Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Before the routine use of endoscopic imaging with capsule endoscopy (CE) and deep enteroscopy, discrete ulcers of the small and large intestine were thought to be rare but associated with a broad spectrum of diseases. Similarly, the classic literature on these ulcers has emphasized symptomatic presentations, typically of diseases not confined to the small or large intestine alone. Our current ability to image the small intestine and, more importantly, biopsy ulcerative lesions, has opened a new chapter in the approach to intestinal ulceration. Similar to its history in the UGI tract, medication-induced injury to the small intestine, mainly from NSAIDs but from other medications as well, is now well-recognized and appreciated to be common. Clinical presentations associated with intestinal ulceration vary widely with location and degree of intestinal involvement, ranging from anemia and hypoproteinemia to abdominal pain, hemorrhage, obstruction, and perforation.
Generally, the disease a patient has does not lead to the identification of ulcers in the small intestine, but rather ulcers, erosions, and mucosal breaks are identified at endoscopic procedures such as ileo-colonoscopy, endoscopy, enteroscopy, and CE done for symptoms, after which the diagnosis is made. The endoscopic appearances of intestinal ulcers vary greatly by longitudinal extent, circumferential extent, and depth. Once, it had been thought that the endoscopic appearance and number of ulcers could be used to diagnose a specific disease. For example, circumferential ulceration was believed at one time to be seen only with NSAID ulceration and as part of the development of diaphragm disease. It is now realized that circumferential ulceration can be seen in many different diseases. Site of ulceration within the small intestine also is not diagnostic of any particular illness. Thus, NSAID ulcers and Crohn disease-related ulcers can be seen proximally or distally in the small bowel, although the presence of an increasing number and larger ulcers as one moves distally in the small bowel highly suggests Crohn disease.
Additionally, there is no common physiologic pathway to small bowel ulceration and our understanding of the formation of ulcerations changes with new discoveries. Thus, for example, it was initially believed that NSAIDs led to ischemia as a result of reduced blood flow in the microcirculation and that ischemia was the underlying etiology. Recent studies, however, raise the possibility that NSAID effects on the microbiota may be the main pathogenic pathway. Indeed, such NSAID ulcerations have been shown to decrease with the use of antibiotics (see later). Another possibility for NSAID enteropathy is inherited deficiencies that lead to susceptibility for ulceration. One patient with “idiopathic” small intestinal ulceration was found to have an inherited deficiency of cytosolic phospholipase A 2 , leading to a global reduction in intestinal eicosanoid levels. As discussed later, genetic mutations in a prostaglandin transporter gene is now felt to be the cause of chronic nonspecific ulceration disease (CNSU). There likely are other rare genetic predispositions to intestinal ulceration that are linked to prostaglandins, and other trophic factors that are critical in maintaining small intestinal integrity and which may explain the actual pathophysiology of small bowel ulceration. In addition to local metabolic causes, there also can be locally toxic effects of medications, radiation, and infection. Finally, there can be localized ischemia in small intestinal arterioles or venules as seen in certain vasculitides or ischemia can develop from large vessel occlusions as seen with mesenteric ischemia.
Thus, when confronted by identified ulcers, erosions, or mucosal breaks in the small intestine, the physician needs to ascertain the cause and diagnosis often requires understanding of the individual patient’s complete medical history. Intestinal ulcerations also are explored elsewhere in this textbook (see Chapter 107, Chapter 115, Chapter 118, Chapter 32, Chapter 38 and 128 ).
Ulcers of the small intestine can develop as a result of a primary disease of the small intestine, noxious chemicals affecting the mucosa, or changes in the splanchnic circulation leading to ischemia and ulceration. The mesenteric circulation is affected by many systemic diseases and physiologic states, but relatively few lead to ischemia and ulceration of the small intestine. Metabolic conditions such as hypertension and diabetes mellitus; hematologic conditions such as hypercoagulable states and vasculitides, either primary or secondary to infections or neoplasms, all can affect the splanchnic vasculature, producing a variety of symptoms. Many biologic and chemical substances of endogenous and exogenous origin also can influence the mesenteric circulation, with either beneficial or catastrophic results.
Pseudoxanthoma elasticum (PE) is a disorder of the composition or metabolism of elastic fibers. PE has a characteristic skin lesion of yellow papules, from which it derives its name and which typically involves the neck and axilla, giving the skin in these locations a typical “plucked-chicken” appearance. In this disorder, elastin fibers become thickened and encased in an amorphous material of calcium and minerals. Calcification of the fibers can ensue, leading to stiffening and loss of elasticity in the media of large arteries. Though rare, small arterioles can be obliterated, leading to ischemia and ulceration of the small bowel. There are 4 forms of PE, and transmission is as either an autosomal dominant or recessive trait. Vascular friability leading to ulceration and GI bleeding is associated with autosomal recessive type I PE and occurs in 16% of these patients. When all patients with PE and GI bleeding were studied, 70% had the type I recessive trait.
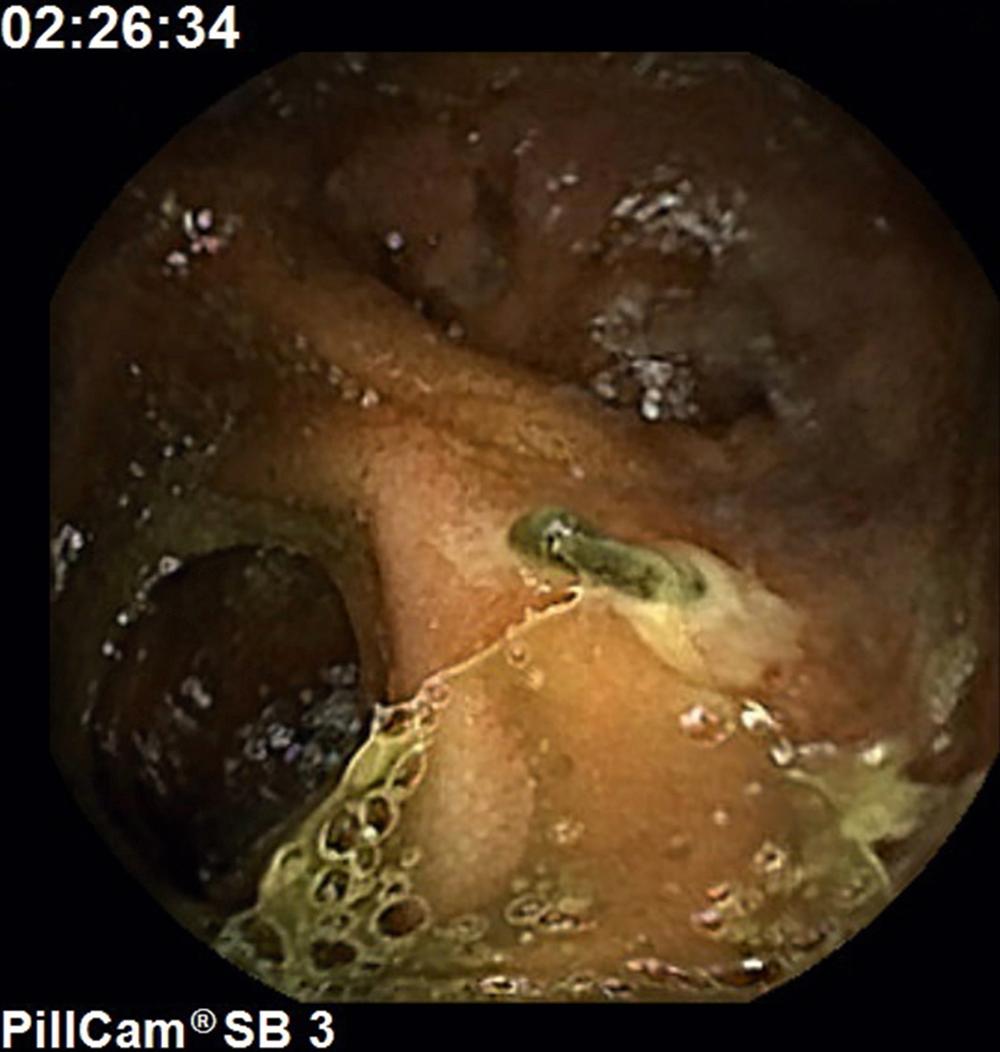
Meckel diverticulum is the most common congenital abnormality of the small intestine. (See Chapter 98 .) Present in approximately 2% of the population, it is a vestigial remnant of the omphalomesenteric duct, also called the vitelline duct or yolk stalk. Meckel diverticulum is the most common cause of GI hemorrhage in children younger than 5 years of age; such hemorrhage is caused by ulcers that develop in the small intestine (ileum) when parietal cell-containing ectopic gastric epithelium within the diverticulum secretes hydrochloric acid ( Fig. 119.1 ). Such patients may complain of RLQ pain when eating because of unbuffered acid in the diverticulum.
The patient with a hypercoagulable state is at increased risk for mesenteric and portal vein thrombosis. Both mesenteric arterial and venous occlusion can produce ischemic bowel injury with subepithelial edema, and hemorrhage followed by ulceration and finally bowel wall infarction (See Chapter 118 ). Superior mesenteric artery (SMA) thrombosis is the most common chronic mesenteric vascular injury, and the location of resultant ulceration is within the jejunum or ileum; such ischemic injury has been demonstrated, though rarely, by CE. There are numerous reports of thromboses in patients with a variety of hematologic disorders, for example, deficiencies of protein C, protein S, and antithrombin III (AT), as well as sickle-cell disease, in addition to iatrogenic causes like the use of hormonal therapy. AT is a potent natural anticoagulant factor that acts by binding to serine proteinases and thereby inactivating factors XIIa, XIa, IXa, Xa, and IIa (thrombin). The prevalence of AT deficiency in the geneal population is estimated at 1 in 5000 to 1 in 20,000; in patients with a history of venous thrombosis or pulmonary embolism, it rises to 1 in 50 to 1 in 286. The disorder is usually congenital but may be acquired. Most patients with congenital AT deficiency present between ages 10 and 35 years with venous thrombosis, usually associated with conditions known to predispose to thrombosis, such as surgery, pregnancy, and delivery. Protein C and protein S are 2 other circulating proteins that function as anticoagulants. Protein S is a cofactor of protein C, and protein C inactivates factors Va and VIIIa. A deficiency of either protein is associated with an increased risk of thromboembolism and is best treated with lifelong anticoagulation using warfarin sodium. Patients on oral contraceptives have been known to be hypercoagulable, and it has been suggested that there is a combined effect of estrogen therapy and deficiency of AT. In addition, hypercoagulability can occur in patients with pancreatitis or neoplastic diseases, particularly pancreatic cancer.
One study has identified a patient with idiopathic small intestinal ulceration who was found to have an inherited deficiency of cytosolic phospholipase A 2 , which led to a global reduction in intestinal eicosanoid levels. There likely are other rare genetic predispositions to ulceration linked to prostaglandins and other trophic factors that are critical in maintaining small intestinal integrity, and which may explain the so-called “idiopathic small bowel ulcer.”
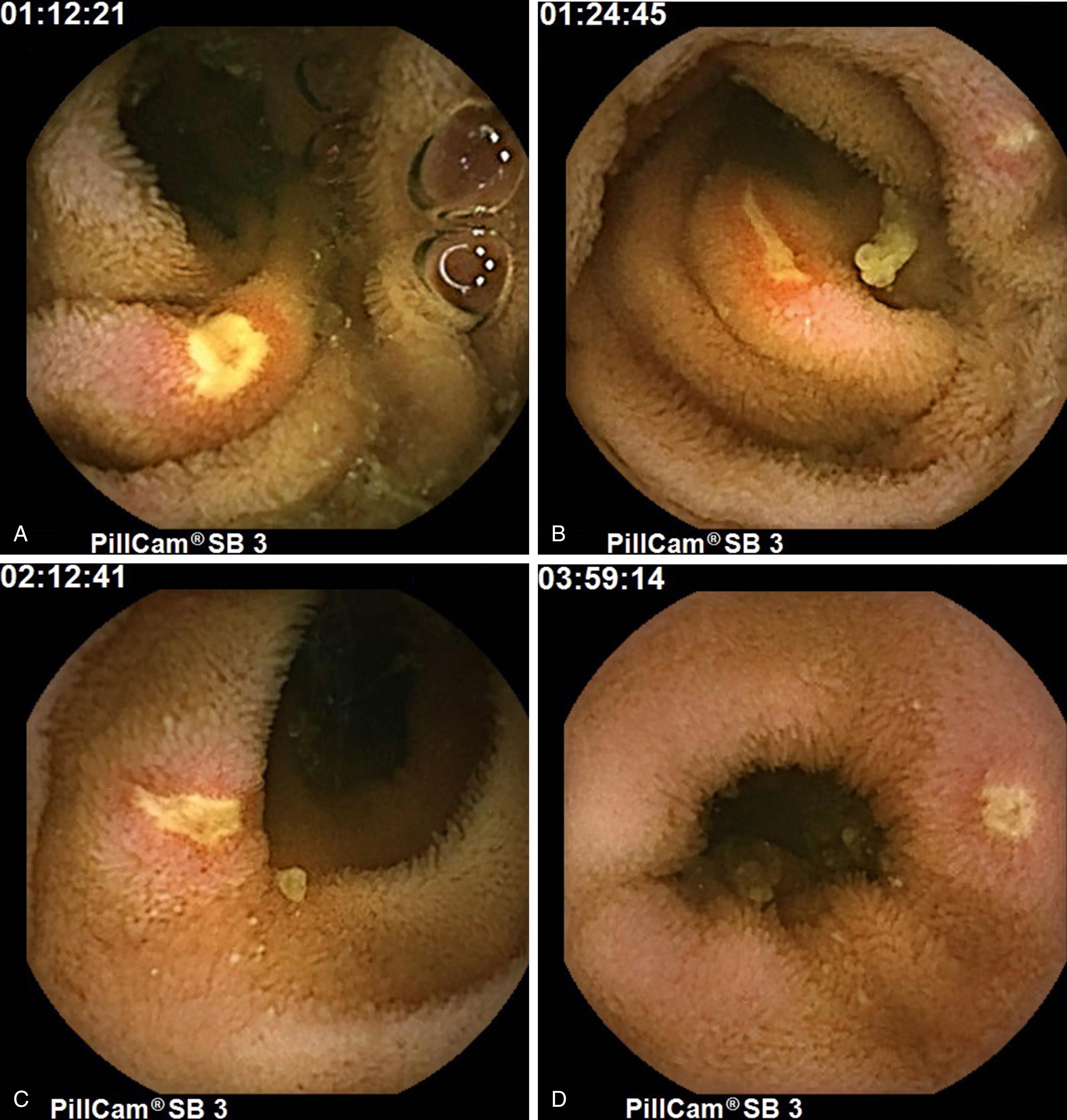
Vasculitis, inflammation, and necrosis can affect both arteries and veins of any size, including the vasa recta and intramural vessels (see Chapters 38 and 118 Chapter 118Chapter 38). Symptoms depend on the size and location of the vessels involved. Generally, when large mesenteric arteries are affected, patients present acutely because of arterial occlusion and subsequent bowel gangrene with perforation. These patients may be indistinguishable from those with mesenteric ischemia from atherosclerotic disease of the splanchnic vessels or mesenteric embolic events. Occasionally, the only elucidating factors will be evidence of systemic disease such as eosinophilia, renal insufficiency, or an elevated rheumatoid factor concentration. Disease of medium-sized arteries can lead to aneurysm formation and subsequent rupture and bleeding. Disease of small arteries, vasa recta, and intramural arterioles presents as pain, fever, ileus, and occult GI bleeding. Ulceration of the small bowel can be seen in all of these, but is most notable in diseases affecting the medium- and small-sized arteries; in such cases, the area of ischemic injury and ulceration caused by the vasculitis is segmental, with a smaller area of involvement than would be seen with disease of larger vessels ( Fig. 119.2 A-D ). Perforation is less common with small-vessel disease, and symptoms are often secondary to either ulceration or peritoneal irritation. Venulitis and obstruction of venous return can lead to mucosal edema with symptoms of malabsorption, ulceration, and ultimately venous infarction.
Various diseases cause vasculitis with either significant or relatively insignificant clinical manifestations. Common to most cases of vasculitis is a necrotizing arteritis of small- and medium-size arteries with perivascular inflammation, edema, and fibrinoid necrosis. Progression of such inflammation can lead to aneurysm formation, vessel perforation and bleeding, vascular occlusion, thrombosis, or fibrosis; these pathologic changes most often result from immune complex deposition within the vessel wall. It is postulated that circulating immune complexes become trapped along the basement membrane of arteries at sites of increased permeability. Complement system activation occurs, and polymorphonuclear leukocytes are attracted to the vessel wall. These cells release their lysosomal enzymes, leading to fibrinoid necrosis. Systemic diseases associated with high levels of immune complexes, such as rheumatoid arthritis (RA) and SLE, are thus associated with vasculitis. Cell-mediated immune reactions also can cause vasculitis but do so less commonly than do immune complex disorders. In these situations, lymphocytes sensitized to specific antigens release lymphokines near the vessel wall. These substances, such as macrophage migration-inhibitory factor, can recruit macrophages, which in turn release lysosomal enzymes, leading to vasculitis.
Classic polyarteritis nodosa (PN) is one of the systemic necrotizing vasculitides, a broad category of disease that includes allergic granulomatous angiitis formerly called (Churg-Strauss syndrome), and syndromes that overlap between this disorder and PN, such as hepatitis B vasculitis. PN is a necrotizing vasculitis of medium-size and small arteries. Vessel involvement is classically segmental and involves the branch points of arteries, with a predilection for forming aneurysms at these sites. Once the disease process begins, it may spread distally to arterioles and venules. The various stages described earlier of blood vessel necrosis, thrombosis, ischemia, and infarction can be seen at the same time at different branch points. The incidence of this uncommon disease is unknown, but it affects persons in middle age, with a mean age of 45 years, and has a male-female ratio of 2.5:1. In more than 50% of cases, patients present with fever, weight loss, and malaise. Forty-four percent of patients with PN will have abdominal involvement. Abdominal pain, nausea, and vomiting are the most common complaints. Solid organs are not spared in this disorder, and pancreatitis, as well as abnormal liver function, have been reported. Ulcerations of the GI tract, especially the small intestine, are well documented (see Fig. 119.2 ). In one autopsy series, the jejunum was involved in 37% of cases, the ileum in 27%, the mesentery in 24%, and the colon in 20% ; gastric ulcerations are less common. Major complications, namely bleeding, perforation, and infarction, are uncommon, being seen in 6%, 5%, and 1.4% of patients, respectively. Perhaps the most unusual presentation of PN is intestinal angina, which has been reported in only 15 patients. Arthralgias and renal insufficiency are seen in 64% and 70% of patients, respectively, and should suggest the diagnosis of vasculitis. The diagnosis is supported by finding aneurysms as large as 1 cm in diameter within the mesenteric, renal, and hepatic vasculature on angiography, but this finding is no longer considered pathognomonic.
Allergic granulomatous angiitis is similar to PN with the addition of pulmonary involvement, eosinophilia, and granuloma formation. Vasculitis of the pulmonary vessels, which is not seen in PN, is the hallmark of allergic granulomatous angiitis. Patients often present with wheezing and have severe asthma. Abnormal chest films with infiltrates are seen in 93% of patients. Eosinophilia is present in 85% of cases. Biopsy samples of involved blood vessels reveal both intravascular and extravascular granulomas not seen in PN. Prognosis and therapy are similar to those of PN. GI involvement is seen in 50% of patients. CE has been used to document the ulcerations in allergic granulomatous angiitis, which tend to be deep and to involve the jejunum and ileum.
Overlap syndrome is a catch-all term for disease not conforming to either PN or allergic granulomatous angiitis. Within this group is the vasculitis associated with the HBV. Such HBsAg-positive patients can present with classic PN or only some of its features. Unlike classic PN, however, only small arteries are involved. Fifty percent of patients with classic PN are HBsAg positive, attesting to the frequent incidence or association of systemic necrotizing vasculitis with this particular immune complex. A similar vasculitis may be seen with serous otitis media.
Several other collagen vascular diseases can also result in vasculitis. RA may lead to a vasculitis with a PN-like picture and intestinal ulcerations. Patients with RA who develop PN usually have severe joint disease and low serum complement levels and often are found to have circulating cryoglobulins. These patients have a high titer of circulating immune complexes, with resultant vasculitis. Initially, it was believed that glucocorticoid therapy was causative in the development of vasculitis, but it is now recognized that such usage is not an independent risk factor. Rather, use of glucocorticoids merely correlates with the severity of the arthritis, which in turn is associated with the development of vasculitis. Still, however, most ulcerations of the small intestine in patients with RA are secondary to NSAID use. A study of 37 patients with RA taking NSAIDs and presenting with anemia or occult blood in their stool reported that 68% had ulcers or erosions in the small intestine.
Vasculitis is common in SLE secondary to the high incidence of circulating immune complexes. Twenty percent of patients with SLE develop cutaneous vasculitis. Patients with SLE and vasculitis of the mesenteric circulation can present with a variety of symptoms, including bloating, nausea, diarrhea, GI bleeding, or acute abdominal pain. Mesenteric vasculitis is the most serious GI complication of SLE. GI bleeding occurs in 46% of patients with small- or large-bowel vasculitis, one-half of whom do not survive. Intestinal ischemia or bowel wall infarction with perforation also occurs and usually presents with signs of an “acute” abdomen. The outcome in cases of perforation is poor, with a mortality rate of more than 66% in most reports. In a series of 107 patients with SLE, 5 died from intestinal perforation, the most common cause of death in this series. Less severe symptoms such as nausea, vomiting, bloating, diarrhea, ileus, obstruction, or malabsorption also may occur. Symptoms often are masked by the concurrent use of glucocorticoids. Double balloon enteroscopy in lupus enteritis has been reported to show multiple ulcerated stenoses and large ulcerations in both the jejunum and ileum ( Fig. 119.3 ).
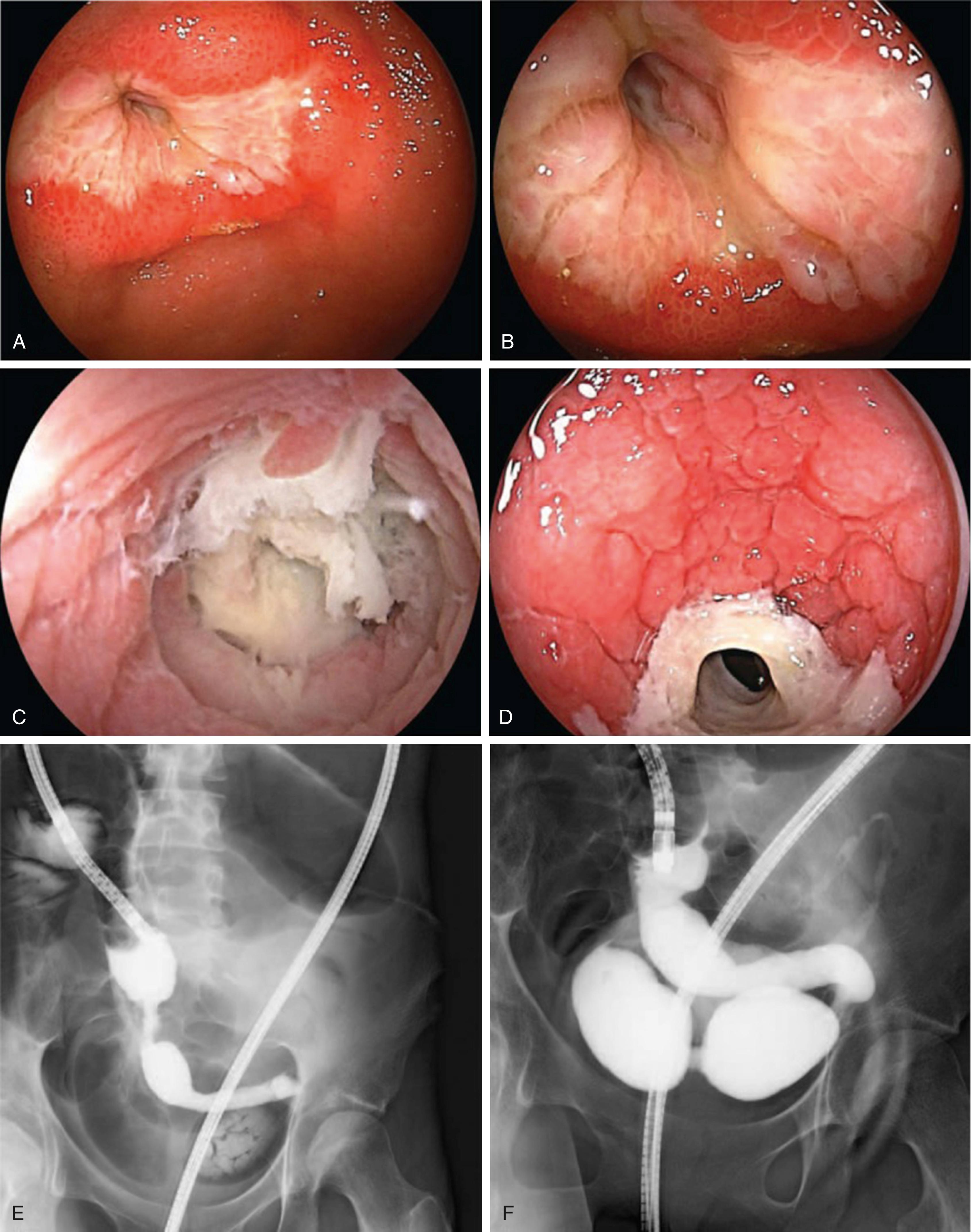
Histologically, both small-vessel arteritis and venulitis can occur. A PN-like picture also can be encountered. Patients with SLE may develop intestinal ischemia secondary to thrombosis, as the lupus anticoagulant and antibodies to phospholipid and to cardiolipin can lead to thrombosis. A complication of vasculitis and ischemic bowel injury is pneumatosis linearis. Furthermore, patients with SLE can present with protein-losing enteropathy and malabsorption secondary to vasculitis. Generally, venulitis more often is associated with malabsorption than is arterial disease. Venous outflow obstruction leads to thickening of small-bowel folds with or without ulcerations. It is also postulated that immune complex deposition can lead to increased permeability of intestinal capillaries and subsequent protein-losing enteropathy. Vasculitis associated with other collagen vascular diseases also has been reported including Sjögren syndrome, dermatomyositis, and systemic sclerosis.
Another vasculitis is the hypersensitivity type. This group of diseases involves the arterioles, capillaries, and venules, and, as with PN, is an immunologic response to antigens. The most common difference between hypersensitivity and necrotizing vasculitis is the site of the inflammatory changes. In hypersensitivity vasculitis, the post-capillary venules are most commonly involved, whereas the small- and medium-size arteries are the major targets in necrotizing vasculitis. Often, hypersensitivity vasculitis is associated with dermatologic findings. A large variety of causative factors are known, including infectious agents, drugs, and chemicals. GI tract involvement is considered uncommon, being described in only 15% of cases. Bleeding is the most common symptom, and life-threatening complications such as massive bleeding, perforation, and infarction have not been described.
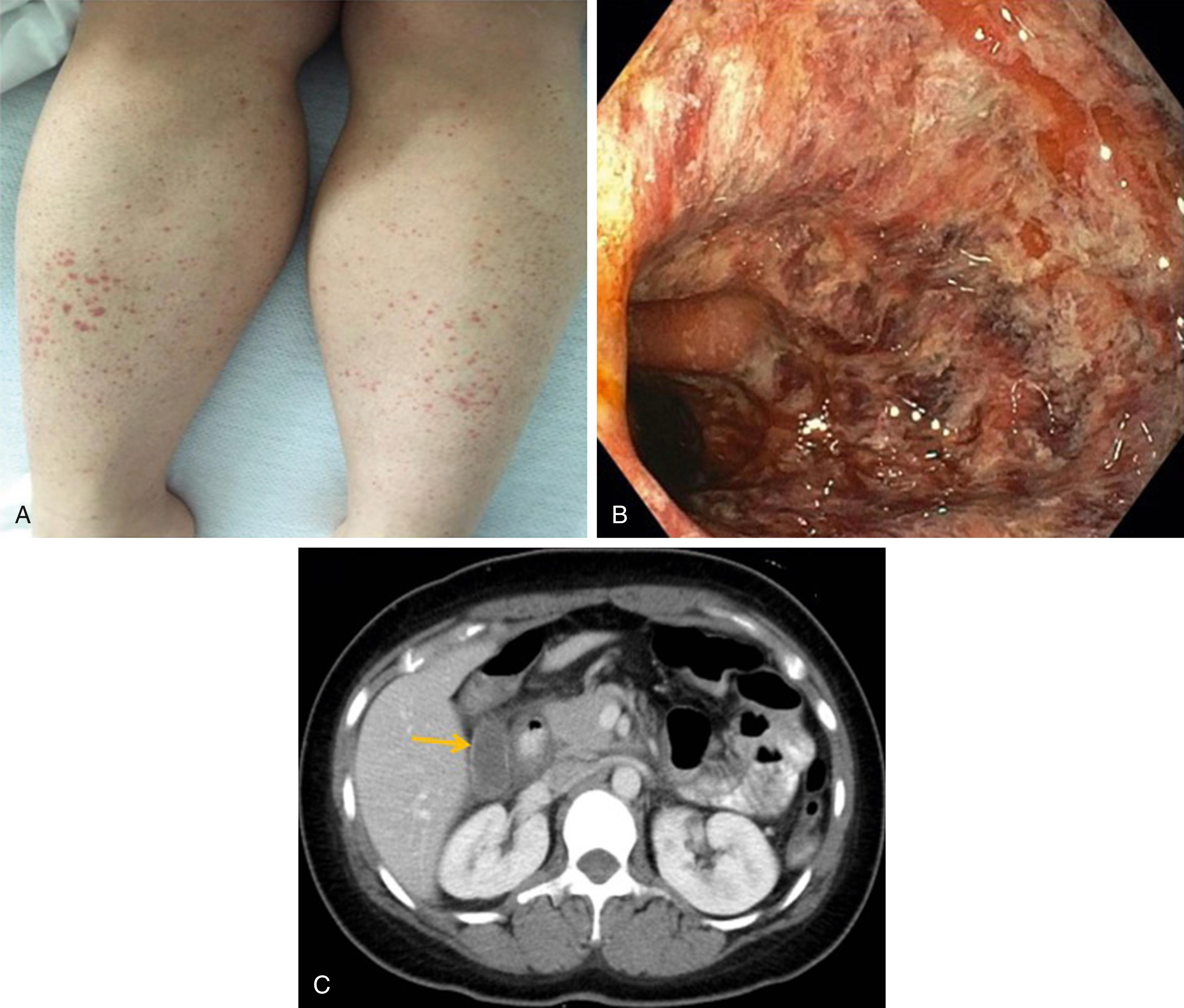
Henoch-Schönlein purpura (HSP) is a small-vessel vasculitis that frequently involves the GI tract. It is characterized by non-thrombocytopenic purpura, renal involvement, and abdominal pain with GI bleeding. HSP is most often seen in children between the ages of 4 and 7 years. Although the inciting agent for HSP is not known, IgA immune complexes have been identified within the vessel walls in this disease. GI symptoms are seen in more than half the patients, with colicky abdominal pain in 40%. Although bleeding is common (40% of patients), major hemorrhage is not (less than 5% of patients). Abdominal pain may be secondary to vasculitis-associated intramural hemorrhage. Pain, nausea, and vomiting also may develop if hematoma formation or resultant edema leads intussusception. Pancreatitis has been reported, as has protein-losing enteropathy. Both upper endoscopy and CE have been used to diagnose HSP ( Fig. 119.4 ), the latter having shown multiple ulcers of the small bowel.
Behçet disease (BD) is characterized by oral and genital ulcers as well as recurrent iritis or chorioretinitis and skin lesions. Venulitis is the primary disorder with BD. Its etiology is unknown, although theories about a viral cause have been proffered. Circulating immune complexes have been found in BD, and immunofluorescence studies have shown deposition of immunoglobulins within blood vessel walls, especially venules. BD is more common in men from Eastern Mediterranean countries and has been associated with HLA-B5. GI symptoms are present in 50% of patients and include abdominal pain, nausea, vomiting, diarrhea, and constipation; malabsorption also may occur. Mucosal ulceration is secondary to venulitis and gives rise to pain, bleeding, and perforation. Patients with a predominance of GI symptoms are said to have “entero-Behçet’s syndrome.” CE has shown erosions and aphthous ulcers in the small bowel in a series of 20 patients with BD. Although the mortality rate of BD is low (3% to 4%), intestinal perforation is one of the 3 most common causes of death, along with CNS involvement and aortic aneurysm rupture. There is no accepted therapy for BD; glucocorticoids, immunosuppressive agents, and colchicine have been tried with varying success.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here