Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Intestinal and multivisceral transplantation remains a dynamic process, moved forward by advances in the multidisciplinary care of intestinal failure, surgical techniques, innovative immunosuppressive strategies, and an improved understanding of intestinal transplantation immunology. More recently, great strides have been made in the medical and surgical management of patients with intestinal failure, which has resulted in a decrease in the number of patients added to the intestinal transplant waiting list and subsequently undergoing intestinal transplantation ( Figs. 147.1 and 147.2 ). , Multidisciplinary dedicated intestinal rehabilitation and transplantation units have become established at high-volume centers, where patients are managed with an available armamentarium of medical therapy, intestinal rehabilitation surgeries, and intestinal transplantation. The goal of these multidisciplinary dedicated intestinal rehabilitation and transplantation units is that patients likely to require transplantation will be listed, followed, and transplanted at the optimum time after weighing the risks and benefits and complications of each of these modalities (medical therapy, intestinal rehabilitation surgery, intestinal transplantation).
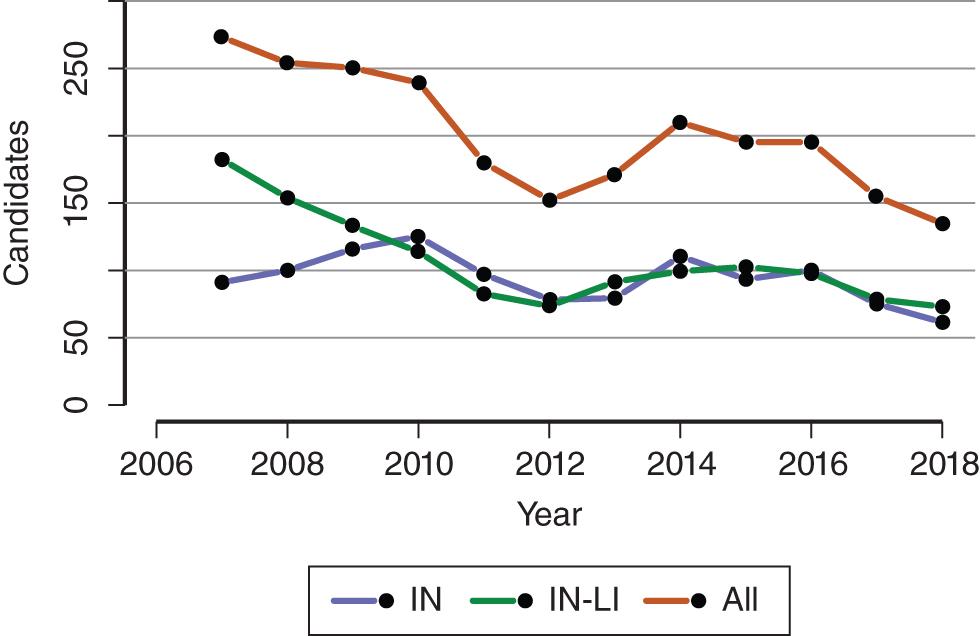
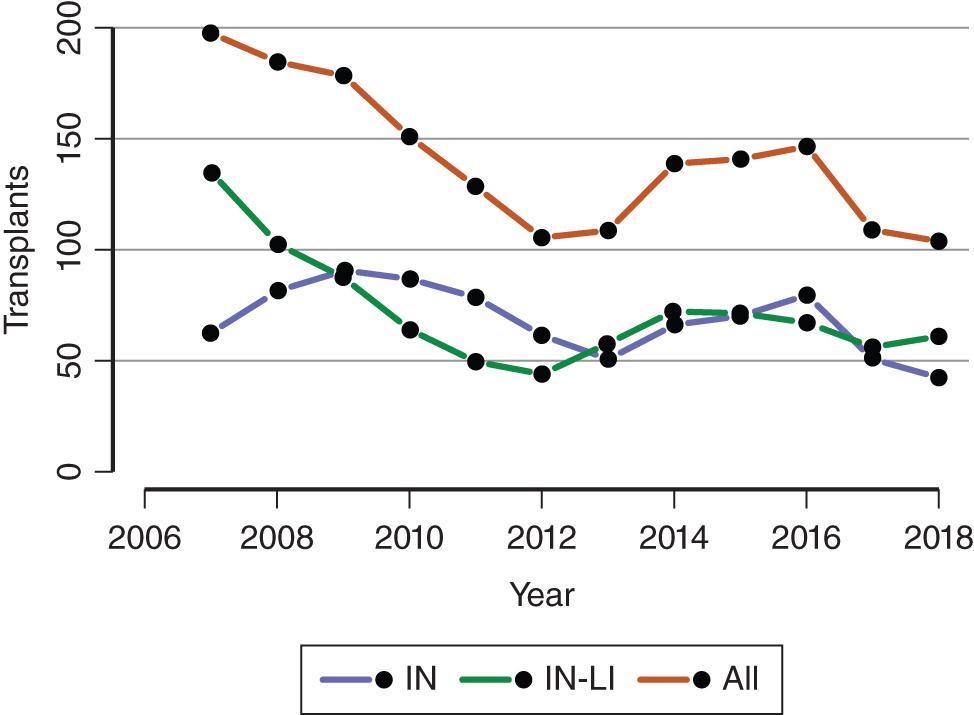
In the United States alone, 1204 patients were alive with a functioning intestinal allograft as of June 2018. Immunosuppression for intestinal and multivisceral transplantation commonly involves some form of perioperative (antibody) induction protocol, although many centers now tailor induction and maintenance protocols to individualize strategies based on each patient’s situation, often by employing immunologic testing (crossmatching and donor-specific antibody testing) to help determine optimum strategies.
The future of intestinal transplantation depends on the prevention and treatment of chronic rejection, which disproportionately affects isolated intestinal allografts. Chronic rejection continues to be a fundamental barrier to achieving successful long-term outcomes and is the ongoing subject of rigorous investigation. Long-term data on nutritional outcomes and transplantation morbidity help further determine the optimal timing and role of intestinal and multivisceral transplantation in patients with intestinal failure.
As per the most recently updated guidelines published by the European Society of Clinical Nutrition and Metabolism (ESPEN) in 2018, intestinal failure is defined as “the reduction of gut function below the minimum necessary for the absorption of macronutrients and/or water and electrolytes, such that intravenous supplementation is required to maintain health and/or growth.” , Patients with intestinal failure are initially managed by administration of parenteral nutrition (PN) through central venous access.
The duration of intestinal failure is variable and, in certain patients, unpredictable, from shortterm to lifelong, and depends largely on the adaptation capacity of the remaining viable intestine, which is dependent on variable factors such as patient age and underlying diagnosis. As such, intestinal failure can be functionally classified based on ESPEN guidelines published in 2015 into three categories:
Type 1—acute intestinal failure, short-term, self-limiting
Type 2—prolonged acute intestinal failure requiring PN over periods of weeks to months
Type 3—chronic intestinal failure requiring PN over months to years
Furthermore, 2015 ESPEN guidelines further help conceptualize intestinal failure by classifying it into five major pathophysiologic classifications: (1) short bowel syndrome (reduced absorptive mucosal surface), (2) intestinal fistula (bypass of large areas of absorptive mucosal surface), (3) intestinal dysmotility (intolerance to oral/enteral nutrition because of nonmechanical intestinal obstruction), (4) mechanical obstruction, and (5) extensive small bowel mucosal disease (inefficient absorptive and/or nutrient-losing mucosal surface).
Optimal management of patients with intestinal failure is achieved after a detailed multidisciplinary evaluation. Obtaining a comprehensive history is critical and must include birth details (such as prematurity) and disease history; complicating comorbid medical history; past surgical procedures; infections; number and location of previous central venous lines; presence of central venous thrombosis; a detailed nutrition history, including duration of PN, details of PN prescriptions, and maximal enteral feeding tolerance and its trajectory (still advancing or static); medication history; and frequency/volume of stools. A careful history and physical examination performed by the intestinal rehabilitation team is critical to the process of achieving a complete pretransplant work-up. Further investigations include blood tests, upper gastrointestinal (GI) contrast study with small bowel follow-through; contrast enema if indicated; abdominal sonogram to assess for hepatosplenomegaly; and an examination of central venous anatomy either by ultrasound, computed tomography (CT) or magnetic resonance (MR) venography, or traditional venography. Occasionally, endoscopy with small intestinal aspiration for quantitative microbial culture and mucosal biopsy is recommended. In equivocal cases, liver biopsy is recommended if there is evidence of liver dysfunction or portal hypertension to determine if concomitant liver-containing transplantation is required.
Management of patients with intestinal failure focuses on optimization of gut adaptation and recovery of intestinal function to achieve enteral autonomy. With improved management of PN and minimization of complications, longer durations of PN can be achieved to allow for gut adaptation. Clinical trials have shown favorable outcomes with the use of the glucagon-like peptide analogue (GLP-2) teduglutide in increasing intestinal adaptation and increasing the likelihood of weaning off PN in short bowel syndrome patients. As a result, the Food and Drug Administration (FDA) has approved the use of teduglutide for the management of patients with short bowel syndrome over the last decade. , A small portion of patients have shown excellent response with the ability to greatly reduce or even completely eliminate parenteral support. However, the high cost of the medication, estimated at >$400,000 per year per patient, limits its general use, especially without guidance on which patients will achieve enteral autonomy with therapy.
Simultaneously, progress has been made in intestinal rehabilitative surgical therapies, including autologous reconstructive surgeries (repairing enterocutaneous fistulae, restoring enteral continuity with primary reconstruction or use of alimentary conduits) and bowel-lengthening procedures such as the longitudinal lengthening procedure described by Bianchi and the serial transverse enteroplasty (STEP). A recent study focusing on outcomes of the full spectrum of intestinal rehabilitative surgeries and bowel-lengthening procedures on 500 consecutive patients with intestinal failure has shown remarkable results, with restored nutritional autonomy in 82% of patients with a 5-year survival of 70%.
Alternatively, if gut dysfunction is considered irreversible, management of these patients concentrates on maintaining optimal growth in children and nutritional repletion in adults to prepare them for likely intestinal transplantation. Recently, there has been a shift in timing of intestinal transplantation, focused more on the individualized risks and complications of PN versus intestinal transplantation. In other words, if patients, especially children where there still may be hope for further adaptation, are stable on PN with no significant complications, then deferring transplant is quite reasonable.
Small bowel bacterial overgrowth (SBBO) is a common clinical problem in patients with intestinal failure and is treated with a variety of antibiotic regimens. To date, no comparative studies are available to enable an evidence-based approach to treat SBBO. The use of metronidazole for anaerobic overgrowth, combined with trimethoprim and sulfamethoxazole or an oral aminoglycoside for gram-negative organisms, is a common theme. Metronidazole monotherapy is used if the dominant symptoms suggest predominantly anaerobic overgrowth (such as bloating, increasing diarrhea, and D-lactic acidemia). The extreme sensitivity of anaerobes to oxygen makes the use of small bowel aspirate cultures relatively unreliable as a means of microbial surveillance or indication to treat SBBO. Probiotics such as Lactobacillus and Saccharomyces have been used in an attempt to limit SBBO. Given the absence of randomized evidence to support the efficacy of probiotics, coupled with reasonable concerns about impurities and possible contamination with other bacteria (e.g., Leuconostoc ), the use of probiotics is controversial and mostly discouraged in patients with central venous catheter access and intestinal failure and especially after transplantation because of increased immune suppression.
Parenteral nutrition–associated liver disease (PNALD), also referred to as intestinal failure–associated liver disease (IFALD), remains a critical problem in this patient population, affecting infants disproportionately. The 1-year mortality of patients with PNALD exceeds 20% in the absence of PN weaning or transplantation. Although not always feasible, the best strategy to prevent and treat PNALD involves a commitment to the advancement of enteral nutrition. Despite a conscientious approach to PN therapy, many children and adults still develop cholestasis relatively early in their clinical course. Prevention and timely treatment of infection, minimizing SBBO, preventing overfeeding with dextrose, providing adequate amino acids, cycling PN, or providing PN-free days when possible are probably important measures to slow the progression of PNALD. Stasis of bile in the nonstimulated biliary system and gallbladder can lead to sludge buildup and cholelithiasis. In the authors’ experience, cholecystectomy may be beneficial to minimize the risks of developing biliary obstructive episodes and pancreatitis and should be considered early in any operative course as part of a combined procedure. Cholecystectomy on its own is rarely done, as it seldom improves liver function and is not indicated for PNALD alone. Bowel reconnection, even in patients with very short guts, may relieve some of the high pressure in the upper GI tract and issues of reflux cholangitis, which may allow for long-term improvements in liver function.
Rates of PNALD have significantly decreased with novel methods to reduce phytosterol exposure from parenteral lipid emulsions through lipid minimization protocols with the reduction of traditional soy-based lipid solutions and even their eventual removal by substituting with Omegaven (a fish oil–based, intravenous [IV] lipid solution rich in omega-3 fatty acids). Omegaven has recently been FDA approved (as of July 2018) for use in neonatal and pediatric patients suffering from PNALD. Another option in patients with PNALD is the use of a mixed oil lipid-based emulsion such as SMOF (soybean oil, medium-chain triglycerides, olive oil, fish oil) lipid, which is currently approved for use in adults in the United States with an ongoing pediatric trial. SMOF lipid may offer another alternative in pediatric patients with PNALD who demonstrate poor weight gain or develop essential fatty acid deficiencies while on fish oil–based emulsions. , Although biochemical improvement in liver disease has been shown with these alternative lipid formulations, it is unclear whether they actually alter histologic progression of liver disease.
In addition to PNALD, patients on long-term PN are at risk of developing metabolic bone disease (MBD). Associated with an insidious onset of bone pain that can become quite severe, patients with MBD will present with normal serum calcium, phosphorus, vitamin D, and parathyroid hormone, but with hypercalciuria. Nontraumatic spinal and rib fractures have been reported in these patients. To optimize bone maintenance in patients on PN, it is important to include calcium in parenteral formulations, prevent metabolic acidosis, and minimize aluminum contamination. Symptoms of MBD tend to resolve only after stopping PN.
Many intestinal transplant recipients will require intensive care unit (ICU) management during the pretransplant period, although the number of patients being transplanted from home (outside of a hospital setting) has increased with improved medical management and resulted in improved outcomes in this subset of patients. Sepsis and GI hemorrhage are common reasons for ICU admission in patients with intestinal failure. Blood products, though necessary in the resuscitation of GI hemorrhage, should be used judiciously in the absence of acute bleeding. Pretransplant exposure to blood products, particularly platelets, can predispose intestinal transplant recipients to developing antibodies and can place them at a higher risk of developing antibody-mediated rejection after transplantation. Leukoreduced blood products may be preferable in patients awaiting transplant.
Central line–associated bloodstream infections (CLABSIs) unfortunately are common in PN-dependent patients. All attempts should be made to avoid this event, with fastidious central line care at the core of the matter. Centers have employed central line care bundles to aid in sterile central line care at home. Lock therapy has played an important role in further reducing the incidence of CLABSI with ethanol, taurolidine, and sodium EDTA locks all showing various degrees of effectiveness. One of the determining factors to move forward with transplantation is loss of venous access. Hence, although infection (especially fungal and life-threatening gram-negative infections) may necessitate removal of a tunneled central venous catheter, all attempts at salvaging the line, or at least the venous site, should be considered. Smaller pediatric patients and patients with a history of thrombosis may have limited venous access, necessitating preservation and treatment through an infected line. Percutaneous lines should be placed with caution in these patients, as great vessels may no longer be patent, and the trauma to the remaining vessels may have serious consequences. Many surgeons, anesthesiologists, and intensivists now place central lines under ultrasound guidance, although the more difficult and chronic-issue lines are best served in interventional radiology, with their vast cadre of specialized techniques. Ultrasound evaluation of deep veins to assess for patency is notoriously variable and often unreliable, and in difficult or concerning cases venography remains the gold standard. Many centers now use cross-sectional imaging through MR or CT venography in place of traditional fluoroscopic venography. Mapping should be considered before excessive loss of venous access occurs.
Nutritionally deplete patients are relatively immune suppressed and prone to severe community-acquired infections. Pediatric patients with intestinal failure and IFALD are at increased risk of respiratory failure even with common viral infections. Because children have a compliant chest wall, increased abdominal girth creates a mechanical disadvantage even during normal tidal volume breathing. In the setting of pulmonary infection, volume overload, or decreased cardiac output, the work of breathing can lead to fatigue.
Neurodevelopmental outcomes of children with intestinal failure have been understudied. Conflicting evidence exists regarding neurodevelopmental outcomes, with some reports of gross motor and cognitive deficits identified within the first few years of life.
The goals of the transplant team are to determine whether the patient may benefit from transplantation, assess alternatives to transplant, evaluate any contraindications to transplantation, and provide education to the patient and caregivers about the complex process of undergoing transplantation. After evaluation by the multidisciplinary team and open and frank discussion among all the team members, the usual result is one of three choices: (1) the patient may be followed closely as an intestinal care patient and listing status deferred depending on progress; (2) the patient may be listed as a status 2 recipient to accrue time on the list while awaiting possible medical improvement or bowel adaptation but realizing that transplantation is still quite likely; or (3) the patient may be listed as a status 1 intestinal recipient if the patient has met criteria for complications of irreversible intestinal failure, or as a liver intestine recipient if the patient is deemed to have irreversible intestinal failure and associated irreversible significant liver dysfunction for immediate consideration where criteria make transplantation inevitable.
In October 2000, the Centers for Medicare and Medicaid Services approved intestinal, combined liver-intestine, and multivisceral transplantation as a standard of care for patients with irreversible intestinal failure who could no longer be maintained with PN. As experience with intestinal rehabilitation and transplantation has increased over the last two decades, there has been debate about updating the original guidelines published in 2001 about indications of intestinal transplants. As a result, a working group of worldwide experts on intestinal failure and transplantation was assembled at the XIV International Small Bowel Transplant Symposium in 2015. Based on this deliberation, a recent consensus statement about updated indications of intestinal transplantation was published in 2019. Most notably, extreme short bowel and transplantation for quality of life are absent from the revised indications, largely because of the advances in intestinal rehabilitation and resultant improvements in quality of life on PN. Based on these updated 2019 guidelines, indications for intestinal transplantation in the current era can be summarized as follows:
Evidence of advanced or progressive IFALD defined as either (a) hyperbilirubinemia >4.5 mg/dL that persists for more than 2 months despite IV lipid modification strategies or (b) any combination of elevated serum bilirubin, reduced synthetic function (low albumin or elevated international normalized ratio [INR]), and laboratory indications of portal hypertension and hypersplenism (especially low platelets) persisting for more than 1 month in the absence of a confounding infectious event(s).
Progressive loss of central venous access, typically defined by loss of at least two upper body central venous sites (left subclavian, left internal jugular, right subclavian, or right internal jugular) or occlusion of a brachiocephalic vein in children. In adults, this criterion should be evaluated on a case-by-case basis.
Life-threatening morbidity in the setting of indefinite PN dependence of either anatomic or functional etiology, as suggested by (a) in children, two admissions to the ICU (after initial recovery from the event resulting in intestinal failure) because of cardiorespiratory failure (mechanical ventilation or inotrope support) resulting from sepsis or other complications related to intestinal failure and (b) in adults, on a case-by-case basis.
Invasive intraabdominal desmoids in adolescents and adults.
Acute diffuse intestinal infarction with hepatic failure.
Failure of first intestinal transplant.
Owing to the particularly high morbidity and mortality of children with IFALD, increasing efforts have been made by the pediatric medical community to optimize timing of referral of these patients to specialized intestine failure rehabilitation centers and transplant centers to improve overall outcomes. In addition to the earlier criteria for intestinal transplantation, a recent expert consensus panel recommended the following patients be evaluated for intestinal transplant: (1) children with massive small bowel resection, (2) continuing prognostic or diagnostic uncertainty, (3) microvillous inclusion disease or intestinal epithelial dysplasia, and (4) the request of the patient or family. This initiative, to a large extent, has been relatively successful in breaking down previously held prejudices and allowed for prompt and, at times, very early evaluation. This in turn has allowed for more coordinated expert multidisciplinary management of these patients. With fewer patients now heading into liver failure with improved preservation of the liver, it gives the medical and surgical teams more time to let the native bowel adapt, so that more patients, even after 5 or more years of PN therapy, are eventually able to wean off of PN who previously would never have been given this chance.
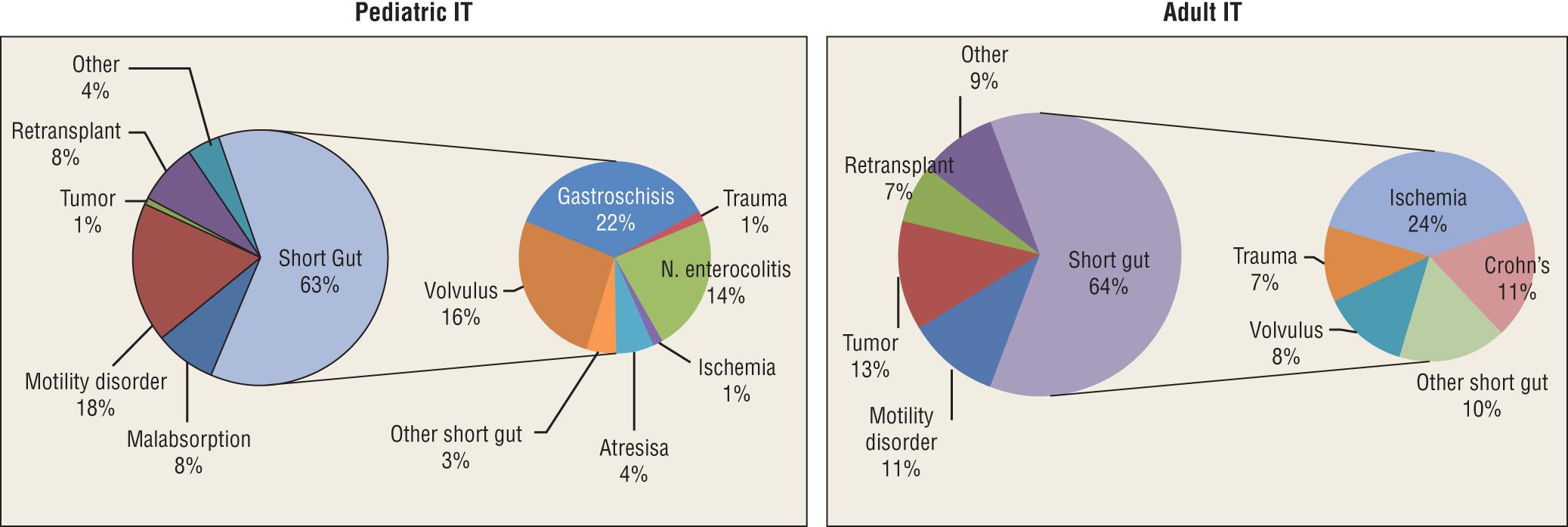
The initial underlying pathophysiologic disorders leading to intestinal failure and subsequently intestinal transplantation in children and adults are summarized in Table 147.1 . Based on data from the Intestine Transplant Registry published in 2015, short gut syndrome is the most common pathophysiologic mechanism responsible for intestinal failure requiring intestinal transplantation in children and adults. The relative distribution of various pathophysiologic indications leading to intestinal failure and subsequently to intestinal transplantation in children and adults is summarized in Fig. 147.3 .
| Pediatric Patients | Adult Patients |
|---|---|
| Volvulus | Superior mesenteric artery thrombosis |
| Gastroschisis | Crohn disease/irritable bowel disease (IBD) |
| Necrotizing enterocolitis | Desmoid tumor |
| Pseudo-obstruction | Volvulus |
| Intestinal atresia | Trauma |
| Microvillous inclusion disease | Familial polyposis |
| Hirschsprung disease | Budd-Chiari disease |
| Trauma | Intestinal adhesions |
| Pseudo-obstruction | |
| Radiation enteritis |
Transplantation is unlike any other surgical procedure in that it is rarely an isolated event, unlike general surgical procedures, and has significant short- and long-term medical and surgical issues that need to be considered. Nowadays, as the overall management of PN improves, the indication and timing of transplantation become a balancing act, trying to weigh the risks and complications of PN versus the potential short- and long-term risks of intestinal transplant.
For both children and adults, the evaluation of intestinal and multivisceral candidates usually begins as an inpatient process because of the complexity of the case and the need for many different services to see the patients and investigations that may be required. Most intestinal failure units use multidisciplinary teams that assess patients during evaluations. This comprises the GI team, other medical teams as indicated (cardiac, renal, genetic, immunology, pulmonology, etc.); general surgery (especially pediatric surgery); transplantation; and many allied services including psychology, social work, pharmacy, and others as indicated in each case. Transplant teaching is a vital component, as is determining fiscal ability and supportive structures for potential recipients. Each team provides recommendations, and, in particular, the GI team may make recommendations with the PN and IV fluids and medications that may be quite beneficial to the patient and the referring team. At times, especially in a multidisciplinary center with both good pediatric surgical and transplant skills, these patients may undergo surgical exploration and attempts at reconstruction and/or bowel-lengthening procedures before transplantation. These are done with an attempt to either avoid transplantation, or at least minimize the need for PN and concomitant liver disease and delay the need for transplantation, or to make the conditions at transplant better (i.e., obtain colonic growth and health in previously disconnected patients or resecting enteric fistulae/intraabdominal abscesses that may be contributing to sepsis). Having a surgeon well versed in medical management strategies, able to operate as a general surgeon to try to help restore enteral autonomy by surgical means, and be able to set the patient up for transplant is advantageous for both the patient and intestinal failure unit, allowing for more aggressive surgical interventions to try to avoid or delay transplantation.
Determining which type of allograft to use in patients with intestinal failure involves a comprehensive evaluation of the function and anatomy of the remaining bowel along with other abdominal organs. Intestinal failure patients are considered candidates for isolated intestinal transplant (with or without colon), combined liver and intestine transplant, multivisceral transplant (including liver, stomach, duodenum, pancreas, and small bowel with or without colon), or modified multivisceral transplant that excludes the liver. There has been ongoing debate in the transplant community regarding the previously mentioned nomenclature; however, from a practical and immunologic point of view, the major differentiation is the inclusion of the allograft liver as part of the transplant bloc. Whether to perform simultaneous hepatic replacement remains a challenging decision even to experienced transplant surgeons, particularly for patients with borderline liver dysfunction and a biopsy that shows fibrosis (Ishak level 3–4 and above) and some degree of synthetic dysfunction suggestive of portal hypertension. Nevertheless, it must be realized that the classic symptoms and signs of impending liver failure may not be the same in patients with limited or no intestine, and the use of Omegaven or SMOF may normalize the bilirubin but not affect fibrosis. This also affects the Model of End-Stage Liver Disease (MELD) and Pediatric End-Stage Liver Disease (PELD) score in these potential recipients; hence the essential need for “supplemental” points to be added to their total when the intestine is added to the United Network for Organ Sharing (UNOS) liver transplant listing. This has been allowed in the pediatric population for some time now but remains a vexed issue in the adult population.
The key factors in determining whether to perform liver transplant in patients with intestinal failure are the extent of portal hypertension and the severity of parenchymal liver disease. In general, patients with mild portal hypertension should be cautiously considered for isolated intestinal transplant. Under these circumstances, it is mostly recommended that venous outflow from the intestinal allograft bypass the portal circulation and be drained to the recipient systemic circulation through the inferior vena cava. This is especially relevant as there has been no documented proof that portal drainage is more beneficial or that relative portal and mesenteric hypertension may have deleterious effects on intestinal allograft function. The other predicament is the patient with marginal intestinal length who may, under ideal circumstances and more time, be able to adapt and come off of PN but the liver has undergone such considerable damage that it needs to be replaced. In this circumstance, liver transplantation alone in the face of short gut syndrome can and has been done but is fraught with potential dramatic consequences and the potential need for liver and intestine retransplantation if it is not successful.
Brief descriptions of recipient operations are provided here. The multivisceral donor procurement operation has previously been well described. It is important to note that to a large extent, the future course after transplant is significantly determined by the quality of the donor and success of the donor recovery and back-table preparation. The intestine is perhaps the most sensitive donor organ and prone to ischemic events either by nature of the cause of death or because of pharmacologic agents (vasopressors) that are used to support the donor leading up to and after neurologic determination of death. It is thought that damage to the bowel sets up an inflammatory cascade that in turn makes the organ more immunogenic, or at least more susceptible to a rejection episode once it is reperfused in the recipient. Hence, the recipient surgeon is extremely judicious in the selection of a suitable donor and the number of acceptable donors is restricted. Other factors that play into this decision include blood group (generally identical), size-matching issues because of restricted abdominal domain (reduced grafts have been shown to have short- and long-term issues), and some potential crossmatch issues (especially in retransplant patients). Because of stringent donor selection criteria, the intestinal allograft is one of the more infrequently recovered allografts from available donor pool.
For isolated intestinal transplants ( Fig. 147.4 ), the donor intestinal graft (jejunum and ileum) is procured along with donor vascular conduits, including an artery (iliac and/or carotid) and a vein (iliac, occasionally jugular). The donor superior mesenteric vessels are occasionally anastomosed directly to the recipient superior mesenteric artery and vein if adequate length is achieved. More commonly, interposition vascular conduits are anastomosed to the recipient infrarenal aorta and recipient superior mesenteric vein (portal drainage) or inferior vena cava (systemic drainage) to provide sufficient length and proper orientation for the allograft.
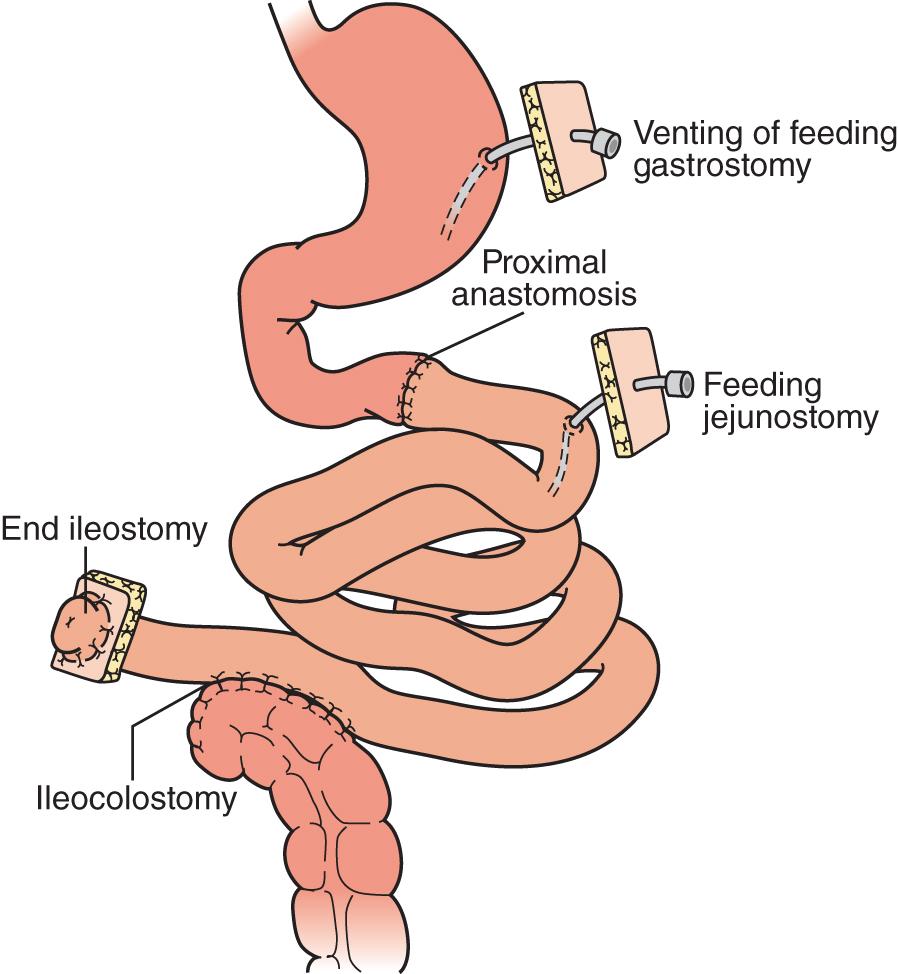
The intestinal reconstruction involves a proximal duodenojejunostomy or jejunojejunostomy, depending on individual recipient considerations of remnant bowel viability and anatomy. The distal end of the intestinal allograft may be used as a permanent end ileostomy if the recipient has no remaining viable colon or may be anastomosed to the remnant colon, leaving a short portion of allograft distal to the enterocolic anastomosis to bring out as a temporary end ileostomy (Bishop-Koop ileostomy) that allows access to the bowel for endoscopic surveillance and mucosal biopsies.
Colonic transplantation with the small bowel has been used more frequently, especially in situations where the native colon is very short or nonfunctional (such as Hirschsprung disease or pseudo-obstruction). In this case, a loop ileostomy is formed so that the small bowel can be biopsied and the colon normally brought out as an end colostomy, or if anastomosed to the remaining colon, a loop allograft colostomy may be formed as well. Inclusion of colonic graft with small bowel graft has been shown in a recent study to provide improved postoperative renal protection along with increasing rate of interval ileostomy reversal in intestinal transplant recipients.
In patients with gastric dysmotility, although a stomach-inclusive allograft may be ideal, because of donor availability (size and/or anatomic vascular aberrations), it may not be recoverable; hence a gastrojejunostomy may also need to be performed as an alternative to drain the stomach. Single or multiple feeding tubes (gastrostomy tube, jejunostomy tube, and combined gastrojejunostomy tube) may be placed based on multiple considerations, including recipient pretransplant oral intake capacity and dysmotility issues.
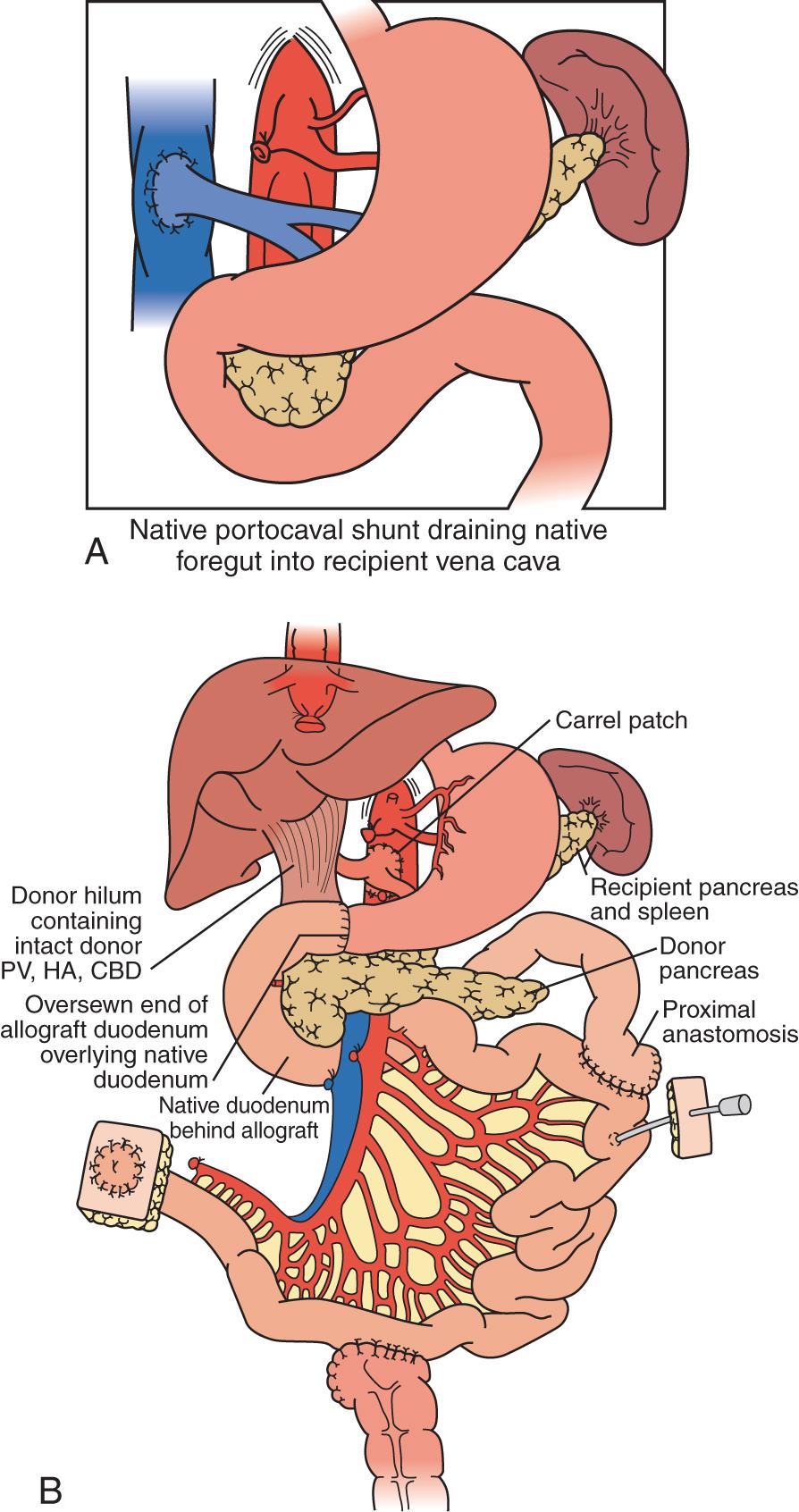
For combined small bowel and liver transplants ( Fig. 147.5 ), the recipient hepatectomy is routinely performed with preservation of the native retrohepatic inferior vena cava. The recipient foregut, including stomach, native pancreas, and proximal duodenum, is also preserved and its venous outflow maintained with a permanent end-to-side portocaval shunt. The composite donor allograft includes the primary organs (liver and small bowel) and the donor duodenum and pancreas, allowing for maintenance of donor hepatobiliary continuity. Arterial inflow to the composite donor allograft is achieved using an arterial interposition conduit from the recipient infrarenal aorta to the reconstructed donor aorta placed onto the Carrel patch of donor celiac artery and superior mesenteric artery. Allograft venous outflow via the suprahepatic inferior vena cava commonly involves the well-described “piggyback” technique, anastomosing the donor suprahepatic inferior vena cava/hepatic veins cuff to the confluence of the recipient hepatic veins and cava. Occasionally, a “standard” bicaval anastomosis is performed. Intestinal reconstruction is performed in a similar fashion to an isolated intestinal transplant, mostly with the upper anastomosis being a jejunojejunostomy. Feeding tubes are placed as indicated.
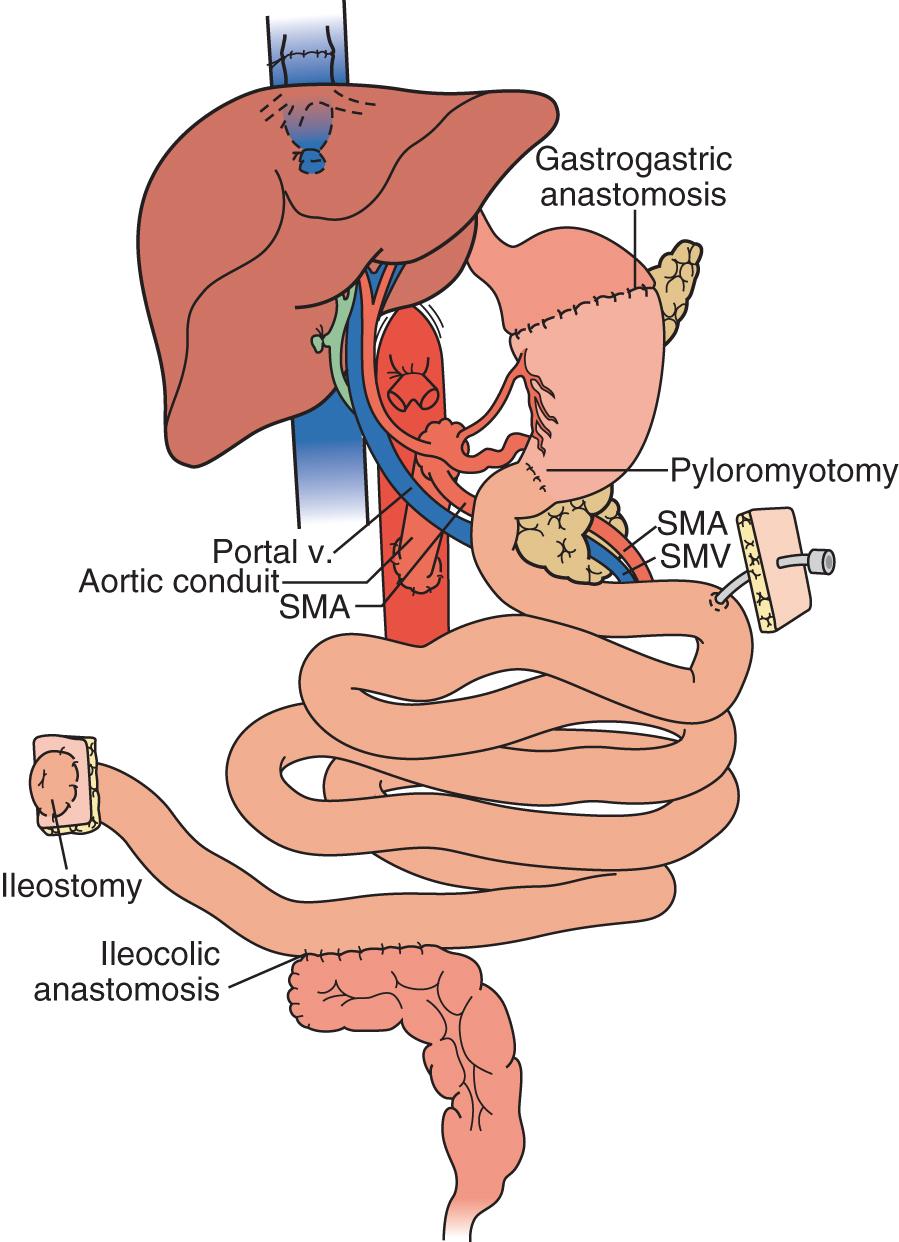
In the majority of cases of full multivisceral transplant procedures ( Fig. 147.6 ), before implantation, the recipient distal stomach, duodenum, pancreas, liver, and remaining small bowel are resected. The recipient inferior vena cava is routinely meticulously preserved. The absence of remaining foregut or midgut precludes the need for a portocaval shunt. Vascular inflow is similar to composite liver-bowel transplant but now includes celiac inflow to the stomach as well. Vascular outflow is identical to composite liver-bowel transplant. The donor spleen is removed from the composite allograft on the back table before reperfusion. Intestinal reconstruction is performed proximally with a gastrogastrostomy anastomosis, and the distal anastomosis is similar to previously described intestinal transplants. To avoid gastric outlet obstruction resulting from vagal denervation, a Heineke-Mikulicz pyloroplasty is routinely performed after reperfusion. Feeding tubes are placed as indicated.
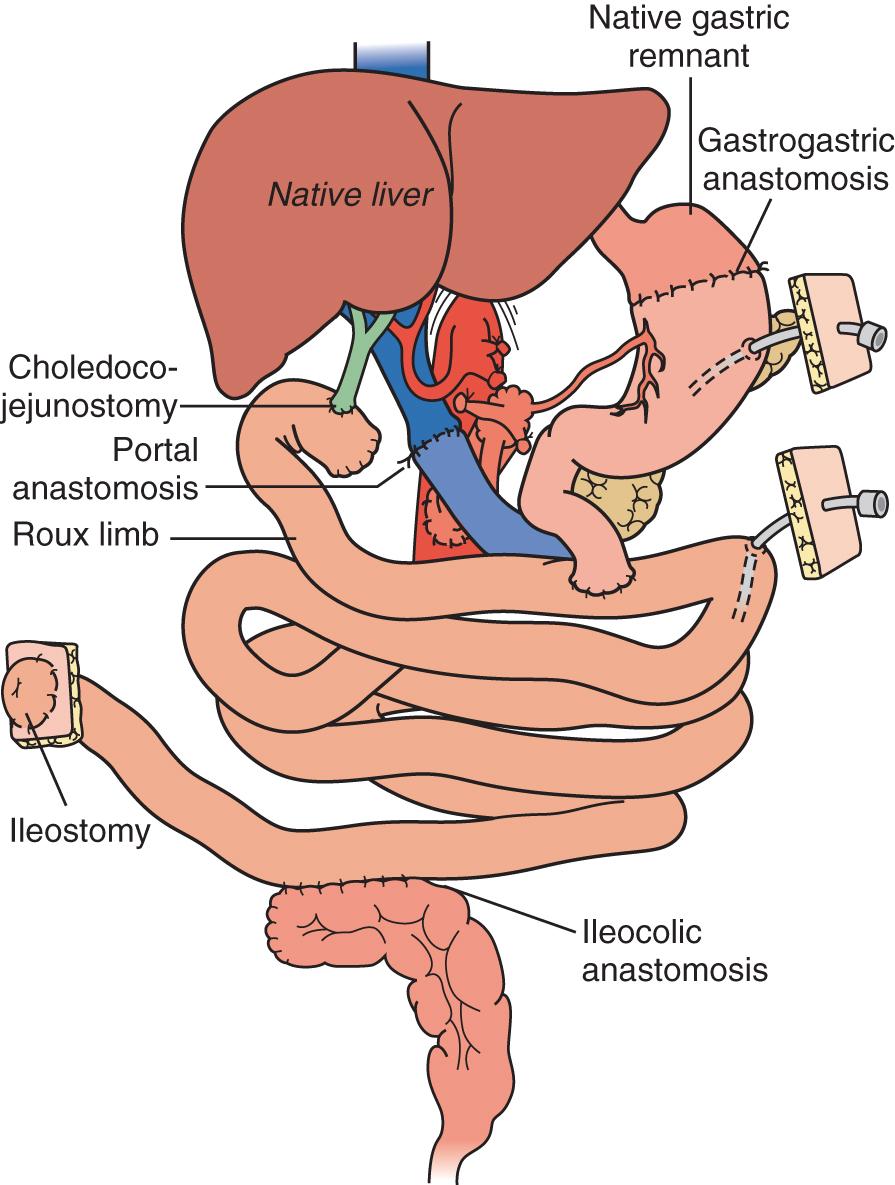
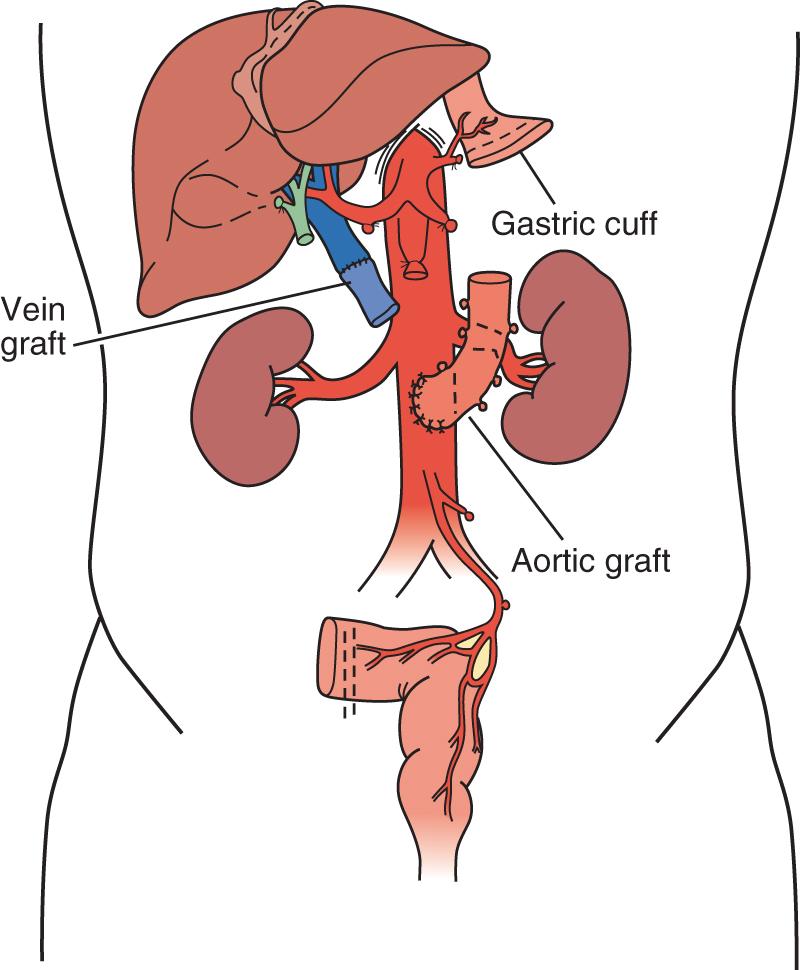
A “modified” multivisceral transplant ( Fig. 147.7 ) involves transplantation of a full composite allograft without a liver. The recipient liver is preserved along with its vasculature and the extrahepatic biliary system with duodenum, pancreas, and spleen. Vascular conduits are used routinely ( Fig. 147.8 ). If the native enterohepatic biliary system is intact, the native duodenum or jejunum is drained into the allograft duodenum or jejunum. If, however, the procedure involves disruption of hepatobiliary continuity, the native bile duct can be drained via Roux-en-Y hepaticojejunostomy constructed from donor intestinal allograft (often the case in children where the donor bile duct is very small), or in adults and older children via a choledochoduodenostomy (duct-to-duct) anastomosis.
Fascial closure after intestinal transplant can be difficult because of reduced abdominal domain, loss of fascial compliance from prior laparotomies, and possible concern for abdominal compartment syndrome. Previous studies have shown that abdominal fascia cannot be closed in about one-third of intestinal transplant cases. Low donor to recipient weight ratio (<0.8) has been shown to be an important predictive factor for allowing fascial closure after intestinal transplant. , A recent study identified high-risk recipient factors (preexisting enterocutaneous fistulas, male sex, and age over 4 years) contributing to an inability to achieve sustained abdominal wall closure after intestinal transplant. Currently available options for abdominal closure in these cases include skin-only closure, advanced reconstructive surgical options including use of biologic mesh and delayed skin grafting, and consideration of abdominal wall transplant at the time of intestinal transplant.
Advances in the technical aspects of intestinal and multivisceral transplantation have occurred in parallel with improvements in intraoperative monitoring and postoperative critical care management of these challenging patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here