Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Infection is the greatest killer in human history. The microbial theory of disease is probably the greatest advance in medicine, for it has made possible the identification of the cause of many diseases, the development of diagnostic modalities (i.e., cultivation of microorganisms), treatment with antimicrobial agents, and the development of vaccines. The achievements of the microbial theory of disease in medicine and public health are unparalleled with regard to any other contribution in medicine. The formulation of the Koch postulates to establish causality between a microbe and a disease provided a major breakthrough that has influenced the philosophy of science. Although during the 20th century some considered that infectious diseases had been conquered, the pandemic of coronavirus disease 2019 (COVID-19), epidemics of acquired immunodeficiency syndrome (AIDS), Ebola virus, swine flu, and the like tell a different story. The recent understanding that infections during pregnancy may predispose to schizophrenia, autism spectrum disorders, and cerebral palsy , in the offspring suggests that insults occurring during intrauterine life (infection-induced inflammation) can have lasting effects.
Fetal and neonatal physicians and scientists have been interested in intra-amniotic infection as a cause for preterm labor and delivery, neonatal morbidity and mortality, and long-term disorders. This chapter will focus largely on the microbiology, the innate immune response, and the fetal inflammatory response syndrome (FIRS).
Four major transitions occur at birth: (1) emergence from an aquatic environment where oxygen is acquired through the placenta to a dry environment in which respiratory exchange occurs through the lungs; (2) adaptation from a warm environment in which the fetus has a temperature that is 1°C higher on average than that of the mother to a cooler environment at room temperature; (3) exchange of a continuous supply of nutrients through the placenta for intermittent feedings during the neonatal period; and (4) transition from a sterile bacterial environment to the establishment of the neonatal microbiome (e.g., gut, respiratory tract, skin).
The fetus normally lives in an environment devoid of bacteria, as determined by cultivation and molecular microbiologic techniques of amniotic fluid. , Therefore, birth represents a critical stage for acquisition of the first microbiota. During labor, the fetus becomes exposed to the vaginal microbiota and such bacteria become the pioneer microorganisms that invade the formerly sterile body of the infant to establish the first neonatal microbiota. , Such microbiota may differ when an infant is born via vaginal or cesarean delivery.
Under pathologic conditions, however, microbial invasion of the amniotic cavity due to bacteria, fungi, and viruses can predispose to fetal infection. Microorganisms can enter the fetus through different sites, including the mucous membranes of the airways, gastrointestinal tract, tympanic membrane, the conjunctiva, or skin. Once bacteria are in contact with the mucous membranes, they can gain access to the fetal circulation and cause bacteremia, which then can lead to sepsis, septic shock, or even death. The fetal immune system, after the second trimester of pregnancy, is capable of mounting both an innate and an adaptive immune response, which play a key role in host defense against infection. Indeed, recent studies show that the amniotic fluid harbors innate and adaptive immune cells of fetal origin, which play a central role in host defense against microbes invading the amniotic cavity.
Microbial invasion of the amniotic cavity has been causally linked to preterm labor with intact or ruptured membranes. One-third of preterm neonates are born to mothers with microbial invasion of the amniotic cavity. , , Moreover, the earlier the gestational age at delivery, the greater the frequency of intra-amniotic infection and the greater the burden of disease. ,
Table 12.1 and Fig. 12.1 define terms often used to refer to entities related to intra-amniotic infection. Microbial invasion of the amniotic cavity is defined as the presence of organisms in the samples of amniotic fluid retrieved by transabdominal amniocentesis and detected by cultivation methods and/or molecular microbiologic techniques. Intra-amniotic inflammation is the presence of an inflammatory response in the amniotic cavity, which can be detected by the presence of inflammatory cells, for instance, white blood cells, in an elevated concentration of an inflammatory mediator, such as interleukin (IL)-6, 36 or in an elevated concentration of matrix metalloproteinase (MMP). Rapid tests that can determine these biomarkers have become available recently as point-of-care tests, and results can be obtained in less than 20 minutes without specialized laboratory equipment.
| Condition | Definition |
|---|---|
| Microbial invasion of the amniotic cavity | The presence of organisms in the amniotic fluid retrieved by transabdominal amniocentesis and detected by cultivation methods and/or molecular microbiologic techniques |
| Intra-amniotic inflammation | An increased number of inflammatory cells (white blood cells) or cytokines or matrix metalloproteinase 8 (MMP-8) in amniotic fluid
|
| Intra-amniotic infection | The combination of microbial invasion of the amniotic cavity and intra-amniotic inflammation |
| Sterile intra-amniotic inflammation | The presence of intra-amniotic inflammation with the absence of microorganisms detected by both cultivation methods and molecular microbiologic techniques |
| Clinical chorioamnionitis | Maternal fever (temperature ≥37.8°C or ≥38.0°C) and the combination of at least two of the following clinical signs: (1) maternal tachycardia (heart rate >100 beats/min); (2) fetal tachycardia (heart rate >160 beats/min); (3) uterine tenderness; (4) purulent or foul-smelling amniotic fluid or vaginal discharge; and (5) maternal leukocytosis (white blood cells count >15,000/mm 3 ) |
| Acute histologic chorioamnionitis | The presence of neutrophils in the chorioamniotic membranes or the chorionic plate. This condition represents a maternal inflammatory response |
| Acute funisitis | Inflammation of the umbilical cord (umbilical vein, umbilical artery, and Wharton’s jelly). This condition represents a fetal, not a maternal inflammatory response |
![Fig. 12.1, Microbial invasion of the amniotic cavity, intra-amniotic inflammation, sterile intra-amniotic inflammation, and intra-amniotic infection. Microbial invasion of the amniotic cavity is characterized by the presence of microorganisms in the amniotic fluid obtained by transabdominal amniocentesis and detected by cultivation methods and/or molecular microbiologic techniques. Intra-amniotic inflammation is defined by the presence of inflammatory cells (white blood cell count greater than or equal to 50 cells/mm 3 ) or an elevated concentration of a biomarker of inflammation (an interleukin-6 [IL-6] concentration ≥2.6 ng/mL or a matrix metalloproteinase-8 [MMP-8] concentration >23 ng/mL). Sterile intra-amniotic inflammation is defined as the presence of intra-amniotic inflammation in the absence of microorganisms. When intra-amniotic inflammation is accompanied by the presence of microorganisms in the amniotic cavity, this condition is referred to as intra-amniotic infection. Fig. 12.1, Microbial invasion of the amniotic cavity, intra-amniotic inflammation, sterile intra-amniotic inflammation, and intra-amniotic infection. Microbial invasion of the amniotic cavity is characterized by the presence of microorganisms in the amniotic fluid obtained by transabdominal amniocentesis and detected by cultivation methods and/or molecular microbiologic techniques. Intra-amniotic inflammation is defined by the presence of inflammatory cells (white blood cell count greater than or equal to 50 cells/mm 3 ) or an elevated concentration of a biomarker of inflammation (an interleukin-6 [IL-6] concentration ≥2.6 ng/mL or a matrix metalloproteinase-8 [MMP-8] concentration >23 ng/mL). Sterile intra-amniotic inflammation is defined as the presence of intra-amniotic inflammation in the absence of microorganisms. When intra-amniotic inflammation is accompanied by the presence of microorganisms in the amniotic cavity, this condition is referred to as intra-amniotic infection.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/IntraAmnioticInfectionInflammationandtheFetalInflammatoryResponseSyndrome/0_3s20B9780323712842000124.jpg)
The term intra-amniotic infection is used when there is a combination of microbial invasion of the amniotic cavity and intra-amniotic inflammation. Most cases of microbial invasion of the amniotic cavity without evidence of inflammation are due to specimen contamination by skin flora, laboratory bacteria, or reagents. When intra-amniotic inflammation is present in the absence of microorganisms, the condition is referred to as sterile intra-amniotic inflammation. , This term should be reserved for samples that are negative for microorganisms detected by both cultivation methods and molecular microbiologic techniques. , Sterile intra-amniotic inflammation has been attributed to “danger signals” (or alarmins ) that are released during cellular stress or cell necrosis.
Clinical chorioamnionitis refers to a syndrome characterized by the presence of a maternal fever (temperature ≥37.8°C or ≥38.0°C) and two or more of the five following clinical signs: (1) maternal tachycardia (heart rate >100 beats/min); (2) fetal tachycardia (heart rate >160 beats/min); (3) uterine tenderness; (4) purulent or malodorous amniotic fluid or vaginal discharge; and (5) maternal leukocytosis (white blood cell count >15,000/mm 3 ).
Clinical chorioamnionitis represents the clinical manifestation of a maternal systemic inflammatory response and is present in only 10% to 20% of all patients with proven intra-amniotic infection. Acute histologic chorioamnionitis is the presence of neutrophils in the chorioamniotic membranes or the chorionic plate and represents a maternal host response. Funisitis is inflammation of the umbilical cord (umbilical vein, artery, and Wharton’s jelly) and is a fetal host response. , Chorionic vasculitis consists of inflammation of the fetal vessels on the surface of the chorionic plate and is evidence of fetal inflammation. Acute histologic chorioamnionitis, more common than clinical chorioamnionitis, is the pathologic expression of intra-amniotic inflammation. , , However, acute histologic chorioamnionitis should not be considered synonymous with intra-amniotic infection, since a fraction of patients with sterile intra-amniotic inflammation have acute histologic chorioamnionitis and, sometimes, funisitis. The term acute refers to a specific inflammatory lesion in which the predominant cell is the neutrophil. Chronic inflammatory lesions of the placenta are characterized by the infiltration of lymphocytes, plasma cells, and macrophages. Although chronic inflammatory lesions could be due to specific infectious agents (such as viruses), the main cause of chronic inflammatory lesions is maternal antifetal rejection, and interested readers are referred to a recent review by the authors on this subject.
Microorganisms may gain access to the amniotic cavity through three main pathways : (1) ascension from the vagina and cervix , ; (2) hematogenous dissemination through the placenta (transplacental infection) , ; and (3) accidental introduction at the time of invasive procedures, such as amniocentesis, percutaneous fetal blood sampling, chorionic villus sampling, or shunting.
The most common pathway of intra-amniotic infection is the ascending route, , , , evidenced as follows:
The bacteria isolated in the amniotic fluid of patients with intra-amniotic infection are frequently found in the vagina. ,
In twin gestations, acute histologic chorioamnionitis, more common in the first-born twin, is rarely seen in the second twin. Moreover, when intra-amniotic infection is detected, the presenting sac is nearly always involved.
In virtually all cases of congenital pneumonia, inflammation of the chorioamniotic membranes is present. , ,
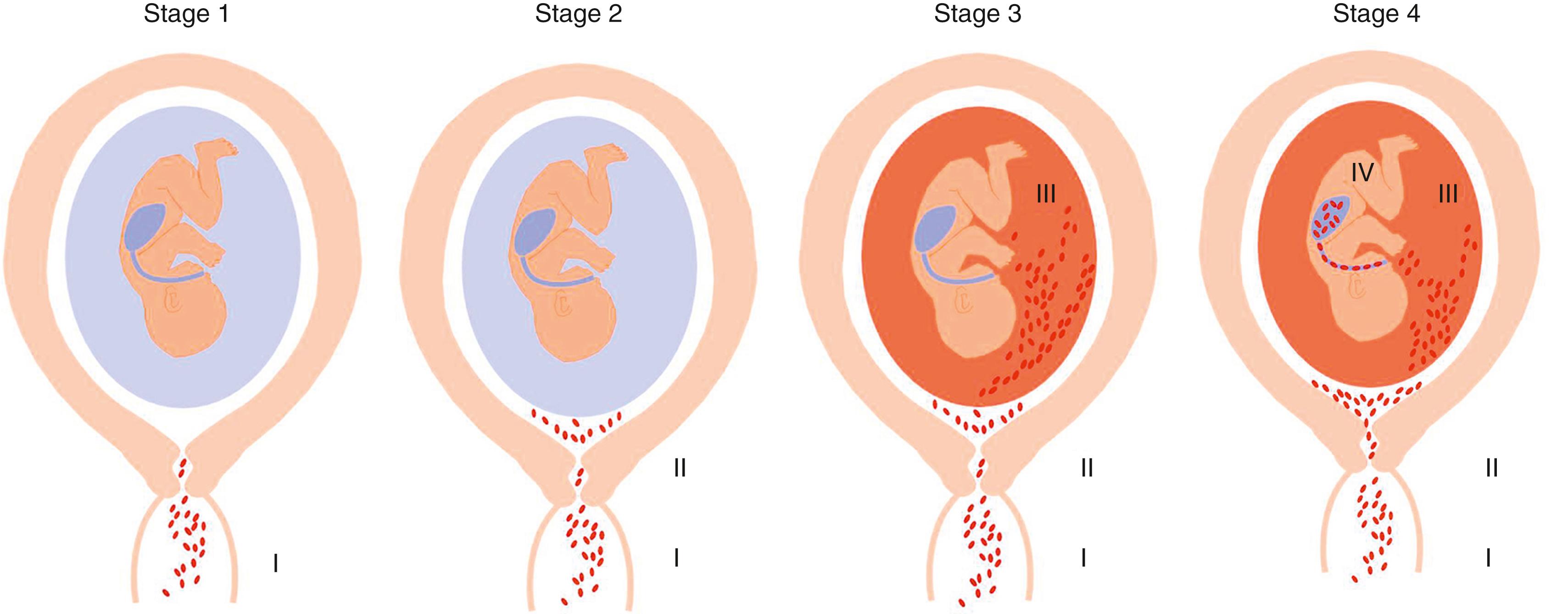
The stages of ascending infection are listed in Fig. 12.2 . Stage 1 corresponds to a change in the vaginal/cervical microbial flora or to the presence of pathologic organisms (e.g., Neisseria gonorrhoeae ) in the cervix. Some forms of bacterial vaginosis may be an early manifestation of this initial stage. An abnormal vaginal microbiota (even in the absence of bacterial vaginosis) can correspond to the first stage and predispose to preterm delivery. The specific changes in the vaginal microbiota have been recently described by using sequence-based techniques. , In stage 2 , microorganisms gain access to the uterine cavity and reside in the lower pole of the uterus between the membranes and chorion, where they can elicit a localized inflammatory reaction. In stage 3, bacteria have penetrated the chorion and the amnion in the lower pole of the uterus into the amniotic cavity, leading to microbial invasion of the amniotic cavity and intra-amniotic infection when an inflammatory response is established in the amniotic fluid. Rupture of the membranes is not a prerequisite for intra-amniotic infection because microorganisms are capable of crossing intact membranes. Finally, in stage 4 , bacteria may gain access to the fetus through different sites. Aspiration of infected fluid by the fetus may lead to congenital pneumonia. Otitis, conjunctivitis, and omphalitis may occur by the direct spreading of microorganisms from infected amniotic fluid. Seeding from any of these sites to the fetal circulation may result in fetal bacteremia and sepsis.
The traditional concept has been that bacteria invade the decidua from the lower genital tract and, from there, are localized to the space between the chorion and amnion before invading the amniotic cavity. , However, molecular data bring into question whether most cases of intra-amniotic infection are preceded by a stage in which the infection is in the decidua or in the chorioamniotic space.
The most common microbial isolates from the amniotic cavity of women with intra-amniotic infection are Ureaplasma spp. ( Ureaplasma parvum and Ureaplasma urealyticum ), Fusobacterium spp., and Mycoplasma hominis. a
a References 29, 31, 53, 102, 121, 128–136.
These microorganisms are frequently found as members of the vaginal microbiome in normal pregnant women. Why these specific microorganisms ascend into the amniotic cavity and not others, which are more frequently present, such as Lactobacilli spp., is unknown. Table 12.2 displays the most frequent microorganisms found in the amniotic fluid of patients with intra-amniotic infection. Most microorganisms are also found in the lower genital tract of normal pregnant women.
| Ureaplasma urealyticum |
| Fusobacterium nucleatum |
| Mycoplasma hominis |
| Streptococcus agalactiae |
| Sneathia species |
| Gardnerella vaginalis |
| Lactobacillus species |
| Staphylococcus aureus |
| Peptostreptococcus species |
| Streptococcus viridans |
| Bacteroides species |
In approximately 50% of patients with intra-amniotic infection, more than one microorganism are isolated from amniotic fluid. The inoculum size differs considerably, and in 71% of cases, more than 10 5 colony-forming units per milliliter are found. Chlamydia trachomatis is rarely found in the amniotic fluid.
Viral invasion of the amniotic cavity has been found in 2.2% of women (16 of 729) undergoing second-trimester amniocentesis for genetic indications. Human herpes virus 6 is the most common organism, followed by human cytomegalovirus, parvovirus B19, and Epstein-Barr virus. Studies of the presence of viral genomes in amniotic fluid that used targeted polymerase chain reaction (PCR) approaches showed that viruses are extremely rare in patients with preterm prelabor rupture of the membranes (PROM). , Characterization of the amniotic fluid virome is a frontier.
Preterm labor and preterm PROM account for approximately two-thirds (one-third each) of preterm deliveries, and the remaining third are the result of indicated delivery because of maternal or fetal indications (i.e., preeclampsia, fetal growth restriction). Microbiologic studies suggest that intra-amniotic infection (stage 3 of ascending intra-amniotic infection) occurs in 25% to 40% of preterm births ; however, this may be an underestimation because some pathogens are difficult to identify by conventional cultivation methods. , Indeed, with the use of molecular microbiologic techniques, bacterial DNA has been identified in culture-negative amniotic fluid from women in preterm labor. , ,
Approximately 12% of patients presenting with an episode of preterm labor and intact membranes have microorganisms in the amniotic fluid detected by cultivation techniques. , , , Women with amniotic fluid cultures positive for microorganisms generally do not have clinical evidence of infection on presentation (i.e., fever and other signs of clinical chorioamnionitis). However, such patients are more likely to develop clinical chorioamnionitis (37.5% vs. 9%), to be refractory to tocolysis (85.3% vs. 16.3%), and to have spontaneous rupture of the membranes (40% vs. 3.8%) than patients with negative amniotic fluid cultures. Moreover, the earlier the gestational age at preterm birth, the more likely that microbial invasion of the amniotic cavity is present, and at 21 to 24 weeks of gestation, most spontaneous births are associated with acute histologic chorioamnionitis, compared to approximately 10% at 35 to 36 weeks. The rate of neonatal complications is higher in infants born to women with intra-amniotic infection than in those born to women without infection. , ,
The rate of amniotic fluid cultures positive for microorganisms at admission is approximately 32.4% in patients with preterm PROM , ; however, clinical chorioamnionitis is present in only 29.7% of patients with proven microbial invasion. , The rate of microbial invasion in preterm PROM reported by these studies is probably an underestimation of the true prevalence of intra-amniotic infection. Indeed, available evidence indicates that the frequency of intra-amniotic infection is higher among women with preterm PROM and a severely reduced volume of amniotic fluid than among those without oligohydramnios. , Because women with oligohydramnios are less likely to undergo an amniocentesis, the bias in these studies is to underestimate the prevalence of infection. In addition, women with preterm PROM admitted in labor generally do not undergo amniocentesis. These patients have a higher rate of microbial invasion of the amniotic cavity than those admitted without labor (39% vs. 25%; p = .049). Moreover, of patients who are not in labor at admission, 75% will have an amniotic fluid culture positive for microorganisms when amniocentesis is performed at the onset of preterm labor. This important observation confirms the traditional clinical view that when a patient with preterm PROM goes into labor, this most likely reflects the presence of intra-amniotic infection. Recent studies, utilizing a combination of cultivation methods and molecular microbiologic techniques, indicate that 50% of patients with preterm PROM have microorganisms detected in the amniotic cavity.
Women presenting with a dilated cervix, intact membranes, and few, if any, contractions before 24 weeks of gestation are considered to have clinical cervical insufficiency. Of such patients, 51.5% have an amniotic fluid culture positive for microorganisms. The most common microorganism is U. urealyticum, which is found in 22.4% of patients with cervical insufficiency. Whether intra-amniotic infection is the cause or a consequence of cervical dilatation or a short cervix has not been determined. Shortening of the cervical canal or silent cervical dilatation with protrusion of the membranes into the vagina may lead to a secondary intra-amniotic infection. The outcomes for patients with microbial invasion of the amniotic cavity are uniformly poor. These patients develop subsequent complications such as rupture of the membranes, clinical chorioamnionitis, or pregnancy loss. The clinical implication of this observation is that it is prudent to assess the microbial state of the amniotic cavity before the placement of a cerclage in patients with cervical insufficiency.
A sonographic short cervix in the second trimester, defined as a cervical length of 25 mm or less, is a powerful predictor of spontaneous preterm delivery. , Subclinical microbial invasion of the amniotic cavity was detected in 9% of asymptomatic patients with a sonographic cervical length of less than 25 mm. , Some of these patients have “sludge” in the amniotic cavity, which is often evidence of the presence of a microbial biofilm.
Microbial invasion of the amniotic cavity occurs in 11.9% of twin gestations. This finding is in contrast to the 21.6% rate of positive amniotic fluid cultures observed in singleton gestations with preterm labor and delivery. These data suggest that intra-amniotic infection is a possible cause of preterm labor and delivery in twin gestations but they do not support the hypothesis that intra-amniotic infection is responsible for the high rate of preterm delivery observed in twins. , Moreover, when intra-amniotic infection is detected, the presenting sac is nearly always involved.
Clinical chorioamnionitis is the most common infection-related pregnancy complication at term , and is associated with adverse maternal , and neonatal outcomes. , Neonates born to mothers with clinical chorioamnionitis at term are at an increased risk for cerebral palsy (odds ratio [OR] 9.3, 95% confidence interval [CI] 2.7 to 31.0).
The microbiology of clinical chorioamnionitis was originally described in 1982 by means of cultivation of amniotic fluid obtained with a transcervical catheter. However, such method of amniotic fluid collection is associated with high rates of contamination. Clinical chorioamnionitis is a syndrome in which 60% of patients have proven intra-amniotic infection, 24% have sterile intra-amniotic inflammation, and 15% have no evidence of intra-amniotic infection or inflammation. , , The cause of such a systemic inflammatory response in the mother without an intra-amniotic inflammatory response has been attributed to maternal neuroinflammation associated with the administration of epidural anesthesia.
Neonates born to mothers with clinical chorioamnionitis at term but without intra-amniotic inflammation often have systemic intravascular inflammation, suggesting that intrapartum fever (maternal systemic inflammation) alters the fetal immune response. Accumulating evidence demonstrates that maternal systemic inflammation can predispose the fetus to neuroinflammation.
Compelling evidence from studies of the microbiologic state of the amniotic cavity, using traditional cultivation methods at the time of genetic amniocentesis, suggests that intra-amniotic infection may be chronic in nature. Cassell and colleagues reported the recovery of genital mycoplasmas from 6.6% of amniotic fluid samples (4/61) collected by amniocentesis between 16 and 21 weeks of gestation. Two patients had cultures positive for M. hominis , and two had cultures positive for U. urealyticum. Subsequently, Gray and colleagues reported a prevalence of cultures positive for U. urealyticum of 0.4% (9/2461) in amniotic fluid samples obtained during second-trimester genetic amniocenteses. After exclusion of one patient who had a therapeutic abortion, all patients (8/8) with positive amniotic fluid cultures had either a fetal loss within 4 weeks of amniocentesis ( n = 6) or preterm delivery ( n = 2). Furthermore, all had histologic evidence of acute histologic chorioamnionitis. A similar finding was reported by Horowitz and colleagues, who detected U. urealyticum in 2.8% of amniotic fluid samples (6/214) obtained between 16 and 20 weeks of gestation. The rate of adverse pregnancy outcomes (fetal loss, preterm delivery, and low birth weight) was significantly higher in patients with a positive amniotic fluid culture than in those with a negative one (50% vs. 12%; p = .04). Of interest, patients with a positive amniotic fluid culture were more likely to have an obstetric history that included more than three previous spontaneous abortions than those with a negative culture (33% vs. 4%; p = .03). In a retrospective study of 2718 women undergoing genetic amniocentesis, 1.8% of samples (49/2718) were positive for Ureaplasma / Mycoplasma; 34 patients were treated with orally administered erythromycin. Second-trimester pregnancy loss was significantly higher in the untreated group than in the treated group (44.4% vs. 11.4%; p = .04); however, no significant differences were observed between the untreated and treated groups in the rate of preterm delivery (20% vs. 19.4%; p = .7) and adverse pregnancy outcomes (55.6% vs. 28.6%; p = .1). These observations suggest that microbial invasion of the amniotic cavity could be subclinical in the second trimester and that pregnancy loss or preterm delivery may take weeks to occur.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here