Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The kidneys are among the organs most commonly affected by systemic vasculitis. This chapter reviews the etiology and pathogenesis of forms of vasculitis that most often affect the kidneys. The emphasis is on vasculitis that targets predominantly small vessels because this category of vasculitis most often involves the kidneys. A broad spectrum of cellular and humoral inflammatory mediator systems are involved in the pathophysiology of vasculitis. The two most common pathways to renal vasculitis are immunoglobulin-rich immune complex–mediated vasculitis and immunoglobulin-poor pauci-immune antineutrophil cytoplasmic autoantibody (ANCA)–mediated vasculitis. Immune complexes in vessel walls cause inflammation by activating humoral and cellular inflammatory mediator systems in the blood, including the complement, kinin, and coagulation systems. ANCA cause vasculitis by activating cytokine-primed neutrophils by both Fc and Fab’2 binding, resulting in the release of destructive oxygen radicals and lytic enzymes that injure tissue. This inflammation is amplified by activation of the alternative complement system with generation of C5a, which is a pivotal factor in mediating intense necrotizing inflammation.
Key Words
vasculitis, immune complexes, anti-neutrophil cytoplasmic autoantibodies, inflammation, leukocytes, autoantibodies, autoimmunity, arteritis
The kidneys are among the most vascularized tissues of the body. Thus, it is not surprising that they are among the organs most commonly affected by many forms of systemic vasculitis. Vasculitis is inflammation of vessel walls that shares many of the pathophysiologic characteristics of inflammation in any tissue. The seminal pathogenic event is the recruitment and activation of leukocytes in vessel walls with resultant injury. This always involves elements of the innate inflammatory response. In some, if not most, forms of vasculitis, elements of the adaptive immune response also are involved in the induction of the inflammation. This may be an appropriate adaptive immune response (e.g., against rickettsial organisms in the vasculitis of Rocky Mountain spotted fever), an idiosyncratic allergic hypersensitivity response (e.g., in some drug-induced vasculitides), or a maladaptive autoimmune response (e.g., vasculitis caused by antineutrophil cytoplasmic autoantibodies or autoantibodies to capillary basement membrane collagen).
This chapter will review the etiology and pathogenesis of several of the variants of vasculitis that most often affect the kidneys. The focus will be on vasculitides that are not known to be caused by direct invasion of vessel walls by infectious pathogens (i.e., the noninfectious vasculitides). This does not mean that an infection is not involved in the pathophysiology of the vasculitis, only that there is no evidence that the inflammation is directly targeted against intact microbes in the vessel wall. Infection still could be involved indirectly, for example by providing antigens that form pathogenic immune complexes with antibodies or by stimulating a pathogenic autoimmune response. The greatest emphasis will be on vasculitis that predominantly targets small vessels, including glomerular capillaries, because of the frequency and clinical importance of renal involvement in this category of vasculitis.
Ideally, a classification system for vasculitis would be based on specific etiologies or distinct pathogenic mechanisms especially in a discussion of the pathophysiology of vasculitis. Table 83.1 lists some of the putative etiologies and pathogenic mechanisms for vasculitides that may involve the kidneys. Unfortunately, the etiology and pathogenesis of many forms of vasculitis remain poorly understood and controversial, and thus cannot be used as the sole basis for classification systems that are used for the diagnosis of patients. Instead, categorization of vasculitides is based on a combination of features that includes the types of vessels involved, the pattern of inflammation, the evidence for various immunopathologic processes, and the clinical expression of disease ( Table 83.2 , Figure 83.1 ). The major general subdivisions of vasculitis are large-vessel vasculitis, medium-sized vessel vasculitis, and small-vessel vasculitis. Each subdivision is characterized by a pathologic pattern of inflammatory injury that can be caused by more than one clinicopathologic form of vasculitis and that is a final common pathophysiologic pathway of injury that can result from more than one etiology.
| Mediated by Immune Complex Deposition or in Situ Formation |
|---|
| Henoch-Schönlein purpura (IgA vasculitis) |
| Cryoglobulinemic vasculitis |
| Lupus vasculitis |
| Rheumatoid vasculitis |
| Serum sickness vasculitis (Serum-sickness-associated immune complex vasculitis) |
| Infection-induced immune complex vasculitis (e.g., hepatitis B and C) |
| Anti-glomerular basement membrane (anti-GBM) disease |
| Kawasaki disease (possibly via anti-endothelial antibodies) |
| Hypocomplementemic urticarial vasculitis (anti-C1q vasculitis) |
| Mediated by antineutrophil cytoplasmic autoantibodies (ANCAs) |
|---|
| Microscopic polyangiitis |
| Wegener granulomatosis (Granulomatosis with polyangiitis) |
| Churg-Strauss syndrome (Eosinophilic granulomatosis with polyangiitis) |
| Renal-limited vasculitis (pauci-immune crescentic glomerulonephritis) |
| Cell mediated |
|---|
| Giant cell arteritis |
| Takayasu arteritis |
| Large-Vessel Vasculitisa | |
|---|---|
| Giant cell arteritis | Granulomatous arteritis of the aorta and its major branches, with a predilection for the extracranial branches of the carotid artery. Often involves the temporal artery. Usually occurs in patients older than 50 and often is associated with polymyalgia rheumatica. |
| Takayasu arteritis | Granulomatous inflammation of the aorta and its major branches. Usually occurs in patients younger than 50. |
| Medium-Sized Vessel Vasculitisa | |
|---|---|
| Polyarteritis nodosa | Necrotizing inflammation of medium-sized or small arteries without glomerulonephritis or vasculitis in arterioles, capillaries, or venules. |
| Kawasaki disease | Arteritis involving large, medium, and small arteries and associated with mucocutaneous lymph node syndrome. Coronary arteries are often involved. Aorta and veins may be involved. Usually occurs in children. |
| Small-Vessel Vasculitisa | |
|---|---|
| Wegener's granulomatosis (Granulomatosis with polyangiitis) | Granulomatous inflammation involving the respiratory tract, and necrotizing vasculitis affecting small to medium vessels (e.g., capillaries, venules, arterioles, and arteries). Necrotizing glomerulonephritis is common. |
| Churg-Strauss syndrome (Eosinophilic granulomatosis with polyangiitis) | Eosinophil-rich and granulomatous inflammation involving the respiratory tract and necrotizing vasculitis affecting small to medium-sized vessels, and associated with asthma and blood eosinophilia |
| Microscopic polyangiitis | Necrotizing vasculitis with few or no immune deposits affecting small vessels, i.e., capillaries, venules, or arterioles. Necrotizing arteritis involving small and medium-sized arteries may be present. Necrotizing glomerulonephritis is very common. Pulmonary capillaritis often occurs. |
| Henoch-Schönlein purpura (IgA vasculitis) | Vasculitis with IgA-dominant immune deposits affecting small vessels (i.e., capillaries, venules, or arterioles). Typically involves skin, gut, and glomeruli and is associated with arthralgias or arthritis. |
| Cryoglobulinemic vasculitis | Vasculitis with cryoglobulin immune deposits affecting small vessels (i.e., capillaries, venules, or arterioles) and associated with cryoglobulins in serum. Skin and glomeruli are often involved. |

Large-vessel vasculitis affects the aorta or its major arterial branches or both, and is characterized pathologically by chronic inflammation that often includes multinucleated giant cells. Giant cell arteritis and Takayasu arteritis are the two primary categories. When the kidneys are affected by large-vessel vasculitis, the most commonly involved vessels are the main renal artery and interlobar arteries, and the most common clinical manifestation is renal artery stenosis with renovascular hypertension. The pattern of inflammation in large-vessel vasculitis with a predominance of T lymphocytes, monocytes, and macrophages (including multinucleated giant cells) suggests a type IV cell–mediated immune pathogenesis, however, no exogenous or endogenous (auto) antigen target for a pathogenic T-lymphocyte response has been identified.
Medium-sized vessel vasculitis is necrotizing inflammation of arteries in the absence of involvement of capillaries or venules. The primary clinicopathologic categories are polyarteritis nodosa and Kawasaki disease. Although the pathophysiology of these two diseases appears to be quite distinct, both are characterized in the acute phase by arteritis with localized transmural necrosis and infiltration by activated neutrophils and monocytes that cause lytic destruction of the vessel wall. Within several days, the acute inflammation and necrosis are replaced predominantly by lymphocytes and macrophages that orchestrate the progressive replacement of the necrosis with fibrosis. In the kidneys, medium-sized vessel vasculitis has a predilection for the lobar and arcuate arteries, but any artery, including the main renal artery, may be involved. Common intrarenal manifestations include pseudoaneurysm formation, thrombosis, infarction, and hemorrhage.
Small-vessel vasculitis is characterized by acute, often necrotizing, inflammation of many small vessels in multiple organs. Involvement of glomerular capillaries causing glomerulonephritis is a frequent component of most forms of small-vessel vasculitis, and may be the dominant feature. Involvement of dermal venules causing leukocytoclastic angiitis with palpable purpura also is a frequent feature. Small-vessel vasculitis caused by antiglomerular basement membrane antibodies (anti-GBMs) and antineutrophil cytoplasmic autoantibodies (ANCAs) represent life-threatening forms of small-vessel vasculitis that have a predilection for attacking glomerular and pulmonary capillaries. Although the defining feature of small-vessel vasculitis is the involvement of capillaries and venules, small-vessel vasculitis also may affect small and even medium-sized arteries resulting in a necrotizing arteritis that is histologically indistinguishable from a medium-sized vessel vasculitis such as polyarteritis nodosa.
The kidneys are affected much more often by small-vessel vasculitis than by medium-sized vessel vasculitis or large-vessel vasculitis. Thus, the focus of this chapter will be on the pathophysiology and immunopathology of small-vessel vasculitis. The two major immunopathogenic categories of small-vessel vasculitis are immune complex–mediated vasculitis and antineutrophil cytoplasmic autoantibody (ANCA)–mediated vasculitis. Immune complex vasculitis is characterized immunohistologically by conspicuous localization of immunoglobulin in vessel walls, whereas ANCA vasculitis is characterized by the absence or paucity of immunoglobulin in vessel walls. Although these two categories of vasculitis have different etiologies, they share many common histologic features and use common inflammatory mediators and pathways. The next section of this chapter will review some of these common inflammatory pathways. The subsequent two sections will review the pathogenic features of immune complex vasculitis and ANCA vasculitis.
The seminal pathophysiologic event in small-vessel vasculitis is segmental acute inflammation of small vessels, often accompanied by necrosis ( Figure 83.2 ). Many of the basic inflammatory processes that occur in small-vessel vasculitis also occur in the necrotizing arteritis of medium-sized vessel vasculitis ( Figure 83.3 ). The primary initial effector cells in small-vessel vasculitis are neutrophils and monocytes, although macrophages and T lymphocytes are rapidly recruited in response to the injury and are active participants in the subacute and chronic progression of injury. Targeting of leukocytes to specific vascular segments involves multiple cell adhesive and cell signaling events in both the leukocytes and the vessel walls, particularly the endothelium. Collectively these events (discussed below) comprise a leukocyte adhesion cascade. They have been characterized most thoroughly at sites of physiologic diapedesis of neutrophils and monocytes through small vessels, especially postcapillary venules, as they traffic toward tissue sites of innate and adaptive immune surveillance or sites of immune response to microbial invasion or other pathogenic stimuli. The events of the leukocyte adhesion cascade are generally accepted to occur in most tissues. However, recent work has demonstrated that some tissue- and organ-specific variation does occur, although there is no current evidence to suggest that such variation accounts for the vascular and organ predilections of certain of the vasculitides.
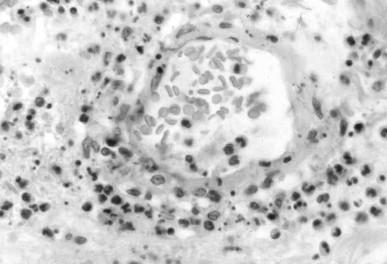
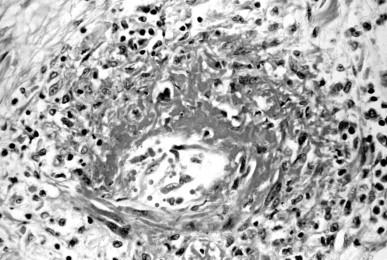
In physiologic diapedesis, leukocyte transmigration through vessel walls causes only minor and transient disruption of the vessel wall structure. In vasculitis, the leukocytes undergo full activation as they attach to and enter the vessel wall rather than waiting until after they have transmigrated into the tissue. Neutrophils survive for only a few minutes, or at most a few hours, at sites of acute inflammation. Monocytes transform into activated macrophages after entry into sites of acute inflammation. Activated macrophages release cytokines that modulate the inflammation. For example, macrophages release factors that enhance T-lymphocyte adhesion to nearby venular endothelial cells and induce targeted chemotaxis of T lymphocytes toward the site of the macrophage activation. If the stimulus for acute inflammation does not persist, neutrophils and monocytes are quickly replaced by macrophages and T lymphocytes at the site of small-vessel vasculitis. Thus, sites of small-vessel vasculitis in biopsy specimens, including renal biopsy specimens, often have predominantly macrophages and T lymphocytes in the injured vessels although the initial injury was induced earlier by neutrophils and acutely activated monocytes.
Chemoattractants for neutrophils and monocytes play a central role in mediating vasculitis not only by attracting leukocytes but also by activating leukocytes and endothelial cells to participate in inflammatory events. Modulation of the expression of adhesion molecules and cytokine/chemokine molecules by endothelial cells, leukocytes in the circulation, and leukocytes in the tissue orchestrates the inflammation at sites of vasculitis. In small-vessel vasculitis, the balance is tipped toward neutrophil recruitment and activation in the acute phase, but within only one or two days the balance shifts toward recruitment of predominantly monocytes and T lymphocytes. However, if the pathogenic process that initiated the initial injury persists, as often is the case, lesions will include a mixture of acute and chronic inflammatory cells for extended periods of time.
Important chemoattractants for neutrophils and monocytes include C5a (which can be generated by activation of either the classical, lectin or alternative complement pathway), leukotriene B4, platelet-activating factor (PAF), and CXC chemokines such as interlukin-8 (IL-8, CXCL8), platelet factor 4 (PF4, CXCL4), GRO1 oncogene (GROα, CXCL1), and stromal cell-derived factor-1 (SDF-1, CXCL12). Progression of vasculitic lesions from an acute to a subacute or chronic phase is mediated primarily by a different set of chemoattractants that preferentially recruit monocytes and T lymphocytes, such as the CC chemokines macrophage inflammatory protein-1α (MIP-1α, CCL3), monocyte chemotactic protein-1 (MCP-1, CCL2), and regulated upon acitivation, normal T-cell expressed, and secreted (RANTES, CCL5). Chemoattractants not only attract leukocytes through direct engagement of ligands on the leukocytes, but also by enhancing the expression and binding of leukocyte adhesion molecules (e.g., β2 and β1 integrins) to their ligands on endothelial cells and elsewhere in the tissue.
Chemokines on the surface of endothelial cells attract leukocytes to the cells in concert with the adhesion molecules reviewed below, and provide a gradient for directional migration toward the intercellular junction (or transcellularly through the endothelial cytoplasm). Once through the endothelium, gradients of bound and free chemokines in the extracellular matrix and interstitial fluid continue to provide directional signals until the leukocyte encounters enough stimulation to undergo full activation. In physiologic diapedesis of leukocytes across postcapillary venules, full activation is not attained until after the leukocyte has migrated away from the vessel wall. In the absence of an adequate pathogenic stimulus in the extravascular compartment, a leukocyte eventually will undergo senescence and apoptosis without becoming activated. In vasculitis, leukocytes are activated as they attach to or enter the vessel wall resulting in inflammatory vascular damage. There are many etiologies for this mural activation, including the presence of infectious organisms in the vessel wall (e.g., Rickettsia rickettsii in Rocky Mountain spotted fever), immune complexes in the vessel wall (e.g., cryoglobulins in cryoglobulinemic vasculitis), or activation by antineutrophil cytoplasmic autoantibodies (e.g., in microscopic polyangiitis).
Leukocyte recruitment into the vessel wall is accomplished by the events of the leukocyte adhesion cascade. This is a multistep process that sequentially involves leukocyte tethering to endothelium followed by leukocyte rolling, activation, arrest, spreading and crawling on the endothelium, and finally paracellular or transcellular migration (diapedesis) across the endothelium ( Figure 83.4 ). Important initial events are the tethering and rolling of leukocytes on the surface of endothelial cells. This allows the leukocytes to sample the endothelial surface for chemokine signals derived from the underlying tissue and for upregulated endothelial adhesion molecules that can modulate rolling and mediate firm adherence. Engagement of these molecules allows static adhesion to the endothelium followed by crawling toward endothelial cell junctions where transmigration between the cells can be accomplished by further engagement of adhesion molecules in the junction. Transmigration through endothelial cells may also occur. Once through the vessel wall, movement into the perivascular interstitial tissue involves chemoattractant gradients and further engagement between integrins and ligands on extracellular matrix molecules.
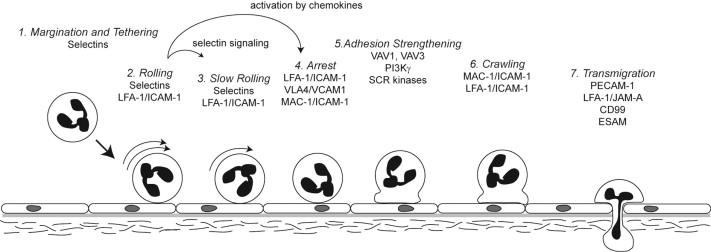
The first step in the exit of leukocytes from flowing blood within the vessel lumen is margination toward the endothelial surface. The resulting juxtaposition of these cells facilitates capture or tethering of the leukocyte to the endothelial surface. Once tethered, shear stress pushing on the leukocyte, combined with the formation of new adhesive bonds at the downstream leukocyte edge and disruption of adhesive bonds at the upstream leukocyte edge, allows for rolling of the leukocyte along the endothelial surface. The adhesive interactions necessary for leukocyte tethering and rolling are mediated primarily by the binding of selectin adhesion molecules to their ligands. E-selectin and P-selectin are expressed on activated endothelium, and P-selectin is also expressed on activated platelets. L-selectin is constitutively expressed on most leukocytes. P-selectin is pre-formed and stored in Weibel-Palade bodies or alpha granules, respectively, and therefore is rapidly translocated to the cell surface quickly in response to mediators released at sites of acute inflammation, such as histamine and thrombin. In contrast, cell surface expression of E-selectin is dependent on RNA and protein synthesis stimulated by inflammatory cytokines such as tumor necrosis factor (TNF) and interleukin-1 (IL-1), and is therefore delayed for several hours after an inflammatory stimulus is received.
A number of selectin ligands (or counter receptors) have been described. The best characterized is P- selectin glycoprotein ligand-1 (PSGL-1). PSGL-1 was originally described as a P-selectin ligand but is now known to function as a ligand for all three selectins. It is expressed on most leukocytes, endothelial cells, and platelets. Ligands for E-selectin also include E-selectin ligand -1 (ESL-1) and correctly glycosylated forms of CD44, both of which are expressed on leukocytes. L-selectin ligands include PSGL-1 on other leukocytes, and peripheral node addressins including glycosylation-dependent cell adhesion molecule-1 (GLYCAM-1), podocalyxin, and CD34, which are found on peripheral lymphoid tissue high endothelial venules (HEV).
Binding of the selectins to their ligands is modulated by several molecular features. High affinity selectin binding is dependent on ligand glycosylation, in part requiring the sialyl Lewis X tetrasaccharide as a recognition epitope. The final step in the synthesis of this critical binding epitope is the addition of an α(1,3)-linked fucose, mediated by α(1,3)-fucosyltransferases type VII and IV. L-selectin binding to ligands on peripheral lymph node HEV, or at sites of chronic inflammation, utilize a sulphated version of this epitope, 6-sulph-sialyl Lewis X. Lastly, high affinity binding of P-selectin or L-selectin to PSGL-1 requires sulphation of certain tyrosine residues near the N-terminus of the molecule. The molecular features of the selectins and their ligands result in binding interactions with very high on- and off-rates, allowing bonds to form and break quickly. In addition, shear stress is required and also enhances L-selectin and P-selectin binding. Combined, these binding features result in interactions called catch bonds that are particularly suited to mediating the tethering and rolling of leukocytes on the endothelium.
At sites of small-vessel vasculitis, increased expression of P-selectin and E-selectin on endothelial cells is induced by inflammatory chemokines. Subsequent binding to their ligands on leukocytes allows for and modulates primary leukocyte tethering and rolling. Rolling may also be triggered by secondary tethering. In this process, leukocytes previously captured onto the endothelium bind, via their PSGL-1, to L-selectin on other marginated leukocytes and mediate their capture and tethering to the endothelium. The physiologic significance of secondary tethering remains unclear, although it can be observed in vivo.
In addition to selectin-mediated rolling, it is known that some integrin adhesion molecules can mediate and modulate this process. For example, the β 1 integrin very late antigen 4 (VLA4) has been shown to mediate rolling of monocytes and T cells. Similarly, the β2 integrin lymphocyte function-associated antigen 1 (LFA-1) can interact with endothelial intercellular adhesion molecule 1 (ICAM-1) to modulate slow rolling. In this process, neutrophil signaling events triggered by binding to E-selectin result in expression of an intermediate-affinity form of LFA-1 that transiently interacts with endothelial ICAM-1 to decrease rolling velocity. A second β2 integrin, macrophage-1 antigen (MAC-1) can also mediate slow rolling. Thus, the additive ligation of more than one class of adhesion molecule may be required for efficient transition from fast rolling to a state of arrest.
Leukocyte transition from rolling to arrest is the next step in leukocyte transmigration. Leukocyte activation is rapidly triggered by chemokines and other cell activators, and leukocyte arrest is mediated primarily by binding of leukocyte integrin-type adhesion molecules to members of the immunoglobulin superfamily expressed on the endothelium. Leukocyte rolling facilitates activation by slowing transit time which allows for increased exposure to soluble activating factors and also exposure to endothelial-bound activating factors. Evidence suggests that chemokines that facilitate engagement of integrins and direct endothelial transmigration are bound to glycosaminoglycans (GAGs) or the Duffy antigen/receptor for chemokines (DARC) on the surface of endothelial cells. The type of chemokine that occupies these binding sites depends on the relative availability and concentration of the chemokines. These sites can engage different cytokines depending on the type of leukocyte that is being recruited for a particular physiologic or pathophysiologic process. Thus, if high levels of CXC chemokines are present in the vessel wall, CXC chemokines will occupy the endothelial binding sites and will attract predominantly neutrophils. In contrast, if high levels of CC chemokines are present in the vessel wall, CC chemokine will occupy the endothelial binding sites and will attract predominantly T lymphocytes. Chemokines can reach the luminal surface of endothelial cells by vesicular abluminal-to-luminal transcytosis, or be deposited there by circulating platelets or microparticles, or the exocytosis of intracellular granules derived from other cells.
Integrins with the β 1 or β2 subunit are particularly important in the arrest of neutrophils and monocytes and their recruitment to sites of acute vascular inflammation. These include CD49d/CD29 (VLA-4), CD11a/CD18 (LFA-1), CD11b/CD18 (Mac-1), and CD11c/CD18. The avidity of certain integrins on leukocytes can be rapidly upregulated by chemokines at sites of inflammation and enhance arrest and accumulation of inflammatory cells. Ligation of specific heterotrimeric G-protein coupled receptors (GPCRs) by chemokines triggers a complex intracellular signaling cascade that results in integrin transition to intermediate and high affinity forms to promote arrest. This process, termed inside-out signaling, is incompletely understood but is thought to sequentially involve phospholipase C signaling, small GTPase activation, and induction of integrin conformational changes.
Several members of the immunoglobulin superfamily are the ligands on the surface of endothelial cells for the leukocyte integrins. Important examples are the intracellular adhesion molecules (ICAMs) that include ICAM-1, ICAM-2, and ICAM-3. Up-regulation of these ligands, especially ICAM-1, on endothelial cells at sites of vascular inflammation works in concert with the up regulation of integrins on circulating leukocytes to induce leukocyte infiltration at sites of vasculitis. Ligation of CD11a/CD18 (LFA-1) on neutrophils to ICAM-1 on endothelial cells causes adhesion and ligation of CD11b/CD18 (MAC-1) to ICAM-1, promoting arrest and diapedesis. Ligation of β2 integrins to ICAMs is particularly important for neutrophil influx at sites of acute vascular inflammation, whereas lymphocyte recruitment to sites of inflammation depends more on the binding of the β4 integrin VLA-4 (CD49d/CD29) to a different immunoglobulin, vascular cell adhesion molecule-1 (VCAM-1 [CD106]).
Many of the leukocyte adhesion cascade processes and events described thus far can contribute to the specificity of leukocyte recruitment and arrest. Specificity can be accounted for by the combinatorial effects of differential expression of integrins and their receptors, differential expression of the chemokines and their receptors, and varied intracellular signaling to regulate integrin function.
Recent work has begun to delineate events following integrin engagement that are thought to strengthen binding to the endothelium and thereby promote leukocyte recruitment into the inflamed vascular wall. These events are triggered by integrin-mediated outside-in signaling, characterized by ligand-induced integrin conformational changes, and cell surface integrin clustering that result in the recruitment of protein tyrosine kinases (PTKs) and initiation of PTK-dependent signaling pathways. Also, the signaling adaptor molecule paxillin has been shown to bind to the α 4 integrin chain of VLA-4 only in its high-affinity conformation, suggesting that ligand binding initiates outside-in signaling through this adapter protein. In addition, studies have shown that neutrophils lacking phosphatidylinositol 3-kinaseγ (PI3Kγ) or the guanine nucleotide exchange factors VAV1 and VAV3 exhibit accelerated detachment. Although these signaling pathways are not fully elucidated, these studies provide evidence that integrin binding events that mediate leukocyte arrest also trigger intracellular signaling processes that stabilize post-arrest leukocyte adhesion.
Static leukocyte adhesion to the endothelium is followed by transmigration of the cell across the endothelial barrier. Adherent leukocytes crawl over the endothelial surface (termed lateral migration) to locate a preferred site at which to cross the endothelium. Neutrophil crawling on venular endothelium is dependent MAC-1 and its ligand ICAM-1, and disruption of these binding events results in a delay of transmigration. Crawling is likely to also occur as an integral part of the prominent leukocyte recruitment that occurs on the arterial side of the circulation in the vasculitides.
Once at an endothelial cell junction, neutrophils and monocytes are able to crawl through the junctions between endothelial cells (paracellular transmigration) by progressive engagement and disengagement of a series of adhesion molecules on the surface of the leukocytes with ligands on the junctional surfaces of the endothelial cells. Platelet/endothelial cell adhesion molecule-1 (PECAM-1) (CD31), an immunoglobulin superfamily member, is expressed on leukocytes and on endothelial cell surfaces and intercellular junctions. Homophilic PECAM-1 adhesion is particularly important for transmigration of leukocytes through intercellular junctions between endothelial cells. Transmigration of neutrophils and monocytes also involves the interactions of LFA-1, VLA4, or MAC1 on the leukocytes with immunoglobulin superfamily adhesion molecules in the endothelial intercellular junction, JAM-A (junctional adhesion molecule-A), JAM-B, or JAM-C, respectively. Distal to the PECAM-1-mediated and JAM-mediated events, homophilic binding between CD99 on leukocytes and CD99 in the intercellular junction between endothelial cells takes over to facilitate completion of diapedesis. Recent work also shows that endothelial cell-selective adhesion molecule (ESAM) at the endothelial junction facilitates transmigration of neutrophils but not lymphocytes, possibly by activation of the Rho GTPase and destabilization of the tight junction. Leukocytes may also cross the endothelial barrier by a transcellular route through the body of the endothelial cell (transcellular migration). The significance of this route in vivo , and the underlying mechanisms involved in this process are currently poorly understood.
During the process of diapedesis, there are endothelial cytoskeletal and junctional events that result in localized retraction of endothelial cytoplasm and opening of the intercellular junction. These events result from signals transduced from the ligation of endothelial adhesion molecules (e.g., ICAM-1 and PECAM-1). Endothelial stimulation by inflammatory cytokines and adhesion molecule ligation, activation of membrane bound and interstitial matrix metalloproteinases (MMPs), and release of serine proteinases at the surface of migrating leukocytes cause altered distributions and integrity of junctional adhesion molecules (JAMs), vascular endothelial cadherins, and other junctional complex components so that leukocytes can crawl between endothelial cells into the intima, muscularis, or perivascular interstitium. Once past the endothelial barrier, leukocyte migration through the underlying lamina and matrix involves progressive engagement of matrix molecules by leukocyte adhesion molecules, especially PECAM-1, β1 integrins (e.g., VLA-4, VLA-5) and carcinoembryonic antigen–related cell adhesion molecules (CEACAMs).
At sites of vasculitis, the endothelium may be absent because of inflammatory damage. Denudation of endothelium will expose surfaces that contain ligands for neutrophils, monocytes and other inflammatory cells ( Table 83.3 ). At sites of acute necrotizing vasculitis, the lumen often is immediately adjacent to zones of fibrinoid necrosis that contain fibrin that has been generated by the activation of the coagulation cascade by tissue factor and other thrombogenic materials at the site of injury. Mac-1 and other neutrophil and monocyte adhesion molecules can adhere to fibrin and begin the process of activation and migration into the inflamed tissue. Matrix molecules exposed by endothelial denudation, such as fibronectin, laminin, entactin, and collagen also present ligands for leukocyte adhesion molecules, especially β1 integrins such as VLA-4 and VLA-5. Serine proteinases (e.g., elastase) released at the surface of migrating leukocytes act synergistically with β1 integrins and upregulated leukocyte PECAM-1 to allow the leukocytes to migrate into the tissue.
| Leukocyte Event | Leukocyte Receptor | Endothelial Ligand |
|---|---|---|
| Rolling | L-selectin (CD62L) | PSGL-1 (CD162; on leukocytes not endothelium) |
| PSGL-1(CD162) | P-selectin (CD62P) | |
| PSGL-1 (CD162), ESL-1, CD44 | E-selectin (CD62E) | |
| LFA-1 (CD11a/CD18) (β2 integrin) | ICAM-1 (CD54) | |
| VLA-4 (CD49d/CD29) (β 1 integrin) | VCAM-1 (CD106) | |
| Arrest | LFA-1 (CD11a/CD18) (β 2 integrin) | ICAM-1 (CD54) |
| VLA-4 (CD49d/CD29) (β 1 integrin) | VCAM-1 (CD106) | |
| Mac-1 (CD11b/CD18) (β 2 integrin) | ICAM-1 (CD54) | |
| Crawling | Mac-1 (CD11b/CD18) (β 2 integrin) | ICAM-1 (CD54) |
| LFA-1 (CD11a/CD18) (β 2 integrin) | ICAM-1 (CD54) | |
| Transmigration | Mac-1 (CD11b/CD18) (ß2 integrin) | ICAM-1 (CD54) |
| LFA-1 (CD11a/CD18) (β 2 integrin) | JAM-A CD321) | |
| VLA-4 (CD49d/CD29) (β 1 integrin) | JAM-B (CD322) | |
| Mac-1 (CD11b/CD18) (β 2 integrin) | JAM-C | |
| PECAM-1 (CD31) | PECAM-1 (CD31) | |
| MIC2 (CD99) | MIC2 (CD99) | |
| TISSUE LIGAND | ||
| Tissue infiltration | Mac-1 (CD11b/CD18) (β 2 integrin) | Fibrin, fibronectin |
| PECAM-1 (CD31) | Basement membrane | |
| α6β1 integrin (β 1 integrin; CD151) | Laminin | |
| VLA-4 (CD49d/CD29) (β 1 integrin) | Fibronectin | |
| VLA-5 (CD49e/CD29) (β 1 integrin) | Fibronectin | |
| CEACAM (CD66) | Fibronectin | |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here