Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pathologists are asked to evaluate colorectal biopsy specimens for a variety of reasons, but often only a pattern of injury can be identified, at best. This evaluation is performed with the hope that a specific diagnosis can be rendered once appropriate clinical, radiological, and laboratory information is obtained. However, some forms of colitis, such as lymphocytic colitis, collagenous colitis, and ischemic colitis, do have specific histological features, and a diagnosis can be rendered in the absence of clinical information. Many histological features are characteristic of chronic inflammatory bowel disease (IBD). However, it is often difficult or impossible to distinguish ulcerative colitis (UC) from Crohn’s disease (CD) on the basis of colorectal biopsy specimens only, particularly after the patient has been treated medically, in which case the features of these two disorders overlap considerably.
Perhaps the most important aspect of evaluating colorectal biopsy specimens is to differentiate normal from abnormal colitis, which can often be difficult because of the presence of bowel preparation and biopsy procedure artifacts. Artifacts include surface epithelial degeneration, edema, hemorrhage and congestion ( Figs. 17.1 and 17.2 ), aggregation of inflammatory cells, pseudolipomatosis (intramucosal air; Fig. 17.3 ), mucin depletion, and even neutrophilic cryptitis, among others. Pathologists should not be reluctant to render a diagnosis of normal colon if the histological features fall into one or more of the artifact categories, particularly if the patient is asymptomatic. After all, normal is the most common diagnosis in the general population. Terms such as nonspecific (or increased) chronic inflammation, nonspecific colitis, and increased acute and chronic inflammation are inappropriate pathological diagnoses that often cause confusion for clinicians and thus should be avoided.
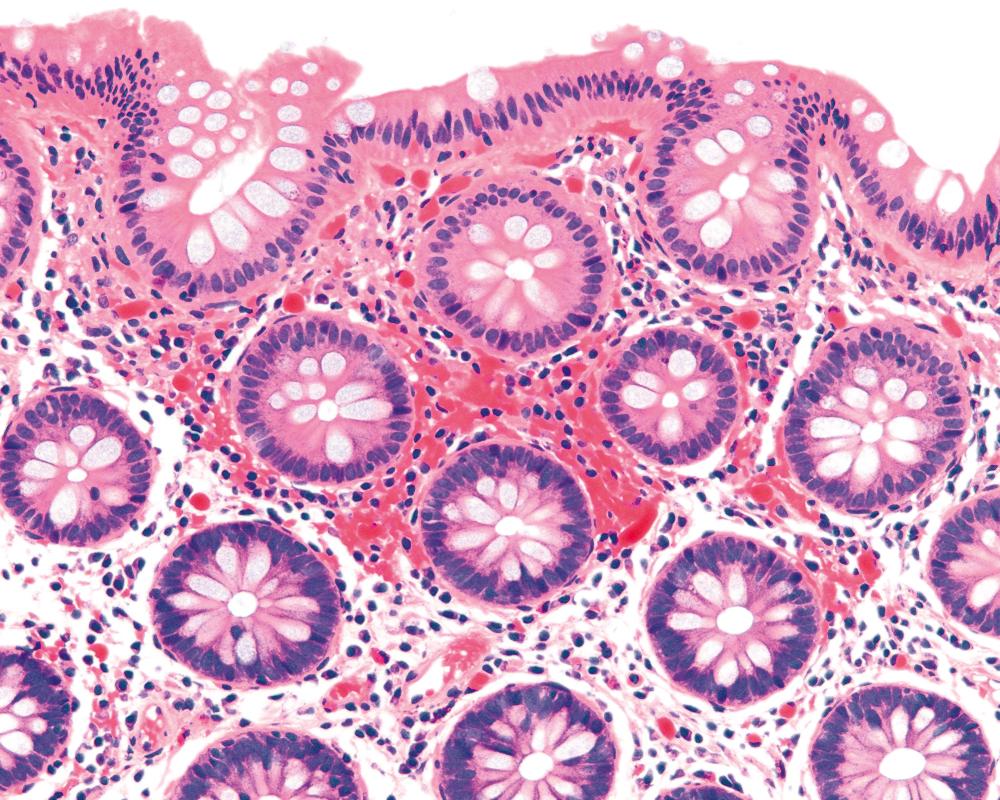
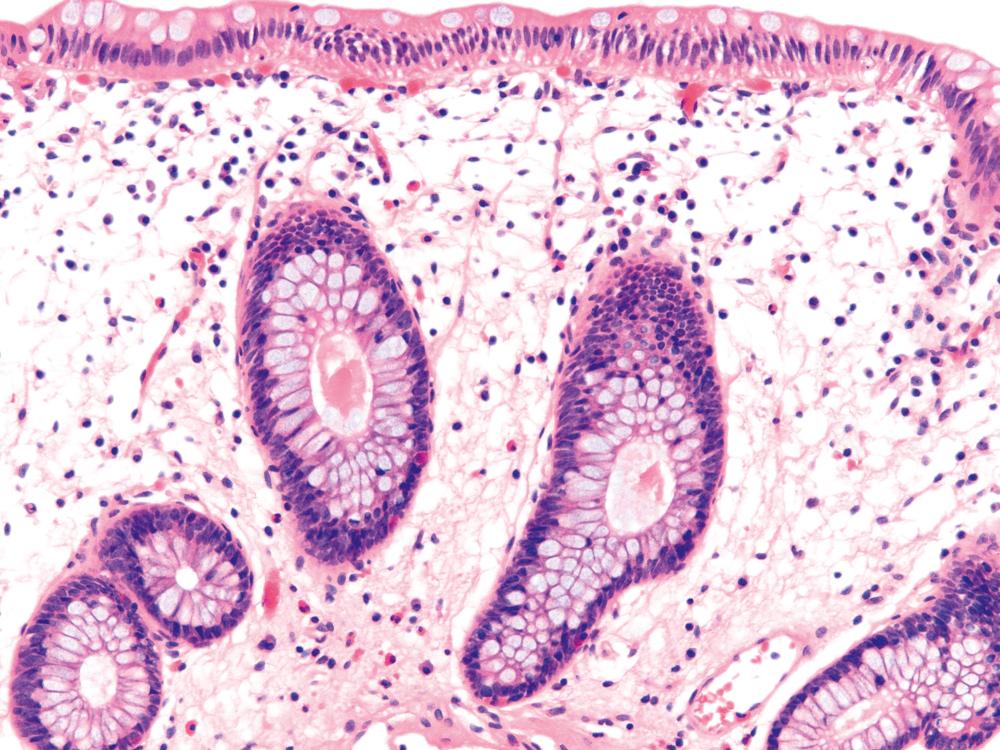
Pathologists should be aware of several important points when evaluating colorectal biopsy specimens, particularly with regard to histological findings that are considered normal. For example, lymphocytes and plasma cells are always present in the lamina propria of colorectal mucosa, regardless of the anatomic location. However, the density of lamina propria inflammatory cells varies among the different anatomic locations. In general, the cecum and right colon are more cellular than other segments of the colon. A progressive decrease in the cellular constituents of the lamina propria is normal from the right to the left colon ( Fig. 17.4 ). In addition, although the colonic crypts are mostly arranged in a straight and tubular configuration, show an even distribution, and typically extend to the level of the muscularis mucosae, the distal rectum may show some variation in crypt architecture under normal circumstances ( Fig. 17.5 ). Lymphocytes are normally present in the surface epithelium of the colorectal epithelium and number approximately 5 per 100 epithelial cells. Surface intraepithelial lymphocytes are generally more prominent in the cecum and right colon than in the remainder of the distal colon. In addition, intraepithelial lymphocytes are more numerous in areas of mucosa overlying lymphoid follicles ( Fig. 17.6 ). , Eosinophil counts also vary substantially in different portions of the colon (more in the right colon than in the left), and “normal” numbers depend on other factors, such as the geographic location and the latitude of the patient’s principal habitat. , Individuals who live in the southern states of North America, or closer to the equator, have a higher number of lamina propria eosinophils compared with individuals who live in more northern states. Knowledge of the anatomic location of the colonic biopsy specimens is also important, but this has become increasingly difficult in recent times mainly because gastroenterologists have a tendency to place biopsy specimens from different sites into one specimen container.
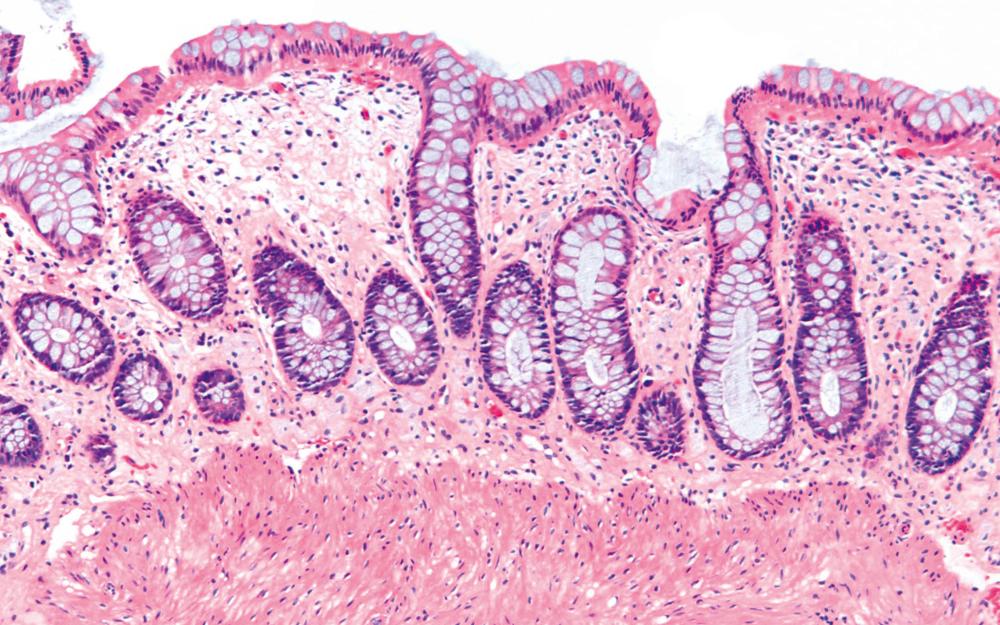
Evaluation of patterns of injury in colorectal biopsy specimens is often best performed at low magnification (see Chapter 13 ). For example, lamina propria cellularity and crypt architecture are easier to evaluate at low magnification than at high magnification. In addition, it is easier to compare histological changes among different fragments of tissue within the same specimen block under low-power examination.
After a biopsy is determined to be abnormal, distinction of acute from chronic changes is important clinically. The most consistent and reliable markers of chronic injury are crypt architectural distortion, basally located lymphoid aggregates, basal plasmacytosis, diffuse mixed inflammation, Paneth cell (or pyloric gland) metaplasia (or hyperplasia in biopsy specimens from the right colon), and lamina propria fibrosis ( Table 17.1 ). Many types of colitides, including acute infectious colitis, may result in expansion of the lamina propria by plasma cells; however, basal plasmacytosis, wherein plasma cells fill the space between the bases of the crypts and the muscularis mucosae, is an excellent histological marker of chronic colitis. This feature is also helpful to differentiate acute infectious colitis from acute-onset IBD in most circumstances. In IBD, features of chronicity are almost always present in the cecum, right colon, and proximal portion of the transverse colon ; even in these portions of the colon, an increase in the number and a change in the distribution of Paneth cells helps indicate and confirm the presence of chronic injury.
| Feature | Acute Colitis | Chronic Colitis |
|---|---|---|
| Crypt architecture | Preserved | Often distorted |
| Expansion of lamina propria | Usually superficial, predominantly neutrophils ± eosinophils | Diffuse (superficial and deep), mixed lymphocytes and plasma cells |
| Basal lymphoid aggregates | Usually absent | Often present |
| Basal plasmacytosis | Usually absent | Almost always present |
| Granulomas | Usually absent | Present in Crohn’s disease; related to crypt rupture in ulcerative colitis |
| Cryptitis and crypt abscesses | Present, superficial | Present, superficial and deep |
| Pyloric or Paneth cell metaplasia | Absent | Often present |
| Lamina propria fibrosis | Absent | May be present |
Features of active injury in the colon include, most importantly, regenerative and degenerative epithelial changes, even in the absence of increased inflammatory cells. However, often other inflammatory infiltrates are present. The most common are neutrophil- or eosinophil-mediated injury in the form of cryptitis, crypt abscesses, mucosal erosions, and ulceration. Edema and hemorrhage also may be prominent in some forms of colitis, and there are several other forms of active injury as well (see Chapter 13 ). These changes may be superimposed on a background of chronic colitis, in which case the diagnostic term chronic active colitis is used.
UC is a chronic, episodic inflammatory disease of the colon. It has a propensity to develop in adolescents and young adults, although there is a second incidence peak among middle-aged men. The incidence and prevalence rates of UC are highest in North America, England, northern Europe, and Australia. Estimates of the annual incidence of UC in North America and Europe range from 1.5 to 20.3 cases per 100,000 individuals. The incidence of UC appears to have stabilized during the past 25 years and is no longer increasing, unlike that of CD, which seems to be increasing in incidence. It has been estimated that UC will develop in approximately 1% of the U.S. and European population during their lifetime. There is marked ethnic variation in the incidence of UC, with a high incidence in the Jewish population. In the United States, the annual incidence of UC among Jewish individuals is 13 per 100,000 person-years, compared with 3.8 per 100,000 among non-Jewish whites. Recent data indicate a prevalence of 286 cases per 100,000 population in the United States. UC is more common in industrialized countries compared with less-developed countries, and in urban compared with rural populations. The incidence rate of UC among immigrants who have moved to high-risk geographic regions is higher than that of the same ethnic groups in their native countries.
The clinical symptoms of UC vary depending on the phase and extent of disease. They include urgency, passage of mucus, tenesmus and rectal bleeding in patients with proctitis, and diarrhea (mainly bloody), rectal bleeding, abdominal pain, fever, and weight loss in patients with extensive colitis. Among patients with fulminant colitis, symptoms include fever, generalized abdominal pain, rectal bleeding, and abdominal distention. Patients may also complain of symptoms related to anemia and hypoalbuminemia, such as fatigue, dyspnea, and peripheral edema. In general, the clinical symptoms correlate with the severity of disease. However, on occasion, there may be evidence of histologically or endoscopically active disease in asymptomatic patients. The onset of symptoms is typically slow and insidious. In most cases, patients are symptomatic for weeks or months before seeking medical attention. Some patients with UC present clinically more acutely and thus show symptoms that mimic acute infectious colitis. In fact, in some instances, infection such as with Salmonella or Clostridium difficile precedes an initial episode of UC (see Acute Self-Limited [Infectious] Colitis).
Extraintestinal manifestations of UC can affect any organ system but are most common in the skin, eyes, mouth, joints, and liver. Cutaneous hypersensitivity, photosensitivity, and urticarial rashes may occur in response to medical therapy (especially sulfasalazine) rather than the underlying disease itself. Erythema nodosum occurs in 2% to 4% of patients with UC. It manifests as single or multiple, tender, erythematous nodules on the extensor surfaces of extremities. Pyoderma gangrenosum is less common, occurring in 1% to 2% of patients with UC. The lesions may be single or multiple and may occur on the trunk, extremities, face, breast, and stoma sites. Less common skin manifestations include Sweet’s syndrome and oral aphthous ulcers.
The two most common ocular manifestations of UC are episcleritis and uveitis; these occur in 5% and 8% of patients, respectively. Seronegative arthropathy (type 1, pauciarticular or type 2, polyarticular) occurs in 5% to 20% of individuals with UC and is more common than axial arthropathy; the latter manifests as sacroiliitis and ankylosing spondylitis. In terms of liver involvement, most patients with UC have mild elevations of serum aminotransferase and alkaline phosphatase levels. The most important complication is primary sclerosing cholangitis (PSC), which occurs in almost 3% of patients with UC. Unlike all of the complications listed previously, which typically follow the colonic disease activity, PSC may follow an independent progressive course, even when UC has been stable or inactive for years, and it may appear before onset of UC as well.
Patients with mild or moderately severe disease usually exhibit minimal signs on physical examination. The affected portion of the colon may be tender on abdominal palpation, but abdominal rigidity or guarding is highly unusual. Severe (fulminant) colitis is usually associated with generalized abdominal tenderness, with either normal or hyperactive bowel sounds, which decrease with disease progression. Distention of the abdomen with absent bowel sounds is an ominous sign that suggests peritoneal irritation in cases of fulminant colitis. Other signs that may be associated with UC include aphthous ulceration of oral mucosa, clubbing of fingernails (typically in long-standing UC), peripheral edema, and mild perianal disease. Digital rectal examination is often normal but may occasionally reveal velvety and edematous mucosa. In addition to anemia that results from acute or chronic gastrointestinal (GI) blood loss, patients with UC are predisposed to hypercoagulability and its complications, such as deep vein thrombosis, pulmonary embolism, renal artery thrombosis, cerebrovascular accidents, mesenteric vein thrombosis (and consequent ischemic colitis), and coronary thrombosis.
Laboratory findings in UC depend on disease activity. Anemia, leukocytosis, thrombocytosis, increased erythrocyte sedimentation rate (ESR), elevated C-reactive protein (CRP) level, and hypoalbuminemia are typically associated with active disease. Stool cultures for organisms such as C. difficile, Campylobacter species, and Escherichia coli are usually performed to exclude an infectious cause or complication. Perinuclear antineutrophil cytoplasmic antibodies (pANCA) are positive in 60% to 80% of patients with UC. Immunoglobulin A (IgA) anti– Saccharomyces cerevisiae antibody (ASCA) is found in fewer than 1% of patients with UC, whereas IgG ASCA may be seen in as many as 20% of patients (see Ancillary [Serologic] Diagnostic Tests for Inflammatory Bowel Disease).
Radiological studies help provide a general assessment of the extent of disease and complications associated with UC. Plain radiographs are indicated in cases of severe UC to assess for the presence of intraperitoneal air. The finding of marked colonic dilation suggests fulminant colitis. Because the transverse colon is the least dependent part of the colon, a diameter larger than 5 cm is highly suggestive of toxic megacolon. In the earliest stage of UC, a double-contrast barium enema may show a fine granular appearance of the colon. With advanced disease, deep submucosal ulcers result in characteristic “collar-button” ulcers. Diffuse absence of mucosal haustrations, thumbprinting, and narrowing or shortening of the colon are some of the features associated with pancolitis. Computed tomography (CT) is not very helpful in detecting mucosal changes in early disease. However, in advanced UC, the hallmark finding is the presence of mural thickening. In almost 70% of patients with UC, CT with contrast reveals the classic target or double halo sign caused by inhomogeneous enhancement of the thickened bowel wall. Rectal narrowing and widening of the presacral space are typical findings of long-standing UC.
Assessment of disease activity and prognosis is based on clinical, endoscopic, or histological findings or a combination of these indices. Although it is not standardized, a widely accepted clinical classification is that of Truelove and Witts. Frequency of bowel movements, rectal bleeding, fever, tachycardia, anemia, and elevated ESR are used to classify disease activity as mild, moderate, or severe. Because this classification does not correlate with disease status in patients with limited colitis, a numerical disease activity score, known as the Sutherland index or Ulcerative Colitis Disease Activity index, is now more commonly used, especially in clinical trials. It combines scores from four components (stool frequency, rectal bleeding, sigmoidoscopic findings, and physician’s global assessment).
The exact etiology of UC still remains unknown. However, its pathogenesis is related to a combination of three major elements: genetic susceptibility of the host, immunity, and environmental factors. Although specific agents may incite an inflammatory response in a susceptible host, such agents have not yet been identified. However, studies have shown that luminal microorganisms, their metabolic byproducts, and interactions with normal epithelial structures play key roles in stimulating a host immune response in UC.
The observation that 10% to 20% of patients have at least one other affected family member lends support to the role of genetic factors in the development of UC. The strongest evidence of a genetic influence is derived from three European studies wherein 6% to 16% of monozygotic twin pairs had concordant UC, compared with 0% to 5% of dizygotic twin pairs. The lifetime risk of developing disease is higher among first-degree relatives of a patient of Jewish descent and among relatives of patients with early-onset disease.
A wide array of genes are responsible for conferring genetic susceptibility, disease specificity, and phenotype in patients with UC. Linkage analyses have demonstrated that chromosomes 1, 2, 3, 5, 6, 7, 10, 12, and 17 harbor susceptibility genes for UC. Specifically, the IBD2 locus on chromosome 12 has a strong association among families with UC. Additionally, the C3435T polymorphism of the human multidrug resistance 1 (MDR1) gene is also linked to susceptibility to UC.
Besides susceptibility genes, human leukocyte antigen (HLA) alleles also influence disease behavior in UC. A significantly increased frequency of HLA-A11 and HLA-A7 has been observed to occur in patients with UC. Specifically, HLA-DR1 (DRB1∗0103) has been associated with severe colitis. HLA-DR2 (DRB1∗1502) has been associated with UC in Japanese and Jewish populations. ,
Recent studies in patients with very-early-onset IBD and in those with both a family history of IBD and severe manifestations of the disease have helped identify rare genetic variants that interfere with biochemical cellular pathways that ultimately may lead to increased inflammation. For example, a rare mutation that affects the regulatory function of the X-linked inhibitor of apoptosis (XIAP) gene was found to result in early-onset refractory IBD in a 15-month old boy. Furthermore, mutations in the interleukin (IL)-10 receptor (IL10RA) gene region, which can ultimately manifest as early-onset IBD, have also been found to be characterized by Mendelian-like inheritance with highly penetrant variants. Nevertheless, these mutational events only account for 10% to 25% of IBD cases, and not all cases have been associated with these types of genetic abnormalities, which suggests that IBD is very likely a complex polygenic disease.
It is now widely accepted that continuous antigenic stimulation by commensal bacteria, fungi, or viruses leads to chronic inflammation in individuals who have defects in immunoregulation, mucosal barrier function, and microbial killing. The distal terminal ileum and colon contain the highest concentrations of bacteria (almost 10 12 organisms per gram of luminal content), and they are a source of constant antigenic stimulus to the host immune system. Animal studies have shown rapid development of colitis when germ-free HLA-B27 transgenic rats and IL-10–deficient mice are populated with normal specific pathogen-free bacteria. Administration of antibiotics effectively prevents and reduces the severity of colitis in these animal models. There are several postulated mechanisms by which gut flora may initiate or contribute to the development of colitis (see Immune Factors). By virtue of their ability to adhere to or invade the surface epithelium or to produce enterotoxins, microbial organisms stimulate production of inflammatory cytokines. Alteration of the balance between protective and harmful bacteria (e.g., Bacteroides species) reduces the concentration of short-chain fatty acids, which provide nourishment to colonocytes. The most frequently observed changes include a decrease of Firmicutes, an increase in Proteobacteria and Bacteroidetes, and an increase in mucolytic bacteria and sulfate-reducing bacteria, such as Desulfovibrio. An impaired mucosal barrier and inability to kill microbes because of impaired host defense mechanisms also contribute to hyperresponsiveness and production of high levels of inflammatory cytokines. Abnormal antigen processing, loss of tolerance, autoimmunity, and an abnormally excessive T-cell response are some other mechanisms that influence the severity of inflammation.
A T-cell α-chain receptor knockout mouse model of colitis has demonstrated lack of development of inflammation after appendectomy in animals at 3 to 5 weeks of age. Subsequent case-control studies on humans have also suggested that appendectomy may have a beneficial effect on the disease course. However, there have been no prospective studies confirming a possible protective effect.
The best-characterized environmental factor associated with UC is cigarette smoking. UC is more common in nonsmokers than in current smokers. In fact, the second incidence peak in middle-aged men may, in part, be linked to patients who have stopped smoking later in life. A recent prospective study of a cohort of 229,111 women who were followed over a period of 32 years (Nurses Health I and II cohort) showed that the risk of UC is highest during the first 2 to 5 years after cessation of smoking but remains elevated for more than 20 years. The postulated mechanisms for the protective effect of smoking include modulation of cellular and humoral immunity, increased generation of oxygen free radicals, and alteration of cytokine levels.
The use of medications, most notably antibiotics, has also been associated with an increased risk of IBD. This association may result from changes in the intestinal microbiome after use of antibiotics during early stages of life, when the microbiota plays a critical role in shaping immune cell development. Nonsteroidal antiinflammatory drugs (NSAIDs), contraceptives, and statins are medications that have been associated with a nearly twofold increased risk of UC. ,
The intestinal barrier, consisting of intestinal epithelial cells (IECs) and innate immune cells, maintains the equilibrium between luminal contents and the mucosa. Enterocytes, goblet cells, neuroendocrine cells, Paneth cells, and M cells each appear to play distinct roles in maintaining this equilibrium. Genetic deletion of Muc2 (mucin 2), a major goblet cell-derived secretory mucin, results in spontaneous colitis in murine models. Dendritic cells have been found to accumulate in the mucosa of patients with IBD. Blockage of CD40/CD40L interactions between dendritic cells and effector T-cell populations prevents experimental T-cell–mediated colitis.
Both humoral and cell-mediated immunological mechanisms play major roles in the pathogenesis of UC. UC is associated with an increase in the synthesis of IgG, notably the IgG 1 and IgG 3 subclasses. Most patients have circulating antibodies to a variety of dietary, bacterial, and self antigens that are of the IgG 1 subclass, and these are polyclonal in nature. Because serum antibody titers usually do not correlate with disease activity or course, it has been postulated that cross-reactivity between antibodies to bacterial antigens and colonocyte epithelial epitopes may help trigger an immunological response that leads to mucosal inflammation.
UC is associated with several autoimmune diseases such as diabetes mellitus, pernicious anemia, and thyroid disease. The possibility that UC is an autoimmune disease is supported by the fact that patients with UC have serum antibodies directed against lymphocytes, ribonucleic acid, smooth muscle, gastric parietal cells, and thyroid tissue. Patients also have antibodies to epithelial cell–associated components, notably an autoantibody against a 40-kDa epithelial antigen found in normal colonic epithelium. This IgG autoantibody was eluted specifically from colonic mucosa of patients with UC and was not found in patients with CD or other colonic inflammatory conditions. This antigen also shares epitopes with antigens found in the bile ducts, skin, eyes, and joints, sites that are commonly associated with extraintestinal manifestations of UC. The other autoantibody associated with UC is pANCA. It is found in 60% to 80% of UC patients and belongs to the IgG 3 subclass. The exact antigen to which pANCA is directed is unknown. There is some evidence that the antigen is a 50-kDa nuclear envelope protein that is specific to myeloid cells. The pathogenic relevance of pANCA is unclear. It appears to be associated with an aggressive disease course and development of pouchitis.
Bacterial antigens also trigger innate immunity by activation of pattern-recognition receptors, which include Toll-like receptors (TLRs) and nucleotide-binding oligomerization domain (NOD)-like receptors (NLRs). Activation of TLRs and NLRs results in downstream activation of nuclear factor κB (NF-κB), which further stimulates production of various proinflammatory cytokines and chemokines. Defects in any of these pathways can result in abnormal bacterial processing and possibly IBD.
Colonic epithelial cells express class II major histocompatibility complex (MHC) antigens and can initiate an inflammatory response by acting as antigen-presenting cells. Increased turnover of colonic epithelium, reduced metabolism of short-chain fatty acids, abnormal membrane permeability, and altered composition of mucosal layers contribute to the pathogenesis of UC. , Animal models of colitis produced by disruption of colonic epithelium further support the role of epithelial cells in the pathogenesis of IBD.
Release of various cytokines from the T-cell inflammatory pathways may also lead to increased epithelial cell permeability and alteration of the endothelium, contributing to diarrhea and localized ischemia, respectively.
In untreated cases, the extent of colonic involvement depends on the clinical severity of disease. UC classically involves the rectum with variable, but continuous, involvement of the colon more proximally ( Fig. 17.7 ). According to the Montreal classification, the extent of UC is divided into ulcerative proctitis (involvement limited to the rectum), left-sided or distal UC (involvement of the rectum and sigmoid but not beyond the splenic flexure), and extensive UC or pancolitis (involvement of the rest of the colon proximal to the splenic flexure). At the initial onset of disease, pancolitis occurs in approximately 20% of patients, left-sided colitis in 50% to 60%, and proctitis or rectosigmoiditis in approximately 45% of patients. Skip lesions, in the form of appendiceal, periappendiceal, or ascending colon/cecal involvement, have been observed in as many as 80% of patients with subtotal UC (see Unusual Morphological Variants of Ulcerative Colitis). Based on a long-term follow-up study by Farmer and colleagues, pancolitis eventually develops in almost one-half (46%) of patients with proctitis or rectosigmoiditis and in more than 70% of those with left-sided colitis.
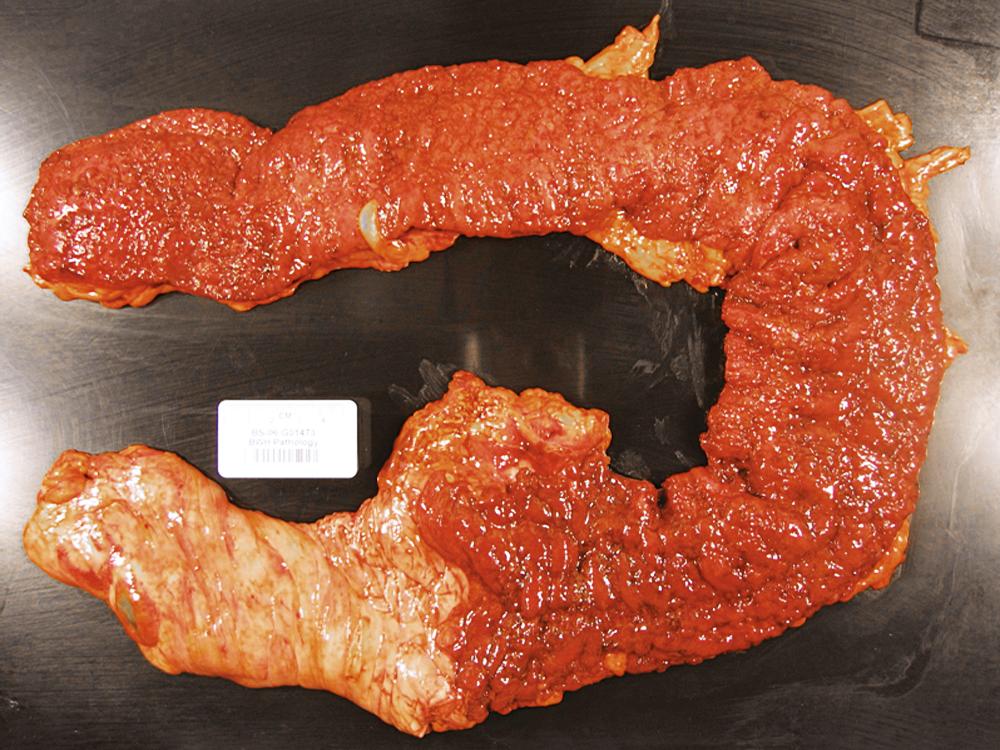
In the active phase of disease, the mucosa usually appears diffusely congested, granular, and edematous. Ulcers, when present, are usually small and oriented longitudinally in relation to the teniae coli. They often appear to undermine adjacent areas of mucosa, which leads to the formation of polypoid mucosal folds or inflammatory pseudopolyps. In such cases, the mucosa may have a cobblestone appearance, similar to that observed in CD. Rarely, an exaggerated form of pseudopolyp formation, known as filiform polyposis, may be present ( Fig. 17.8 ). It is characterized by the presence of elongated, slender, villiform, wormlike, polypoid mucosal projections and usually spares the rectum. In severe cases, ulcers may be extensive, involve large segments of bowel, and lead to near-total or total mucosal loss.
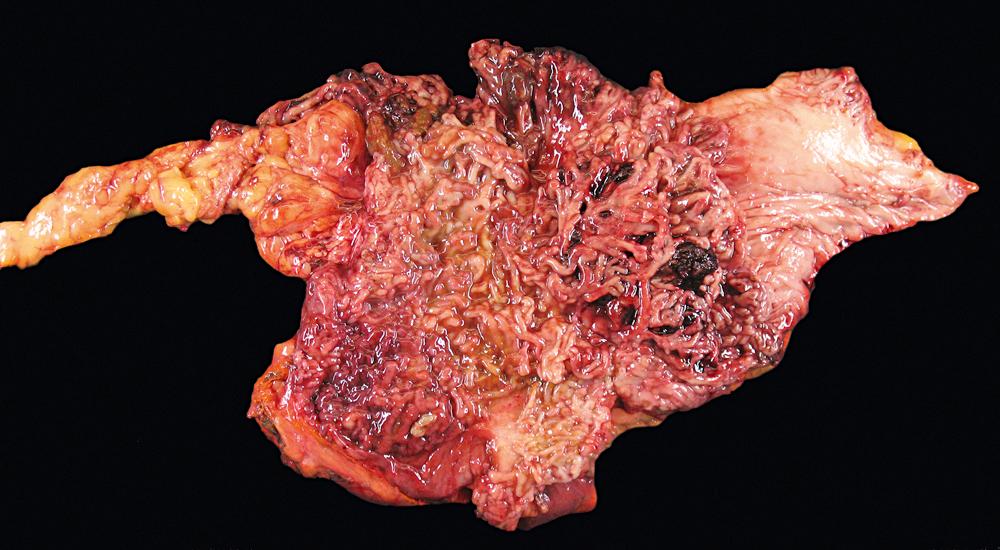
In cases of toxic megacolon, the bowel wall appears extremely thin, dilated, and congested. The serosal surface usually demonstrates fibrinous or fibrinopurulent exudate. Rarely, there is evidence of perforation. The mucosal surface in these cases is extensively denuded, hemorrhagic, ulcerated, and often covered with purulent exudates.
In the quiescent (inactive) phase of UC, the mucosa may appear completely normal, or it may show diffuse granularity, either with or without inflammatory pseudopolyps. In some cases of long-standing UC, the bowel wall is thickened and contracted (“colonic foreshortening”), and the mucosal surface may appear atrophic.
In treated UC, especially when patients have been given steroid enemas, the rectal mucosa may show minimal or no abnormalities on gross examination. Similarly, in patients who have received medical treatment before surgical resection, there may be focal, diffuse, or even widespread areas of grossly normal-appearing bowel between areas of affected bowel.
Depending on the phase of disease and the degree of inflammatory activity, UC is categorized histologically as chronic inactive, chronic active, or active (without features of chronicity) for the purpose of diagnostic sign-out. Chronic colitis (regardless of “activity”) is defined by the presence of histological features of chronicity ( Box 17.1 ), such as crypt architectural distortion, crypt atrophy, diffuse mixed lamina propria inflammation, basal plasmacytosis, basally located lymphoid aggregates, and Paneth cell metaplasia (in the left colon). Other changes of chronicity include lamina propria fibrosis, pyloric gland metaplasia, and Paneth cell hyperplasia in the right colon. Common changes of “activity” include neutrophilic or eosinophilic cryptitis, crypt abscesses, regenerative or degenerative epithelial changes, hemorrhage, necrosis, erosions, and ulceration.
Neutrophilic (or eosinophilic) cryptitis
Crypt abscesses
Necrosis
Regenerative and degenerative epithelial changes
Erosions
Ulcers
Crypt architectural distortion
Crypt atrophy
Crypt foreshortening
Irregular spacing of crypts
Irregular size of crypts
Crypt branching or budding
Loss of crypt parallelism
Villiform surface contour
Basal plasmacytosis
Basal lymphoid aggregates
Diffuse mixed lymphoplasmacytic infiltrate within the lamina propria
Paneth cell metaplasia in the left colon (hyperplasia in the right colon)
Pyloric gland metaplasia
Lamina propria or submucosal fibrosis
Thickening or duplication of the muscularis mucosae
Unfortunately, there is no universally accepted method of grading histological activity in biopsy specimens from patients with UC. At least 18 different histological scoring systems have been proposed to categorize activity, but these are mainly used in research protocols and do not have much relevance to clinicians when deciding how to manage their patients. Scoring systems that have been used in clinical studies range from use of stepwise methods, in which disease activity is divided into rather subjectively assessed grades, to others that prefer use of a more quantitative methods, by using numerical scores that correspond to specific histological features. For instance, the Riley scoring system uses a four-point score ( none, mild, moderate , and severe ) to assess six histological features: presence of an acute inflammatory cell infiltrate (neutrophils in the lamina propria), crypt abscesses, mucin depletion, surface epithelial integrity, chronic inflammatory cell infiltrate (round cells in the lamina propria), and crypt architectural irregularities. This scoring system was applied in a prospective study that was aimed at predicting recurrence in 82 outpatients with asymptomatic UC in endoscopic remission. This scoring system was later modified (Modified Riley score) to rank the degree of inflammation hierarchically, and to exclude crypt architectural changes, which according to the authors, are not responsive to clinically relevant changes in inflammation. This system has never been validated but has been used in multiple randomized control trials.
Geboes et al. developed a scoring system that categorizes histological changes as grade 0 (structural change only), grade 1 (chronic inflammation), grade 2 (2a, lamina propria neutrophils and 2b, lamina propria eosinophils), grade 3 (neutrophils in the epithelium), grade 4 (crypt destruction), and grade 5 (erosions or ulcers). This system generates a score from 0 to 5.4 that increases with disease severity or activity. The Geboes scoring system has been shown to be reproducible to some degree, where the authors found moderate to good interobserver agreement among three pathologists (kappa 0.59 to 0.70).
More recently, two histological scoring methods have been developed and validated: the Nancy index and the Robarts histopathological index (RHI). , Both of these scoring systems demonstrate mostly a high degree of intraobserver and interobserver agreement. The RHI was developed by scoring biopsies according to the Geboes score and then determining which features were reproducible and correlated best with the pathologists’ interpretation of disease severity using a visual analog scale. Four features were found to correlate best with overall disease severity, including lamina propria chronic inflammation, lamina propria neutrophils, neutrophils in the epithelium, and the presence of ulcers and erosions. The Nancy index is a stepwise 5-item method that assesses lamina propria lymphoplasmacytic inflammation, neutrophilic inflammation, and ulcers to arrive at the final grade.
One simple and reproducible type of grading system that is commonly used in clinical practice by the authors of this chapter is as follows. The degree of activity is graded as mild if less than 50% of the mucosa shows evidence of activity, and the lamina propria is only mildly expanded by inflammatory cells. It is graded as moderate if more than 50% of the mucosa shows activity and/or if the lamina propria inflammatory component is moderately or severely dense, but there are no surface erosions or ulcers. It is graded as severe if surface erosion or ulceration is present, regardless of the density of inflammation in the lamina propria or epithelium. In this simple classification, “activity” includes any of the features outlined earlier in this chapter, and in this manner, it takes into account the fact that patients who are in an active phase of disease clinically may not necessarily show peak neutrophil influx at that point in time (see Figs. 17.10 to 17.12 ).
Histologically, previously untreated UC in adults typically involves the colon in a diffuse and continuous manner, always beginning at the distalmost portion of the rectum and extending proximally to the point at which inflammation stops, which in most cases is rather abrupt ( Table 17.2 ). Typically, specimens from involved regions of the colon have a similar appearance. Usually, each biopsy fragment shows a homogeneous and diffuse pattern of injury, although the severity of inflammation may vary from region to region in the bowel (usually worse distally) or between individual biopsy fragments from one area of the colon, especially posttreatment (see discussion on posttreatment features later in this chapter).
| Untreated Ulcerative Colitis | Crohn’s Disease |
|---|---|
| Diffuse, continuous disease | Segmental disease |
| Rectal involvement | Variable rectal involvement |
| Disease worse distally | Disease severity is variable from segment to segment |
| No fissures (except in fulminant colitis) | Fissures, sinuses, fistulous tracts common |
| No transmural aggregates | Transmural lymphoid aggregates |
| Either no or only mild distal ileal involvement (less than 5 cm in general) | Often moderate to severe ileal involvement, either segmental or diffuse and usually more than 5 cm |
| Upper GI tract involvement (stomach, duodenum) less common | Upper GI tract involvement (esophagus, stomach duodenum, jejunum) much more common |
| Anal disease less common | Anal tags, fissures and fistulae are more common |
| Granulomas are less common, and when present are crypt-rupture (mucin) associated | Epithelioid granulomas unrelated to ruptured crypts common |
| Appendiceal involvement is more common and resembles chronic active colitis seen in the rest of the colon | Appendiceal involvement is less common |
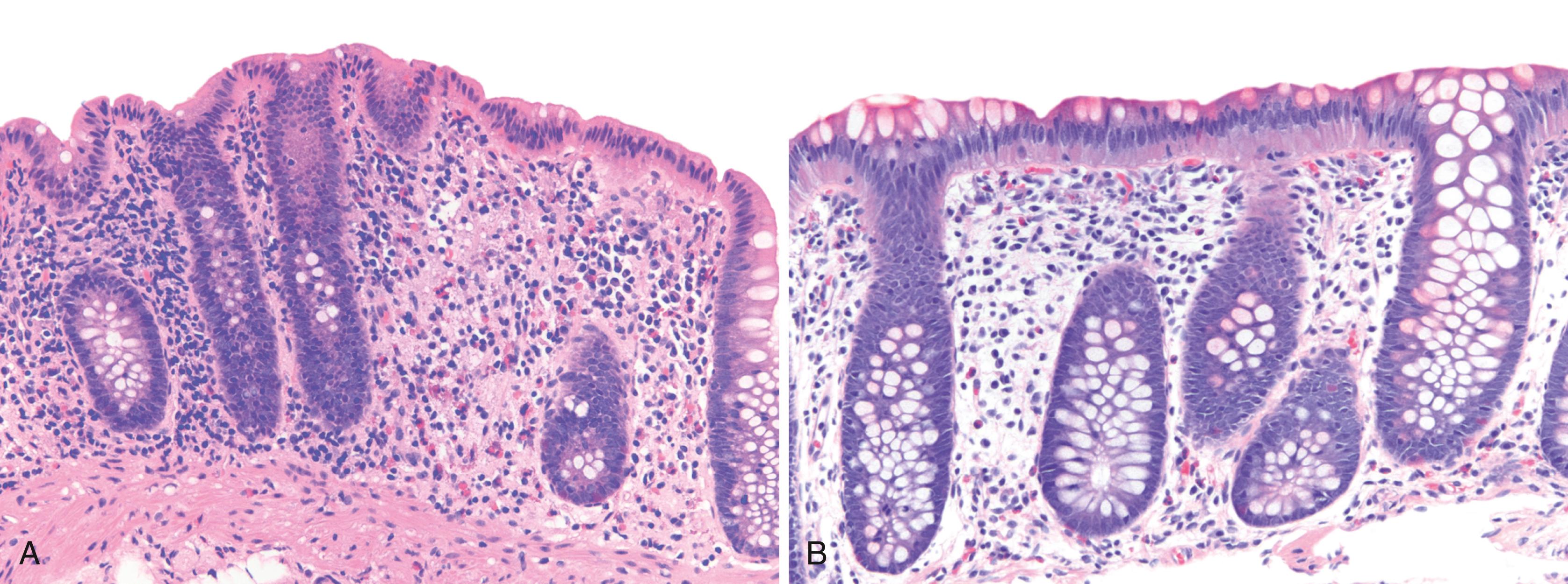
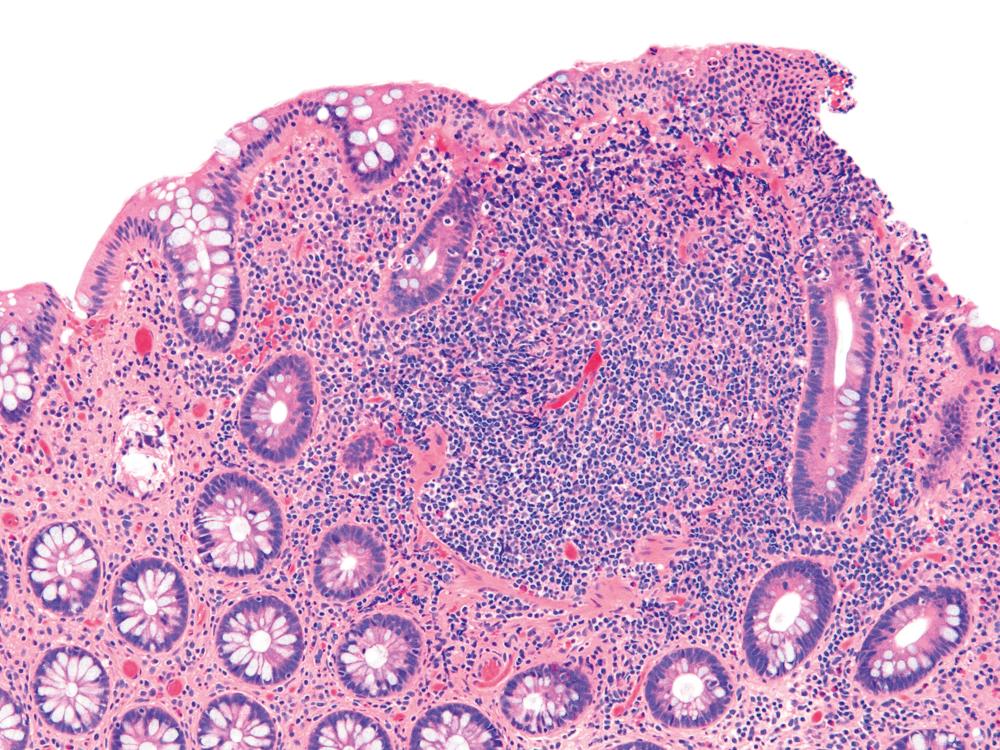
UC characteristically involves the mucosa and sometimes involves the superficial submucosa as well ( Fig. 17.9 ). The histological findings vary depending on the clinical phase of disease. Ultimately, in periods of clinical activity, UC is predominantly a lymphoplasmacytic inflammatory process with superimposed neutrophils, hemorrhage, and epithelial degeneration and regeneration. A dense, homogeneous lymphoplasmacytic infiltrate typically expands the lamina propria ( Fig. 17.10 ). The density of plasma cells is usually greatest in the basal region of the lamina propria (termed basal plasmacytosis ). Basally located lymphoid aggregates (situated between the bases of the crypts and the muscularis mucosae) are also common. They may show germinal centers. Expansion of the lamina propria and the presence of basal lymphoid aggregates contribute to irregular spacing of the crypts.
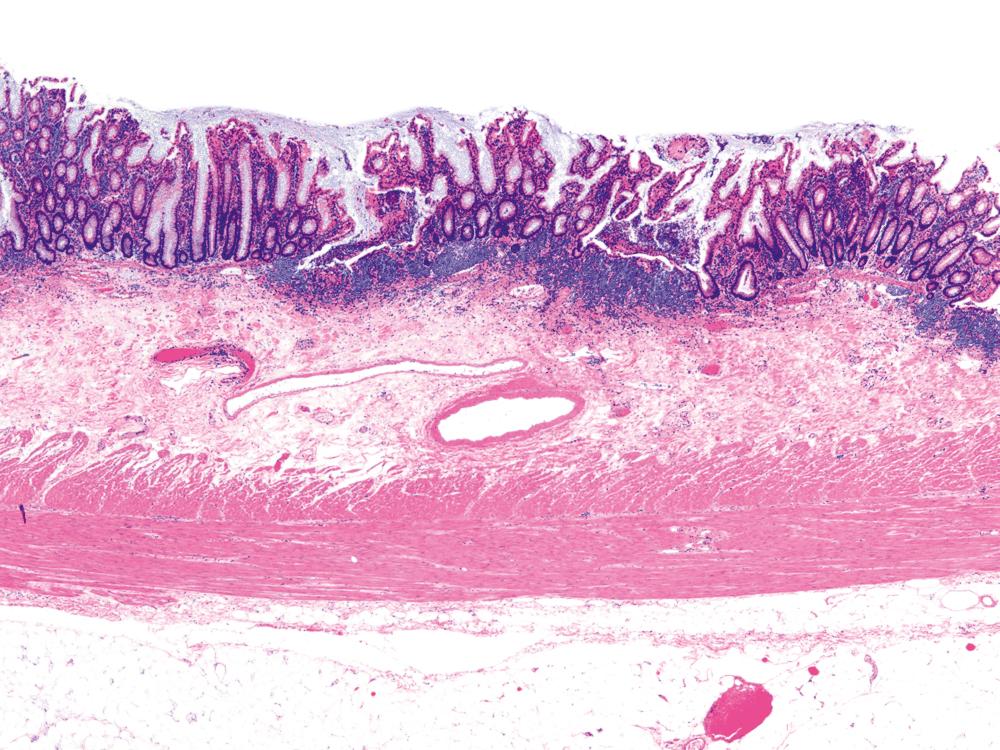
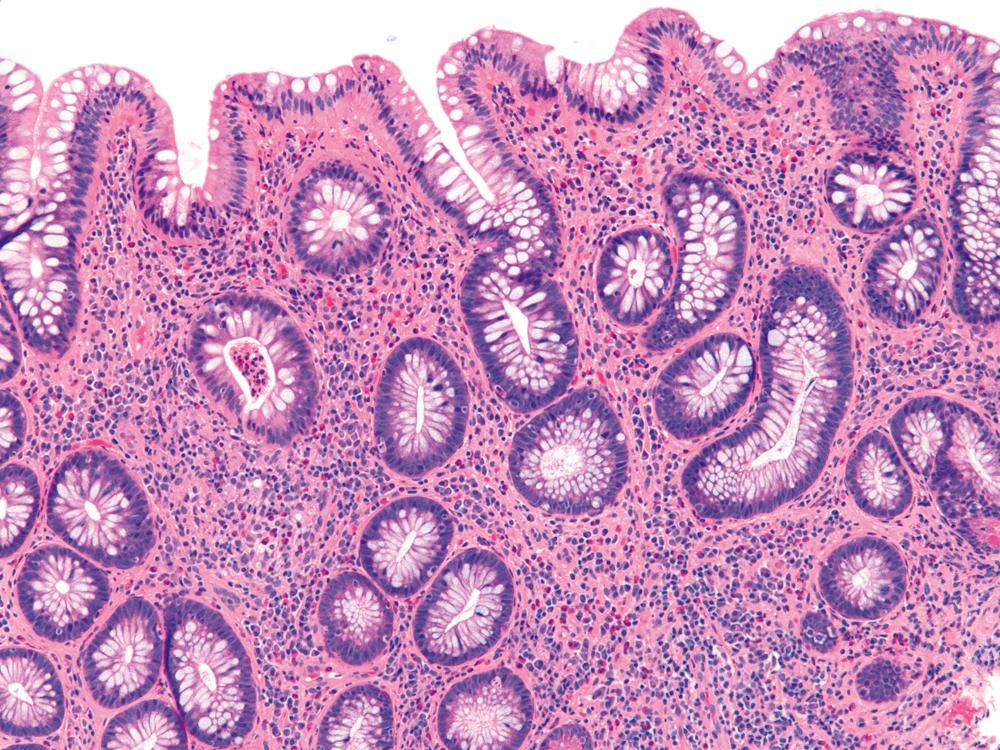
One characteristic and frequent morphological feature of UC is crypt architectural distortion, which is characterized by irregularly arranged, branched, dilated, and/or shortened crypts. In some circumstances, branching and shortening of crypts represents morphological manifestations of crypt regeneration. Most patients with new-onset (pretreatment) UC have experienced several weeks to months of subclinical or minimal inflammation, during which time the lamina propria has been inflamed, plasma cells have congregated in the basilar region of the lamina propria, and significant crypt injury with regeneration has occurred. Crypt architectural distortion is considered a hallmark of chronic injury. However, it may develop in any inflammatory disease of the colon that manifests as periods of repeated bouts of injury and repair before full mucosal healing. Thus when the inflamed mucosa is repeatedly exposed to bouts of injury followed by repair, the epithelium does not always heal with perfectly well-formed and aligned crypts. Crypt distortion is therefore not a specific feature of UC (or CD). It occurs in many other types of disorders, such as chronic recurrent ischemia, persistent or recurrent infections (e.g., C. difficile ), radiation colitis, drug-induced colitis, graft-versus-host disease (GVHD), and even microscopic colitis. Furthermore, not all patients with UC, even in an active phase of disease, reveal crypt distortion in every portion of the colonic mucosa. For this reason, absence of crypt distortion does not necessarily rule out a diagnosis of UC.
Depending on the severity of active disease, a neutrophilic inflammatory cell infiltrate (with or without eosinophils) may be seen within the lamina propria and/or in the surface and crypt epithelium (cryptitis); it may be minimal, focal and patchy, or diffuse and severe. Similarly, aggregates of neutrophils within the crypt lumina (crypt abscesses) may be focal or diffuse ( Figs. 17.11 and 17.12 ). Rupture of crypts caused by inflammation can lead to the development of aggregates of histiocytes, foreign-body giant cells, and even well-developed granulomas as a response to extravasated mucin ( Fig. 17.13 ). These mucin granulomas (crypt rupture–associated granulomas) are usually present in the deeper portions of the mucosa, where crypts tend to rupture more often compared with the superficial mucosa. Often, deeper cuts through tissue blocks are necessary to determine whether or not a granuloma is related to a ruptured crypt. Some patients show a marked foreign-body–type giant cell response or even a fully developed granulomatous response, which can be seen at the base of the mucosa. Distinguishing granulomas in UC from those in CD can be challenging, but the latter are more often randomly located in the mucosa, are more often superficial in location, and are typically unrelated to injured crypts (see Crohn’s Colitis). When a mucosal granuloma is identified, serial sections should be evaluated to determine whether it is located immediately adjacent to a ruptured crypt.



In areas of activity, the surface and crypt epithelium always shows regenerative and/or degenerative changes. Features of regenerative changes include loss of mucin, enlarged and variably sized nuclei either with or without nuclear stratification, hyperchromasia, prominent nucleoli, and increased mitotic activity. Features of degenerative changes include cytoplasmic vacuolization and heterogeneity, nuclear fragmentation and pyknosis, and lack of mitotic activity. In addition, the crypts may show increased apoptotic activity. In areas adjacent to erosions and ulcers, degenerative cells may acquire a syncytial appearance, with abundant and prominent eosinophilic cytoplasm ( Fig. 17.14 ). In some instances, the syncytial epithelium overlies stroma that is devoid of crypts and contains actively inflamed granulation tissue. Regenerating surface epithelial cells, which are cuboidal initially and then columnar with maturation, acquire a slightly more basophilic cytoplasm. On occasion, the surface epithelium may be villiform in appearance, resembling small-intestinal mucosa. Regenerative changes may mimic dysplasia (see Dysplasia in Ulcerative Colitis and Crohn’s Disease). Mucin reappears slowly, first in cuboidal cells and later in goblet cells. With time, goblet cells may become numerous.

Paneth cell metaplasia in the left colon and pyloric gland metaplasia are reliable histological indicators of chronic injury. In active disease, even the ascending colon, cecum, and transverse colon may show irregularity in the distribution and increased numbers of Paneth cells ( Fig. 17.15 ). Pyloric gland metaplasia is less common in UC than in CD. It is more commonly observed in samples from the proximal colon and is often seen in close proximity to ulcerated mucosa.

Occasionally, the rectum is completely free from inflammatory disease (absolute rectal sparing) or shows less activity than in the proximal colon (relative rectal sparing). This phenomenon is usually caused by the effects of prior medical treatment, either orally or, more commonly, with enemas (e.g., steroid enemas). However, on careful inspection, one often finds one or more subtle features of chronicity, which is evidence of prior inflammation and injury.
Grossly normal–appearing colonic mucosa proximal to regions of active colitis may also reveal a spectrum of abnormalities. Most commonly, there is a mild lymphoplasmacytic infiltrate within the lamina propria. However, the crypts are usually more evenly arranged and lack distortion. A few neutrophils may be present in the lamina propria or occasionally within a crypt, but neutrophilic crypt abscesses are rare. Eosinophils may also be increased and may produce small eosinophilic crypt abscesses on occasion. In the context of a patient with classic UC in the more distal colon, these changes in the proximal bowel are a reflection of the underlying inflammatory disorder.
Most patients with UC eventually enter a resolving, or healing, phase of disease, characterized by decreasing activity (and symptoms) after an active colitis episode. This phase of disease is characterized morphologically by less activity and less crypt injury, but higher levels of crypt regeneration and remodeling ( Fig. 17.16 ). Injured crypts typically heal from the base of the mucosa progressively upward toward the luminal surface. Neutrophils and other active components of crypt injury decrease first, followed by a reduction in lamina propria lymphocytes and plasma cells. During this initial healing phase, there is often much variability in the type and degree of mucosal inflammatory changes within biopsy fragments from different regions of the colon and even within individual fragments of mucosa from a single site. Neuroendocrine cell hyperplasia occurs in some patients, whereas others develop prominent lymphoid follicles that are more common in the distal colon and rectum (follicular proctitis). Follicular proctitis appears to identify a subgroup of patients who have a less favorable response to medical therapy. ,

The resolution period of UC, characterized by decreasing activity and increasing repair, may last for several weeks or months. Thereafter, UC patients may be symptom free for variable but often long periods. This is the “inactive” or “quiescent” period of disease. During this time, the mucosa may be completely normal, or it may show various degrees of chronic inactive disease (mainly crypt distortion), either with or without mild patchy activity ( Fig. 17.17 ). Architecturally distorted crypts represent a” biomarker” of prior bouts of colitis. Alternatively, the colon may heal completely and thus appear normal histologically. The rate at which this occurs is variable among patients. For instance, patients with only mild active colitis of short duration can show complete restitution of architecturally normal mucosa within several months after the initial active episode. The pace of crypt remodeling is usually slow in most patients; such remodeling occurs typically over many weeks to months.

A summary of the causes of unusual morphological patterns of disease in UC is provided in Box 17.2 . It is important that pathologists recognize these variants so they can avoid falling into diagnostic traps and misdiagnosing UC as CD. This section provides a summary of many of the causes of Crohn’s-like changes that may occur in UC as a result of a variety of factors such as treatment effect, age of the patient, systemic manifestations of disease, or simply a result of long-term waxing and waning of inflammatory activity. These include most commonly segmental or patchy involvement of the colon, rectal sparing, skip lesions, ileal or upper GI tract inflammation, aphthous ulceration, and granulomas, among others.
Effects of oral or topical therapy
Low-grade disease in remission
Cecum or ascending colon inflammation in left-sided colitis
Appendiceal involvement as a skip lesion
Pediatric ulcerative colitis (initial presentation)
Ileitis in ulcerative colitis (backwash ileitis)
Upper gastrointestinal tract involvement (e.g., duodenitis)
Crohn’s-like aphthous ulcers in ulcerative colitis
Granulomas (usually related to crypt rupture)
Fulminant colitis
Some UC patients with either subtotal or limited left-sided colitis may show patchy, mild, chronic, or active inflammation in the cecum or ascending colon that may be falsely interpreted as CD because of the impression of segmental involvement of the colon, which is a classic manifestation of CD in the colon. Up to 65% of patients with UC have limited left-sided involvement initially. However, proximal extension occurs in 29% to 58% of such patients. , , For instance, in one study by D’Haens and colleagues of 20 patients with established left-sided UC, 6 showed a sharp demarcation between affected and unaffected portions of colon, whereas 14 showed a more gradual transition. The area of transition in such cases may appear somewhat patchy, giving a false impression of skip lesions. Seventy-five percent of this latter group of patients showed an area of inflammation in the cecum, primarily in the periappendiceal mucosa, that was separate from the distal inflamed segment. In a study by Mutinga et al., 14 patients with both left-sided UC and pathologically confirmed patchy right-sided chronic inflammation were compared with 35 control patients who had limited left-sided UC only. The two groups had similar demographic features, extraintestinal manifestations, severity of disease, prevalence of progression to pancolitis, and natural history, which suggests that patchy right-sided inflammation in patients with left-sided colitis has little clinical significance. This phenomenon should be recognized by pathologists so a false diagnosis of CD can be prevented when there is an initial suspicion for segmental disease in the colon.
In a prospective study of 271 patients with UC, including 63 with inactive left-sided or subtotal colitis, periappendiceal cecal mucosal involvement was identified in 32% of patients. Similarly, since the original description by Davison and Dixon in 1990 of “discontinuous involvement” wherein the appendix was found to be inflamed in 21% of 62 cases of patients with only distal colonic UC, several other studies have shown that the appendix may be involved as a “skip lesion” in this disease, , although at least one other study failed to confirm this finding. In another study by Groisman et al., ulcerative appendicitis was present in 86% and 87% of patients with “nonuniversal” and “universal” UC, respectively. Their study included two cases with limited left-sided involvement combined with appendiceal involvement. Overall, the role of the appendix in UC is poorly understood. Patients with prior appendectomy have been shown to have a lower risk for UC. , In one study, the severity of appendiceal inflammation (ulceration) in patients with UC was a strong predictor of the development of pouchitis after a total proctocolectomy and ileoanal pouch procedure. In summary, involvement of both the appendix and the cecal or ascending colon can occur in patients with subtotal colitis. This phenomenon should be recognized by pathologists as an acceptable potential skip lesion in UC.
Several studies have shown that pediatric patients with untreated UC at initial clinical presentation may show evidence of relative, or even complete, rectal sparing or even patchy colonic disease on biopsy studies. Markowitz et al. reported 12 pediatric patients with untreated UC, 5 (42%) of whom showed patchy, mild active inflammation and mild crypt changes in the rectum, but diffuse involvement in the more proximal regions of the colon. In fact, one patient had a completely normal rectal biopsy specimen. A study by Glickman et al. compared the rectal mucosal biopsy appearance of 70 pediatric patients who had UC with that of 44 adult patients, all at initial presentation before medical treatment. Compared with adults, the pediatric patients showed significantly fewer cases of chronic active disease and a greater number of patients with microscopic skip areas and relative rectal sparing. In this study, 2 of the 70 pediatric patients had completely normal rectal biopsy specimens at initial clinical presentation, in contrast with none of the adult patients. Thus an absence of features of chronicity or the presence of mild active disease and microscopic skip areas at initial presentation in pediatric patients should not exclude a diagnosis of UC. In adults, relative (but not absolute) rectal sparing (i.e., less severe inflammation in the rectum compared with the proximal colon) may be seen, rarely, at initial presentation before treatment as well. , In one study, a 31% prevalence rate of relative rectal sparing was noted in a series of 46 adult patients with UC at initial presentation, but even in those cases, histological features of chronicity were almost always present in the rectal mucosa at the time of initial diagnosis.
Patients with UC, and particularly those with involvement of the cecum and ileocecal valve, may also show active inflammation in the distal few centimeters of the terminal ileum; a condition that has been historically termed backwash ileitis (BWI). The hypothetical mechanism for this phenomenon is that when inflammation of the proximal cecum/ileocecal mucosa is severe enough, it may cause malfunction of the ileocecal junction (ICJ), retrograde flow of colonic contents into the distal terminal ileum, and secondary inflammation of the terminal ileum as a result of a “toxic” reaction to the luminal refluxate. , This theory was originally proposed in the early 20th century by Crohn and Rosenak in an effort to explain the observation that some patients had a previously undescribed “combined form of ileitis and colitis,” a constellation of findings that was unusual in “regional enteritis” or “ulcerative colitis.” In a more contemporary study of the ileum in UC patients, Haskell et al. reviewed 200 UC resection specimens and found active ileitis in 17% of cases; in most cases, the inflammation was confined to the distal 1 cm of the ileum. Ninety percent of cases consisted of mild, patchy neutrophilic inflammation in the lamina propria, focal cryptitis or crypt abscesses, and patchy villous atrophy and regenerative changes ( Fig. 17.18A ). In rare instances, surface ulceration or even pyloric (mucous gland) metaplasia was present as well (see Fig 17.18B ). Most interestingly, in their study, a “backwash” mechanism for ileitis in UC was questioned because some patients showed a conspicuous lack of cecal involvement. The authors of that study suggested that ileitis may have developed as a result of drug toxicity (such as NSAIDS) or perhaps as a result of the bowel preparation procedure, rather as a result of backwash. More recently, in a critical evaluation of the literature on BWI by Patil et al., the concept of BWI was discarded in favor of a theory that ileitis in UC patients may more likely be related to primary involvement of the underlying disease, rather than as a result of mechanical backwash of colonic contents into the ileum. The originally proposed backwash theory has never been proven experimentally and, in fact, was postulated during a time when little was known regarding the broad pathological manifestations of CD in the GI tract, including the colon, or the variability of involvement of the upper GI tract in either CD or UC. Regardless, most cases of ileitis in UC, regardless of the precise etiology, show only mild inflammation. Thus when a patient with presumed UC shows more severe ileal findings, such as deep or even fissuring ulceration, abundant submucosal inflammation, non–crypt-rupture related granulomas, or long lengths of ileal involvement (generally >5 cm), a strong suspicion for CD should always be entertained, particularly when the patient does not have inflammation of the proximal cecum and ileocecal valve.

In a large case-control study, Arrossi et al. evaluated pouch outcome in patients with UC and in those with IBD of the indeterminate type and reported that the presence of BWI was not a significant risk factor for the development of pouchitis. This has been confirmed in other studies as well.
Previous studies have suggested that the condition referred to as backwash ileitis may be associated with an increased risk of colonic adenocarcinoma. For instance, in a study by Heuschen et al., 590 UC patients were classified into those with and without BWI, or limited left-sided colitis and evaluated for the presence of colorectal cancer (CRC) in resection specimens. Twenty-nine percent of 107 BWI patients had CRC compared with 8% and 1.8% of patients without BWI or left-sided colitis, respectively. However, one of the major limitations of this study was that cancer was an indication for proctocolectomy in 25% of their BWI cohort compared with 8% of patients without BWI. Therefore the conclusion regarding the causal association of BWI and CRC cannot be justified based on the data presented in this study. Larger studies performed over the past 20 years , , have shown no significant association between BWI and risk of CRC. In a retrospective case-control study by Navaneethan et al., 178 proctocolectomy specimens with BWI were compared with 537 controls to evaluate the association of BWI and colonic neoplasia. In patients with BWI, colon cancer was seen in 18% of patients in contrast with 12% and 9% in those with extensive colitis without BWI or left-sided colitis, respectively. Within the BWI group with colon cancer, low-grade dysplasia and high-grade dysplasia was noted in 4% and 8% cases, respectively. With dysplasia as the end-point, multivariate analysis revealed that BWI with extensive colitis was associated with an increased risk of any colonic neoplasia (odds ratio [OR] = 3.53; 95% confidence interval [CI] 1.01–12.30, P = 0.04). It is important to note that dysplasia/colon cancer was an indication for colectomy in 13% cases with BWI and 10% cases without BWI. Unfortunately, the retrospective nature of this study precludes evaluation of the causal association of BWI and colorectal neoplasia.
There are other factors that may lead to the development of active inflammation in the distal ileum (“active ileitis”) in patients with UC. These include various infections, drugs (e.g., NSAIDs), and, most commonly, the effects of certain bowel preparation agents. For instance, in a prospective study consisting of 50 patients with active UC, 16% had ileal inflammation, but without involvement of the cecum, indicating that backwash was surely not the cause of the ileitis (“non-BWI”). These patients revealed higher levels of ileal inflammatory cytokines (IL-6, IL-8, and tumor necrosis factor-α [TNF-α]) and also had extraintestinal manifestations of UC (e.g., arthritis, pyoderma gangrenosum) at the time of clinical presentation. This condition should not be confused with CD of the terminal ileum. In CD, the distal ileum typically shows longer lengths of involvement (often >5 cm) and is associated with histological features of chronicity, such as pyloric gland metaplasia, ulceration, and established radiological abnormalities.
Gastric and/or duodenal involvement may rarely occur in patients with clinically and pathologically confirmed UC. For instance, Valdez et al. described four patients with chronic active inflammation in the duodenum similar in appearance to the patients’ colonic disease. In another study, five patients had chronic active gastritis, and four had chronic active duodenitis; in these cases, the upper GI findings resolved after colectomy. Several similar cases have been reported in the Japanese literature. In a study by Lin and colleagues, esophageal, gastric, and duodenal biopsies from 69 patients with proven UC were compared with those of 97 control subjects. The most common pattern of inflammation in the upper GI tract was “focal gastritis,” followed by the presence of mixed inflammation in the basal aspect of gastric mucosa, and superficial plasmacytosis ( Fig. 17.19 ). Diffuse chronic duodenitis was observed in 10% of UC patients, and these changes persisted after colectomy. The authors did not find specific mucosal changes in esophageal biopsy specimens from patients with UC. Until such cases have been followed for longer periods, it is difficult to know with certainty whether the finding of upper GI inflammation in UC represents a primary manifestation of UC or simply an unrelated, coincidental inflammatory disorder. Ultimately, more precise characterization of these types of UC cases will likely require long-term follow-up to help establish specific criteria of upper GI involvement in patients with UC.

Although aphthous type ulcers are common in CD, they may occur in other types of colitides and even in UC, as well. For instance, they have been reported in infectious colitis, diverticular disease–associated colitis, and diversion colitis, among other conditions. In one study, aphthous-type ulcers were present in 17% of UC resection specimens. In this study, manifestations of CD did not develop in any of the patients, nor was the presence of aphthous ulcers associated with the subsequent development of pouchitis on follow-up.
Some cases of active UC, particularly the more severe ones, may develop granulomas in the deep mucosa, and these are most often associated with ruptured crypts (see Fig. 17.13 ). This finding may be prominent in some UC patients and may, in fact involve several contiguous crypts in the deep mucosa. Granulomas may also develop in UC from degenerated collagen, particulate matter, superimposed infections, or as a result of a drug reaction, therefore not all are related to ruptured crypts. In equivocal cases, “mucin granulomas” related to ruptured crypts may be distinguished from primary granulomas in CD by the use of histochemical stains or by evaluation of multiple deep tissue sections to find the ruptured crypts. In general, granulomas located in the superficial aspects of the mucosa (and certainly in the submucosa) are more likely to be CD-related compared with basally located mucosal granulomas, which are more often crypt rupture–related in etiology.
Current therapeutic strategies for UC are separated into those that treat active disease (induction therapy) and those that prevent disease recurrence once remission has been achieved (maintenance therapy). Medical therapy focuses on agents that alter the host’s immune response in an effort to decrease mucosal inflammation. First-line therapy consists of oral 5-ASA preparations such as sulfasalazine, Pentasa, Asacol, and balsalazide. Sulfasalazine induces remission in 39% to 62% of patients with mild to moderate UC. Topical 5-ASA enemas can be used to treat disease located as much as 20 cm from the anal verge. Moderate to severe flares of UC are treated with systemic glucocorticosteroids. Azathioprine and 6-mercaptopurine are two purine analogue immunomodulators that interfere with nucleic acid metabolism and exert a cytotoxic effect on lymphocytes. Cyclosporine A, another potent inhibitor of cell-mediated immunity, is primarily indicated in patients with severe steroid-refractory disease.
Several recent advanced biological therapies and small molecules have shown clinical efficacy in patients with moderate to severe colitis who have previously failed to improve with corticosteroids and/or thiopurines. These therapies include anti–TNF agents like infliximab, adalimumab, and golimumab, anti-α 4 β 7 -integrin antibody (vedolizumab), anti-IL-12/IL-23p40 antibody (ustekinumab), and small-molecule antibody preferentially targeting JAK1 and JAK3 (tofacitinib).
The goal of medical therapy is to achieve clinical remission. In current clinical practice, disease activity is monitored by assessing patients’ clinical symptoms and severity of colonic inflammation by colonoscopy. However, there is evidence to suggest that endoscopic findings do not necessarily correlate with histological disease, especially after treatment. ,
In patients who have received medical therapy (oral or enema), mucosal histological changes can vary considerably. Portions of mucosa may heal completely, whereas others may still be active. Healing occurs in a segmental or patchy fashion. This pattern of healing gives an impression of segmental or patchy disease (skip lesions), which may be mistaken for CD. In this circumstance, exceptions to the classic principles of UC pathology may lead to diagnostic confusion.
Classic teaching emphasizes that UC is characterized morphologically by the presence of diffuse fixed architectural or cellular mucosal changes (or both) that categorize the process as chronic. However, in 1993, Odze and colleagues prospectively evaluated 123 rectal mucosal biopsy specimens from 14 patients with pathologically confirmed UC treated with either 5-aminosalicylic acid (5-ASA) or placebo enemas. During the course of treatment, 29% of rectal biopsies from 64% of patients were histologically normal, showing no evidence of chronic or active disease. Patients treated with 5-ASA showed a significantly higher percentage of normal biopsy specimens (obtained from areas of mucosa previously shown to be involved with chronic active disease) than did the placebo group. This was the first report to demonstrate that “fixed” chronic features in UC may revert to normal in the natural course of the patient’s illness and that topical therapy may enhance this phenomenon. Subsequent studies by Kleer and Appelman, Bernstein and colleagues, and Kim and colleagues, all of whom evaluated patchiness of disease and patterns of involvement in UC colorectal biopsy specimens with time, confirmed and expanded the initial findings of Odze’s group.
In these studies, 30% to 59% of patients, some of whom were treated with oral sulfasalazine or steroids (or both), showed either patchiness of disease or rectal sparing on follow-up surveillance biopsies. Awareness of these data should prevent misinterpretation of the findings of a normal rectal biopsy specimen or patchiness of disease in medically treated patients with UC as evidence against this diagnosis or as representing skip areas characteristic of CD. In addition, patients with low-grade indolent disease, particularly those in clinical and pathological remission, may show minimal architectural features of chronicity or perhaps even a completely normal-appearing biopsy specimen during the natural waxing and waning course of their illness. However, it must be emphasized that these data relate primarily to biopsy material from treated patients. They do not apply to patients whose UC has not yet been treated or in whom a diagnosis is being considered on the basis of the evaluation of a resection specimen. Evaluation of disease “continuity” by analysis of mucosal biopsies is not useful to distinguish UC from CD of the colon in previously treated IBD patients. In contrast, large portions of mucosa from a resection specimen with a normal histological appearance are an indication of true segmental disease and normally provide reliable evidence in support of an alternative diagnosis such as CD.
The European Crohn’s and Colitis Organization and the International Organization for the Study of Inflammatory Bowel Disease (IOIBD) define remission as complete resolution of clinical symptoms and endoscopic mucosal healing. Endoscopic mucosal healing in UC is defined by resolution of visible mucosal inflammation and ulceration, often assessed by using the Mayo endoscopic scoring method. As discussed earlier Chronic Inactive (Quiescent) Colitis, as mucosal healing occurs, there is quite a lot of variability in the type and degree of inflammatory changes such that at any given point of time, mucosal biopsies may show changes ranging from active colitis, chronic active colitis, chronic inactive colitis, or completely normal mucosa on histological examination. In fact, in a prospective observational study of 103 patients with UC in clinical remission, histological inflammation was found in 54% of patients receiving maintenance therapy, and 37% had at least moderate inflammation based on histology scores. Of the 52 patients with endoscopic evidence of left-sided colitis, 34% had histological inflammation in the proximal colon.
Deep remission is a more recent concept in IBD management. It is currently defined as the combination of clinical remission and mucosal healing. In a large retrospective study consisting of 252 IBD patients who were treated with anti–TNF-α therapy for ≥11 months, 122 patients achieved deep remission. The majority of patients in deep remission (81%) had “inactive disease,” while 19% had active disease histologically.
Much emphasis has recently been placed on the importance of mucosal healing as an outcome for therapies in patients with IBD. Few studies in UC have shown that despite endoscopic mucosal healing, active histological disease is associated with poor long-term outcomes. , , In two studies by Bitton et al. and Bessissow et al. in which biopsies from UC patients with endoscopically inactive disease (defined as a Mayo endoscopic score of 0) and a follow-up period of 12 months were evaluated, basal plasmacytosis, whether present in a focal or diffuse pattern, was found to be an independent predictor of clinical relapse in UC patients with mucosal healing. In a more recent study of 646 UC patients, 60% had endoscopic mucosal healing, 40% had histological quiescence, and 10% had histological normalization on follow-up colonoscopy. Of the 310 patients who were in clinical remission, histological normalization was independently associated with increased odds of relapse-free survival compared with histological quiescence (hazard ratio [HR], 4.31; 95% CI, 1.48 to 12.46; P = 0 .007) and histological activity (HR, 6.69; 95% CI, 2.16 to 20.62; p = 0.001). Another study by Zezos and colleagues found increased numbers of lamina propria eosinophils in patients with active UC, and severe eosinophilic infiltration was the most significant predictor of treatment failure in these patients. In a recent study consisting of 281 UC patients, histological features of UC activity were associated with increased rates of systemic corticosteroid use, colectomy, and hospitalization in the entire cohort (P <0.05 for all) and with increased rates of systemic corticosteroid use in an analysis limited to patients in endoscopic remission (p <0 .001); in patients in endoscopic remission, only histological activity was independently associated with use of systemic corticosteroids (multivariate OR, 6.34; 95% CI, 2.20–18.28; p <0 .001). Compared with patients without histological evidence of UC activity, patients with only a small number of mucosal neutrophils still had higher rates of systemic corticosteroid use (p <0.001). Interestingly, in contrast with the aforementioned studies, this study did not find any association between basal lymphoplasmacytosis or intramucosal eosinophilia and poor clinical outcome.
Lastly, in a large meta-analysis that included 28 studies contributing 2677 UC patients, histologically active disease was associated with an overall increased risk of relapse (OR 2.41, 95% CI, 1.91 to 3.04), with a similar effect noted in the subgroup with an endoscopic Mayo score of 0 versus 0 or 1. More rigorous Geboes cutoffs demonstrated numerically stronger impact on relapse rates: Geboes <3.1 (OR 2.40, 95% CI 1.57–3.65), Geboes <2.1 (OR 3.91, 95% CI 2.21–6.91), and Geboes 0 (OR 7.40, 95% CI 2.00–18.27). Among individual histological features, basal plasmacytosis (OR 1.94), neutrophilic infiltrations (OR 2.30), mucin depletion (OR 2.05), and crypt architectural irregularities (OR 2.22) predicted relapse.
Persistent histological inflammation has been associated with an increased risk for development of colorectal neoplasia in patients with UC. In a case control study of 204 patients with UC, histological and endoscopic activity predicted the development of neoplasia during the 14-year study period on univariate analysis. However, only histological inflammation was predictive on multivariate analysis (OR, 4.69; 95% CI, 2.10 to 10.48; p <0.001). In another cohort study of 418 UC patients, 15 developed advanced neoplasia during a median follow-up period of 6.7 years. In this study, the average histological inflammation score correlated with neoplasia development on multivariate analysis (HR, 3.8; 95% CI, 1.7 to 8.6). In a more recent meta-analysis of 1443 patients, Flores et al. showed that the pooled odds ratio for colorectal neoplasia was 3.5 (95% CI, 2.6 to 4.8; p <.001) in those with any mucosal inflammation and 2.6 (95% CI, 1.5 to 4.5; P = 0.01) in those with histological inflammation, when compared with those with mucosal healing. These findings could have implications on surveillance for CRC screening in patients with long-standing UC where clinicians may consider adjusting screening intervals in individuals who lack mucosal healing at surveillance colonoscopy.
Surgical therapy is indicated in cases of medically refractory disease, recurrent systemic complications, unacceptable side effects of medical therapy, colonic perforation, colonic dysplasia, or carcinoma. Surgical choices include subtotal colectomy with ileostomy, colectomy with ileorectal anastomosis, and proctocolectomy with ileal pouch–anal anastomosis (IPAA). A variety of configurations of the ileal pouch have been designed, with J- pouch, S- pouch, and K- pouch being the most common. The J- pouch procedure is commonly performed in patients with refractory UC or UC-associated neoplasia. In patients with UC-associated dysplasia of the rectum or sigmoid colon, mucosectomy of the rectal cuff is often performed during the pouch construction.
The typical course of UC consists of periods of remission interrupted by flares of activity. In approximately 5% of patients, the course is complicated by toxic megacolon, defined as acute colonic dilation (with a transverse colon diameter >6 cm radiologically) with loss of haustration in a patient with a severe attack of colitis. It is usually encountered early in the course of disease and in some cases may be the initial presentation of UC. Approximately 50% of patients respond to medical therapy alone. Perforation is the most important predictor of mortality. Surgery is recommended for patients with perforation or clinical deterioration after 48 to 72 hours of medical therapy.
Colonic strictures develop in almost 5% of UC patients. Their presence should always prompt a high index of suspicion for malignancy, especially if the stricture is located proximal to the splenic flexure. In a retrospective study comprising 1156 patients, 24% of strictures were found to be malignant. Most of the malignant strictures were located proximal to the splenic flexure, were clinically symptomatic, and developed late in the course of disease (after 20 years). Cancers associated with strictures tend to be more advanced than those not associated with a stricture.
Patients with UC have an increased risk for CRC, and the primary risk factor is the duration and extent of the disease (see Dysplasia in Ulcerative Colitis and Crohn’s Disease).
CD is a chronic inflammatory condition that can affect any part of the GI tract but has a propensity to involve the distal small and proximal large intestine. Descriptions of CD date back to more than three centuries, when it was termed terminal ileitis, regional enteritis, and granulomatous enterocolitis. Crohn, Ginzburg, and Oppenheimer are credited with the first modern description of CD in 1932.
The clinical classification of CD, known as the Montreal classification, is based on age at disease onset, principal anatomic location, and clinical behavior ( Box 17.3 ). It distinguishes disease of the ileum from that of the colon or both the ileum and colon. Approximately 30% to 40% of patients have small bowel involvement only, and 30% to 40% have ileocolonic involvement; only 10% to 20% have exclusive involvement of the colon (Crohn’s colitis). In patients with ileal disease, colonic lesions develop in fewer than 20% of patients during a period of 10 years. Similarly, ileal involvement occurs in 20% of patients with colonic disease. In a retrospective analysis of 84 patients with Crohn’s colitis, 52% had right-sided colitis, 40% had left-sided colitis, and 6% had pancolonic involvement. Small bowel involvement was more frequently associated with right-sided disease, whereas proctitis and perianal lesions were more frequent in patients with left-sided disease.
Age at diagnosis
A1 younger than 16 years
A2 between 17 and 40 years
A3 older than 40 years
Location
L1 ileal
L2 colonic
L3 ileocolonic
L4 isolated upper tract disease
Behavior
B1 nonstricturing, nonpenetrating
B2 stricturing
B3 penetrating
p perianal disease
Accurate assessment of the incidence and prevalence of CD worldwide is limited because of inconsistencies in diagnostic criteria, lack of thorough clinical evaluation with the use of modern radiological techniques, and, in some cases, an inability to differentiate UC from CD pathologically. Despite these limitations, reproducible epidemiological trends have been discerned. The age-adjusted annual incidence rate was reported to be 9 per 100,000 persons in Olmsted County, Minnesota. More recently, the prevalence of CD in the United States was estimated at 201 per 100,000 adults or 43 per 100,000 people younger than 20 years of age. There is a higher incidence of CD in northern latitudes (e.g., Denmark, 9/100,000; Nova Scotia, 20/100,000) than in southern Europe (e.g., Spain, 0.9/100,000; Italy, 3.4/100,000). In Asian, South American, and most African countries, the incidence is very low. Although all ethnic groups may be affected, CD is more prevalent among white North Americans, northern Europeans, Ashkenazi Jews, Scandinavians, and the Welsh.
Women are slightly more commonly affected than men (female-to-male ratio, 1.3:1). Most patients with CD are diagnosed during the second to fourth decades of life, although there is a smaller peak between the fifth and seventh decades. There is no relationship between pathological findings and age at onset, but some studies have identified a greater proportion of colonic and distal colonic disease among older patients and a predominance of ileocolonic disease in younger patients. , In one recent study that evaluated 118 patients with either isolated colonic or ileocolonic CD, those with isolated colonic CD were significantly older at disease onset and had a shorter interval from initial diagnosis to surgery. Compared with patients with ileocolonic disease, those with isolated colonic CD more often had subtotal or total colitis and were more likely to have left-sided colitis.
The clinical presentation of CD varies substantially depending on the principal location of disease, the intensity of inflammation, and the presence or absence of specific intestinal and extraintestinal complications. In some cases, weight loss and fever may be the only presenting features, especially in children. Crohn’s colitis often manifests with diarrhea, either with or without blood. Depending on the extent of colonic involvement and the severity of inflammation, patients may have a range of initial findings, ranging from minimally altered bowel habits to fulminant colitis. Intermittent and colicky abdominal pain is a common presenting symptom. Although most patients have either relative or complete sparing of the rectum, proctitis may be the initial or even the only area of involvement at presentation in some cases. Perianal skin tags, anal fissures, or ulcers are often present at the time of diagnosis as well. In a subset of patients (almost 24%), perianal disease preceded intestinal symptoms by a mean period of as long as 4 years.
The clinical pattern of disease is typically divided into aggressive fistulizing, fibrostenosing, and cicatrizing disease. Fistulas are a common finding in CD and may involve different segments of the bowel; more rarely, they may involve adjacent organs as well (coloduodenal, cologastric, or rectovaginal fistula). Symptoms of intestinal obstruction or jaundice, or both, are more common in the fibrostenotic form of disease.
Extraintestinal manifestations of CD occur in 6% to 25% of cases and are more common among patients with colonic involvement. , They include musculoskeletal disorders such as pauciarticular arthropathy (6%), polyarticular arthropathy (4%), and peripheral arthralgias (16% to 20%). Axial arthropathies, granulomatous vasculitis, periostitis, and amyloidosis are other rare rheumatological complications. Mucocutaneous lesions include pyoderma gangrenosum, erythema nodosum, and oral aphthous ulcers. Episcleritis and uveitis tend to occur in association with active intestinal disease and occur in as many as 6% of patients. Among hepatobiliary manifestations, more than 25% of cases manifest with symptomatic cholelithiasis. Although it is more commonly associated with UC, PSC may develop in as many as 4% of patients who have colonic CD. Hyperoxaluria with calcium oxalate stone formation, interstitial nephritis, and cardiomyopathy have associations with CD as well.
Patients with CD have a prothrombic tendency and therefore may present with venous thromboembolism or, less commonly, arterial thrombosis. In more than 50% of patients, a predisposing factor cannot be identified. ,
As in UC, there is no specific clinical or laboratory test that establishes a definite diagnosis of CD. The diagnosis is usually established based on a compilation of clinical findings combined with radiological, endoscopic, and pathological findings. Laboratory tests may be completely normal. In some patients, the white blood cell count is elevated, which suggests a pyogenic complication. The presence of anemia, elevated ESR, and elevated CRP in a patient with abdominal pain is not specific for CD but should always prompt a workup for IBD. Stool studies, including culture, examination for ova and parasites, C. difficile toxin assay, and serological testing for Entamoeba histolytica, are usually performed to exclude an infectious cause. Serological testing shows elevated ASCA levels in 41% to 76% of patients. ,
Barium studies are a popular method of investigation for patients with suspected CD. They are especially helpful in delineating late transmural complications of CD. Aphthous ulcers, thickened mucosal folds, submucosal edema, fistulas, sinus tracts, and fixed strictures are some of the findings that may be detected by barium studies. Currently, CT or magnetic resonance enterography is preferred over barium studies. Radiological findings that correlate with disease activity include mural enhancement and increased density of pericolonic fat. ,
Common endoscopic findings of CD include aphthous ulcers, edema, cobblestoning, and luminal narrowing. Segmental involvement is characteristically present in early-stage disease. Rectal sparing is often present in untreated cases.
Once a diagnosis of CD has been established, clinical disease activity is usually monitored with the use of a composite scoring system. The Crohn’s Disease Activity index is a commonly used scoring system that evaluates eight variables (stool count, abdominal pain, general well-being, features of extraintestinal disease, opiate intake for diarrhea, presence of abdominal mass, hematocrit value, and body weight).
The cause of CD is unknown; similar to UC, it probably involves a combination of environmental factors (e.g., luminal bacteria, infectious agents), abnormalities in immune regulation, and genetic predisposition for development of disease.
Many infectious agents, including Chlamydia, Listeria monocytogenes, Pseudomonas species, paramyxovirus, and Mycobacterium paratuberculosis, have been etiologically linked to CD as a cause of granulomatous vasculitis and bowel injury. Molecular techniques have detected M. paratuberculosis in tissues of some patients with CD. One study by Lamps and colleagues found Yersinia species DNA in 31% of Crohn’s resection specimens. There are many histological similarities between these infections and CD, and the possibility that one or more of them triggers the development of CD is an appealing hypothesis. However, thus far, there is no conclusive evidence to implicate any one specific organism in the pathogenesis of CD.
The relative risk (RR) for development of IBD among first-degree relatives of patients with CD is 14 to 15 times higher than in the general population. Ethnicity also appears to play a significant role. Eastern European (Ashkenazi) Jews have a two-fold to four-fold higher risk for CD than non-Jews from the same geographic location. Further support for a genetic predisposition is provided by data on monozygotic and dizygotic twins. , The concordance rate for CD is 67% among monozygotic twins and 8% among dizygotic twins, suggesting a strong genetic influence. Although results are inconsistent and vary with the population being studied, numerous studies have found both positive and negative associations between HLA antigens and the development of CD. ,
Genome-wide association studies and computerized meta-analyses have identified 71 susceptibility loci for CD on 17 chromosomes thus far. Three important pathways have been highlighted by these studies. The first susceptibility locus was identified in 2001 as nucleotide-binding oligodimerization domain 2 (NOD2) or caspase-recruitment domain 15 (CARD15). A homozygous carrier of disease-specific allelic variants has a 17.1-fold increased risk for CD, whereas the OR for a heterozygous carrier is 2.5. Genetic polymorphisms in NOD2/CARD15 are present in 20% to 30% of patients with CD, and the abnormalities correlate with younger age at onset, ileal location of disease, and an increased likelihood of stricture formation. , NOD2/CARD15 mutations are more common in white patients with CD but are rare in Asians and Africans. The NOD2/CARD15 gene product binds to muramyl dipeptide, a component of bacterial peptidoglycan found in both gram-positive and gram-negative bacteria. , NOD2/CARD1 is expressed in Paneth cells that produce endogenous antimicrobial peptides known as defensins. NOD2/CARD15 gene variants interfere with binding to muramyl peptide, resulting in decreased antibacterial defense.
The second important pathway implicated in CD pathogenesis is related to autophagy, a unique process by which cytoplasmic constituents are isolated within a membrane-bound vesicle and then delivered to lysosomes for elimination. Misfolded or misaggregated proteins are eliminated via this pathway without inciting an inflammatory or autoimmune response. Variants in at least two autophagy-related genes have been associated with CD, namely, the autophagy-related 16-like 1 (ATG16L1) gene and the immunity-related guanosine triphosphatase family member M (IRGM) gene on chromosome 5.
The third pathway associated with CD is related to IL-23. IL-23 is a cytokine produced by dendritic cells and macrophages in response to various antigenic signals. In response to IL-6 and transforming growth factor-β (TGF-β), naïve CD4+ T cells upregulate the IL-23 receptor (IL-23R), which results in autocrine generation of effector T cells that produce IL-17. Although most common single nucleotide polymorphisms (SNPs) in the IL23R gene are associated with an increased risk for CD and UC, rare variants appear to be protective against the development of CD. ,
The gradually increasing incidence of CD has also been attributed to a variety of environmental factors. Higher socioeconomic status, use of oral contraceptives, NSAID use, increased intake of refined sugars, and decreased intake of dietary fiber have all been implicated as risk factors for CD. Zinc deficiency is associated with immunological dysfunction in patients with CD, and some data suggest that an elemental diet may improve CD by reducing intestinal permeability.
In contrast with UC, CD is more prevalent among smokers. Smoking is an independent risk factor for clinical, surgical, and endoscopic recurrences in CD and also appears to influence disease activity after surgery. Although the pathogenesis is unclear, it is believed that smoking causes alteration of intestinal permeability, induces cytokine production, and promotes production of microvascular thrombi. ,
Similar to UC, disturbance of the intestinal epithelial barrier and innate immunity also plays a role in the pathogenesis of CD. Analysis of intestinal biopsies from patients with CD shows downregulation of the junctional protein epithelial cadherin, which composes the tight junctions of this physical barrier.
Interaction of effector T cells and antigen-presenting cells is vital to the pathogenesis of CD. Processing of luminal antigens by dendritic cells located within the lamina propria and the subsequent interaction between MHC class II molecules and T-cell receptors leads to activation and differentiation of T cells. The helper T-cell Th1 and Th17 responses that characterize CD are influenced by cytokines IL-23, IL-6, and TGF-β. Within mononuclear cells, NF-κB plays a key role in regulating transcription of IL-1, IL-6, IL-8, TNF, and other peptides that generate an inflammatory response. TNF is not only essential in formation of granulomas, but also causes neutrophil activation and, along with interferon-γ, induces expression of MHC class II molecules on IECs.
TNF and other proinflammatory cytokines also promote expression of adhesion molecules on endothelial cells, which leads to trafficking of inflammatory cells into mucosa. Integrins α 4 β 7 and α E β 7 have ligands (mucosal addressin cellular adhesion molecule and E-cadherin, respectively) that are specific to the intestinal environment. Antibodies to the α 4 subunit of integrin have a therapeutic role in the management of CD.
Tissue destruction (especially penetrating ulcers, fistulas, and sinuses) ultimately results when proinflammatory substances such as prostaglandins and matrix metalloproteinases are elaborated by mononuclear cells and granulocytes. Mural fibrosis, another characteristic finding of CD, is a result of TGF-β that is released in the presence of inflammation. It stimulates production of type III collagen, which not only promotes healing of ulcers but also contributes to the formation of strictures. Fat wrapping (“creeping fat”) is an indicator of transmural disease that is often identified intraoperatively. It results from upregulation of peroxisome proliferator–activated receptor-γ (PPAR-γ), which regulates homeostasis of adipose tissue. Histologically, the finding of pyloric gland metaplasia in the lower GI tract indicates chronic mucosal injury. Pyloric glands are a form of ulcer-associated cell lineage (UACL); they represent budlike glandular structures that develop from the base of intestinal crypts at sites of chronic ulceration. The UACL expresses a variety of peptides implicated in the repair of damaged GI mucosa, notably epidermal growth factor and members of the trefoil peptide family, which restores epithelium in areas of mucosal ulceration.
The pathophysiology of diarrhea in CD is related to multiple factors. Increased mucosal permeability caused by inflammation and production of prostaglandins, biogenic amines, neuropeptides, and reactive oxygen metabolites results in exudation of proteins and fluids. Bacterial overgrowth and altered colonic mobility in areas of strictured bowel also contribute to diarrhea.
CD is classically characterized by segmental involvement of the affected areas of bowel, although pancolitis, and even proctitis as the only manifestation of colonic involvement may occur in a small proportion of cases ( Fig. 17.20A ). The serosal surface often appears congested and may be covered with a fibrinous exudate. Fat wrapping (creeping fat) along the antimesenteric border is a common finding in CD. In contrast with UC, the bowel wall in CD is typically thickened. It does not lie flat on opening. Mucosal aphthous ulcers overlying lymphoid aggregates are an early feature of CD. A zone of hyperemia often surrounds larger ulcers. As the disease progresses, ulcers enlarge to form discontinuous, serpiginous, or bear claw–type longitudinal furrows (see Fig. 17.20B ). Areas of edematous, mildly inflamed, or even normal mucosa are located between areas of longitudinal ulcers; this results in the development of a cobblestone appearance of the mucosa. Inflammatory pseudopolyps are most commonly encountered in the transverse colon and splenic flexure. In segments of bowel involved by disease, the wall of the bowel is usually thick and fibrotic. Fissures, sinuses and fistulous tracts, and mural or pericolonic abscesses may be present in complicated cases.

Strictures are more common with long-standing disease. Depending on the degree of obstruction, the bowel wall proximal to the stricture may be secondarily dilated and congested ( Fig. 17.21 ). Perforations are uncommon in CD, occurring only in 1% to 3% of cases. , They occur as a result of superimposed ischemia or infection or as a complication of fissures, sinus tracts, or fistulas. Fibrous adhesions may seal off sites of perforation, so they may not be visible on gross examination of the bowel.

Histologically, colonic CD (whether isolated or combined with ileal disease) classically shows skip areas of involvement, both grossly and microscopically. Areas of involvement alternating with areas of normal mucosa are characteristic (“segmental” colitis). However, as mentioned earlier, pancolitis occurs in a small proportion of cases. Overall, CD is characterized by the presence of a wide variety of mucosal and mural changes ( Box 17.4 ). Other major pathological features include aphthous and fissuring ulcers, sinuses and fistulas, transmural lymphoid aggregates, and nonnecrotizing granulomas. All of these features may affect the bowel wall in a patchy and segmental distribution. Other, less characteristic but common features include submucosal fibrosis, neural hypertrophy, muscularis mucosae and muscularis propria hypertrophy, neural plexitis, perivascular lymphoid aggregates, serositis, and pyloric gland metaplasia.
Segmental disease
Creeping fat
Fistulas/sinuses
Skip lesions
Granulomas
Transmural lymphoid aggregates
Aphthous and fissuring ulcers
Submucosal fibrosis
Neural hypertrophy
Muscularis mucosae hypertrophy
Muscularis propria hypertrophy
Neural plexitis
Perivascular lymphoid aggregates
Serositis
Pyloric gland metaplasia
Similar to UC, and depending on the phase of disease, the mucosa may show a wide spectrum of changes ranging from completely normal to diffuse and severe chronic active inflammation with ulceration. In biopsies, mucosal disease is categorized similar to UC: chronic inactive, chronic active, or active (see earlier discussion). Histological features of chronicity include crypt architectural distortion, crypt atrophy, diffuse mixed lamina propria inflammation, basal plasmacytosis, basally located lymphoid aggregates, pyloric gland metaplasia, and Paneth cell metaplasia (in the left colon). Other changes of chronicity include lamina propria fibrosis and Paneth cell hyperplasia in the right colon. “Activity” is characterized by the presence of neutrophilic or eosinophilic cryptitis, crypt abscesses, regenerative and/or degenerative epithelial changes, necrosis, erosions, and ulceration. Grading of the degree of inflammation in patients with CD is difficult and has not been standardized. This is also because some areas of the bowel may show severe ulcerating disease, and others adjacent to it may be completely normal. However, in individual biopsies, grading the degree of activity can be done similar to the system proposed by the authors in the Ulcerative Colitis section earlier in this chapter.
In active CD, the disease may be patchy, with foci of injured and inflamed crypts situated adjacent to completely normal crypts. Two types of ulcers are characteristic of active CD: aphthous and fissuring ulcers. Aphthous ulcers arise in focal, mildly active CD. They are well-delineated, small, superficial lesions that overlie lymphoid aggregates. They usually involve a portion of mucosa occupied by about two to four crypts in length. The earliest stage is a mild neutrophilic infiltrate in the superficial half of the lymphoid aggregate. Neutrophils infiltrate the crypts and form small basilar crypt abscesses, producing epithelial necrosis and an intraluminal exudate ( Fig. 17.22 ). Concomitant neutrophilic infiltration and erosion of the superficial epithelium develops into a small microabscess that covers the lymphoid aggregate as the ulcer expands. Irregularly shaped crypts with regenerative epithelial changes are typically found at the edges of older (healing) ulcers. Aphthous ulcers can continue to expand and connect to form serpiginous or longitudinally oriented ulcers.

Crypt disarray is a feature of mucosal involvement in CD, similar to UC. There is significant variation in the size and shape of crypts. Features such as crypt branching and shortening are most easily appreciated at medium or low magnification ( Fig. 17.23 ). Heterogeneity in the density and distribution of lymphoplasmacytic inflammation within the lamina propria is a common finding in CD, as it is in UC. Well-circumscribed, focal collections of lymphocytes that surround several crypts (lymphoid aggregates) simulate normal lymphoid follicles. However, lymphoid aggregates in CD may reveal crypts within their center, whereas in normal lymphoid follicles, the crypts are pushed toward the periphery.

Although nonnecrotizing epithelioid granulomas are characteristic of CD, they are neither specific nor sensitive for this diagnosis. , , The prevalence of granulomas in endoscopic biopsy samples ranges from 13% to 50%; in resections, it ranges from 40% to 60%. Granulomas are more frequently encountered early in the course of disease. They are typically sarcoid-like and composed of aggregates of epithelioid histiocytes admixed with lymphocytes and neutrophils ( Fig. 17.24 ). Occasionally, giant cells are present. In some cases, they may be very sparse and poorly formed, consisting only of small, pericryptal collections of closely arranged histiocytes, referred to as pericryptal microgranulomas . Serial step sections enhance the likelihood of detecting pericryptal microgranulomas. They may be present in involved as well as in uninvolved segments of colon. They may be present in any layer of the bowel wall or within pericolonic lymph nodes ( Fig. 17.25 ).


Granulomas in CD should be distinguished from mucin granulomas that form around ruptured crypts, which is common in UC (see Pathological Features in the Ulcerative Colitis section earlier in this chapter). Macrophages within mucin granulomas usually have a greater amount of bubbly or clear cytoplasm, which is caused by phagocytosis of mucin and crypt contents. Foreign-body giant cells usually are not a component of CD-related granulomas, but they may be found in mucin granulomas. Mucin stains are not always helpful because a small amount of mucin may also be present in the cytoplasm of macrophages in CD-related granulomas. Thick-walled capillaries, pericryptal fibroblastic sheaths, and tangential sections of germinal centers may mimic the well-formed granulomas of CD. The presence of necrotizing granulomas or a large number of granulomas should prompt a workup for an infectious etiology such as tuberculosis, histoplasmosis, yersiniosis, or sarcoidosis.
Pyloric gland metaplasia is far more common in CD than in UC, and it is frequently present in the small bowel. In colonic CD, pyloric gland metaplasia is more common in the cecum and right colon ( Fig. 17.26 ).

Among the common mural changes of CD are knifelike fissuring ulcers and sinus tracts, which typically occur at right angles to the longitudinal axis of the bowel. They may extend through the bowel wall and may develop into a fistula or result in pericolonic abscess formation ( Fig. 17.27 ). They are usually lined by acute inflammatory cells, necrotic and granulation tissue, and loose aggregates of epithelioid histiocytes resembling early granulomas. Multinucleate giant cells may be present as well. Dense lymphoid aggregates, with or without germinal centers, are usually found at the mucosal-submucosal junction. However, transmural lymphoid aggregates that are located in the deeper aspects of the bowel wall, including the subserosal adipose tissue (“Crohn’s rosary”), are characteristic of CD ( Figs. 17.28 and 17.29 ). These are defined as collections of predominantly small, mature-appearing lymphocytes of variable size that are distributed throughout the bowel wall. Some cases of CD show prominent perivascular lymphoid aggregates, which may or may not be associated with granulomas, particularly in the submucosa. This feature has led some authorities to postulate that CD may represent a vascular disorder.



Abnormalities in smooth muscle, such as splaying and hypertrophy of the muscularis mucosae and distortion (and hypertrophy) of the muscularis propria, are common in long-standing CD. Proliferation of smooth muscle that obliterates the submucosa (“muscularization of submucosa”) occurs more commonly in stricturing CD. CD also affects the neural tissue. There is both hypertrophy of nerve trunks and an increase in the number of ganglion cells. , Large, abnormal fusiform nerve bundles may be seen throughout the affected bowel wall. Additionally, the myenteric plexuses may show infiltration by lymphocytes (myenteric plexitis) ( Fig. 17.30 ). Some studies have shown that myenteric plexitis in the proximal margins of an ileocolonic resection is more likely to be present in resection specimens of patients with a previous surgery for CD and of those with a shorter duration of disease before surgery. The severity of inflammation also appears to correlate with severity of endoscopic recurrence.

Some studies suggest that colonic CD less frequently shows major pathological features than does ileal CD. A study by Soucy and colleagues showed that patients with isolated CD had significantly fewer major pathological features of CD, such as strictures/stenosis, pericolonic adhesions, segmental disease, the finding of proximal disease worse than distal disease, perivascular lymphoid aggregates, and pyloric gland metaplasia, compared with those with ileal CD. A small proportion of patients from both groups showed inflammatory changes limited to the mucosa, similar to what is seen in UC (see later discussion).
Perianal involvement occurs in approximately 74% of CD patients, typically within 10 years of the initial diagnosis, and it generally increases in frequency in patients with more distal colonic disease. In 20% to 36% of patients with CD, the patients’ perianal disease actually precedes the intestinal disease. Skin tags or fibroepithelial polyps are the most common type of perianal lesion in CD, being present in 40% to 70% of cases. Histologically, they resemble fibroepithelial polyps that occur in non-CD patients. Other manifestations of anal CD include anal fissures, stenosis, and anorectal abscesses and fistulas, the latter comprising 18% of all CD-related perianal lesions. The histological features of perianal CD are similar to CD elsewhere in the GI tract in that the specimens from excisional biopsies, limited resections during fistula repair, or proctocolectomy for severe or medically refractory perianal disease show mucosal ulceration accompanied by transmural lymphoid aggregates and mural fibrosis. One note of caution is that the fistulae may be associated with a foreign-body–type giant cell reaction, and this should be distinguished from true granulomas when evaluating whether the patient has CD. Medical management remains the main type of therapy for most patients with perianal CD.
As mentioned previously, the colon is involved in almost 50% of CD cases, either alone or in combination with the small intestine. However, in some studies, up to 20% of CD patients have disease limited to the colon. Although some cases of Crohn’s colitis show classic features of CD (e.g., fissuring ulcers, sinus tracts, transmural lymphoid aggregates, submucosal fibrosis, granulomas), others show few or none of these features and resemble UC, both clinically and, particularly, histologically. These cases have been termed superficial Crohn’s disease or UC-like Crohn’s disease because the inflammatory changes are limited to the mucosa and, in some cases, the superficial submucosa, similar to UC ( Fig. 17.31 ). In these cases, a diagnosis of CD may be established by noting the presence of typical CD in other regions of the GI tract, such as the distal small intestine or the perianal region, or by showing other cardinal features of CD such as granulomas unrelated to ruptured crypts, segmental involvement of the colon with skip areas unrelated to treatment effect, or absolute rectal sparing from the initial onset of disease. In addition, CD also should be suspected in patients who have UC-like features in the colon and severe anal or perianal disease (e.g., fissures, fistulas). UC-like CD is rare. In one of the earlier studies that evaluated 10 patients with “superficial” CD, the diagnosis of CD was rendered based on the association with classic CD in other segments of the same resection specimens, a previous history of resection for CD, small bowel involvement, segmental or irregular disease distribution, and presence of granulomas.

In a comprehensive case-control study by Soucy et al. comprising 73 patients with isolated colonic CD and 45 patients with ileocolonic CD, all of whom had detailed pathological evaluation of a wide variety of UC and CD features, the incidence of UC-like CD was 14% and 13%, respectively, in the two groups. Patients with UC-like CD were younger than those who had classic CD with mural involvement. In this study, 50% of patients with superficial UC-like CD had only left-sided colitis, whereas 19%, 13%, and 6% showed right-sided, total, and subtotal colitis, respectively. Granulomas were observed in 44% of cases. Of the four UC-like CD patients who underwent an IPAA procedure, none showed an anastomosis breakdown. When the UC-like colonic CD cohort was compared with patients with non–UC-like colonic CD, the mean age at diagnosis was significantly younger in the former group (25 vs. 35 years; P = 0.02). However, there were no differences in the pathological (nonmural) features or in overall outcome between these two groups.
In another study consisting of 21 CD patients with UC-like pancolitis at presentation who underwent a pouch procedure, only 14% had pouch complications that necessitated pouch resection. Furthermore, recurrent CD in the small intestine developed in only 5%. Therefore it appears that many patients with UC-like colonic CD have good success after a pouch procedure, and this approach may be a viable option in affected patients who are either resistant to permanent ileostomy or prefer a pouch procedure.
The natural history of CD is quite variable, and the type of therapy administered also affects the disease course. Two population-based studies that evaluated patient outcome—from Olmsted County, Minnesota (225 patients), and Copenhagen County, Denmark (373 patients)—found that a relapse or exacerbation of disease developed in 10% to 30% of CD patients, respectively, after the first year of diagnosis, 15% to 25% experienced low disease activity, and 50% to 65% were in remission. On long-term follow-up (>10 years), 10% to 13% of patients remained in remission, 67% to 73% experienced a chronic intermittent course, and a chronic course with continuous activity developed in 13% to 20%. , Penetrating/fistulizing CD, young age at presentation, short duration of disease before first surgery, and ileocolonic disease are high risk factors for CD recurrence after surgery. Based on the outcome data from a detailed clinicopathological study consisting of 118 patients with isolated and ileocolonic CD, colonic recurrence will develop in as many as 28% of patients after surgery, noncolonic recurrence will develop in 10%, and recurrent disease at both sites will develop in 6%, during a mean follow-up interval of more than 5 years. A recent meta-analysis consisting of 21 studies (2481 patients) that assessed positive resection margins, 10 studies (808 patients) that assessed plexitis, and 19 studies (1777 patients) that assessed granulomas in patients undergoing resection for CD showed that positive resection margins increased the risk of clinical recurrence (RR 1.26, 95% CI, 1.06 to 1.49) and surgical recurrence (RR 1.87, 95% CI, 1.14 to 3.08), granulomas increased the risk of clinical recurrence (RR 1.31, 95% CI, 1.05 to 1.64) and endoscopic recurrence (RR 1.37, 95% CI, 1.00 to 1.87), and plexitis increased the risk of endoscopic recurrence (RR 1.31, 95% CI, 1.00 to 1.72).
The goal of therapy is to induce and maintain clinical remission. Aminosalicylates are used in the treatment of mild to moderate CD. Antibiotics, especially metronidazole and ciprofloxacin, are used to treat perianal disease, fistulas, and active luminal CD. Moderately severe CD is treated with tapered doses of prednisone. The thiopurine analogues, azathioprine and 6-mercaptopurine, inhibit cell-mediated immunity and therefore play a role in management of CD. Methotrexate induces remission in patients who do not tolerate thiopurine analogues. Infliximab and adalimumab are monoclonal anti-TNF antibodies that benefit approximately 60% of patients with luminal disease. Forty percent of these patients maintain a good response after 1 year. Among newer therapeutic agents is natalizumab, a humanized monoclonal antibody against α 4 -integrin that inhibits leukocyte adhesion and migration into inflamed tissue. It has been found to be an effective induction and maintenance agent for treatment of moderately to severely active CD.
Surgery is most often indicated for patients with intra-abdominal abscess, medically refractory disease, intestinal obstruction, toxic megacolon, hemorrhage, dysplasia, or cancer. Postoperative disease recurrence has been associated with the presence of perianal disease, ileorectal anastomosis, and segmental resection. In a study by Morpurgo and colleagues, patients with either granulomas or segmental colitis at presentation showed a higher recurrence rate when compared with patients without granulomas and patients with pancolitis. In a study by Soucy and colleagues, which specifically evaluated patients with colonic CD at initial presentation, there were no significant differences in the development of adverse outcomes between patients with isolated colonic CD versus those with ileocolonic CD.
Patients with CD are at risk for small-bowel and colorectal adenocarcinoma. The risk is higher in patients with a family history of sporadic CRC, uncontrolled inflammation, shortened colon, or multiple inflammatory pseudopolyps. The range of cumulative prevalence of dysplasia in colonic CD reported in earlier studies was between 2% and 5%. , More recently, the estimated 25-year risk of dysplasia in colonic CD has ranged from 12% to 25% , (see Dysplasia in Ulcerative Colitis and Crohn’s Disease).
In a small proportion of IBD cases (1% to 10%, depending on the study), a definite diagnosis of UC or CD cannot be established with absolute certainty after evaluation of the patient’s resection specimen. This occurs, most commonly, either because the pathologist has insufficient clinical, radiological, endoscopic, or prior (biopsies) pathological data on the patient and/or because of overlapping pathological features between these two disorders, which occurs most commonly in patients with acute fulminant colitis. In these circumstances, the term indeterminate colitis (IC) has been used, but some authorities prefer the term IBD, unknown type or IBD, indeterminate type. In 1998, Swan and colleagues evaluated 95 patients with “fulminant colitis” with the aim of identifying features that could help separate UC from CD. After all pathological material and clinical follow-up information had been reviewed, microscopic examination correctly diagnosed UC or CD in 91% of cases. Granulomas and transmural lymphoid aggregates located distant from areas of ulceration were the most specific indicators of CD. Overall, the diagnosis of indeterminate colitis is a vastly overused term in IBD pathology. Furthermore, in recent years, unfortunately, it has been expanded to use in patients in whom a definite diagnosis of CD or UC cannot be made in endoscopic biopsies. This diagnosis should never be used in biopsies because it is usually not possible to distinguish CD from UC in biopsy studies, especially in IBD patients who have already undergone medical therapy, and in those without CD-like anal disease, clear evidence of CD-like ileal involvement, or multiple granulomas unrelated to ruptured crypts. Furthermore, IC is not a specific disease entity and therefore has no diagnostic criteria. Rather, it represents a provisional term used by pathologists only when a definite diagnosis of UC, CD, or any other cause of chronic colitis cannot be established with complete certainty given the clinical information and pathological tissue available at the time of sign-out of the patient’s resection specimen. In as many as 80% of cases, the true nature of the patient’s underlying IBD becomes apparent within several years of follow-up or when all of the patient’s clinical, endoscopic, and previous pathology material (including all prior mucosal biopsies) is obtained and reviewed in detail.
The term indeterminate colitis was originally applied to cases of fulminant pancolitis (i.e., severe colitis with systemic toxicity often associated with colonic dilation), a disease in which the classic features of UC may be obscured by the presence of severe, diffuse ulceration with early superficial fissuring ulceration, transmural lymphoid aggregates (in areas of deep ulceration), and relative rectal sparing—features that are normally associated with CD. However, it is now recognized that these “CD-like” features may occur in patients with fulminant UC, so most if not all of the types of specimens originally interpreted as IC can now, more conclusively, be diagnosed as fulminant UC. This is based on data suggesting that with time, patients with fulminant UC who have one or more CD-like features do not develop CD in other portions of the GI tract and have a successful rate of IPAA procedures. For instance, in one study of 21 patients with fulminant pancolitis and superficial fissuring ulcers, the incidence of CD-like complications after total colectomy and IPAA was very low and was statistically similar to that of a control group of patients without fissuring ulcers in their resection specimen. Therefore the presence of these ulcers in patients with fulminant colitis is not considered necessarily indicative of CD and should not be used as evidence to deny the patient an opportunity for an IPAA procedure ( Fig. 17.32 ).

More recently, the term IC has been broadened to include any IBD case in which a definite diagnosis cannot be established pathologically in a resection specimen, even in patients who do not necessarily have IBD. There are other potential causes of “IC” other than IBD, such as severe infectious colitis or drug/medication-related colitis requiring surgical treatment. Naturally, the ability to establish a precise diagnosis of either UC or CD is highly dependent on the level of awareness of the pathologist regarding the range of morphological features seen in these disorders and the unusual patterns of injury in each disorder that can mimic the other. In a study by Farmer and associates, 84 IBD colectomy specimens were reviewed by 24 university pathologists, whose diagnostic accuracy was compared with that of a single GI pathologist who had a particular interest in IBD. The GI pathologist rendered a diagnosis that was different from that of the others in 45% of the specimens; in most cases, this decision resulted in a change of diagnosis from UC to CD. Therefore it is highly recommended that pathologists seek the opinion of an expert GI pathologist regarding any case that is ruled indeterminate from their point of view before final sign-out.
The most common (clinical and pathologic) reasons for establishing an interim diagnosis of IC are listed in Box 17.5 . In addition to the reasons described in the table, pathologists often diagnose IC when a definite diagnosis of UC or CD cannot be made by evaluation of biopsy specimens. This practice is strongly discouraged because the cardinal features of CD cannot be seen in biopsies, and the pattern of inflammation in both UC and CD becomes altered and more similar after medical treatment (see earlier discussion). Therefore, as mentioned earlier, IC is a diagnosis that should never be made on biopsy material. In many instances, a diagnosis of IC is made because of a lack of awareness of the many types of unusual variants of UC that can mimic CD and the subtypes of CD that mimic UC (i.e., UC-like CD). Finally, in some cases, a diagnosis of IC is made because of the unwillingness of pathologists to accept a particular finding, such as true segmental disease with skip lesions in untreated patients, granulomatous inflammation unrelated to ruptured crypts, transmural lymphoid aggregates in patients without fulminant colitis, or deep fissuring ulceration, as a definite diagnostic criterion for CD in colonic resection specimens. In reality, any one of these features should be considered as supportive evidence for a diagnosis of CD in the appropriate clinical setting.
Fulminant colitis
Insufficient clinical, radiological, and pathological information
Interpretation of biopsy specimens
Failure to recognize unusual variants of ulcerative colitis
Ulcerative colitis with Crohn’s-like features
Discontinuous disease
Superficial fissuring ulcers
Aphthous ulcers
Ileal involvement
Involvement of the upper gastrointestinal tract
Granulomas
Crohn’s disease with ulcerative colitis–like features
Pancolitis
Superficial colitis
Inflammatory bowel disease complicated by infections
Pseudomembranous colitis
Cytomegalovirus infection
Chronic recurrent (refractory) pouchitis
Failure to accept hard criteria for Crohn’s disease
Transmural inflammation
Granulomas
Deep fissuring ulceration
Ileal involvement
Segmental disease
Unusual pathological manifestations of other forms of colitis
Ischemia
Radiation
Microscopic colitis with features of inflammatory bowel disease
Diverticular disease–associated chronic colitis
Diversion colitis
Nonsteroidal antiinflammatory drug–induced colitis mimicking inflammatory bowel disease
Acute self-limited colitis
Polyposis disorders that may mimic inflammatory bowel disease
Solitary rectal ulcer syndrome
Inflammatory “cap” polyposis
Juvenile polyposis
In most studies of IC, the majority of cases diagnosed as IC represent UC; a small proportion (10% to 40%) are, in fact, CD. However, in some circumstances, other pathological mimics of IBD, such as NSAID-related colitis, diverticular disease–associated colitis (DAC), radiation or ischemic colitis, and infectious colitis, may also show histological features in resection specimens that mimic either UC or CD ( Box 17.6 ). These other disorders should always be considered when pathologists are confronted with an IC resection specimen.
Ischemic colitis
Radiation colitis
Microscopic colitis with features of IBD
Diverticular disease–associated chronic colitis
Infectious colitis
Bacterial infection— Escherichia coli, Salmonella, Shigellosis, tuberculosis, syphilis, Yersinia, lymphogranuloma venereum
Protozoan infection—amebiasis
Diversion colitis
Drug-induced colitis (NSAID colitis)
Vasculitis (Behçet’s syndrome)
Acute self-limited colitis
Solitary rectal ulcer syndrome
Inflammatory “cap” polyposis
Juvenile polyposis
As mentioned earlier, there is a strong clinical need to classify IBD as CD or UC (or some other disorder) because the IPAA (“pouch”) procedure is usually contraindicated for patients with CD. In studies of IPAA in CD, there was a high risk for morbidity related to pouchitis, fistulas, incontinence, or anastomotic leaks. , Many studies have evaluated the pathological features, natural history, and outcome of ileoanal pouches in patients with IC. , , Results vary considerably because most of these studies were retrospective, used varying and poorly defined criteria for IC, and lacked sufficient follow-up information. Nevertheless, in general, approximately 20% of IC patients experienced severe pouch complications, a frequency that is intermediate between that seen in UC (8% to 10%) and in CD (30% to 40%). , , , This finding is not unexpected because in most studies, the IC study group consists of a mixture of true UC and CD patients. In a study by Yu and coworkers of 82 cases of IC and 1437 cases of UC, all of which involved an IPAA operation, patients with IC had higher incidences of pelvic sepsis, pouch fistulas, and pouch failure than patients with UC. However, 15% of the patients with IC ultimately had their diagnosis changed to CD, and when newly diagnosed CD patients were removed from the analysis, the rates of pouch complications in IC and UC patients were statistically similar.
In 1995, McIntyre et al. compared 71 patients with IC and 1232 patients with UC for frequency of bowel movements, incontinence, and prevalence of pouchitis and pouch failure after an IPAA procedure. Although the failure rate in IC was higher than in UC (19% vs. 8%), IC and UC patients had similar overall outcomes, once again suggesting that most patients with IC probably have UC as the cause of the colonic inflammatory disorder. Although a substantial proportion of CD patients who undergo an IPAA operation experience pouch failure (30% to 45%), some studies have suggested that CD patients whose pouches can be retained in situ have acceptable pouch function. ,
In certain circumstances, serological testing for ANCAs and ASCAs may be helpful in classifying IC cases as UC or CD. , In addition to these antibodies, a widely used serological profile by Prometheus Laboratories tests for antibodies against outer membrane porin C (OmpC), a protein belonging to E. coli that regulates metabolite and toxin transport, and CBir1, a flagellin that presumably induces colitis through adaptive immune response. A typical profile for CD includes detectable ASCA IgA and anti-CBir1 IgG antibodies and an absence of anti-OmpC IgA and pANCA. Elevated serum pANCA and low levels (below the reference range) of ASCA IgA, anti-OmpC, and anti-CBir1 IgA are usually associated with UC.
ANCAs are detected in the serum of 60% to 70% of patients with UC but in only 10% to 40% of CD patients. Patients with UC who have a high level of pANCA are at an increased risk of pouchitis after IPAA. Of the CD patients who are positive for ANCAs, most have left-sided colitis with clinical, endoscopic, or histological features of UC. ASCAs are present in 50% to 60% of CD patients. As a serum marker for CD, ASCA has a sensitivity of 67% and a specificity of 92%. In a meta-analysis designed to evaluate the diagnostic precision of ANCA and ASCA in IBD, these serological tests were shown to be quite specific but not very sensitive in distinguishing CD from UC. In this study, an ASCA+/ANCA− test result offered the best sensitivity and specificity for CD (55% and 90%, respectively). In fact, these tests showed even higher rates in the pediatric IBD population. In a study consisting of 135 pediatric patients (81 with CD and 54 with UC), an antibody panel consisting of pANCA, ASCA, OmpC, and CBir1 identified 65% of children with CD and 76% of those with UC, with a specificity of 94%. In CD, OmpC has been correlated with increased risk of fibrostenosing disease, internal penetrating disease, and the need for small-bowel surgery. CBir1 has been shown to identify cases of CD not detected with other markers. Approximately 55% of CD patients are positive for anti-CBir1, and this marker independently helps identify a subset of CD patients (particularly those with fibrostenosing disease) who may also be ANCA positive.
Use of serological testing has not been extensively studied for IC. In a prospective European study of 97 patients with a diagnosis of IC, only a small minority showed characteristic serological patterns that helped establish a diagnosis of ASCA+/ANCA− CD or ASCA−/ANCA+ UC. In most patients, there was insufficient information to accurately classify IC patients as having either CD or UC. Therefore the clinical utility of these markers in IC appears limited.
UC characteristically involves the mucosa in a diffuse and continuous manner. It almost always affects the rectum. The terminal ileum may be affected to a lesser degree and exclusively in patients with pancolitis (i.e., BWI). Infections, drug or medication-related injury (e.g., NSAIDs), and bowel preparation agents can also result in inflammation of the terminal ileum. In contrast, CD shows segmental or patchy involvement of the bowel and often affects longer lengths of terminal ileum, both actively as well as chronically. In a resection specimen, a diagnosis of UC is usually established by the presence of diffuse chronic active colitis or chronic inactive colitis involving the mucosa in the absence of mural changes typical of CD, such as fissuring ulcers, transmural lymphoid aggregates, non–mucin-related granulomas, mural fibrosis, and fistulas or sinus tracts. Neural hyperplasia, hyperplasia of muscularis mucosae, and submucosal fibrosis are also less common in UC than in CD. In addition, involvement of other regions of the GI tract, especially perianal disease, is a sign of CD. Upper GI involvement in UC has been described, but it is unclear whether these changes are specific to UC or represent a nonspecific systemic immune response to a generalized inflammatory condition. A summary of the classic microscopic features of UC and CD is provided in Table 17.2 .
Because most resections are performed for medically refractory disease or disease-related complications and therefore have been treated medically, both UC and CD may show patchy, or segmental, histological changes. One may also see a reduction in the amount of transmural inflammation in CD. Granulomas are also far less common after treatment.
UC and CD demonstrate considerable overlap in morphological features in biopsy samples with chronic and/or active disease. Furthermore, because the characteristic features of CD are typically found deeper in the bowel wall, it is difficult or impossible to distinguish UC from CD based solely on analysis of mucosal biopsy specimens. However, in untreated cases, features in favor of CD are granulomas not associated with crypt rupture, long or multiple segments of small bowel involvement, clear evidence of upper GI involvement (esophagus, stomach, duodenum, jejunum), rectal sparing at disease onset, particularly in adults, fistulas, and CD-like perianal disease, such as recurrent and severe or unremitting fissures, fistulas, anal tags, ulcers, and hemorrhoidal disease. Patchy disease in the same segment before treatment and evidence of deep and prominent submucosal involvement in a patient without fulminant colitis are additional features that favor CD over UC.
After medical therapy, either orally, intravenously, or by enema, an uneven pattern of healing caused by the therapeutic effects of drugs used for the disease often cause the histological features of UC to become patchy in distribution, mimicking the pattern of CD in the colon (see Unusual Morphological Variants of Ulcerative Colitis). Thus in the posttreatment setting, pathologists cannot consider “patchiness” of disease or “rectal sparing” as features necessarily indicative of CD. In medically treated patients, unless there is confirmed radiological evidence of CD-like anal or perianal disease, granulomas unrelated to ruptured crypts, or definite CD-like ileal or upper GI involvement, it is almost impossible to distinguish UC from CD in this setting.
Fewer than 1% of all CRCs in the United States are associated with IBD. Colorectal carcinoma arising in UC was first documented in 1925 by Crohn and Rosenberg. Warren and Sommers were first to document CRC in a patient with CD in 1948. Both UC and CD patients are at increased risk for development of dysplasia and carcinoma. However, estimation of risk is influenced by numerous factors, such as differences in sampling protocols, recognition and classification of disease, treatment regimens, and methods used to detect neoplasia. Evidence from two population-based studies, one from Manitoba, Canada (RR in CD, 2.64; RR in UC, 2.75) and another from Olmstead County, Minnesota (standardized incidence ratio [SIR] in CD 1.9; SIR in UC, 2.4), indicated that the risk of cancer is roughly equivalent in UC and CD in patients with equal lengths of affected colon. In another study, Choi and colleagues found no significant differences in age at diagnosis, duration of IBD, multiplicity and distribution of cancer, presence of dysplasia, or 5-year survival rate in patients with CD-associated versus UC-associated CRC.
The overall incidence of CRC after 25 to 35 years of UC ranges from 3% to 43%. , In two recent population-based cohort studies, UC-associated CRC accounted for only 0.15% to 0.4% of all cases of CRC diagnosed in the general population. A meta-analysis of 116 studies estimated the risk of cancer in patients with UC at 2% after 10 years, 8.5% after 20 years, and 17.8% after 30 years of disease. Five years later, data from a 30-year surveillance program at St. Mark’s Hospital in the United Kingdom calculated the risk of cancer and dysplasia as 2.5% at 20 years, 7.6% at 30 years, and 10.8% at 40 years. Subsequent population-based studies also suggested that the risk of developing dysplasia/cancer has decreased with time. In a recent study that included 32,911 Danish patients diagnosed with UC, the RR of CRC (1.07; 95% CI, 0.95 to 1.21) was comparable to that of the general population. The change in incidence rate, especially during the first 1 to 10 years after diagnosis, is likely the result of improved surveillance, early use of colectomy for medically refractory disease, and significant improvements in medical therapy.
Features associated with an increased risk of dysplasia and carcinoma in patients with UC include increased duration of disease, increased anatomic extent of disease, associated PSC, a family history of sporadic cancer, young age at onset, and severity of endoscopic and histological disease activity. The RR for dysplasia or cancer significantly increases after 8 to 10 years of disease duration.
Most cases of dysplasia/cancer arise in a background of pancolitis or at least subtotal colitis. Limited left-sided colitis cases carry an intermediate risk, whereas proctitis or proctosigmoiditis carries either little or no risk of cancer at all. , Compared with RRs of 1.7 and 2.8 for patients with proctitis and left-sided colitis, respectively, patients with pancolitis have an RR of 14.8 for neoplasia.
PSC develops in approximately 2% to 5% of UC patients. In a meta-analysis that included 22 studies, patients with PSC and UC were found to have a fourfold increased risk of dysplasia/cancer. UC patients who have undergone liver transplantation for PSC also have a higher risk of dysplasia/cancer, estimated at 1% per year. For these reasons, it is highly recommended that patients undergo annual colonoscopy from the onset of PSC.
A positive family history of sporadic colon cancer is a well-documented predisposing factor for colon cancer, with a more than twofold associated risk. Younger age at onset of colitis has been associated with an 8.6-fold increased risk of dysplasia/cancer. , However, this estimate must be interpreted with caution because of the lower rates of CRC observed in the general population used for comparison in this age group. Nevertheless, this increased risk likely reflects the high proportion of pediatric IBD patients who have extensive disease. ,
Two previous studies from the United Kingdom demonstrated that the severity of endoscopic and histological inflammation is related to colon cancer risk. , In a recent meta-analysis, a diagnosis of extensive colitis was associated with a 4.8-fold increased risk of dysplasia/colon cancer.
There is wide variability in the reported risk of dysplasia/CRC in CD, primarily because of inconsistencies in documentation of the duration and location of CD (colonic vs. isolated small-bowel CD). , In CD, there is also an increased risk for dysplasia and adenocarcinoma in excluded segments of bowel and also in the small intestine.
In a large, population-based survey of 1655 patients with long-standing and extensive CD, the reported RR of CRC was 2.5 (95% CI, 1.3 to 4.3). A meta-analysis of 12 studies showed a higher RR (4.5) for patients with colonic disease. In a recent Danish study, the RR of dysplasia/colon cancer among 14,463 patients diagnosed with CD did not differ significantly from that in the background population, nor did it change over a period of 30 years. This has mostly been attributed to improved medical therapy and surveillance programs.
As in UC, patients with long-standing CD and a family history of sporadic colon cancer are at increased risk for dysplasia/cancer. , , There is a convincing association between degree of inflammation and dysplasia/cancer in CD as well. A population-based study from Sweden showed that the RR for colon cancer was 5.6 in CD patients with disease restricted to the colon. The RR was even greater in patients who were younger than 30 years of age at the time of diagnosis. Therefore extent of colitis, duration of disease, family history of sporadic colon cancer, and young age at presentation are risk factors for development of dysplasia and cancer in colonic CD. In contrast with UC, PSC does not seem to increase the risk of colon cancer in patients with colonic CD. This was recently demonstrated in a case-control study consisting of 114 patients with colonic CD.
Dysplasia is defined as unequivocal neoplastic epithelium confined to the basement membrane. At present, it is the best and most reproducible marker of malignancy risk in patients with IBD. It is present in up to 90% of patients with UC and up to 87% of patients with CD who have carcinoma. It may occur adjacent to and/or distant from the primary tumor. Dysplasia may occur in any portion of the colon, but it most often parallels the location of cancer; it may occur as an isolated focus, but more often is multifocal. Rarely, it is diffuse.
Before the advent of image-enhancing endoscopic techniques, such as high-definition colonoscopy or chromoendoscopy, dysplasia was classified as either flat or elevated; the latter types of lesions were termed dysplasia-associated lesion or mass (DALM). This classification was considered important because these two types of dysplasia were managed differently. There is inconsistency in the literature with regard to the criteria used to define raised, endoscopically visible, dysplastic lesions as DALMs. For instance, the term flat dysplasia has been used to describe endoscopically detectable, but slightly raised, lesions in some studies, but not in others.
Raised dysplastic lesions in UC (and CD) were further separated into those that resembled non–IBD-related sporadic adenomas (“adenoma-like”) and those that did not resemble adenomas, which were termed non–adenoma-like. Endoscopically resectable well-circumscribed, smooth or papillary, nonnecrotic, sessile or pedunculated polyps were called adenoma-like dysplastic lesions (also known as adenoma-like polyp, adenoma-like dysplastic polyp, adenoma-like low-grade or high-grade dysplasia, polypoid dysplasia, or adenoma-like mass ) , ( Fig. 17.33 ), while velvety patches, plaques, irregular nodules, wartlike thickenings, carpet-like lesions, stricturing lesions, and broad-based masses that could not be resected by endoscopic methods, and therefore required surgery were categorized as non–adenoma-like dysplastic lesions ( Fig. 17.34 ).


Recently, the endoscopic classification system of dysplasia in IBD has changed. The new guidelines have proposed an endoscopic classification system of dysplasia that conceptually aligns with the clinical management of dysplasia. This new classification system is the Surveillance for Colorectal Endoscopic Neoplasia Detection and Management in Inflammatory Bowel Disease Patients: International Consensus Recommendations (SCENIC) ( Table 17.3 ).
| Term | Definition |
|---|---|
| Visible dysplasia | Dysplasia identified on targeted biopsies from a lesion visualized at colonoscopy |
|
|
|
|
|
|
|
|
|
Lesion with protrusion but <2.5 mm above the lumen (less than the height of the closed cup of a biopsy forceps) |
|
|
|
Lesion with at least a portion depressed below the level of the mucosa |
| Invisible dysplasia | Dysplasia identified on random (nontargeted) biopsies of colon mucosa without a visible lesion |
This classification is based on the use of high-definition colonoscopy and chromoendoscopy to aid in the visualization of mucosal abnormalities. Based on these recommendations, the terms DALM, adenoma-like, and non–adenoma-like are no longer used in clinical practice. Instead, dysplasia is now categorized as either visible or invisible endoscopically. Visible dysplasia is defined as dysplasia identified on targeted biopsies from a lesion visualized at colonoscopy, while invisible dysplasia is identified on random (nontargeted) biopsies of colonic mucosa, without a visible lesion. This classification scheme (modified from the Paris Classification) further divides visible lesions into polypoid (pedunculated or sessile) or nonpolypoid (superficial elevated, flat or depressed) types (see Table 17.3 ).
Retrospective studies have shown that most dysplastic lesions in IBD are, in fact, polypoid rather than flat or invisible. , For instance, in a large retrospective review of 2204 surveillance colonoscopies in UC patients, 77% of the 104 dysplastic lesions found were elevated or polypoid (or both).
Regarding the anatomic distribution of dysplasia in UC, low-grade dysplasia was found in one study to be more commonly localized to the distal colorectum (68%). Similarly, in a pooled analysis, Choi reported that 52% (range, 47% to 62%) of colitis-related CRCs occurred in the rectosigmoid region. Additionally, a 20-year prospective study and a 30-year analysis of surveillance colonoscopy showed that 44% and 58.6% of dysplastic lesions occurred within the rectosigmoid region, respectively. , A retrospective study of flat dysplasia by Goldstone and colleagues confirmed that in patients undergoing surveillance for long-standing UC, 74.6% of neoplasia (flat low-grade dysplasia, flat high-grade dysplasia, or carcinoma) occurred in the distal colon (64.4% in the rectosigmoid and 10.2% in the descending colon), whereas only 25.4% occurred in the ascending colon, cecum, or transverse colon.
In CD, dysplasia also occurs more often in areas close to, rather than distant from, the primary tumor. Dysplasia distant from cancer has been identified in 38% to 41% of specimens with colonic CD. , Regardless of the location of the primary tumor, dysplasia/cancer always develops in areas of either prior or current colitis, as in UC. In a study by Sigel and associates, dysplasia was found adjacent to carcinoma in 87% of cases and distant from carcinoma in 41% of cases. In their study of 30 cases of CD-related adenocarcinoma, 27% occurred in the small intestine and 73% occurred in the colon. In a study that evaluated 50 cases of Crohn’s colitis-associated dysplasia, 44% of patients were found to have multifocal dysplasia/cancer in segments not affected by colitis.
According to the grading system proposed by Riddell and colleagues in 1983, dysplastic changes in UC and CD are separated into three distinct categories: negative for dysplasia, indefinite for dysplasia, and positive for dysplasia (low and high grade). The Vienna grading system, which denotes five diagnostic categories, is used by many pathologists in Japan and Europe. A comparison of the two systems is outlined in Table 17.4 . Both visible and invisible dysplastic lesions are graded in the same manner.
| Vienna | Riddell |
|---|---|
| 1. Negative for neoplasia/dysplasia | Negative for dysplasia |
| 2. Indefinite for neoplasia/dysplasia | Indefinite for dysplasia |
| 3. Noninvasive low-grade neoplasia (low-grade adenoma/dysplasia) | Low-grade dysplasia |
| 4. Noninvasive high-grade neoplasia 4.1 High-grade adenoma/dysplasia 4.2 Noninvasive carcinoma (carcinoma in situ) 4.3 Suspect for invasive carcinoma |
High-grade dysplasia |
| 5. Invasive neoplasia | Adenocarcinoma ∗ |
| 5.1 Intramucosal adenocarcinoma | Intramucosal |
| 5.2 Submucosal carcinoma or beyond | Invasive |
The grade of dysplasia is determined by evaluating a combination of cytological and architectural alterations of the epithelium. A diagnosis of “negative” is reserved for nondysplastic or regenerating epithelium ( Fig. 17.35 ).

Cases are considered “indefinite for dysplasia” when the features of the atypical epithelium resemble dysplasia, but because of other factors such as inflammation, ulceration, or technical artifact, a definite diagnosis cannot be established with complete certainty ( Fig. 17.36 ). Inflammation and ulceration make interpretation of atypical changes difficult (i.e., reactive vs. neoplastic). Regenerating epithelium often shows enlarged and variably sized nuclei, nuclear stratification, prominent nucleoli, and increased mitoses. These changes overlap with those observed in true dysplasia. In some cases, tangential sectioning of the tissue or severe cautery or processing artifact may also render an atypical focus difficult to interpret. In general, the presence of surface maturation is usually (but not always) indicative of a regenerative process. Thus the presence of this feature should always evoke great caution in diagnosing true dysplasia ( Table 17.5 ).

| Feature | Regenerative Changes | Dysplasia |
|---|---|---|
| Nuclear enlargement | Usually minimal | Always present |
| Nuclear hyperchromasia | May be present | Always present |
| Nuclear stratification | May be present near healing ulcers | Present in adenomatous (intestinal)-type dysplasia |
| Nuclear pleomorphism | Usually minimal | Always present |
| Irregular nuclear contour | Usually minimal | Always present |
| Loss of nuclear polarity | Absent | Present (in high-grade dysplasia) |
| Prominent nucleoli | May be present | Present in high-grade dysplasia |
| Vesicular chromatin | Usually absent | Present (especially in high-grade dysplasia) |
| Loss of surface maturation | Absent | Present (except in crypt dysplasia) |
| Increased mitoses | May be present | Usually present |
| Atypical miotic figures | Usually absent | Present |
| Increased nucleus-to-cytoplasm ratio | May be present | Always present |
| Syncytial appearance of cells with abundant eosinophilic cytoplasm | Present | Usually absent |
| Villous configuration | May be present | Present |
| Mucin loss | Present | Present |
| Epithelial “atypia” overlying inflamed stroma that lacks glands | Present | Usually absent |
| Intraepithelial neutrophils | Usually present | May be present |
| Complex glandular architecture | Absent | Present in high-grade dysplasia |
In low-grade dysplasia, the crypts show a tubular and/or villous configuration and may also demonstrate focal or mild crypt budding and crowding. Cytologically, dysplastic crypts are lined by cells that show nuclear enlargement, elongation (often referred to as pencillate ), increased nucleus-to-cytoplasm (N:C) ratio, hyperchromasia, stratification, a clumped chromatin pattern, single or multiple nucleoli, and hypereosinophilic, mucin-depleted cytoplasm ( Fig. 17.37 ). Dysplastic epithelium normally involves both the crypt and surface epithelium, but early cases may show involvement of the crypts only ( crypt dysplasia; see later discussion). Mitotic figures are often plentiful, but atypical mitotic figures are less common than in high-grade dysplasia. In low-grade dysplasia, the nuclei are usually limited to the basal half of the cell cytoplasm, and there are no significant architectural abnormalities such as glandular crowding, cribriforming, or a back-to-back gland pattern. Other, less common features of low-grade dysplasia include dystrophic goblet cells and endocrine and Paneth cell metaplasia.

With progression from low- to high-grade dysplasia, the cells acquire more cytological atypia, characterized by even more pronounced nuclear enlargement, pleomorphism, hyperchromasia and, in particular, loss of nuclear polarity ( Fig. 17.38 ). The nuclei may show a more “open” chromatin pattern and contain prominent and enlarged nucleoli. Full-thickness nuclear stratification is characteristic when the nuclei are pencil shaped. However, many high-grade lesions show enlarged, but irregular, round nuclei rather than pencil-shaped nuclei. In these cases, nuclear stratification may not be present. Architecturally, back-to-back glands or cribriforming may be present, and this is always considered a high-grade feature, especially when it is prominent and even if the nuclei appear more low-grade cytologically. Mitotic figures, both typical and atypical, are more frequent than in low-grade lesions and are usually present in both the upper and lower portions of the crypts and in the surface epithelium. In general, the features of the most atypical portion of dysplastic mucosa determine the overall grade of dysplasia in any particular biopsy sample. However, there is no uniform agreement regarding the proportion of high-grade dysplastic crypts that is necessary to upgrade a biopsy from low to high grade. A simple rule of thumb is that there should be more than one or two (i.e., more than rare) high-grade crypts present to designate a biopsy as high-grade, but admittingly, this number varies among expert pathologists.

Intramucosal carcinoma is defined by the presence of cells or glands that have penetrated into, but not beyond, the lamina propria or muscularis mucosae. Common patterns of early invasion include single-cell or small-gland infiltration, glands with irregular jagged contours, and extensive cribriforming with pushing borders. Desmoplasia is absent when the carcinoma is limited to the mucosa. Thus its presence is normally diagnostic of submucosal invasion ( Fig. 17.39 ).

Historically, intestinal (“adenomatous”), hypermucinous/villous, and serrated dysplasia were considered the predominant morphological subtypes of dysplasia in IBD. In one study of IBD cancers, 52% of dysplastic lesions were villous, 29% were serrated, 5% were tubular (adenomatous), and 13% showed mixed features. However, more recently, an international group of IBD pathology experts proposed a novel histological classification system of IBD-associated dysplastic lesions that serves to recognize several additional variants that are rare and often difficult to recognize ( Table 17.6 ). This new classification includes four general categories: intestinal, gastric, mixed intestinal and gastric, and unclassifiable . The intestinal type of dysplasia is further separated into tubular/villous (adenoma-like), goblet cell–deficient, crypt cell, sessile serrated lesion (SSL)-like, traditional serrated adenoma (TSA)-like, and serrated, not otherwise specified (NOS) subcategories. The tubular/villous (adenoma-like) intestinal-type of dysplasia remains the most common and most well-characterized type, both histologically and clinically, However, there is emerging data with regard to the other subtypes, and this is discussed in more detail later. Unfortunately, much of the prior dysplasia literature either does not specify clearly the type of dysplasia under study or refers specifically to the intestinal type. As a result, the following discussion applies mainly to the tubular/villous intestinal type of dysplasia, unless otherwise specified.
| Category of Dysplasia | Description |
|---|---|
| Intestinal Type | |
|
|
|
|
|
|
|
|
|
|
|
|
| Gastric Type | |
|
|
|
|
| Mixed Intestinal and Gastric Type |
|
| Unclassifiable |
|
This type of dysplasia is by far the most common type of dysplasia encountered in patients with IBD. Morphologically, it resembles conventional colorectal adenomas. The growth pattern is tubular and/or villous, and the crypts are often crowded. The nuclei are enlarged, elongated, hyperchromatic, and stratified. The density of goblet cells is variable. The intervening columnar cells are enterocyte-like but may contain a small amount of microvesicular mucin. Some lesions contain prominent Paneth cells or endocrine cells. Surface maturation is minimal or absent except near villous tips (see Fig. 17.37 ).
This form of dysplasia is characterized by crypts that either lack completely, or show only rare, goblet cells. The cells show nuclear hyperchromasia and stratification and elongation of nuclei, and it involves the crypts as well as the surface epithelium ( Fig. 17.40 ). In the original study that described this morphological variant of dysplasia, it was associated with the highest interobserver agreement (95% agreement) among all GI IBD pathology experts.

Crypt cell dysplasia is characterized by a noncrowded, uniform flat tubular growth pattern of the epithelium. The crypts are lined by “terminally differentiated” enterocytes interspersed with goblet cells, basally located Paneth cells, and scattered endocrine cells ( Fig. 17.41 ). The nuclei are typically round or oval-shaped, enlarged, hyperchromatic, or vesicular. As opposed to normal or reactive epithelium, crypt cell dysplasia shows lack of maturation to the surface with the nuclei showing either very little or no reduction in overall size. Paneth and endocrine cells may be sparse or completely absent in some cases. The term crypt cell dysplasia is a consensus-based revision of the original term dysplasia with terminal epithelial differentiation in the prior study that described the rare morphological subtypes of dysplasia.

It has been known for quite some time that serrated lesions, both visible and invisible, may develop in IBD patients. In fact, some early studies had suggested that serrated dysplasia is quite common, and in some studies, even more common than the intestinal type. Unfortunately, early studies did not differentiate the various subtypes of serrated dysplasia or recognize that even IBD patients may develop sporadic serrated lesions. Thus the old literature is now difficult to interpret. Serrated lesions may be visible or invisible. Visible lesions are often polypoid, and these range from those that resemble conventional hyperplastic polyps ( Fig. 17.42A ) at one end of the cytological atypia scale to SSLs and TSAs at the other end of the scale. In some cases, serrated lesions may not quite fit nicely into any of these categories, and in these cases the term serrated dysplasia NOS may be used. Invisible (randomly sampled) and nonpolypoid colonic mucosa may also reveal dysplastic lesions with a serrated pattern of growth and even some with cytological and architectural features identical to that of an SSL or TSA.

Sessile serrated lesion–like dysplasia is similar to SSLs seen in the sporadic setting and is characterized by crypts with serrated architecture wherein the serrations may involve the basal or superficial crypts, or both. The crypts show dilation, with or without lateral extension, resulting in L- or T- shaped configurations (see Fig. 17.42B ). However, unlike sporadic SSL, flat crypt bases are not a required feature. The cells have microvesicular cytoplasm and are interspersed with smaller numbers of goblet cells. The nuclei are round to oval or slightly elongated, hyperchromatic, and may contain inconspicuous nucleoli. They are basally located and may be slightly stratified at the crypt base, and there may be maturation at the surface. This category excludes lesions that resemble conventional SSLs without cytological dysplasia.
Traditional serrated adenoma-like dysplasia resembles a sporadic TSA in which the lesion is composed of villiform epithelial proliferation containing slitlike epithelial serrations along with ectopic crypts or aberrant crypt foci. The individual cells show pencillate nuclei with fine chromatin and prominent eosinophilic cytoplasm (see Fig. 17.42C ). In a recent study that evaluated 52 TSA-like lesions in 30 IBD patients, the prevalence of TSA-like lesions in IBD patients was found to be 0.4%. In this series, 23/27 TSA-like lesions identified in colectomy specimens presented as ill-defined plaquelike areas of the mucosa with a granular appearance, and low-grade and high-grade serrated dysplasia was found in 15 of 27 and 10 of 27 cases, respectively.
Little is known regarding the biological characteristics and natural history of serrated lesions in IBD. However, there are some data on hyperplastic changes and hyperplastic-type polyps. In a large clinicopathological and outcome study published in abstract form, 188 polyps from 161 IBD patients with at least 1 serrated polyp were analyzed. Most were hyperplastic polyps (97%) that were detected microscopically, not endoscopically. Adenocarcinoma, adenoma, or flat or elevated dysplasia did not develop in any of the patients, but additional hyperplastic or inflammatory polyps developed in 33% of the patients. In a retrospective study consisting of 94 patients with “hyperplastic polyp-like changes” in biopsies of flat mucosa obtained during routine surveillance (“flat serrated change”), the authors found that 12.8% of these patients had subsequent visible dysplasia on follow-up. However, a multivariate analysis did not show a statistically significant difference in the risk between patients with and without these lesions. Based on these results, the authors suggested that there should be no change in routine surveillance protocols for patients with invisible hyperplastic polyp–like serrated change. One study evaluated serrated lesions in 78 IBD patients and classified them as SSA/P-like (negative for dysplasia), TSA-like (positive for low-grade dysplasia), and cytologically atypical hyperplasia–like (indefinite for dysplasia). The authors found that SSA/P-like visible lesions in IBD occurred preferentially in females and in the right colon, similar to sporadic polyps. Nearly 11% of these nondysplastic SSA/Ps were associated with synchronous or metachronous neoplasia over a follow-up period of 103 months. In contrast, TSA-like and cytologically atypical hyperplasia–like polyps were significantly associated with synchronous or metachronous neoplasia (low-grade dysplasia in 17, high-grade dysplasia in 6, and invasive adenocarcinoma in 3).
IBD patients may rarely show numerous hyperplastic and serrated lesions in their colon, reminiscent of the serrated polyposis syndrome. In one study by Srivastava et al., three patients with IBD (UC in 2, CD in 1) developed a wide spectrum of serrated lesions ranging from invisible serrated dysplasia to a broad range of visible lesions such as hyperplastic-like polyps, sessile serrated lesion–like polyps, and TSA-like polyps throughout the colon. The visible lesions contained various grades of dysplasia ranging from little to no cytological atypia up to adenocarcinoma (two patients). In this study, it was not clear whether the study patients had IBD and superimposed serrated polyposis or whether the serrated lesions developed as a direct result of the underlying IBD; however, the latter was favored by the authors of that study.
Thus, in general, in a patient with a visible lesion that resembles a serrated polyp of any kind, it is important to determine whether the lesion has arisen within or outside the segment of bowel affected by IBD. Similar to sporadic adenomas, patients with IBD may develop “sporadic” serrated polyps that are unrelated to IBD. Regardless, until we know more about the clinical behavior of serrated dysplasia that is related to IBD, the clinical management for visible and invisible lesions is similar to that of serrated lesions seen in non-IBD patients.
Rarely, dysplastic lesions in either UC or CD are composed nearly exclusively of cells with mucinous cytoplasm, similar in appearance to gastric foveolar cells. These cases typically show either no or only a few goblet cells within the dysplastic epithelium. The predominant growth pattern of these lesions may be tubular/villous, completely villous, or even serrated.
Gastric dysplasia has been termed mucinous, hypermucinous, or hypermucinous villous in prior studies. It is a rare form of dysplasia, and when present, it may show such minimal cytological atypia that it may be difficult to recognize it as unequivocally neoplastic. In general, the dysplastic epithelium shows relatively small, hyperchromatic, basally oriented nuclei with minimal, or in some cases, no cytological atypia. , ( Fig. 17.43 ). However, more traditional dysplastic changes may be present in higher-grade lesions. These features include a crowded, back-to-back arrangement of glands lined by cells with hyperchromatic and markedly enlarged nuclei. Prominent nucleoli are common. For the most part, the nuclei maintain their basal orientation or are only partially stratified. In general, the colonic epithelium is rather resistant to mucinous metaplasia, with a few exceptions such as pyloric metaplasia, or focally within hyperplastic polyps (in a microvesicular pattern). Thus the finding of gastric-like mucinous metaplasia should always arouse strong suspicion for gastric dysplasia in an IBD patient. In three recent studies (one published in abstract form) that evaluated the different morphological subtypes of dysplasia in IBD, the prevalence of mucinous dysplasia ranged from 6.8% to 14%. Given the limited number of cases of mucinous dysplasia reported to date, the natural history and biological characteristics of this variant remain largely unknown.

Gastric serrated dysplasia is similar to tubular/villous adenoma–like dysplasia but is distinguished by a strikingly serrated growth pattern involving both the basal and superficial crypts, sometimes resulting in mucin-filled cystic glands. The nuclei are round and vesicular and show little or no reduction in size near the surface ( Fig. 17.44 ).

In many patients, dysplastic lesions show a combination of two or even three morphological subtypes of dysplasia. These may be all within the same general category (e.g., tubular/villous subtype of intestinal dysplasia and traditional serrated adenoma–like dysplasia), or it may occur as a mixture of separate categories (tubular/villous forms of intestinal dysplasia combined with gastric type dysplasia; see Fig. 17.44 ). Histologically, the various subtypes of dysplasia may occur both within the same foci of dysplasia or as spatially distinct lesions. In a study (published in abstract form) consisting of 50 patients all of whom had a colectomy for dysplasia in IBD (UC = 43, CD = 7), of the 67 foci of dysplasia, there were 16 (24%) foci of mixed dysplasia. The most common mixed pattern was intestinal tubular/villous combined with serrated ( N = 9/16; 56%). Two of the 16 mixed cases (13%) showed a combination of intestinal (tubular/villous and serrated) and gastric (tubular/villous) dysplasia. In our experience, the mixed form of dysplasia is more likely to be encountered in large-sized visible lesions rather than in small visible lesions.
Although better characterized in patients with Barrett’s esophagus, rarely patients with IBD may develop early dysplastic changes limited to the bases of the crypts without involvement of the surface epithelium (i.e., surface maturation is present). The lesions usually have a flat configuration, noncrowded crypts, and cytoplasmic features that simulate the repertoire of normal colonic epithelium. This is also known as crypt dysplasia ( Fig. 17.45 ). In our experience, most patients with crypt dysplasia reveal conventional dysplasia (involving both crypt and surface epithelium) elsewhere in the colon at the time of discovery of crypt dysplasia. Because crypt regeneration also shows surface maturation, a diagnosis of crypt dysplasia should not be made in biopsy specimens exhibiting inflammation or ulceration or in those that are not well oriented. At this time, the biological characteristics and natural history of crypt dysplasia in IBD are largely unknown. In a small series of 14 colon biopsies from 7 patients with IBD, aneuploidy was detected in all 14 biopsies with crypt dysplasia in IBD, and 6 patients developed HGD ( n = 5) or adenocarcinoma ( n = 1) in the same colonic segment where crypt dysplasia was diagnosed within a mean follow-up duration of 27 months.

As discussed earlier, the treatment of dysplasia in IBD depends largely on the endoscopic appearance and resectability of the lesion. The SCENIC guidelines define an endoscopically resectable lesion as one that has distinct margins, appears to be completely removed on visual inspection after endoscopic resection, is histologically consistent with complete removal, and the biopsies taken from mucosa immediately adjacent to the resection site are free from dysplasia.
Historically, distinction between adenoma-like DALM (polypoid visible dysplasia in the SCENIC classification) and sporadic adenoma was considered important because the former was usually treated by colectomy, whereas the latter was treated by polypectomy. However, more recent data suggest that patients with UC and an “adenoma-like” lesion may be treated adequately by polypectomy and continued surveillance, regardless of the pathogenesis of the lesion. In a study consisting of 24 UC patients with an “adenoma-like DALM” within areas of colitis, 10 UC patients with an “adenoma-like DALM” outside areas of colitis, and 49 non-IBD (control) patients with sporadic adenomas, 62.5% patients in the first group developed an additional adenoma-like DALM, only 1 patient (4%) developed a single focus of flat dysplasia, and 1 patient (4%) developed a poorly differentiated adenocarcinoma of the cecum by 7.5 years after polypectomy. Among the patients with adenoma-like DALMs outside areas of colitis, 50% developed additional adenoma-like DALMs proximal to areas of colitis, but none developed invisible dysplasia or carcinoma. This group was not significantly different from the control group, 49% of whom developed further adenomas during the follow-up interval. A subsequent study by Kisiel et al. also showed that of 77 UC patients with polypoid visible dysplasia, 36% developed another polypoid low-grade dysplastic lesion, 5% developed invisible low-grade dysplasia, and only 1 patient (1%) developed carcinoma after a median follow-up interval of 20.1 months.
The presence of high-grade dysplasia within an adenoma-like DALM (now referred to as polypoid visible dysplasia ) is not considered a contraindication to polypectomy. In the study by Blonski et al., invisible dysplasia or carcinoma did not develop in any of 9 UC patients with such lesions during a follow-up period of 76.5 months.
In a recent meta-analysis consisting of 1037 IBD patients undergoing endoscopic resection for a total of 1428 colonic lesions, the pooled risk (rate per 1000 person-years of follow-up) of CRC was 2 (95% CI, 0 to 3), the pooled risk of high-grade dysplasia was 2 (95% CI, 1 to 3), and the pooled risk of any lesion was 43 (95% CI, 30 to 57) after complete resection of the lesion. These data highlight the effectiveness of endoscopic resection and follow-up for patients with visible polypoid dysplastic lesions. Unfortunately, most of the previous studies have not addressed differences in outcome between pedunculated and sessile visible dysplastic lesions.
Biopsy specimens from lesions that are deemed nonresectable via endoscopic techniques ( Fig. 17.46 ) often show only dysplastic epithelium, which usually represents surface sampling of a carcinoma. Several studies have detected a strong association between endoscopically unresectable lesions and cancer at colectomy. Given the high risk of synchronous or metachronous adenocarcinoma, the presence of such lesions is a strong indication for colectomy. , ,

Most of the data regarding the natural history and treatment of dysplasia in IBD apply mainly to UC. It has been reported that the 5-year rate of development of either high-grade dysplasia or carcinoma in patients with low-grade dysplasia is 54%. As a result, many institutions recommend colectomy for UC patients with invisible low-grade dysplasia, particularly if it is found on initial colonoscopy, is multifocal, or develops in a metachronous fashion. , , However, some institutions prefer to place patients with invisible low-grade dysplasia under surveillance (see later discussion). Ullman and colleagues showed that 27% of patients with UC who underwent a colectomy within 6 months after a biopsy diagnosis of invisible low-grade dysplasia had unexpected high-grade dysplasia or cancer in their resection specimen. In contrast, invisible high-grade dysplasia is associated with a higher probability of detecting cancer at colectomy (40% to 67%) and of progression to carcinoma (5-year predictive value, 40% to 90%). , , For these reasons, patients with invisible high-grade dysplasia, even if it is unifocal, are referred for colectomy. More recent studies using chromoendoscopy or high-definition white-light endoscopy have reported a 10% incidence rate of invisible dysplasia. Thus random biopsy samples showing invisible dysplasia in older studies may have been obtained from mucosa that may now be recognizable as a distinct mucosal abnormality using modern endoscopic techniques.
In all cases of dysplasia assessment, it is important and highly recommended by the American Gastroenterology Association and the American College of Gastroenterology that the presence or absence of dysplasia be confirmed by at least one pathologist who is skilled in GI pathology or who has broad experience with dysplasia in UC. ,
A study by Kiran and colleagues suggests that instead of segmental colectomy, total proctocolectomy should be strongly considered for CD patients with a preoperative diagnosis of dysplasia because as many as 44% show multifocal dysplasia. Similarly, patients with high-grade dysplasia have a 45% risk of having synchronous cancer.
Patients with biopsy specimens considered indefinite for dysplasia should be treated for their inflammation and have a repeat endoscopy within 6 months, preferably after the inflammation has subsided. ,
Sampling error and interobserver variability are common problems encountered in the interpretation of dysplasia in IBD. Since the original 1983 study by Riddell and coworkers, several investigations have shown only moderate levels of interobserver agreement. , In general, levels of agreement among pathologists are highest for the category of high-grade dysplasia and negative for dysplasia; they are lowest for biopsy specimens showing low-grade dysplasia or indefinite for dysplasia. Recent studies have focused on finding other, more reproducible, adjunctive methods of assessing malignancy risk in UC. These include a variety of histochemical (e.g., mucins, sialosyl-Tn), immunohistochemical (e.g., proliferation markers, TP53), and molecular ( APC, p27, p16, aneuploidy) techniques. α-Methylacyl-CoA racemase has been shown to be sensitive and highly specific for dysplasia in IBD. In one study, 90% of low-grade dysplasia, 80% of high-grade dysplasia, and 71% of adenocarcinomas in IBD were positive, in contrast with none of the foci of nondysplastic regenerating epithelium. Several studies have shown increased expression of TP53 in the progression of neoplasia in IBD, , but a small proportion of nondysplastic cases may also be positive. Furthermore, TP53 immunostaining is fraught with a high false-positive rate, which makes TP53 less useful for differentiating regeneration and true dysplasia in IBD.
Tissue-based studies have demonstrated that IBD-related neoplasia is associated with aberrant methylation in EYA4, ER, CDKN2A, MYOD, CDH1, RUNX3, MINT1, and COX-2 . A tissue-based study showed that methylation of genes in the WNT signaling pathway is an early event in patients with IBD colitis, and there is a progressive increase in methylation of the WNT signaling genes APC1A, APC2, SFRP1, and SFRP2 during development of IBD-associated neoplasia. In an in-depth analysis of IBD-associated CRCs and sporadic cancers performed using whole-exome sequencing, Robles and colleagues showed a lower rate of APC activation and a higher rate of SOX9 inactivating mutations in the IBD-CRC cohort compared with the sporadic CRC cohort. This study also noted that despite both the cohorts showing similar missense mutations within the DNA-binding domain of p53, the identity and molecular distribution of single substitution mutations were different. In another study, Yaeger and colleagues analyzed genomic alterations in over 300 cancer-related genes in 29 UC and 18 CD-associated cancers using a next-generation sequencing platform. They found 6.2 genomic alterations per tumor, with alterations in TP53, IDH1, and MYC being more frequent and mutations in APC being less frequent than those reported in sporadic CRCs in The Cancer Genome Atlas database. (See Chapter 23 for more details on the molecular basis of IBD neoplasia.)
Although these markers show some promise as adjunctive biomarkers, additional studies must be performed before any of this knowledge can be translated into clinical management of patients with long-standing IBD.
The optimal surveillance strategy for patients with UC remains controversial (see Chapter 2 for more details). Much debate centers on the sensitivity of the detection system, the predictive value of dysplasia for assessing the risk for CRC, and the cost. Data supporting the effectiveness of surveillance in patients with UC are not uniform but do suggest a reduction in mortality from carcinoma in those who are willing to undergo prophylactic colectomy should dysplasia be detected. As a result, the overall balance of evidence supports surveillance for dysplasia in UC.
Widespread agreement on appropriate surveillance strategies in patients with UC has not been established. , The American Gastroenterology Association recommends that surveillance should begin after 8 years of disease in patients with pancolitis and after 15 years in patients with colitis involving the left colon only. Colonoscopy should be repeated every 1 to 2 years. , Four-quadrant biopsy specimens should be obtained from every 10 cm of mucosa between the cecum to the sigmoid colon and from every 5 cm more distally. In addition, a biopsy should be performed on any suspicious lesions or masses. In a study by Rubin and associates, 33 biopsy specimens were needed to detect dysplasia with 90% probability, and at least 64 biopsy specimens were needed to reach a 95% probability of detecting dysplasia. The finding of dysplasia, when confirmed by an expert pathologist, is usually an indication for colectomy. For patients for whom a colectomy is not feasible or is unacceptable, frequent surveillance (e.g., every 3 to 6 months) is considered an acceptable alternative. At present, surveillance is not indicated for patients with ulcerative proctitis.
In 2% to 16% of CD patients, dysplasia/cancer is detected during the course of the illness. Overall, the survival benefit of endoscopic surveillance in CD is controversial, primarily because of the lack of prospective studies. , , However, there is a growing trend toward endoscopic surveillance in patients with long-standing CD. The Crohn’s and Colitis Foundation of America consensus conference recommendations for surveillance in patients with colonic CD includes screening colonoscopy for those who have major colonic disease (involving at least one-third of the colon) with a disease duration of at least 8 to 10 years. , However, surveillance is often difficult to perform because of the presence of strictures that often require the use of a pediatric endoscope. Although most strictures are a manifestation of transmural disease, as many as 12% of CD-related strictures are malignant from the onset.
Acute self-limited colitis (ASLC) is defined as a transient, presumably infectious, acute inflammatory disorder of the colon. Patients typically come to medical attention because of abdominal pain and tenderness, low-grade fever, and in some instances bloody diarrhea as well. , The inflammatory process usually resolves completely within 2 to 4 weeks. A wide variety of pathogens, most commonly bacterial organisms such as Campylobacter jejuni, Salmonella, Shigella species, E. coli, and Yersinia enterocolitica, result in this pattern of colonic injury. In immunocompromised patients and very rarely in immunocompetent individuals, viral infections such as enteric adenoviruses (serotypes 40 and 41) and infections by parasitic organisms like Entamoeba histolytica and Cryptosporidium species may also cause a mild, self-limited form of infectious colitis. A detailed description of infectious disorders of the GI tract is provided in Chapter 4 . Here, we discuss the clinicopathological features of the most common organisms that cause ASLC in the context of diagnostic biopsy pathology of the colon ( Table 17.7 ).
| Organism | Clinical Features | Laboratory Findings | Colonoscopic Findings | Location of Disease | Microscopic Findings | Complications |
|---|---|---|---|---|---|---|
| Campylobacter jejuni | Diarrhea, fever, abdominal pain | Fecal blood and leukocytes, positive stool culture | Erythema, edema, multiple superficial ulcers as large as 1 cm | Any part of the colon may be involved | Neutrophilic cryptitis, histiocyte aggregates | GI hemorrhage, toxic megacolon, pancreatitis, cholecystitis, reactive arthritis, Guillain-Barré syndrome, HUS |
| Salmonella spp. ( S. enteritidis and S. typhimurium ) | Nausea, diarrhea, fever, and bloody diarrhea within 48 hours after exposure | Fecal blood and leukocytes, positive stool culture | Mucosal congestion, granularity, friability, and ulceration | Any part of the colon may be involved | Mucosal edema, congestion, cryptitis, crypt abscesses, thrombi in small venules | Toxic megacolon, bleeding, sepsis |
| Shigella spp. | Abdominal cramping, fever, and diarrhea | Fecal blood and leukocytes, positive stool culture | Small, ragged ulcers with normal intervening mucosa; severe cases with diffuse mucosal congestion and erosions | Any part of the colon may be involved | Aphthous ulcers, cellular lamina propria with neutrophils, cryptitis, and crypt abscesses | Appendicitis, HUS, myocarditis, pneumonitis, reactive arthritis, toxic megacolon |
| Escherichia coli (enteroinvasive E. coli and enterohemorrhagic E. coli [EHEC]) | Watery to bloody mucoid diarrhea, tenesmus, fever, abdominal cramps | Fecal blood and leukocytes, positive stool culture (for EHEC) | Segmental involvement with marked mucosal congestion, friability, edema, and diffuse ulcers, rarely pseudomembranes | Right colon is most commonly involved | Neutrophilic cryptitis, crypt abscesses with erosions, ischemic pattern of injury | HUS |
| Yersinia enterocolitica | Fever, abdominal cramps, diarrhea within 1 to 3 weeks after infection | Fecal blood and leukocytes, stool culture, serology | Focal or diffuse mucosal edema, erythema, and ulcers | Proximal colon more commonly involved than rectosigmoid colon | Mucosal edema, cryptitis, and crypt abscesses (necrotizing granulomas are found only in patients with prolonged disease course) | Septicemia, chronic relapsing fever, appendicitis |
The clinical presentation of patients with ASLC includes rapid-onset abdominal pain, diarrhea, and fever. Typical laboratory findings include peripheral blood and fecal leukocytosis. In ASLC related to bacterial infection, stool cultures are positive in only 40% to 60% of cases. Therefore a negative result does not entirely exclude an infectious etiology. , In fact, in most instances, the exact cause of the patient’s colitis is never actually determined. Imaging studies may reveal nonspecific thickening of the bowel wall. However, because of the acute onset, they are seldom performed.
C. jejuni is one of the most frequently isolated stool pathogens in patients with ASLC. In immunocompromised patients, Campylobacter fetus is more frequently isolated. The incidence of C. jejuni –related diarrhea is 4% to 11% in the United States. Bacterial transmission usually occurs via contaminated food. In developing countries, the infection is hyperendemic in children younger than 2 years of age. The most common presenting symptoms are diarrhea (90%), fever (90%), and abdominal pain (70%). These may be accompanied by headache, myalgia, and vomiting. Stool examination typically reveals red blood cells and leukocytes. Campylobacter DNA may be isolated in as many as 19% of affected patients with acute-onset diarrhea.
At least 2000 serotypes of nontyphoid Salmonella species have been identified as potential causative agents of gastroenteritis (nontyphoid salmonellosis). Almost 45,000 cases of Salmonella gastroenteritis are reported annually. In the United States, Salmonella enteritidis and Salmonella typhimurium are the most commonly isolated species. Infection is most likely to develop in children younger than 1 year of age. In addition, patients with achlorhydria, hemolytic anemia, immunosuppression, or malignancy (e.g., disseminated carcinoma, hematolymphoid malignancies) are predisposed to Salmonella infection. Gastroenteritis develops in 75% of patients infected with this organism. Patients usually present with nausea, vomiting, fever, and bloody diarrhea.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here