Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
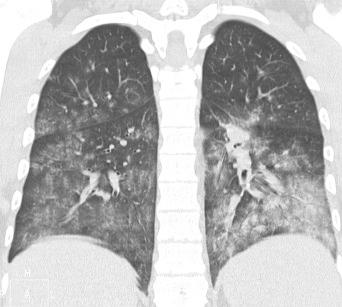
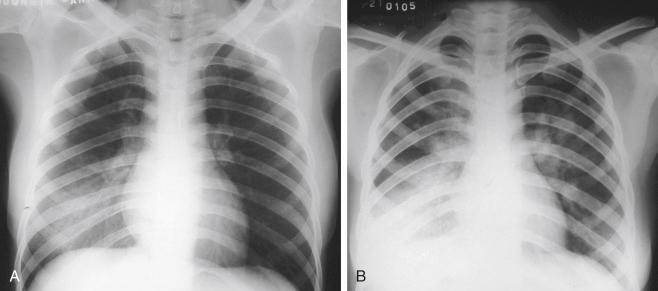
Pneumonia continues to exert a terrible toll on health, especially in childhood, where it is the leading infectious cause of death in under-fives. Airborne infections, whether viral, bacterial or fungal, are principally acquired by inhalation of pathogenic organisms, either directly from the environment as spores or attached to dust particles, or within aerosolized droplets and droplet nuclei generated by the respiratory tract mucosa. Some respiratory tract viral infections may also be transmitted by contaminated hands and fomites.
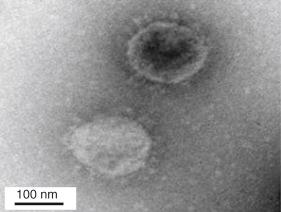
For most of the respiratory viruses the reservoir is human, and there is little demonstration of a capacity to cross species barriers effectively. However, the past 2 decades have witnessed several examples of the potentially devastating effects of cross-species viral transmission: in late 2002 a newly identified animal coronavirus causing a severe acute respiratory syndrome (SARS) gave rise to a major epidemic that disseminated globally through international air travel with alarming rapidity. A decade later another animal virus causing severe respiratory illness was identified in Saudi Arabia—Middle East Respiratory Syndrome coronavirus (MERS-CoV).
These two infections dramatically illustrate the unpredictable epidemic potential of zoonotic viruses that are able to transmit across species barriers and cause human respiratory infection. Indeed, perhaps the most alarming global health threat remains that of a novel transmissible and pathogenic avian influenza strain—for which there are a number of historical pandemic precedents. An array of measures have been developed to limit the pandemic potential of highly pathogenic avian influenza. These include continual sentinel site surveillance, prompt quarantine, flock vaccination and mass culling of domestic poultry when necessary. However, it is clear that humans will continue to be exposed to both old and new zoonotic viral infections for as long as we maintain close exposure to animals.
By contrast, few bacterial pneumonias are zoonotic in origin, the usual reservoir being the respiratory tract of human carriers. Thus, mass population gatherings, whether through displacement caused by strife and famine or voluntary pilgramages such as the Hajj, provide opportunities for transmission and may even play a role in promoting pathogen evolution. Vaccines directed against the virulent bacterial serotypes of Streptococcus pneumoniae have been introduced into mass childhood immunization campaigns, along with vaccines against Haemophilus influenzae serotype B, diphtheria toxin and whooping cough ( Bordetella pertussis ), and these have impacted significantly on the incidence of severe childhood pneumonia. Understandably, much attention is now focused on the evolution of serotype replacement and switching.
Tuberculosis (TB) is currently the leading infectious cause of death worldwide, with over 10 million estimated new cases in 2016, accounting for 1.4 million deaths. Primarily a disease of lower-income populations, its epidemiological map overlaps with that of HIV infection, particularly in sub-Saharan Africa, and TB causes one-third of HIV deaths. Concerted public health drives have reduced both TB incidence and mortality over the past 2 decades, but enormous challenges remain. Affordable diagnostics are insensitive, treatment courses prolonged, and one-quarter of the world’s population is latently infected and remains at persistent risk of reactivation; most worryingly, the prevalence of multi-drug resistance, with its associated difficulties and costs of treatment, is increasing.
The universal distribution of environmental fungi means that inhalation of spores is an unavoidable occurrence. Subsequent development of systemic and potentially life-threatening disease, however, is dependent upon the burden of exposure, the integrity of host immunity and the pathogenic potential of the particular fungal species inhaled—the epidemiology of which will reflect local climatic and geographical conditions.





Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here