Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Lymphoproliferative disorders (LPDs) associated with iatrogenic immunodeficiency constitute a spectrum of lymphoid or plasmacytic proliferations, including a major subset that occurs after solid organ, stem cell, or bone marrow transplantation (post-transplant lymphoproliferative disorders [PTLDs]). A smaller number of cases occur in other situations, such as in patients with rheumatoid arthritis treated with methotrexate or in young patients with Crohn's disease treated with tumor necrosis factor-α (TNF-α) antagonists along with antimetabolites (other iatrogenic immunodeficiency-associated LPDs). Many but not all LPDs are associated with Epstein-Barr virus (EBV). They require further classification because of the great variation in their cytologic composition, degree of destructiveness, immunophenotype, cytogenetic and molecular findings, clinical behavior, and therapeutic approach. Cases range from hyperplastic-appearing lesions to others that are indistinguishable from non-Hodgkin's or Hodgkin's lymphoma in immunocompetent hosts. Even the latter cases, however, are separately designated because reducing or discontinuing immunosuppression, when possible, or administering therapy that would be considered inadequate in immunocompetent hosts may lead to resolution.
The World Health Organization (WHO) classification recognizes four major categories of PTLD ( Box 55-1 ). Which of these disorders is truly neoplastic is debatable, and such a determination is not necessarily of clinical utility. Biopsies performed when there is a question of PTLD should be handled with a standard “rule out lymphoma” protocol that includes all the necessary ancillary techniques required for a complete diagnosis ( Box 55-2 ). Although cytologic and fine-needle aspiration biopsy specimens can be useful in some circumstances, excisional biopsy is preferred because of the importance of assessing architectural features, the need for sufficient material for ancillary studies, and the intralesional heterogeneity present in a moderate number of PTLDs.
Non-destructive PTLD
Plasmacytic hyperplasia
Infectious mononucleosis
Florid follicular hyperplasia
Polymorphic PTLD
Monomorphic PTLD
(Classify according to the lymphoma they resemble.)
B-cell neoplasms
Diffuse large B-cell lymphoma, not otherwise specified
Burkitt's lymphoma
Plasma cell myeloma
Plasmacytoma
Other *
* Small B-cell lymphomas arising in transplant patients are not considered PTLD, with the exception of EBV + marginal zone lymphomas.
T-cell neoplasms
Peripheral T-cell lymphoma, not otherwise specified
Hepatosplenic T-cell lymphoma
Other
Classical Hodgkin's Lymphoma PTLD
Required for diagnosis and best accomplished with histologic sections
Findings are extremely variable: lymphoplasmacytic proliferations with underlying architectural preservation; destructive polymorphic lymphoplasmacytic proliferations with moderately numerous transformed cells or immunoblasts; and lesions fulfilling the criteria for one of the non–small cell B-cell lymphomas, an EBV + marginal zone lymphoma, a plasma cell neoplasm, one of the T-cell lymphomas, or occasionally classical Hodgkin's lymphoma
Required for diagnosis
Findings are extremely variable, including polyclonal lesions, often with many admixed B and T cells, or lesions with the classic immunophenotypic features of one of the lymphoid or plasmacytic neoplasms
Stains for EBV should be performed, with the majority of (but not all) cases positive using EBER in situ hybridization or, in a somewhat smaller proportion of cases, using an EBV LMP-1 immunohistochemical stain
Variably required
Clonal B cells are demonstrable in most cases, except in plasmacytic hyperplasia, some IM PTLDs, and T-cell PTLDs
Clonal T cells are demonstrable in T-cell monomorphic PTLD but also in some other types of PTLD
Various additional molecular cytogenetic and karyotypic findings are related to the type of PTLD, such as MYC rearrangements and TP53 mutations
These studies are most important in recognizing T-cell PTLD and classifying some of the B-cell monomorphic PTLDs
Assessment required for patient care
PTLDs can present with an IM-type illness, like conventional lymphoid or plasmacytic neoplasms, or sometimes with vague or absent symptoms
PTLDs are sometimes discovered only because of routine EBV monitoring or at autopsy
Variable outcomes, ranging from indolent with reduction in immunosuppression to very aggressive in spite of immunochemotherapy; many different factors affect outcome
EBER, EBV-encoded small RNA; EBV, Epstein-Barr virus; IM, infectious mononucleosis; LMP-1, latent membrane protein 1.
PTLDs develop in approximately 2% of all transplant recipients, but there is a significant variation in incidence based on the type of organ transplanted: kidney, 0.5% to 2.5%; marrow or stem cell, 1% to 2%; liver, 1% to 10%; heart and lung, 2% to 10%; and intestinal and multivisceral, 5% to 20%. Some report that the incidence of PTLD has decreased over time, possibly related to increased experience, improved immunosuppressive regimens, and increased use of molecular EBV monitoring with pre-emptive modulation of immunosuppressive therapy, although others have found an increasing incidence.
Many other factors have an impact on the incidence of PTLD. EBV seronegativity at the time of organ transplantation is an extremely important risk factor and explains in part the much higher incidence of PTLD in children than in adults. Transplanting an organ from an EBV-seropositive donor into an EBV-seronegative recipient (EBV mismatch) increases the incidence of PTLD 10- to 75-fold. Lack of previous exposure to cytomegalovirus (CMV) is also associated with an increased incidence of PTLD if the recipient is CMV − and either the donor is CMV + (CMV mismatch) or the recipient experiences a symptomatic primary CMV infection. The effects of EBV mismatch and CMV mismatch appear to be synergistic. Patients who undergo transplantation for hepatitis C–induced cirrhosis reportedly have an increased incidence of PTLD, suggesting that hepatitis C may potentiate the oncogenicity of EBV. Host factors, such as polymorphisms leading to lower expression of proinflammatory cytokines or greater expression of anti-inflammatory cytokines or HLA and other polymorphisms, may also influence the risk of selected PTLDs, although one recent review noted that “it is currently not possible to predict which transplant patients will eventually develop PTLD.” Even after taking into consideration EBV and CMV serostatus, young age remains associated with an increased risk for development of PTLD, particularly early PTLD. In addition, the incidence increases again after the age of 50 years. Overall, there is a higher incidence in males, especially for late-onset PTLD, but a higher incidence in females after small bowel transplantation.
Another important risk factor for PTLD is the immunosuppressive regimen required to maintain or to prepare for the transplant or to treat graft-versus-host disease. The cumulative intensity of immunosuppressive therapy and the specific agents used are associated with the risk for development of early PTLD, whereas the overall duration of immunosuppression is associated with the risk for development of PTLD later. Anti–T-cell antibody preparations, such as OKT3 and antithymocyte globulin (ATG), have been associated with an increased risk of PTLD, as has the use of these agents to remove T cells from bone marrow or stem cell products before transplantation. A combination of ATG, rituximab, and steroids has been suggested to protect against PTLD after small bowel transplantation. Also, depletion of both T cells and B cells is not associated with as great a risk. Some of the newer immunosuppressive strategies may be associated with a lower risk for PTLD. However, the newer novel immunosuppressive regimen belatacept, which was introduced to avoid the nephrotoxic and other side effects associated with long-term use of calcineurin inhibitors, may be associated with an increased risk of PTLD.
Most PTLDs after solid organ transplantation are derived from recipient lymphoid cells, whereas those occurring after bone marrow transplantation are most often donor derived. PTLDs limited to the allograft after solid organ transplantation are more frequently of donor origin. The majority of PTLDs are caused by EBV-infected lymphoid or plasmacytic cells that are not adequately controlled by the immune system because of immunosuppression or, in the case of stem cell and bone marrow transplants, myeloablative regimens. EBV may be acquired from the donor or other sources as a primary infection, superinfection by a second strain of EBV in a seropositive recipient, or, especially in adults, reactivation of latent recipient EBV. EBV-associated PTLD shows variable latency patterns, and individual cells can express different sets of latency proteins. Many cases have a type III latency pattern, similar to that seen in EBV + lymphoblastoid cell lines; a moderate number have a type II latency pattern, and fewer have type I. Classical Hodgkin's lymphoma PTLD may demonstrate pattern type II. Most PTLDs also have at least some replicative EBV activity. Patients with EBV + PTLD lack an effective cytotoxic T-cell response to EBV infection, with decreased EBV-specific CD8 + T cells and CD4 + T cells. Consistent with this, lower levels of EBV-specific T lymphocytes in the face of EBV are associated with a higher likelihood of PTLD. Conversely, PTLD infiltration by cytotoxic T lymphocytes has been associated with a good prognosis. Humoral responses to EBV are also diminished after transplantation, but whether this plays a role in the development of PTLD is not known. Patients with PTLD reportedly demonstrate a T-helper (Th) type 2 serum cytokine profile (interferon-γ/interleukin-2 negative; interleukin-4/interleukin-10 positive) that promotes EBV-induced B-cell proliferation. In fact, monitoring interleukin-10 levels has been proposed as a way to observe patients at risk for PTLD and as a diagnostic tool. Related to these observations, it is of interest that ATG not only causes an overall decrease in lymphocyte counts (predominantly due to decreased CD4 + cells) but also affects Th1 (but not Th2) CD4 + T-cell responses.
Genetic predisposition may also play a role in the development of PTLD and in patients' response to therapy. A polymorphism in interferon-γ resulting in increased synthesis is associated with early-onset and pediatric PTLD, and a TNF gene polymorphism has been associated with the development of PTLD. Polymorphic variants in the HLA system have also been associated with PTLD development, possibly because of their role in interacting with natural killer (NK) cells and cytotoxic T lymphocytes. Interleukin-1 receptor agonist and interleukin-1β alleles associated with a more pronounced response to EBV infection may protect against EBV viremia. A polymorphism in an NK-cell Fc receptor is associated with an increased affinity for immunoglobulin G, resulting in more efficient antibody-mediated cytotoxicity and an improved outcome for patients with PTLD.
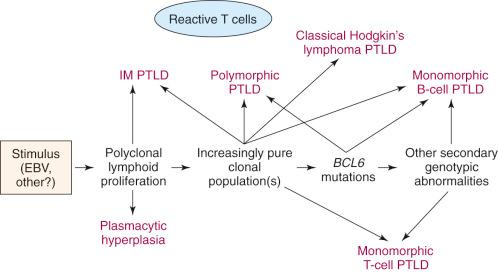
Although very few patients demonstrate a sequential development of the disorder, PTLDs are thought to begin as polyclonal proliferations related to EBV or other stimuli, with the development over time of oligoclonal and then monoclonal B-cell or, much less frequently, T-cell proliferations ( Fig. 55-1 ). Cytogenetic or genotypic abnormalities of the types seen in conventional lymphoid or plasmacytic neoplasms (described later) also occur as the lesions progress, making them less responsive to immune regulation. Antigenic selection may also be important in the development and progression of the B-cell clonal proliferations in PTLDs. Advances in the understanding of the cellular pathways involved in the pathogenesis of PTLD are not just of academic interest as they may suggest new therapeutic strategies, such as JAK/STAT inhibitors.
EBV cannot be demonstrated in approximately 20% to 40% of PTLDs; some report an even higher percentage, with the proportion of EBV − cases greater now than in the past. The cause of EBV − PTLD is uncertain, but at least some cases may represent EBV-related proliferations that have lost the virus after transformation (hit-and-run theory). Others may reflect technical difficulties in the detection of EBV, represent lymphoid proliferations driven by other viral or infectious agents, or be related to chronic antigenic stimulation, possibly by the transplant itself. Rare cases of human herpesvirus 8–positive PTLD have been reported, including polymorphic lesions, a Castleman-like lesion, and primary effusion lymphoma. Other viral associations have been reported rarely and remain to be established. Gene expression profiling studies have suggested a difference in pathobiology between EBV − and EBV + PTLDs, with two studies showing viral-associated changes only in the EBV + cases, suggesting a possible non-viral etiology for the EBV − cases. Some genomic profiling studies have also found that EBV − diffuse large B-cell lymphoma (DLBCL)–type PTLD segregated with DLBCL arising in immunocompetent patients. However, another gene profiling study that included a broad spectrum of PTLDs failed to distinguish EBV + from EBV − PTLDs. Other differences that have been documented between EBV + and EBV − PTLDs include EBV + cases occurring in younger patients, showing a greater proportion of non–germinal center/activated B-cell versus germinal center–type DLBCL PTLD, having downregulation of the BCL2 family proteins Bim and cleaved PARP, and possibly having fewer recurrent lesions by single nucleotide polymorphism analysis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here