoxia
Choline (Cho): a reflection of cell membrane turnover (increased with neoplastic activity)
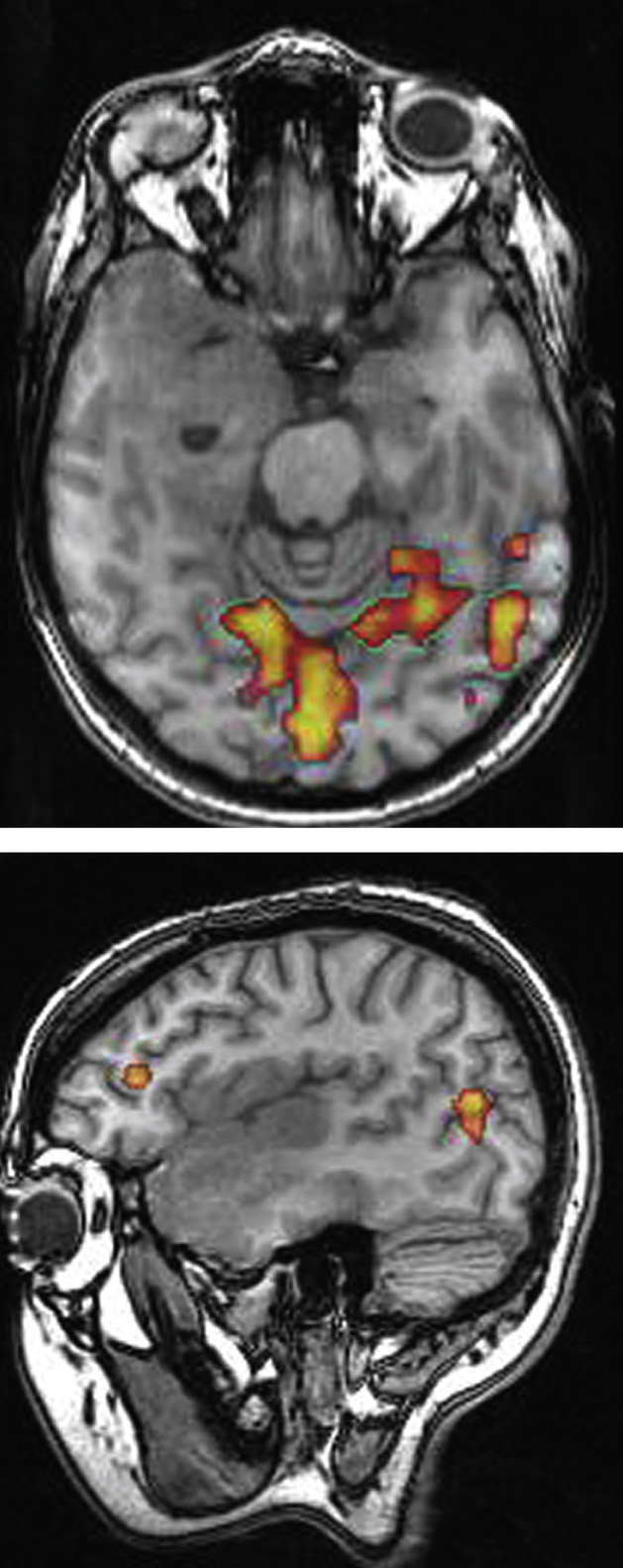
BOLD imaging detects changes in regional cerebral blood flow during various forms of brain activity
This is used for preoperative localization of important cortical regions that may have been displaced by tumour
Tumours arising from the brain parenchyma
Glioma: a broad category including tumours arising from either astrocytes (astrocytoma), oligodendrocytes (oligodendroglioma) or ependymal cells (ependymoma)
Tumours arising from the tissues covering the brain (e.g. the dura or arachnoid) ▸ these occur much more frequently in adults than children (accounting for the majority of the primary infratentorial adult tumours)
Tissues of origin:
Meningioma: meningothelial arachnoidal cells
Haemangiopericytoma: mesenchymal pericytes
Schwannomas and neurofibromas: cranial nerves
Epidermoid and dermoid cysts: developmental cysts or tumour-like lesions
Choroid plexus papillomas: choroid plexus cells
Intracranial tumours are classified according to the WHO classification
Children: primary tumours usually occur infratentorially and within the posterior fossa between the ages of 2 and 10 years (e.g. pilocytic astrocytoma, pontine glioma, ependymoma and medulloblastoma) ▸ below 2 and above 10 years of age supratentorial tumours are more common (paediatric supratentorial tumours will preferentially affect the midline structures) ▸ intracranial metastases are rare
Astrocytoma: this is the most common primary childhood brain tumour (the majority are pilocytic astrocytomas and characteristically occur within the cerebellum, hypothalamus and optic nerves)
Adults: 70% of intracranial tumours are primary (30% are metastases) ▸ the vast majority of tumours are supratentorial – the posterior fossa is rarely affected by a primary tumour (a metastasis is more likely at this location)
Late complication of radiotherapy or gamma knife surgery
Can present as an enhancing mass lesion (difficult to distinguish from recurrent tumour)
Radiation necrosis:
Enhancing area has low FDG uptake/low rCBV
Dynamic contrast enhancement: lower max enhancement slope than recurrence
ADC measurements: enhancing components have higher values than recurrence (lower relative cellular density)
| Extra-axial tumour | Intra-axial tumour | |
|---|---|---|
| ‘Buckling’ and medial displacement of the grey–white matter interface | Yes | No |
| CSF cleft separating the base of the mass from adjacent brain | Yes | No |
| Broad base along a dural or calvarial surface | Yes | No |
| Associated bone changes |
|
Rare |
| Grey–white matter junction | Preserved | Destroyed |
| Tumour | Typical site |
|---|---|
| Colloid cyst | Foramen of Monro/third ventricle |
| Meningioma | Trigone of lateral ventricle |
| Choroid | Fourth ventricle |
| Ependymoma | Lateral ventricle (more common in children) and fourth ventricle |
| Neurocytoma | Lateral ventricles (involving septum pellucidum) |
| Metastases | Lateral ventricles, ependyma and choroid plexus |
| Tumour | Age group |
|---|---|
| Brainstem glioma, optic nerve glioma | 0–5 |
| Medulloblastoma, cerebellar astrocytoma, papilloma choroid plexus, pinealoma, craniopharyngioma | 5–15 |
| Ependymoma | 15–30 |
| Glioma, meningioma, acoustic neuroma, pituitary tumour, hemangioblastoma | 30–65 |
| Meningioma, acoustic tumour, glioblastoma | 65+ |
TUMOURS OF NEUROEPITHELIAL TISSUE
Astrocytic tumours
Anaplastic astrocytoma
Diffuse astrocytoma
Glioblastoma
Gliomatosis cerebri
Pilocytic astrocytoma
Pleomorphic xanthoastrocytoma
Subependymal giant cell astrocytoma
Oligodendroglial tumours
Oligodendroglioma
Anaplastic oligodendroglioma
Oligoastrocytic tumours
Oligoastrocytoma
Anaplastic oligoastrocytoma
Ependymal tumours
Ependymoma
Subependymoma
Anaplastic ependymoma
Myxopapillary ependymoma
Choroid plexus tumours
Choroid plexus papilloma
Choroid plexus carcinoma
Other neuroepithelial tumours
Astroblastoma
Chordoid glioma of the third ventricle
Angiocentric glioma
Neuronal and mixed neuronal-glial tumours
Ganglioglioma and gangliocytoma
Desmoplastic infantile ganglioglioma
Dysembryoplastic neuroepithelial tumour
Central neurocytoma and extraventricular neurocytic tumours
Tumours of the pineal region
Pineoblastoma
Pineocytoma
Embryonal tumours
Medulloblastoma
CNS primitive neuroectodermal tumour
Atypical teratoid/rhabdoid tumour
TUMOURS OF CRANIAL AND PARASPINAL NERVES
Schwannoma (neurilemoma, neurinoma)
Neurofibroma
Perineurioma
Malignant peripheral nerve sheath tumour (MPNST)
TUMOURS OF THE MENINGES
Tumours of meningothelial cells
Meningioma
Mesenchymal tumours
Primary melanocytic lesions
Other neoplasms related to the meninges
Haemangioblastoma
LYMPHOMAS AND HAEMATOPOIETIC NEOPLASMS
Malignant lymphomas
Plasmacytoma
Granulocytic sarcoma
GERM CELL TUMOURS
Germinoma
Embryonal carcinoma
Yolk sac tumour
Choriocarcinoma
Teratoma
Mixed germ cell tumour
TUMOURS OF THE SELLAR REGION
Craniopharyngioma
Granular cell tumour
Pituicytoma
Spindle cell oncocytoma of the adenohypophysis
METASTATIC TUMOURS
| Tumour | Infarct | |
|---|---|---|
| Grey matter changes | This is usually centred on the cerebral white matter and spares the overlying grey matter | This often simultaneously involves the cerebral cortex and juxtacortical white matter |
| Shape | Spherical or ovoid | Wedge or box shaped (with its base towards the brain surface) |
| Distribution | Not confined to a vascular territory | Confined to a vascular territory |
| Contrast enhancement | Gyriform enhancement is rare | Gyriform enhancement can be present |
A benign or malignant tumour arising from an astrocyte
Astrocyte: a structural or supporting cell type within the brain
This is the largest group of primary brain neoplasms (75% of all glial tumours)
Location: supratentorial (50%) ▸ cerebellum (35%) ▸ brainstem (15%)
(the majority will eventually progress to a more malignant type over time):
Grade I (benign pilocytic astrocytoma): non-invasive ▸ occur mainly in young patients ▸ this is potentially resectable with a low proliferative potential (up to 40% of all paediatric intracranial tumours)
It characteristically occurs within the cerebellum in children ▸ it can also occur within the hypothalamus and optic nerves (optic nerve involvement is a feature of NF-1)
Grade II (diffuse astrocytoma): an infiltrating (rather than destroying) low-grade tumour ▸ it results in a relatively mild neurological deficit and a generally good prognosis ▸ typically found in the cerebral hemisphere of young adults
Grade III (anaplastic astrocytoma): although there is increased mitotic activity and anaplasia there is no necrosis
Grade IV (glioblastoma multiforme): this is the commonest primary adult intracranial neoplasm ▸ 90% arise de novo ▸ 10% from lower grade astrocytoma transformation ▸ it is very malignant (with the worst prognosis) ▸ tumour necrosis is a hallmark
It occurs de novo or from a pre-existing lower-grade astrocytoma
Cerebellar pilocytic astrocytoma: this occurs equally within the vermis and cerebellar hemispheres and commonly presents with the effects of hydrocephalus ▸ can be mistaken for haemangioblastoma in adults
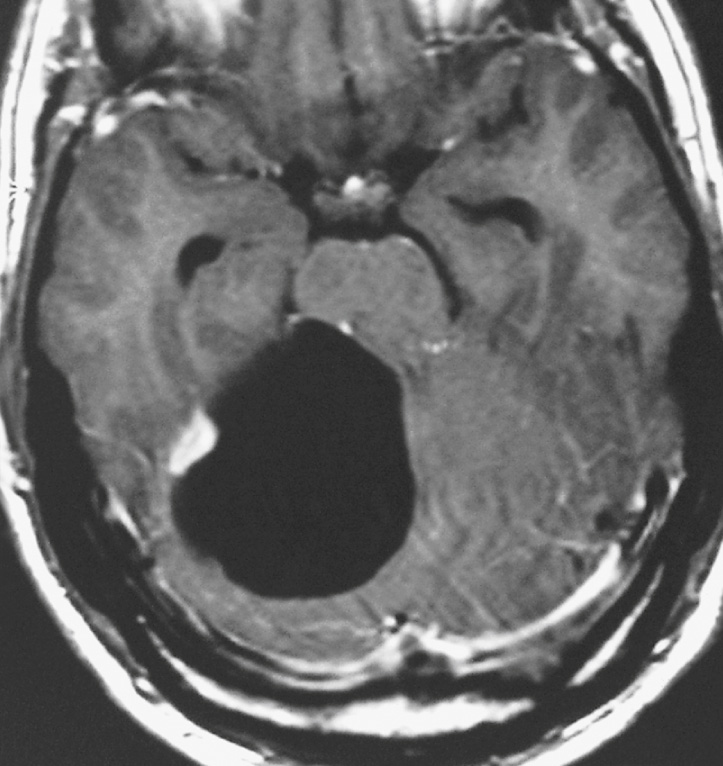
A well-circumscribed and encapsulated large mass ▸ predominantly cystic (70%) or solid (30%) ▸ an associated strongly enhancing mural nodule when cystic ▸ calcification is rare ▸ no adjacent oedema
Differential: a medulloblastoma is a hyperdense solid lesion (NECT)
Cystic component: T1WI: low SI ▸ T2WI: high SI ▸ T1WI + Gad: avid homogeneous enhancement of any solid component
Optic pathway pilocytic astrocytoma: occurs anywhere along the optic tract (usually at the chiasm) ▸ hypothalamic or chiasmatic tumours may be more aggressive
An enlarged optic nerve (variable enhancement) ▸ often large and lobulated when at the chiasm and can extend into the hypothalamus ▸ haemorrhage and necrosis is uncommon
No calcification (unlike an optic nerve sheath meningioma or craniopharyngioma)
An expanded chiasm and hypothalamus ▸ T1WI: low SI ▸ T2WI: high SI
This is less well defined than a pilocytic astrocytoma and with variable mass effect
An iso- or hypodense mass ▸ poor enhancement (there is an intact blood–brain barrier) ▸ calcification in 20%
T1WI: low-to-intermediate SI ▸ T2WI/FLAIR: high SI ▸ T1WI + Gad: enhancement suggests progression to a higher histological grade
More extensive infiltration of peritumoural tissues than with a grade II tumour (+ vasogenic oedema) ▸ ![]() may be non-enhancing
may be non-enhancing
Contrast enhancement and vasogenic oedema are much more extensive than with an anaplastic astrocytoma ▸ although tumours may appear well-defined they are always infiltrative (commonly extending along the white matter tracts)
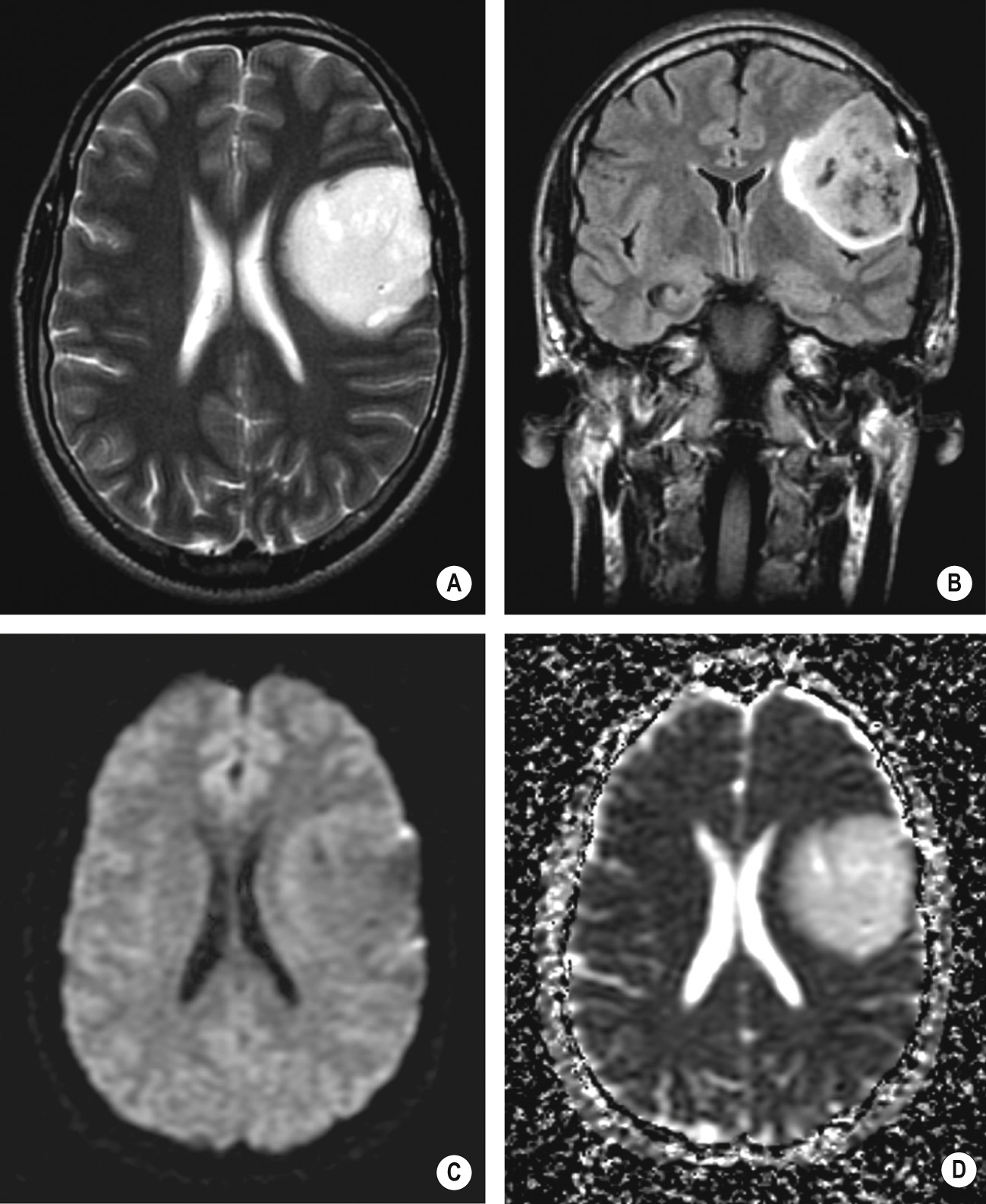
Solid ± central necrosis ± oedema ▸ T1WI/T2WI: heterogeneous SI appearances due to necrosis and haemorrhage ▸ T1WI + Gad: an irregularly thick enhancing peripheral ‘ring’ (active mitosis) ▸ a multicentric tumour with seeding via the CSF space (5%) ▸ a lower ADC than with a low-grade glioma
‘Butterfly lesion’: tumour commonly crosses the midline via the corpus callosum (as can a CNS lymphoma)
Pseudoprogression: due to an inflammatory reaction following chemoradiation (increased enhancement and oedema) ▸ spontaneous improvement
Pseudoresponse: decreased enhancement and oedema without improved survival
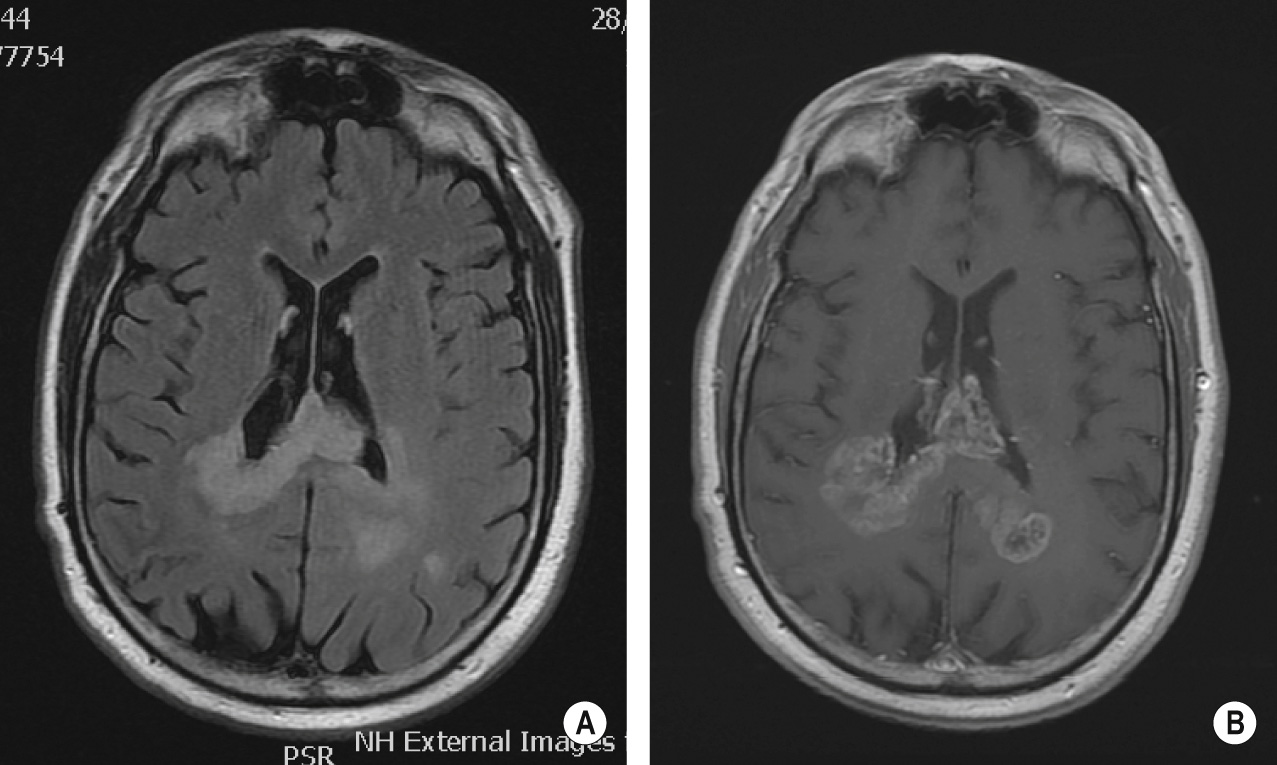
Diffuse infiltration of large areas of brain or spinal cord tissue by glial tumour cells with preservation of the underlying architecture (no definitive mass) ▸ it typically involves the hemispheric white matter ▸ it presents between the 2 nd and 4 th decades (M = F)
A diffuse ill-defined ‘mass-like’ lesion with ventricular effacement ▸ T1WI: a homogeneous intermediate-to-low SI infiltrating mass ▸ T2WI/FLAIR: a homogeneous high SI infiltrating mass ▸ T1WI + Gad: no or minimal enhancement
Differential: lymphomatosis cerebri ▸ viral encephalitis ▸ acute disseminated encephalomyelitis (ADEM) ▸ vasculitis
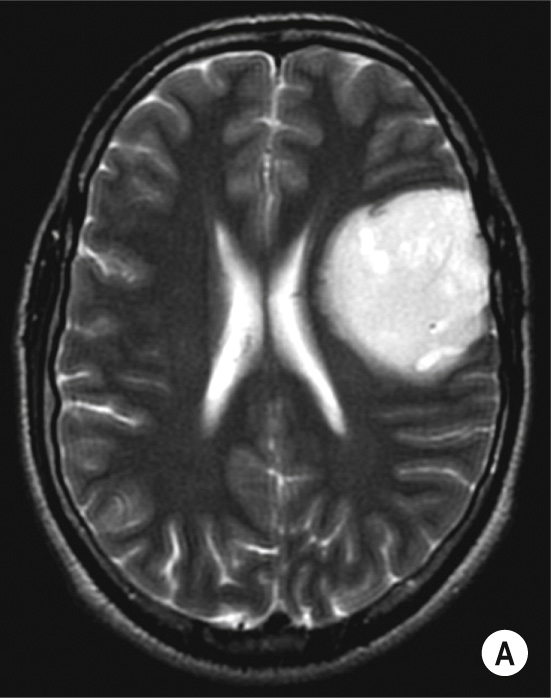
| Pilocytic astrocytoma | Diffuse astrocytoma | Anaplastic astrocytoma | Glioblastoma multiforme | |
|---|---|---|---|---|
| Malignant potential | Benign | Low grade | High grade | Very malignant |
| Age (approximate) | Children | 3 rd or 4 th decade | 5 th decade | 6 th decade |
| Location | Optic chiasm or hypothalamus > cerebellum > brainstem * | Hemispheres (cortex + white matter) | Hemispheres (cortex + white matter) | Hemispheres (cortex + white matter) |
| Enhancement | Mild | Mild | Moderate (ring) | Intense |
| Vasogenic oedema | Minimal | Minimal | Moderate | Significant |
| Calcification | Common | Up to 20% | Occasional | Rare |
* It is typically cystic with a mural nodule and located within the posterior fossa – it tends to be solid or lobulated when seen elsewhere.
A relatively benign slow-growing neoplasm arising from the oligodendrocyte
Oligodendrocyte: a cell that insulates the central nervous system axons and which is equivalent to a Schwann cell within the peripheral nervous system
It is classified as a WHO grade II (well-differentiated, low-grade) or WHO grade III (anaplastic high-grade) tumour ▸ it is chemosensitive
It occurs predominantly in adults (during the 4 th decade) and accounts for 5–10% of all intracranial neoplasms
It is a diffusely infiltrating neoplasm found almost exclusively within the cerebral hemispheres and typically involving the subcortical white matter and cortex (85% are seen within the frontal lobes)
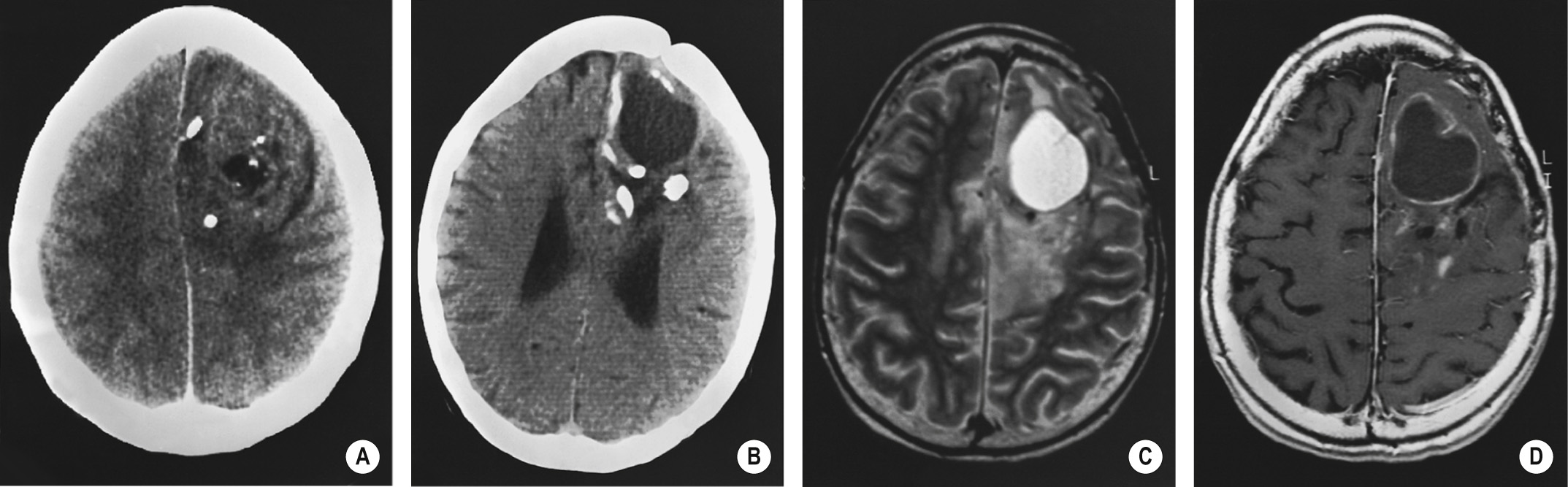
It is well circumscribed, unencapsulated and less infiltrative than a diffuse astrocytoma ▸ it may erode the calvarium
A hypodense lesion which may involve the cortex (with associated cortical thickening) ▸ cysts or haemorrhage can be seen in 20% but necrosis and oedema is rare
50% of tumours will demonstrate variable (and often heterogeneous) contrast enhancement – this is not a reliable indicator of tumour grade (unlike for an astrocytoma)
Intratumoral haemorrhage
Calcification is present in up to 90% of cases – this is central, peripheral or gyriform in nature
T1WI: heterogeneous low-to-intermediate SI ▸ T2WI/FLAIR: heterogeneous high SI ▸ T1WI + Gad: variable and often heterogeneous
A low-grade tumour arising from the ependyma ▸ usually intraventricular – extraventricular rests of ependymal cells may give rise to hemisphere tumours
Ependyma: this forms the epithelial lining of the ventricular system, cerebral hemispheres, brainstem and cerebellum, central canal of the spinal cord and tip of the filum terminale
It accounts for 5% of all intracranial tumours (a higher incidence is seen in the paediatric population)
65% are infratentorial (most commonly arising from the floor of the 4 th ventricle) ▸ 25% are supratentorial (arising from white matter ependymal cells) ▸ 10% arise within the spinal cord
Supratentorial tumours: these are commonly extraventricular (involving the periventricular white matter) ▸ they predominantly affect young adults
Infratentorial tumours: these are commonly intraventricular (affecting the 4 th ventricle) ▸ there are two age peaks at 5 and 35 years of age
Uncommonly disseminates by leptomeningeal spread
An isodense-to-hyperdense, well-demarcated, lobulated mass lesion which takes on the shape of the 4 th ventricle (originating from the roof or floor) and frequently extends through the foramina of Magendie and Luschka to seed via the subarachnoid space (a ‘plastic’ ependymoma) ▸ calcification is seen in >50% of cases and cystic elements can also be demonstrated ▸ there can be an associated obstructive hydrocephalus
Cerebral hemisphere ependymoma: this tends to arise adjacent to the ventricular system (characteristically adjacent to the trigone of the lateral ventricle) and can resemble an astrocytoma ▸ it is more frequently calcified or cystic than an infratentorial tumour
There are mixed signal intensities ▸ T1WI: normal-to-low SI ▸ T2WI: predominantly high SI ▸ T1WI + Gad: mild-to-moderate enhancement (which is often heterogeneous)
Surgical resection (although the tendency of posterior fossa tumours to infiltrate around the cranial nerves makes total resection difficult with associated high recurrence rates)
A variant containing both ependymal and astrocyte cells ▸ it occurs mainly in elderly males and presents as an intraventricular mass in the lateral or 4 th ventricle ▸ it is relatively benign and does not disseminate
An important differential of a posterior fossa ependymoma
It calcifies less frequently ▸ it arises from the roof of the 4 th ventricle ▸ it demonstrates a rounded shape compared with an ependymoma (that moulds to the ventricular margins)
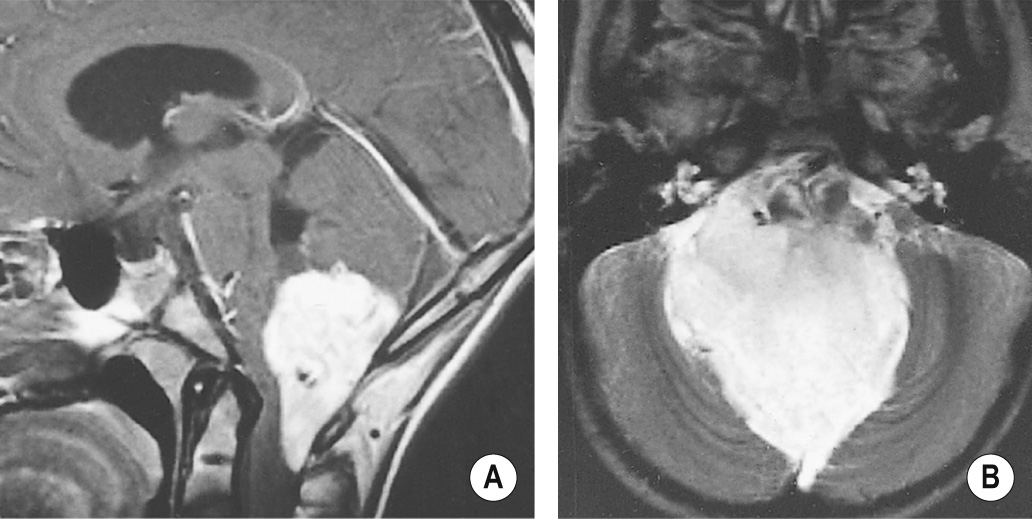
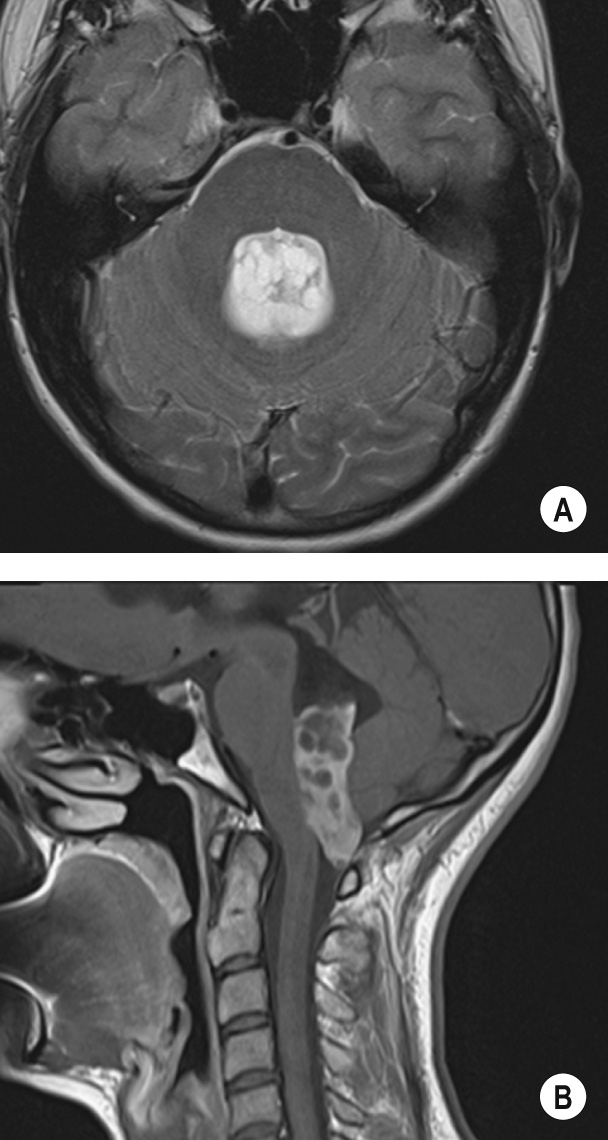
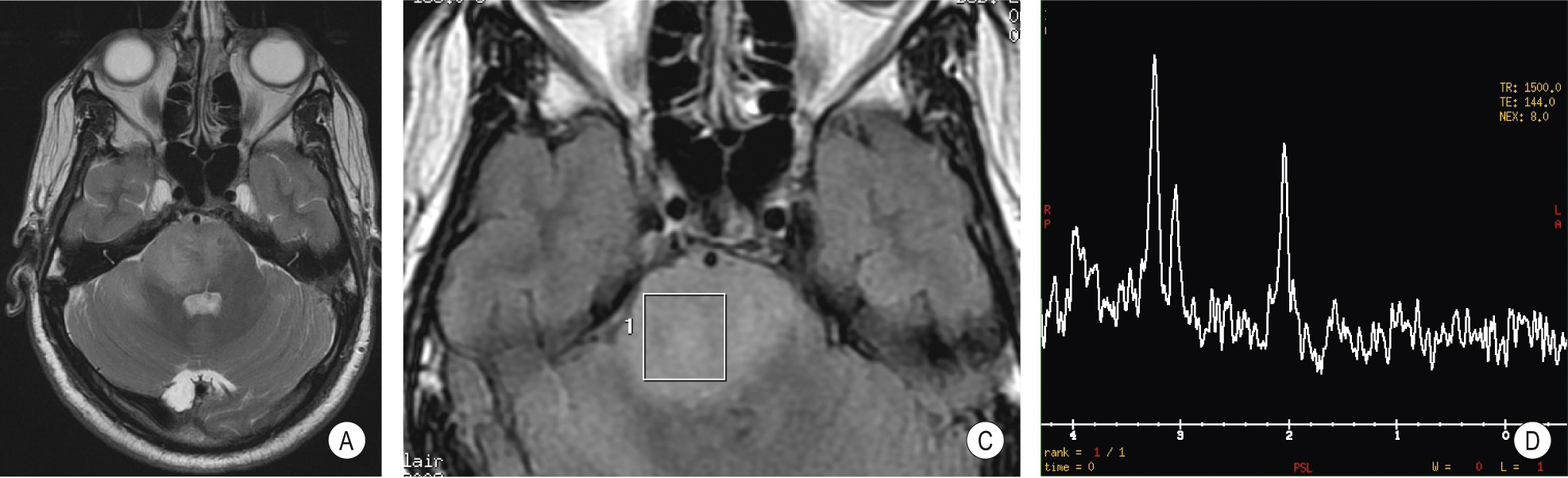
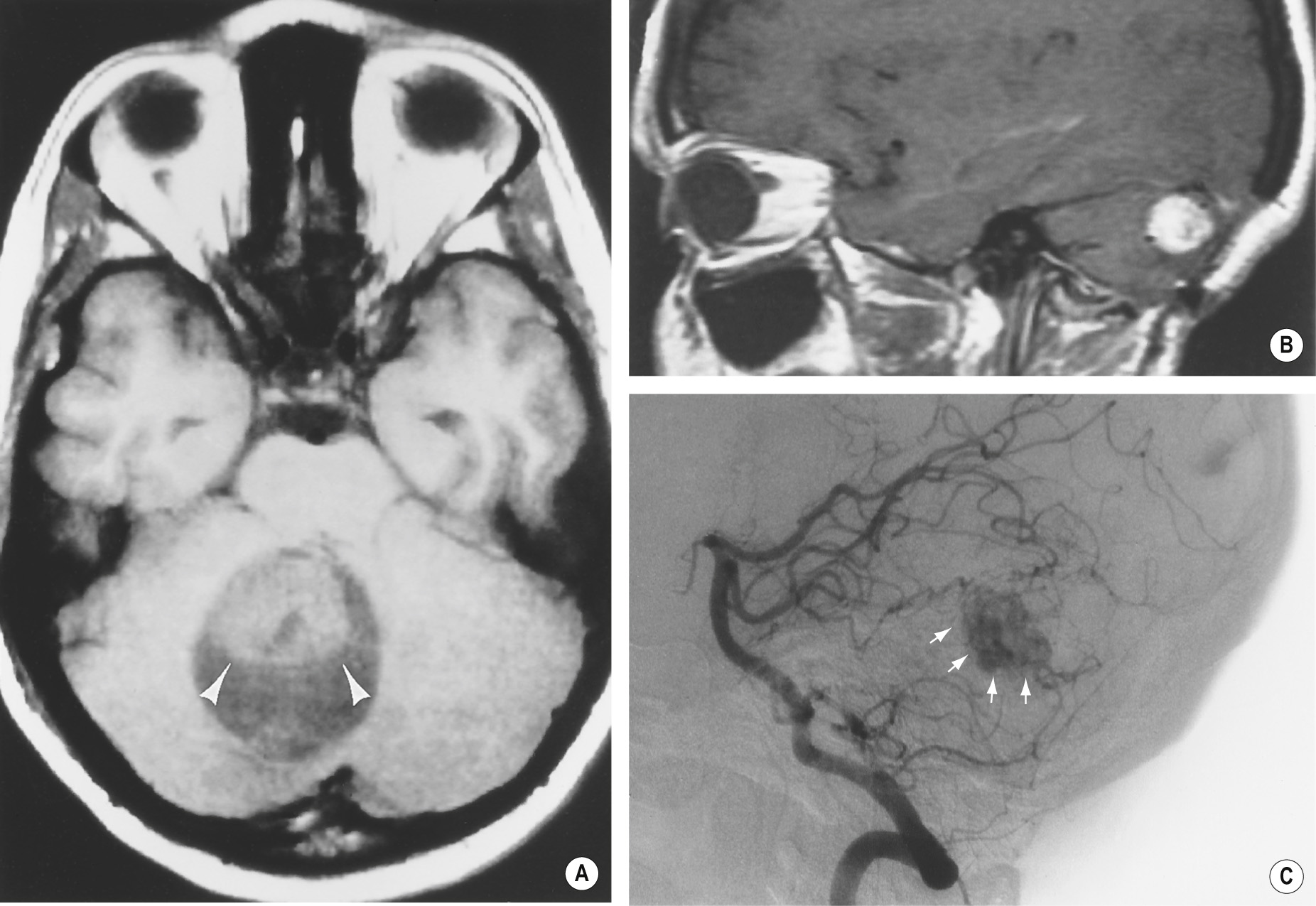
A benign tumour of endothelial origin that is composed of thin-walled blood vessels ▸ it is predominantly found within the posterior fossa (supratentorial lesions are rare) and is the commonest primary intra-axial and infratentorial adult tumour
10% of adult infratentorial masses are haemangioblastomas
It usually presents in young adults (M>F)
Common symptoms include headache, ataxia, nausea, vomiting and vertigo
20% are associated with von Hippel–Lindau (VHL) disease – these generally present at an earlier age
Multiple haemangioblastomas are only seen with von Hippel–Lindau disease ▸ it is an unusual paediatric tumour unless in the context of von Hippel–Lindau disease
It usually appears as a cystic mass with an intensely enhancing mural nodule (± haemorrhage) ▸ there is little surrounding oedema ▸ cyst wall enhancement indicates tumour extension (as for a pilocytic astrocytoma)
It may only consist of strongly enhancing solid components
Multiple signal voids may be seen with MRI (as the lesion is highly vascular)
A vascular nodule within an avascular mass ▸ there may be draining veins present
| Haemangioblastoma | Juvenile pilocytic astrocytoma | |
|---|---|---|
| Age | 30–40 years | 5–15 years |
| Pial attachment | Yes | No |
| A tiny nodule with a huge cystic component | More likely | Less likely |
| Arteriogram | Hypervascular nodule | Hypovascular nodule |
| Multiplicity and association with VHL disease | More likely | Less likely |
This accounts for up to 30% of all paediatric infratentorial tumours (they may occur in adults) ▸ 80% of tumours are high grade, but symptoms occur late as the tumour infiltrates rather than destroys adjacent tissues (hydrocephalus is a late feature)
Pons > midbrain > medulla
This is the most common pontine lesion and has a poor prognosis
It is an expansile and poorly defined pontine lesion (± haemorrhage) ▸ there is poor enhancement ▸ it can encase the basilar artery
This is more common than diffuse disease within the midbrain and medulla
This has similar imaging features to a pilocytic astrocytoma seen elsewhere
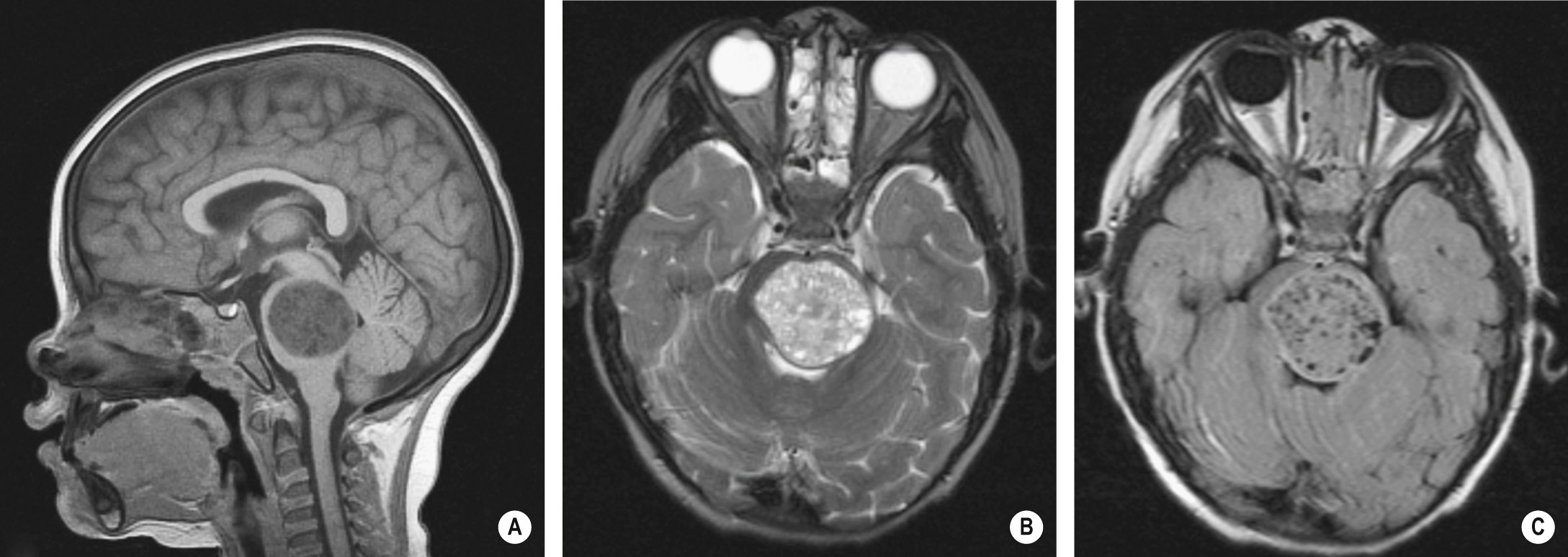
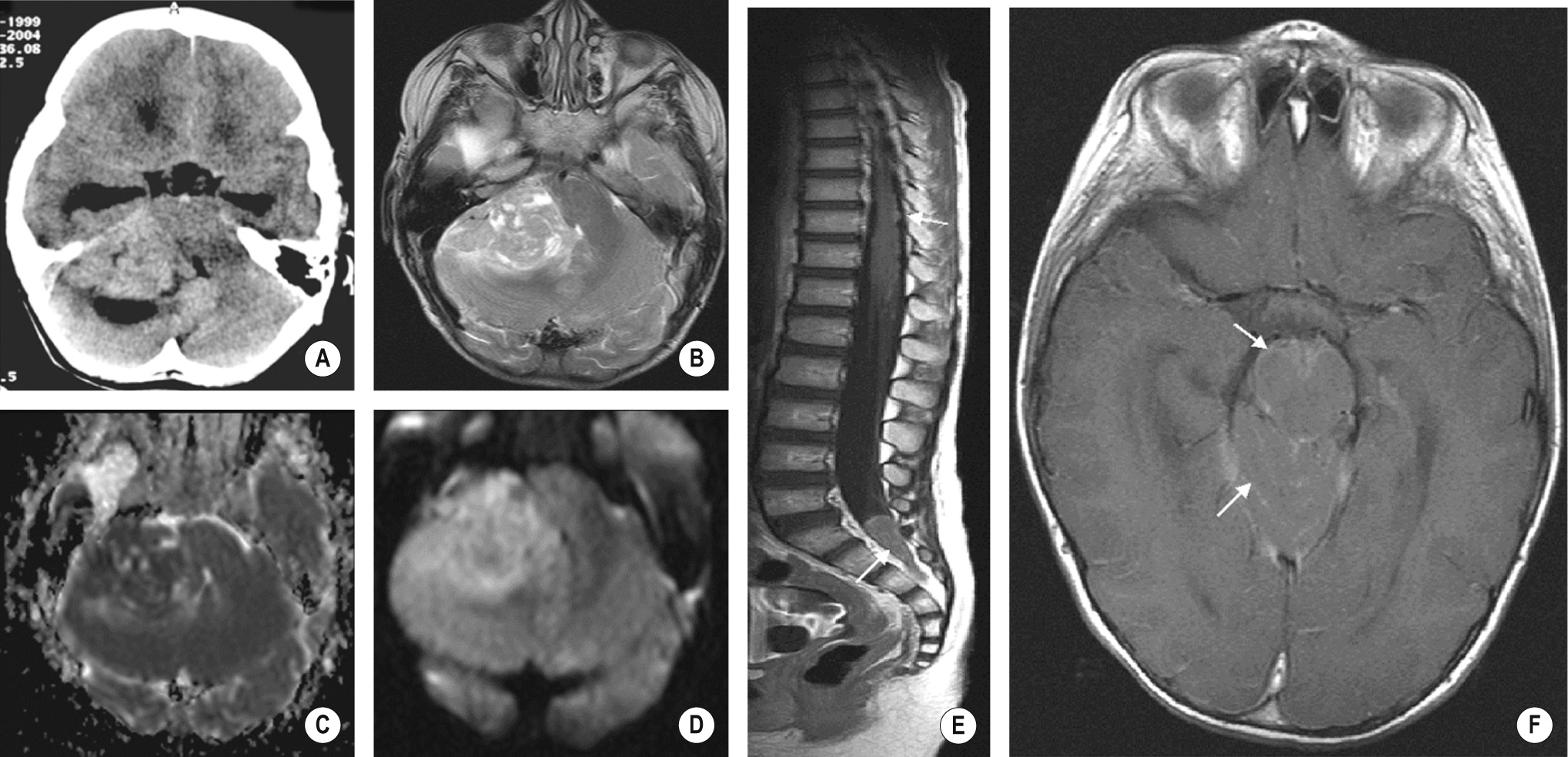
This is an aggressive tumour, accounting for 30-40% of all posterior fossa tumours ▸ it is also known as the PNET of the posterior fossa
It classically arises from the roof of the 4 th ventricle and is therefore usually a midline cerebellar mass (a lateral cerebellar location is more common in older children and adults) ▸ subsequent hydrocephalus is common ▸ occasionally intracranial or intraspinal leptomeningeal disease at presentation
There is a peak age of presentation at 7 years (M>F) ▸ a 2 nd peak is seen in young adults who present with a ‘desmoplastic’ and less aggressive form
A well-defined and hyperdense (due to its high cellular density) midline vermian mass abutting the roof of the 4 th ventricle ▸ there is perilesional oedema (± hydrocephalus) ▸ cystic change, haemorrhage and calcification are frequently seen ▸ brainstem usually displaced anteriorly rather than directly invaded
T2WI: intermediate-to-low SI ▸ T1WI + Gad: variable patchy enhancement ▸ DWI: restricted diffusion
There is a reduced N-acetylaspartate (NAA) peak with an increased choline-to-creatine ratio
Intracranial and intraspinal subarachnoid dissemination is seen in ![]() of patients at presentation ▸ this can appear as:
of patients at presentation ▸ this can appear as:
Irregular, and nodular leptomeningeal enhancement
A communicating hydrocephalus
Nodularity and clumping of the nerve roots
Pial ‘drop’ metastases along the spinal cord surface
Li–Fraumeni, Gorlin's, basal cell naevus, Turcot and Cowden syndromes
Surgical resection + adjuvant radiotherapy (only for those patients who are >3 years old due to the susceptibility of the infant brain)
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here