Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The human herpesviruses (HHVs) are categorized into three groups: alpha, beta, and gamma herpesvirinae ( Table 80.1 ). Each virus contains a core of linear double-stranded DNA, an icosahedral capsid 100–110 nm in diameter, and an envelope with glycoprotein spikes on its surface. The pathogenesis of herpesvirus infections follows the sequence of primary infection, latency, and reactivation. The eight human herpesviruses are discussed in this chapter and include the following: herpes simplex virus types 1 and 2 (HSV-1, HSV-2), varicella–zoster virus (VZV), Epstein–Barr virus (EBV), cytomegalovirus (CMV), and human herpesvirus types 6–8 (HHV-6–8).
| THE EIGHT HUMAN HERPESVIRUSES, THEIR CLASSIFICATION AND DIFFERENTIAL DIAGNOSES | ||||
|---|---|---|---|---|
| Human herpesvirus | Classification | Predominant cell type infected | Differential diagnosis | |
| Lytic infection | Latent infection | |||
| Herpes simplex virus type 1 (HSV-1) (HHV-1) | Alphaherpesvirinae | Epithelial cells | Neurons | Orolabial herpes: aphthous stomatitis, erythema multiforme major, Stevens–Johnson syndrome, herpangina, pharyngitis (e.g. due to EBV), oral candidiasis, mucositis secondary to chemotherapy |
| Herpes simplex virus type 2 (HSV-2) (HHV-2) | Alphaherpesvirinae | Epithelial cells | Neurons | Genital herpes: trauma, chancre (primary syphilis), aphthae, EBV-related ulcers, CMV-related ulcers (AIDS), chancroid, granuloma inguinale, lymphogranuloma venereum |
| Varicella–zoster virus (VZV) (HHV-3) | Alphaherpesvirinae | Epithelial cells | Neurons | Varicella: vesicular viral exanthem (e.g. coxsackievirus A6), PLEVA, rickettsialpox, disseminated HSV, insect bites, drug eruption, scabies, smallpox |
| Herpes zoster: zosteriform HSV, contact dermatitis, phytophotodermatitis, bullous impetigo, cellulitis | ||||
| Epstein–Barr virus (EBV) (HHV-4) | Gammaherpesvirinae | B cells, epithelial cells | B cells | Group A streptococcal infection, acute viral hepatitis, drug reaction with eosinophilia and systemic symptoms (DRESS), toxoplasmosis, lymphoma; primary CMV, HHV-6 and HIV infections |
| For genital ulcers: see genital herpes | ||||
| Cytomegalovirus (CMV) (HHV-5) | Betaherpesvirinae | Lymphocytes, macrophages, endothelial cells | Lymphocytes, macrophages | EBV-induced infectious mononucleosis, toxoplasmosis, viral hepatitis, lymphoma |
| For genital ulcers: see genital herpes | ||||
| Human herpesvirus 6 (HHV-6) | Betaherpesvirinae | CD4 + T cells | Lymphocytes, monocytes | Viral exanthems (e.g. measles, rubella; enterovirus, adenovirus, EBV, and parvovirus infections), scarlet fever, Rocky Mountain spotted fever, Kawasaki disease |
| Human herpesvirus 7 (HHV-7) | Betaherpesvirinae | T cells | T cells | Similar to HHV-6 |
| Human herpesvirus 8 (HHV-8) | Gammaherpesvirinae | Lymphocytes | Lymphocytes, endothelial cells | Acroangiodermatitis (pseudo-KS), bacillary angiomatosis, ecchymosis, hemangioma, angiosarcoma, pyogenic granuloma, pseudolymphoma/lymphoma |
▪ Herpes – herpes simplex ▪ Herpes labialis – cold sore, fever blister, herpes febrilis ▪ Herpes gladiatorum – scrum pox ▪ Herpetic whitlow ▪ Genital herpes – herpes progenitalis
Herpes simplex viruses produce primary and recurrent vesicular eruptions that favor the orolabial and genital regions
The use of sensitive and specific serologic markers has provided insights into the epidemiology of HSV infections
Antiviral therapy significantly influences the course and spectrum of HSV disease
HSV-1 and HSV-2 are ubiquitous pathogens that produce primarily orolabial and genital infections. Worldwide genital herpes is one of the most common sexually transmitted infections. The use of sensitive and specific serologic markers has enabled the epidemiology of HSV infections to be deciphered, while antiviral therapy has allowed clinicians to control the course and extent of HSV disease. Prevention of HSV infections via molecularly derived vaccines remains an important goal.
Herpetic lesions have been described since antiquity, including by the noted physicians Hippocrates and Galen. The word “herpes” was first defined by Greek scholars as “to creep or crawl” in reference to the spreading nature of the skin lesions. The association between orolabial herpetic lesions and genital infection was first recognized during the late eighteenth century. However, better understanding of the pathogenesis of herpes infections did not occur until the twentieth century. Lipschitz was the first to suggest antigenic differences between the two HSV types, but it was not until 1968 that Nahmias and Dowdle demonstrated that HSV-1 was more frequently associated with orolabial and HSV-2 with genital infections . In the past several decades, there have been critical insights into the pathogenesis of HSV infections as well as advances in their diagnosis, treatment, and prevention.
Herpes simplex viruses have a worldwide distribution. Their interaction with human hosts has the following components: (1) primary infection – initial HSV infection, without pre-existing antibodies to HSV-1 or HSV-2; (2) non-primary initial infection – infection with one HSV type in an individual with pre-existing antibodies to the other HSV type; (3) latency – virus in sensory ganglia; and (4) reactivation – recurrent infection with asymptomatic viral shedding or clinical manifestations (recrudescence). Primary, non-primary initial, and recurrent infections can each be symptomatic or asymptomatic. It is estimated that approximately one-third of the world's population has experienced a symptomatic HSV infection.
In children <10 years of age, ~80–90% of herpetic infections are caused by HSV-1. Analyses on a worldwide basis have demonstrated HSV-1 antibodies in 50–90% of individuals 20–40 years old. Although HSV-2 remains the major cause of recurrent genital herpes infections (70–90% overall) , the proportion due to HSV-1 has been increasing in the US, Canada, and the UK. In these countries, HSV-1 now accounts for the majority of genital herpes infections in young adults, especially college students . Recurrences of genital herpes are less common with HSV-1, which lacks HSV-2's specific tropism for genital epithelium.
In the Centers for Disease Control and Prevention (CDC)'s 2005–2008 National Health and Nutrition Examination Survey (NHANES) , the seroprevalence of HSV-2 among individuals 14–49 years of age in the US was 16% overall, with higher rates in women (21%, versus 11.5% in men) and blacks (39%). Of note, 81% of those with positive serology never had a previous diagnosis of genital HSV infection. Other factors associated with acquisition of genital herpes include an age of 15–30 years (period of greatest sexual activity), an increased number of sexual partners, lower levels of income and education, and HIV-positivity; conversely, genital HSV-2 infection also increases the risk of acquiring and transmitting HIV infection .
Transmission of HSV can occur during asymptomatic periods of viral shedding. HSV-1 is spread primarily through direct contact with contaminated saliva or other infected secretions, while HSV-2 is spread primarily by sexual contact. Virus replicates at the mucocutaneous site of infection and then travels by retrograde axonal flow to the dorsal root ganglia, where it establishes latency until reactivation. Latency enables the virus to exist in a relatively non-infectious state for varying periods of time in its host. HSV-1 can induce intracellular accumulation of CD1d molecules in antigen-presenting cells, providing a possible explanation for how the virus evades detection and establishes latency. Normally, CD1d molecules are transported to the cell surface, where they present lipids and stimulate natural killer T cells, thus promoting an immune response . HSV's mechanisms to evade the immune system also include down-regulation of various immunologic cells and cytokines ( Table 80.2 ).
| INTERACTION OF HERPES SIMPLEX VIRUSES WITH THE IMMUNE SYSTEM: HOST RESPONSES AND VIRAL EVASION MECHANISMS |
| Host immune responses to HSV infection |
| Innate immunity |
|
| Adaptive immunity |
|
| HSV's mechanisms of immune evasion |
|
* Viral envelope fusion with the host cell membrane during entry leaves viral proteins on the cell surface.
** Mutations in the genes encoding TLR3 or proteins required for TLR3 signaling (e.g. UNC-93B, TRAF3) lead to particular susceptibility to HSV encephalitis, and polymorphisms in the gene encoding IFN-λ3 (IL-28b) have been associated with increased risk of recurrent orolabial herpes simplex.
The goals of the early host immune response against HSV are to limit viral replication and recruit other inflammatory cells. Toll-like receptors are critical to the first line of innate immune defense against HSV, and their functions include priming CD8 + T cells. HSV-specific memory CD8 + T cells, which are selectively activated and retained in latently infected sensory ganglia, have important roles in controlling the infection and preventing symptomatic recurrences (see Table 80.2 ).
HSV can be reactivated either spontaneously or by a trigger such as emotional stress, ultraviolet light, fever, menstruation, immunosuppression, surgical or dental procedures, and other local tissue damage. Typically, reactivation produces vesicular lesions localized to the skin. However, viremia followed by widespread internal organ involvement can occur in immunocompromised hosts.
HSV infections have a wide range of clinical presentations, and asymptomatic infection is very common. In primary infections, symptoms typically occur within 3 to 7 days after exposure. A prodrome of tender lymphadenopathy, malaise, anorexia, and fever often occurs before the onset of mucocutaneous lesions, which may be preceded by localized pain, tenderness, burning, and tingling. Painful, grouped vesicles appear on an erythematous base and may become umbilicated, followed by progression to pustules, erosions, and/or ulcerations with a characteristic scalloped border. Crusting of lesions and resolution of symptoms typically occurs within 2 to 6 weeks. A similar prodrome can precede recurrent lesions, but these are often fewer in number, with decreased severity and duration compared to those of a primary infection.
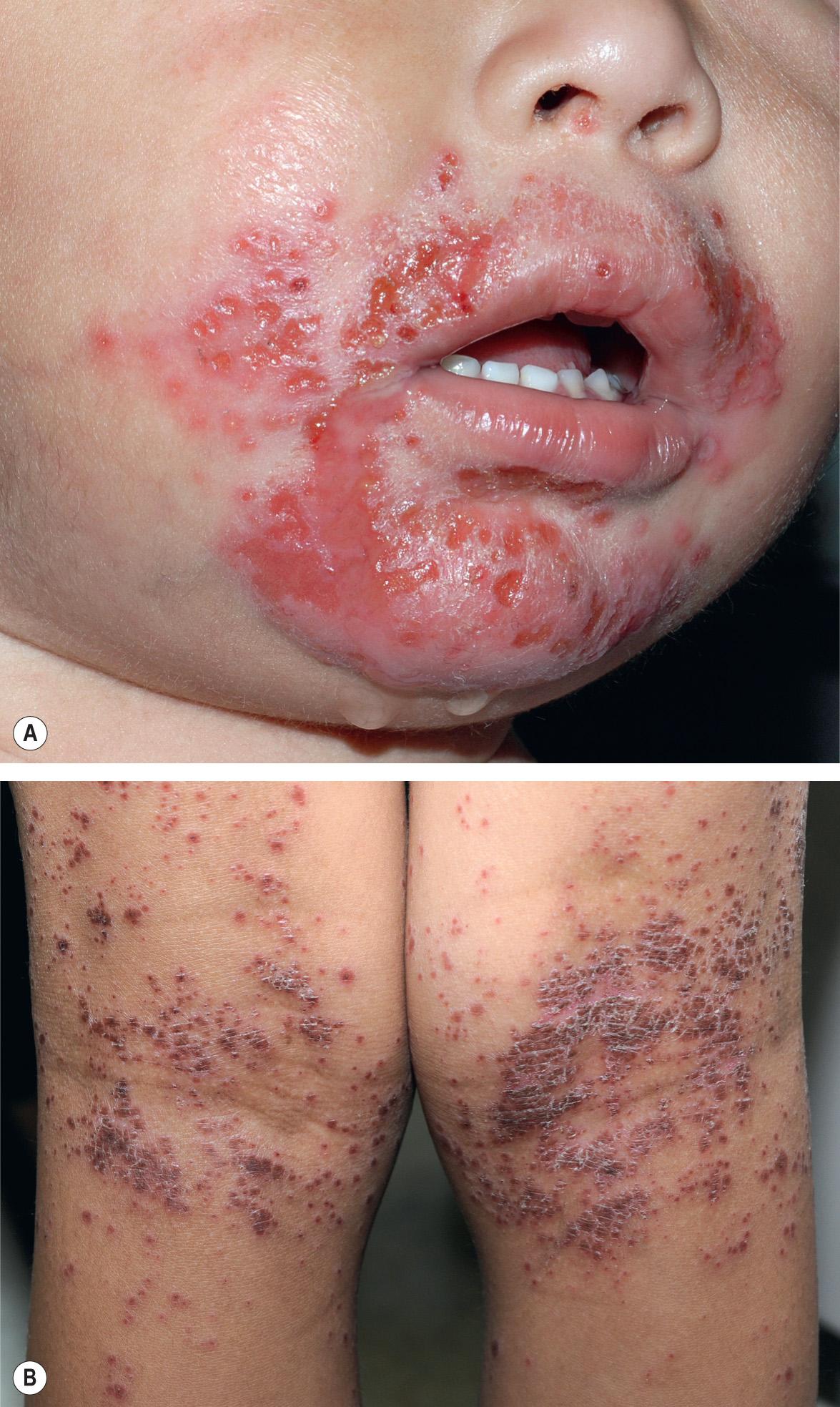
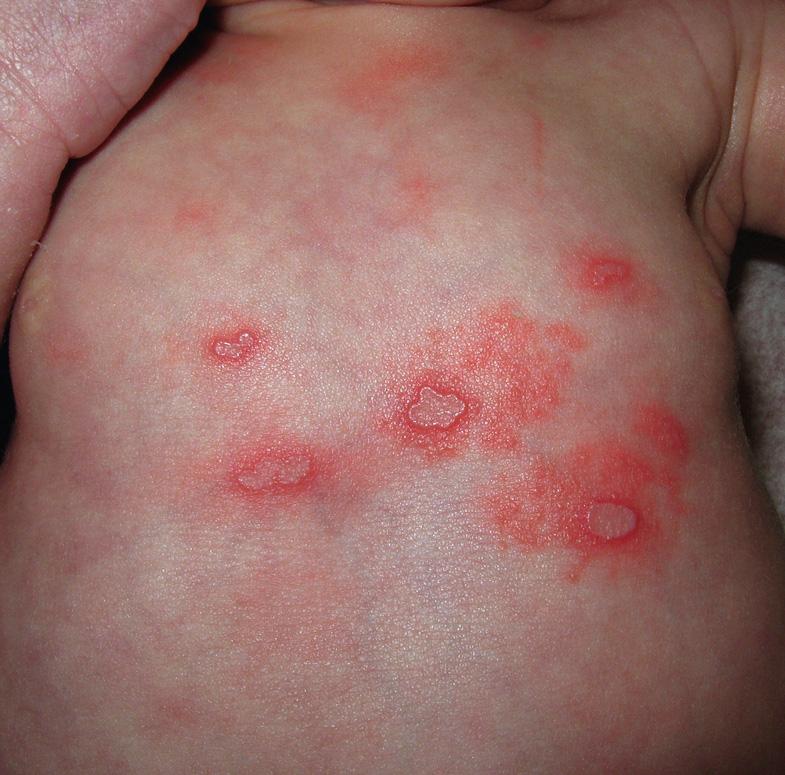
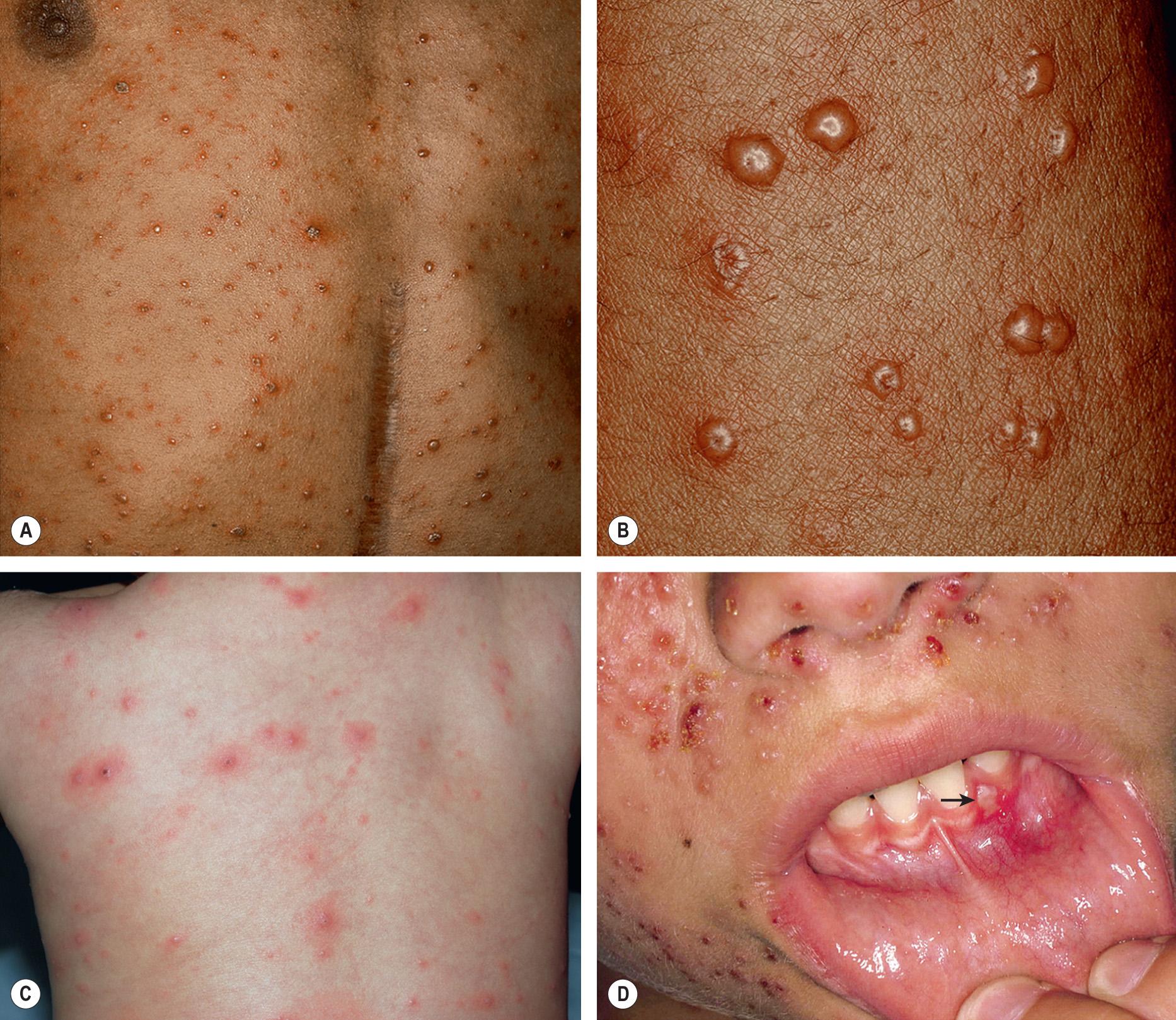
The majority of primary orolabial infections are asymptomatic. Symptomatic infections often present as gingivostomatitis in children or as pharyngitis and a mononucleosis-like syndrome in young adults. The mouth and lips are the most common sites of involvement ( Fig. 80.1A,B ), with oral lesions typically appearing on the buccal mucosa and gingivae. Edema and painful oropharyngeal ulcerations can lead to dysphagia and drooling.
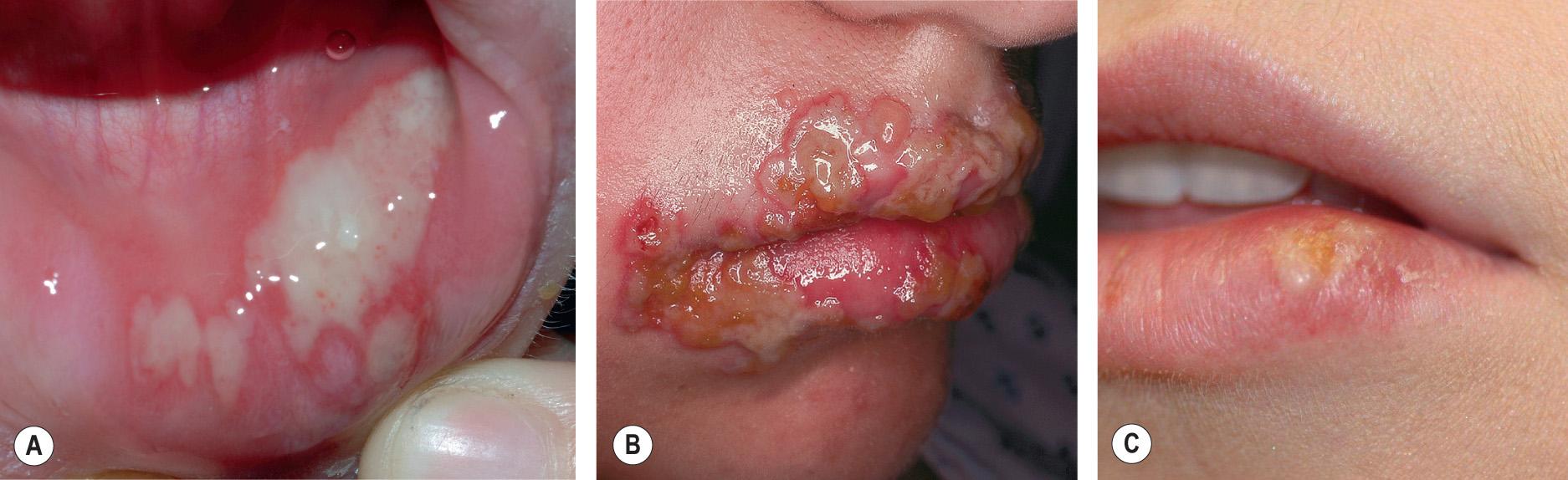
Among individuals with latent HSV-1 infection, 20–40% develop recurrent herpes labialis due to reactivation. Recurrent lesions appear most often on the vermilion border of the lip ( Fig. 80.1C ). Less common sites are perioral skin, nasal mucosa, cheek, and attached oral mucosa overlying bone (e.g. gingiva, hard palate) ( Fig. 80.2 ). In immunocompromised hosts, recurrent herpes infections can involve intraoral movable mucosa not overlying bone (see Fig. 72.1 ).
Primary and non-primary initial genital herpes infections are frequently asymptomatic but (especially with the former) can also present as an excruciatingly painful, erosive balanitis, vulvitis, or vaginitis. In women, lesions often also involve the cervix, buttocks, and perineum and may be associated with inguinal adenopathy and dysuria ( Fig. 80.3 ). Genital lesions in men typically occur on the glans or shaft of the penis, and the buttocks are occasionally affected. Systemic complaints and complications are more common in women. Extragenital lesions, urinary retention, and aseptic meningitis occur in 20%, 10–15%, and 10% of affected women, respectively . In contrast, aseptic meningitis is a rare complication of primary genital herpes infection in men. The presence of more extensive local involvement, regional lymphadenopathy, and fever generally distinguishes primary herpes infection from recurrent disease.
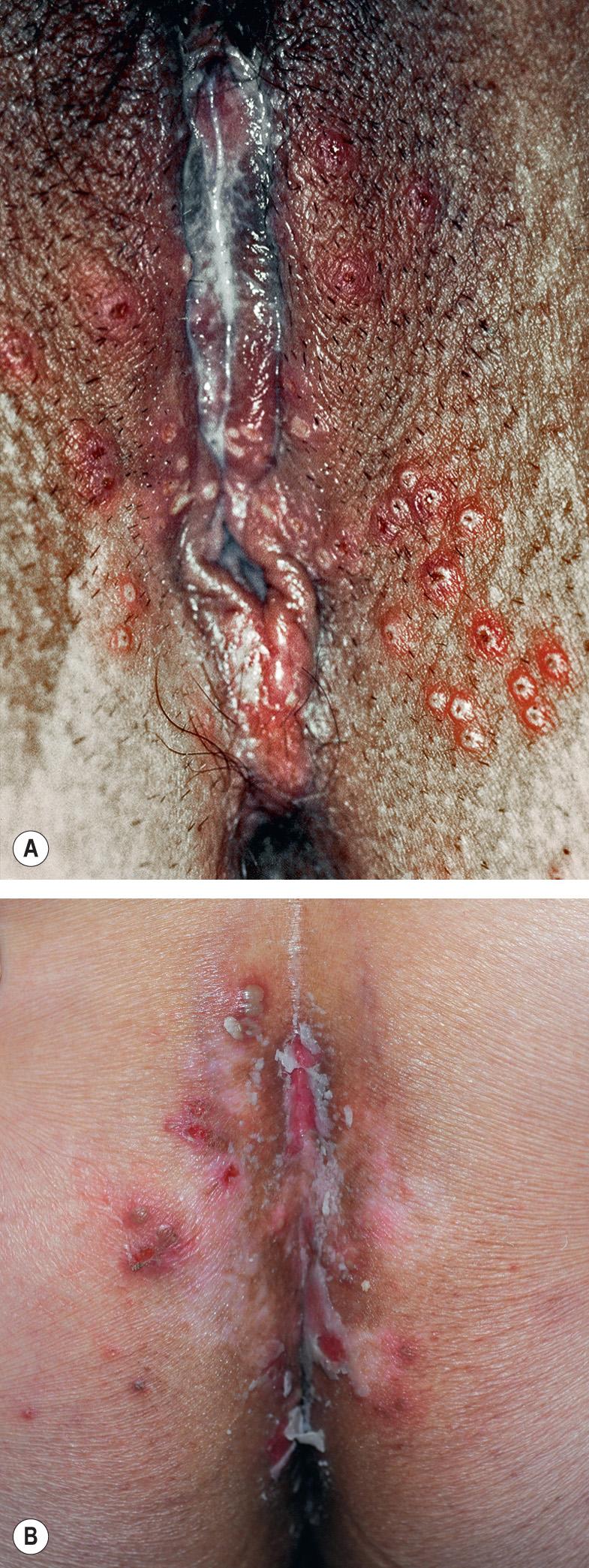
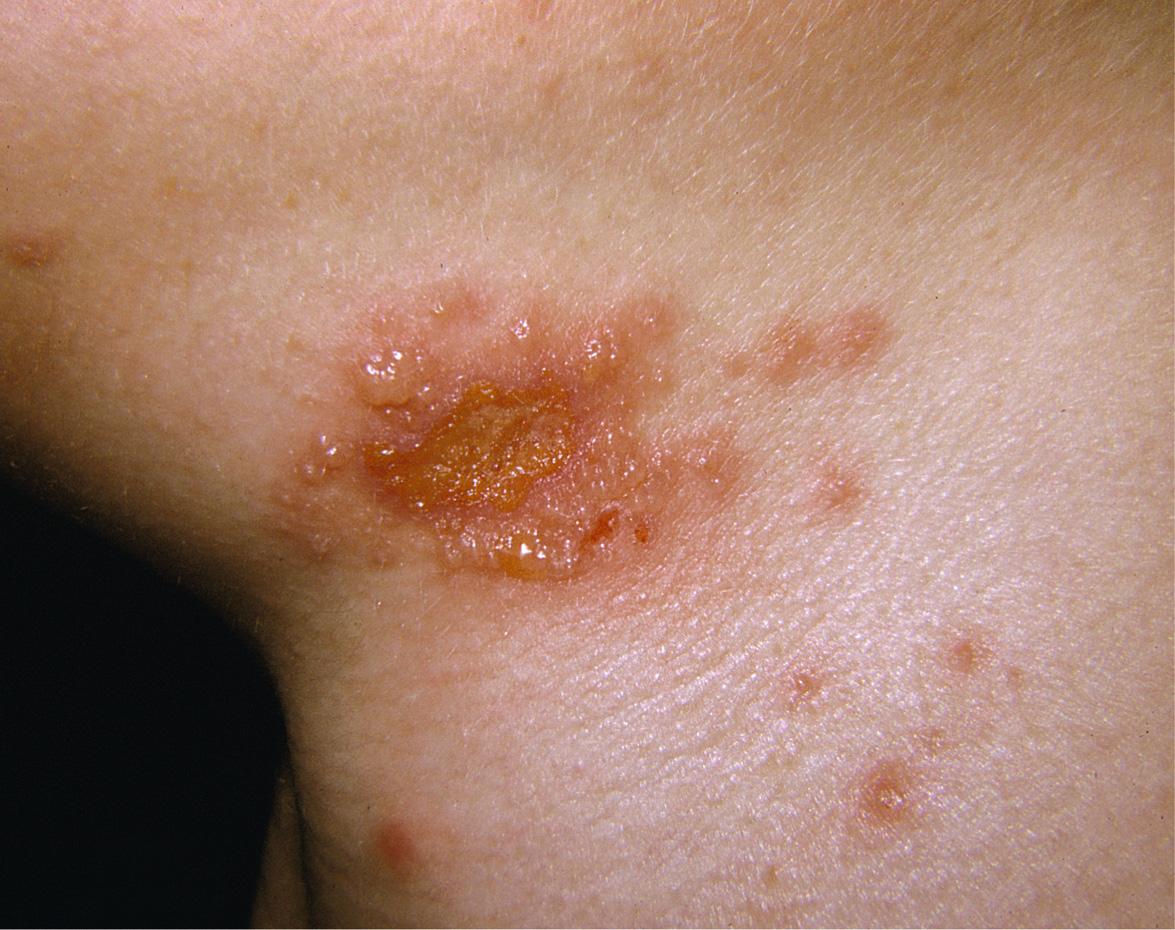
Genital HSV infections can result in both subclinical viral shedding and clinically evident recurrences, which are often relatively mild; recurrences occur more frequently with HSV-2 than HSV-1. Usually, a limited number of vesicles reappears on the genitalia or buttocks ( Fig. 80.4 ), with resolution within 7–10 days, compared to ~20 days for primary infections . Complications are uncommon. The frequency of recurrences correlates directly with the severity of the primary infection and tends to decrease over the next several years. The time interval between recurrences varies greatly, with individuals having an average of four to seven outbreaks annually. Surprisingly, although the majority of individuals with HSV-2 seropositivity report no history of genital herpes infection, symptomatic infection is eventually diagnosed in 50% of this group of patients.
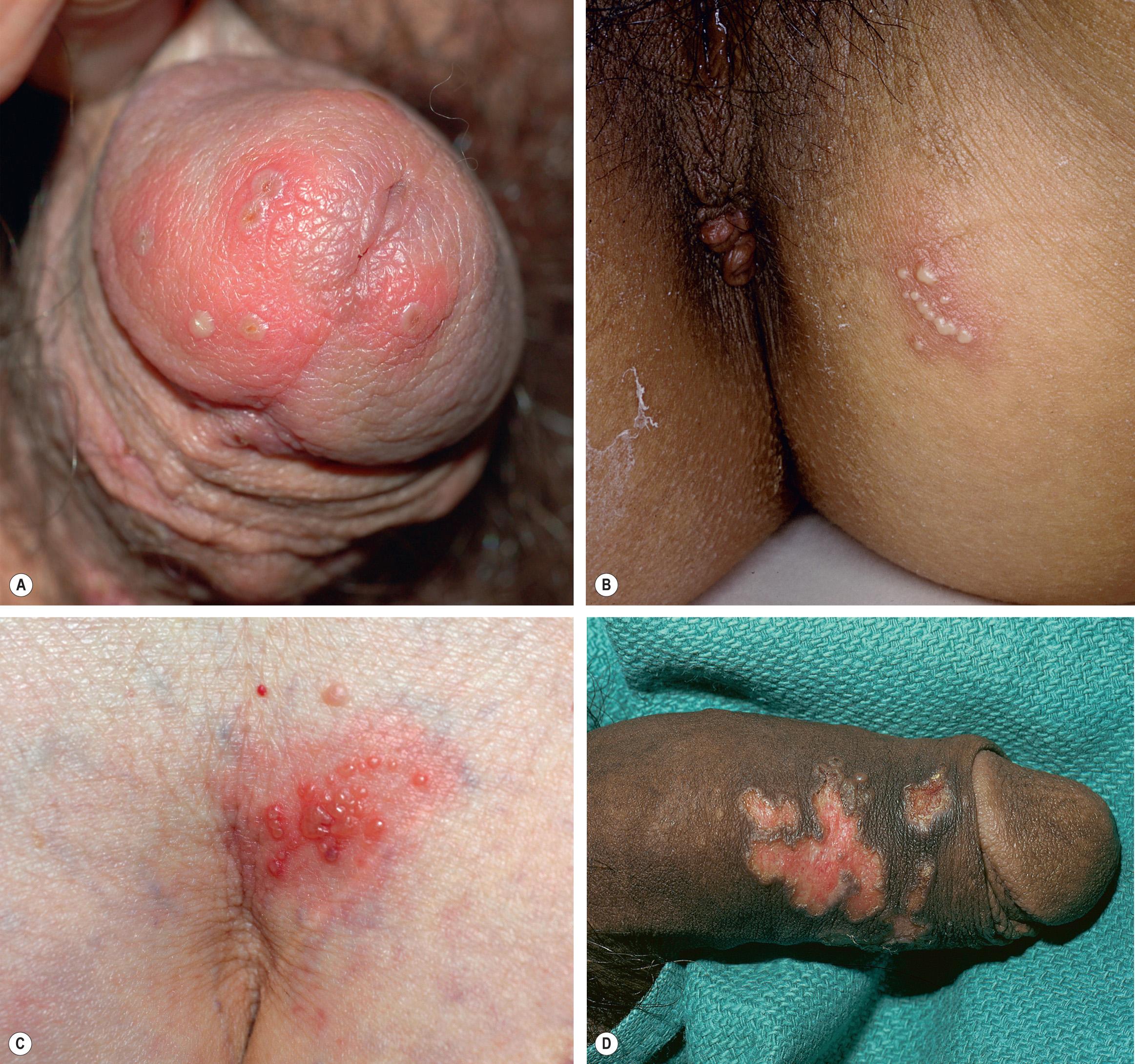
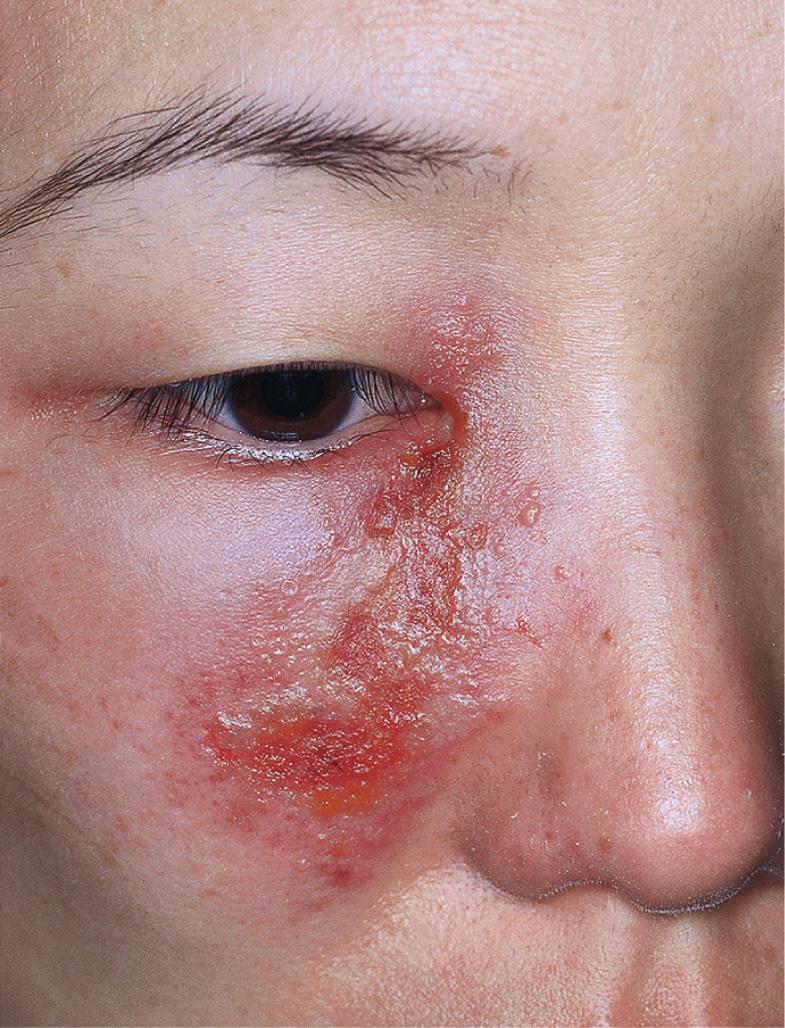
HSV infection can occur anywhere on the skin or mucous membranes. The clinical settings and manifestations of other cutaneous and extracutaneous presentations of HSV infection, including those in immunocompromised individuals and neonates , are summarized in Table 80.3 ( Figs 80.5–80.9 ).
| OTHER CLINICAL PRESENTATIONS OF HERPES SIMPLEX VIRUS INFECTIONS | ||
|---|---|---|
| Form of infection | Clinical settings and HSV type(s) | Cutaneous and/or extracutaneous findings |
| Predominantly mucocutaneous | ||
| Eczema herpeticum (Kaposi varicelliform eruption) |
|
|
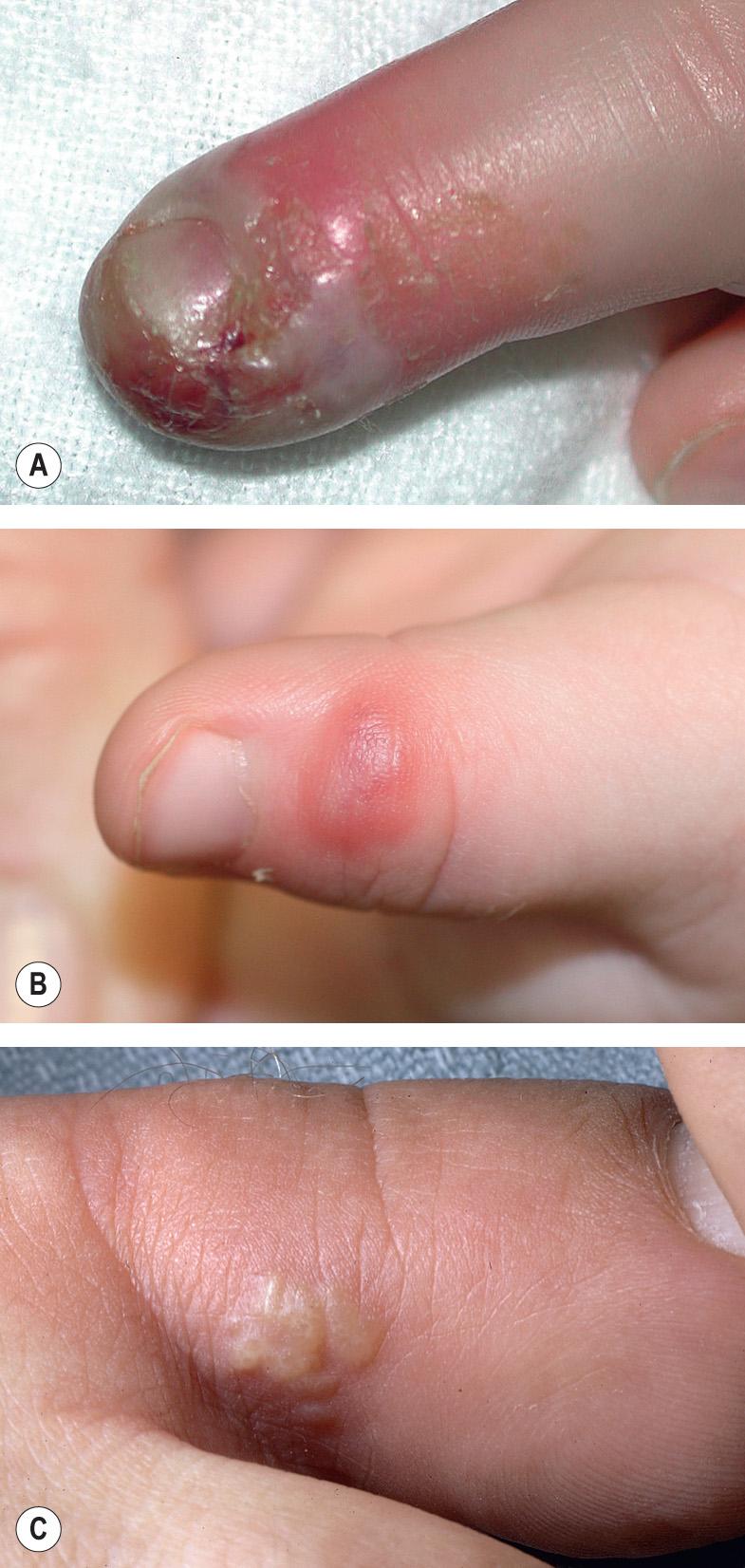
| Herpetic whitlow |
|
|
| Herpes gladiatorum |
|
|
| Herpes simplex folliculitis |
|
|
| Severe/chronic HSV infections |
|
|
| Predominantly extracutaneous | ||
| Ocular HSV infection |
|
|
| Herpes encephalitis |
|
|
| Proctitis |
|
|
| Cutaneous and extracutaneous | ||
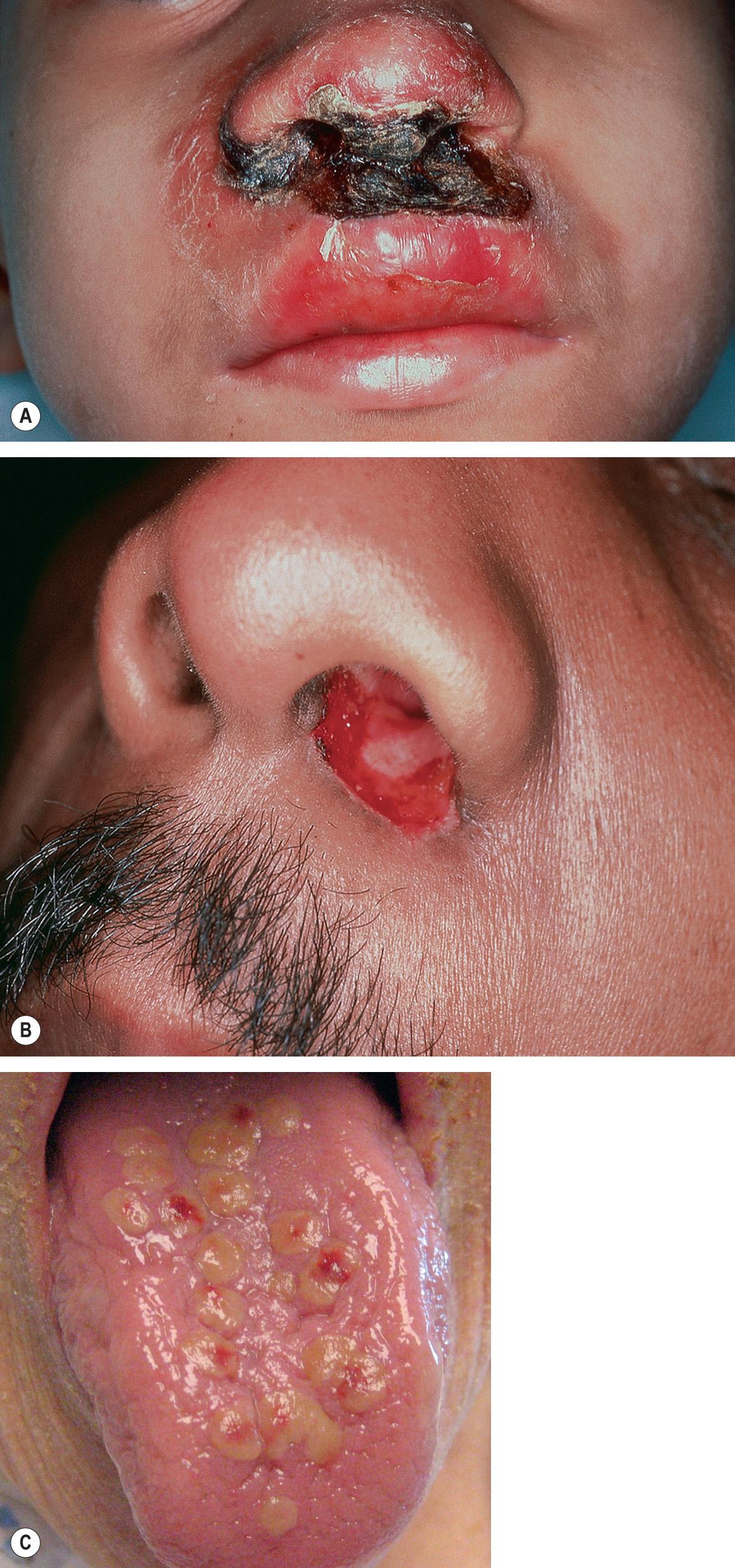
| Neonatal HSV infection |
|
|
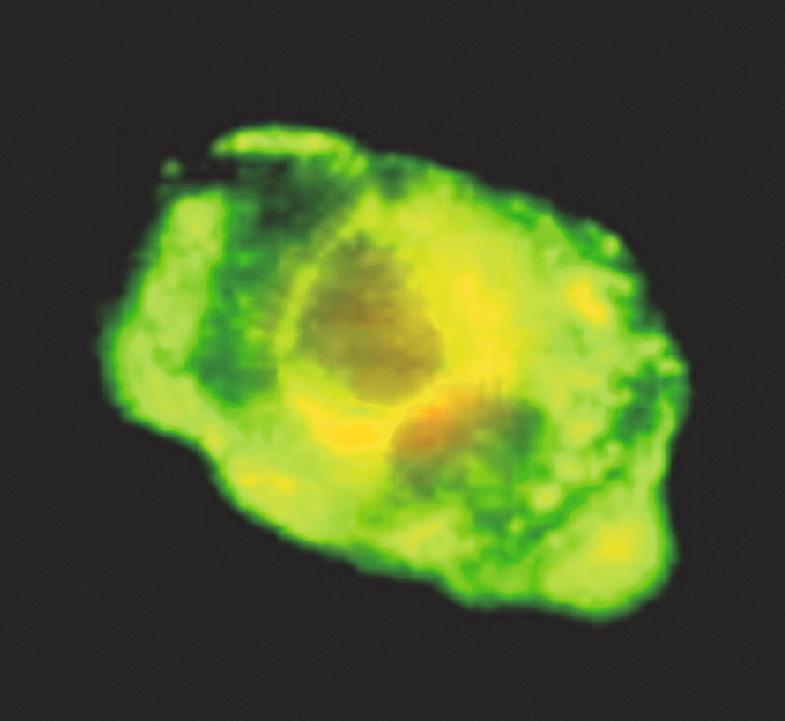
Multiple laboratory tests are available to diagnose HSV infection, including viral culture, direct fluorescent antibody assays (DFA; Fig. 80.10 ), molecular techniques, and serology. Identification of HSV by viral culture usually requires 2–5 days. In addition to being the preferred method for identifying HSV in the cerebrospinal fluid, PCR is increasingly being used as a more rapid, sensitive, and specific method for detecting HSV DNA in specimens from the skin and other organs.
For prevalence or incidence studies and to establish serostatus, the Western blot represents the gold standard for serologic assays; it is 99% sensitive and specific for HSV antibodies. In addition, several commercially available serologic tests can reliably identify patients who have been infected with HSV and are able to distinguish HSV-1 antibodies from HSV-2 antibodies by utilizing type-specific G-1 and G-2 glycoproteins, respectively. However, the sensitivity and specificity of these commercial assays do vary depending on the setting. The mean time to seroconversion following initial infection is 3–4 weeks .
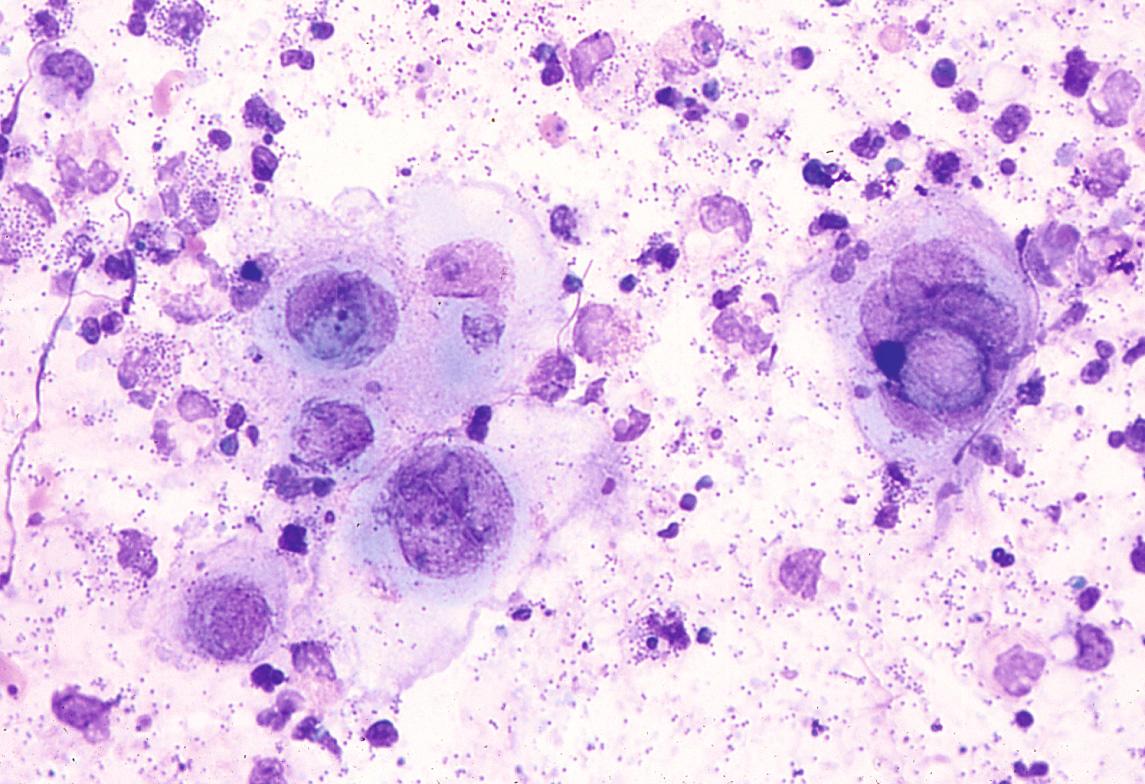
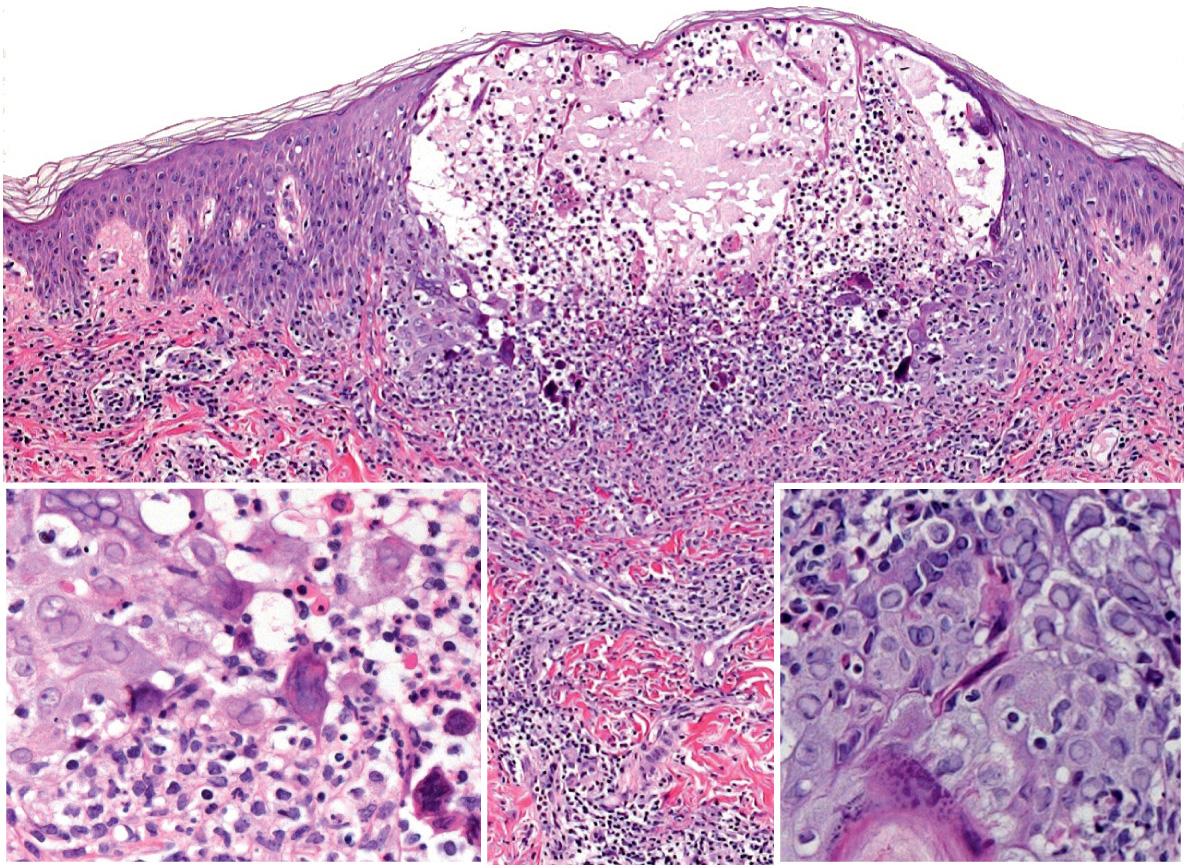
A Tzanck smear of scrapings from early lesions, in particular the base/edges of a freshly unroofed vesicle, reveals multinucleated epithelial giant cells in ~60–75% of HSV outbreaks ( Fig. 80.11 ). In routinely stained biopsy specimens as well as Tzanck smears, HSV-1 cannot be differentiated from HSV-2. Enlarged, slate-gray keratinocyte nuclei with margination of chromatin represent an early histologic finding. This is followed by intraepidermal vesiculation associated with ballooning degeneration of keratinocytes, which is most prominent at the vesicle base ( Fig. 80.12 ). These swollen, pale keratinocytes often fuse to form multinucleated giant cells and may contain eosinophilic intranuclear inclusion bodies surrounded by an artifactual cleft (Cowdry type A inclusions). Ballooning degeneration can affect adnexal structures as well as the interfollicular epidermis. A variably dense dermal infiltrate of lymphocytes, neutrophils, and eosinophils is observed, with a tendency to be milder in recurrent disease. Vascular changes may include areas of hemorrhagic necrosis and perivascular cuffing. Epidermal necrosis is sometimes extensive, especially in the setting of disseminated skin lesions.
Orolabial herpes can sometimes be confused with aphthous stomatitis, erythema multiforme major or Stevens–Johnson syndrome, herpangina and other coxsackievirus infections, pharyngitis (e.g. due to EBV), oral candidiasis, Behçet disease, and chemotherapy-induced mucositis (see Table 80.1 ). Oral aphthae typically present as a single or few lesion(s) on the buccal mucosa without associated vesicles. Erythema multiforme major (most often triggered by HSV) and Stevens–Johnson syndrome can usually be distinguished from HSV by the presence of characteristic cutaneous as well as other mucosal lesions. In herpangina, the posterior pharynx is more commonly involved, unlike in HSV infection, which favors the anterior portion of the mouth. Oral lesions of hand-foot-and-mouth disease are generally accompanied by vesicles that favor acral and (especially with coxsackievirus A6) perioral areas. Exudative tonsillitis, diffuse hyperemia of the oropharynx, and petechiae on the hard and soft palate are common manifestations of EBV-induced mononucleosis. Although a white, curd-like coating of the tongue, buccal mucosa and pharynx is the most common presentation of oral candidiasis, there is also an erythematous form of the disease. In Behçet disease, oral aphthae are recurrent and patients have additional manifestations such as genital aphthae, uveitis, and papulopustular or erythema nodosum-like skin lesions . In immunocompromised hosts, the possibility of coexisting infections must be considered.
Trauma, syphilitic chancres, chancroid, and lymphogranuloma venereum are considerations in the differential diagnosis of genital herpes (see Ch. 82 ). The syphilitic chancre is usually a single lesion that is neither painful nor recurrent. Chancroid often manifests as several tender ulcers with a yellowish-gray exudate overlying a base of granulation tissue, while lymphogranuloma venereum presents with a transient papule, herpetiform vesicle, erosion or ulcer; both conditions typically result in tender inguinal lymphadenopathy. EBV-related genital ulcers and other genital aphthae can be difficult to distinguish from herpetic lesions without viral culture, DFA, or PCR. Additional entities in the differential diagnosis of genital erosions are listed in Table 73.6 .
FDA-approved antiviral agents for treatment of recurrent episodes of orolabial herpes in immunocompetent persons include valacyclovir 2 g twice daily for 1 day, a single 1.5-g dose of famciclovir, and the topical treatments listed in Table 80.4 . Randomized controlled trials have shown that these therapies have modest benefits, decreasing the duration of mucocutaneous lesions, viral shedding, and pain ; efficacy is maximized when treatment is started at the first sign or symptom of a recurrence. Both valacyclovir and famciclovir have also been shown to reduce the frequency of orofacial herpes outbreaks after laser resurfacing.
| ANTIVIRAL THERAPY FOR HERPES SIMPLEX VIRUS AND VARICELLA–ZOSTER VIRUS | |
|---|---|
| Disease context | Drug and dosage |
| Herpes simplex infections | |
| Orolabial herpes * (recurrence) |
|
| Genital herpes (first episode) |
|
| Genital herpes (recurrence) |
|
| Chronic suppression |
|
| Neonatal | Acyclovir: 20 mg/kg iv q8 h × 14–21 days |
| Immunocompromised | |
| Eczema herpeticum/Kaposi varicelliform eruption ** |
|
| Genital herpes, recurrent in the setting of HIV infection | |
| Chronic suppression in the setting of HIV infection | |
| Acyclovir-resistant HSV in immunocompromised patients |
|
| Varicella–zoster virus infections | |
| Varicella |
|
| Herpes zoster |
|
| Immunocompromised patient or disseminated disease | Acyclovir: 10 mg/kg (500 mg/m 2 ) iv q8 h × 7–10 days or until cropping has ceased (depending on the setting, consider continuing until lesions are healed) |
* Indications for and efficacy of antiviral treatment for initial episodes of orolabial herpes in immunocompetent adults have not been defined, but the regimens used for initial genital episodes can be considered in individuals with severe disease; acyclovir 15 mg/kg (200 mg max) po 5×/d × 7 days (initiated within 3 days of disease onset) has been shown to be beneficial in young children with primary herpes gingivostomatitis.
** Not specifically FDA-approved for this indication.
† FDA-approved for ages 2–17 years; pharmacies can compound valacyclovir tablets into an oral suspension (25 or 50 mg/ml).
For treatment of primary and recurrent genital herpes, oral antivirals are the agents of choice (see Table 80.4 ). With initiation within 24–48 hours of onset, acyclovir, famciclovir, and valacyclovir reduce the duration of viral shedding, pain, and time to healing for first-episode and recurrent genital herpes. Intravenous acyclovir is indicated for neonatal HSV infection, severe infections in immunocompromised hosts, severe eczema herpeticum, and patients with systemic complications. In immunocompromised patients, it is important to treat with oral or intravenous antivirals until the cutaneous lesions are completely healed. Systemic antiviral agents need to be dose-adjusted for patients with impaired renal function ( Table 80.5 ).
| DOSE REDUCTIONS FOR ACYCLOVIR, VALACYCLOVIR AND FAMCICLOVIR IN PATIENTS WITH RENAL DISEASE | ||||||
|---|---|---|---|---|---|---|
| Indication | Acyclovir | Valacyclovir | Famciclovir | |||
| Creatinine clearance (ml/min) | Adjusted dose | Creatinine clearance (ml/min) | Adjusted dose | Creatinine clearance (ml/min) | Adjusted dose | |
| Primary genital herpes simplex | <10 | 200 mg po BID | 10–29 | 1 g daily | 20–39 | 125 mg BID |
| <10 | 500 mg daily | <20 | 125 mg daily | |||
| Recurrent genital herpes simplex | <30 | 500 mg daily | 40–59 | 500 mg BID × 1 day | ||
| 20–39 | 500 mg once | |||||
| <20 | 250 mg once | |||||
| Chronic suppression of herpes simplex | <30 | 500 mg q48 h or daily | 20–39 | 125 mg BID | ||
| <20 | 125 mg daily | |||||
| Recurrent orolabial herpes simplex | <10 | 200 mg po BID * | 30–49 | 1 g BID × 1 day | 40–59 | 750 mg once |
| 10–29 | 500 mg BID × 1 day | 20–39 | 500 mg once | |||
| <10 | 500 mg once | <20 | 250 mg once | |||
| Herpes zoster | 10–25 | 800 mg po TID | 30–49 | 1 g BID | 40–59 | 500 mg BID |
| <10 | 800 mg po BID | 10–29 | 1 g daily | 20–39 | 500 mg daily | |
| <10 | 500 mg daily | <20 | 250 mg daily | |||
| Immunocompromised patients with mucocutaneous herpes simplex; for iv acyclovir, also herpes encephalitis and disseminated zoster | 25–50 | 5–10 mg/kg iv q12 h | ||||
| 10–24 | 5–10 mg/kg iv q24 h | 10–29 | 1 g daily * | 20–39 | 500 mg daily * | |
| <10 | 2.5–5 mg/kg iv q24 h | <10 | 500 mg daily * | <20 | 250 mg daily * | |
Chronic suppressive therapy with oral antiviral agents is usually reserved for patients with six or more outbreaks per year (see Table 80.4 ), although clinicians often exercise more lenient criteria for initiation of therapy for individuals with severe or problematic outbreaks, an insufficient prodrome to benefit from episodic therapy, or immunosuppression. In addition to decreasing the frequency of symptomatic outbreaks or even eliminating them, suppressive therapy decreases asymptomatic viral shedding by 95% and can thereby reduce transmission of genital herpes to a susceptible partner . Daily suppressive therapy, together with consistent condom use and avoidance of sexual activity during recurrences, is therefore also recommended for individuals with genital herpes who have a seronegative partner.
The emergence of acyclovir-resistant HSV is an increasing concern for immunocompromised individuals. Foscarnet is the only antiviral drug approved by the FDA for treatment of acyclovir-resistant HSV (see Fig. 127.10 ). Cidofovir is another antiviral agent that has shown efficacy in the treatment of acyclovir-resistant HSV. The use of foscarnet or systemic cidofovir is limited by potentially severe renal toxicity and the requirement for intravenous administration. Although not FDA-approved, compounded topical cidofovir has been advocated by the CDC as a “user-friendly”, albeit expensive, alternative treatment .
Individuals co-infected with HIV and HSV have more severe outbreaks and more frequent viral shedding than those without HIV infection. When added to the medical regimen of HIV-infected patients, antiherpetic suppressive therapy appears to allow the co-infected person to respond better to antiretroviral therapy and to reduce genital and plasma HIV-1 RNA levels, but it does not seem to decrease the risk of HIV-1 transmission . Oral acyclovir, famciclovir, and valacyclovir can be used for genital and orolabial HSV in the setting of HIV infection if there is no evidence of acyclovir resistance.
There is significant interest in prevention of HSV disease. Between 70% and 80% of HSV is transmitted during periods of asymptomatic viral shedding. Sexual abstinence is the only method for absolute prevention of genital herpes, which can be transmitted even with the use of condoms . In addition to antiviral therapy, patient education regarding prevention of genital herpes transmission is essential.
There is currently no licensed vaccine available for HSV, although several vaccines are under development and evaluation for prevention of HSV infection and recurrences. GEN-003, a therapeutic vaccine under investigation for genital HSV-2 infection, was found in a phase I/IIa clinical trial to reduce the rates of outbreaks and asymptomatic viral shedding . In randomized controlled studies, adults with a history of genital herpes due to HSV-2 who received the helicase-primase inhibitor pritelivir had significantly decreased HSV shedding and fewer days with genital lesions than those who received placebo or valacyclovir ; however, drug development in the US has been suspended due to possible adverse effects such as anemia in primate studies.
▪ Varicella – chickenpox ▪ Herpes zoster – shingles
VZV is the etiologic agent of varicella (chickenpox) and herpes zoster (shingles)
VZV has a high morbidity and mortality rate in immunocompromised hosts
Antiviral treatment as well as vaccination can reduce or eliminate serious disease-associated sequelae and decrease the incidence of VZV infection
VZV is the etiology of varicella (chickenpox) and herpes zoster (shingles). Varicella is usually symptomatic, and before the advent of the varicella vaccine, it occurred in 90% of children in the US by the time they reached 10 years of age. Herpes zoster represents reactivation of latent VZV infection and develops in ~20% of healthy adults and 50% of immunocompromised persons, with substantial morbidity and mortality in the latter group. Early initiation of antiviral treatment can reduce or eliminate serious sequelae of VZV infections.
Heberden first distinguished chickenpox from smallpox in 1767. The term “chickenpox” is thought to come from either the French word “chiche-pois” for chickpea (referring to the size of the vesicles) or the Old English word “gigan” meaning “to itch”. The relationship between varicella and herpes zoster was first recognized in 1888, when von Bokay described the development of varicella in children following exposure to those with herpes zoster infection. Kundratitz (1922) and Bruusgaard (1932) more convincingly demonstrated the association between the two diseases by the development of varicella in susceptible children who had been inoculated with vesicle fluid from patients with herpes zoster. This was followed by the identification of the same virus in both diseases, confirming that their etiologies were identical.
VZV has a worldwide distribution and 98% of the adult population is seropositive. In the pre-vaccine era, 90% of children <10 years of age developed varicella and ~4 million cases occurred annually in the US, with large outbreaks during the winter and spring. Since the introduction of the varicella vaccine in 1995, the overall incidence of varicella has decreased by ~85%, with evidence of herd immunity. The age of peak incidence also shifted from 5–9 years to 10–14 years, and a higher rate of breakthrough varicella among immunized children in the latter age group led to the addition of a second varicella vaccine dose to the routine childhood immunization schedule in 2006 .
Although the incidence and severity of herpes zoster increase substantially in middle to late adulthood, it can occur at any age and is more common in younger persons who had a varicella infection within the first year of life. Overall, individuals with a history of varicella have a 20% lifetime chance of developing zoster. The annual incidence of herpes zoster in the US and Europe is classically 2.5/1000 persons between ages 20 and 50 years, 5/1000 between ages 51 and 79 years, and 10/1000 in those >80 years of age. However, there is evidence that exposure to varicella can protect seropositive adults from the development of zoster, and it has been postulated that widespread vaccination against varicella could reduce this immune boosting effect and increase the incidence of zoster. Whether this hypothetical increase will be counterbalanced by the zoster vaccine remains to be determined. Other risk factors for herpes zoster include mental and physical stress, a family history of zoster, use of tofacitinib or proteasome inhibitors (e.g. bortezomib, carfilzomib), and an immunocompromised state, especially when due to HIV infection and allogeneic hematopoietic stem cell transplantation .
Airborne droplets are the usual route of transmission of varicella, although direct contact with vesicular fluid is another mode of spread, and the incubation period is 11–20 days. Varicella is extremely contagious, and 80–90% of susceptible household contacts develop a clinically evident infection. The affected individual is infectious from 1–2 days before skin lesions appear until all the vesicles have crusted.
During varicella infection, primary viremia occurs after an initial 2–4 days of viral replication within regional lymph nodes. A cycle of viral replication in the liver, spleen, and other organs is then followed by a secondary viremia, which seeds the entire body 14–16 days postexposure. During this period, the virus enters the epidermis by invading capillary endothelial cells. VZV subsequently travels from mucocutaneous lesions to dorsal root ganglion cells, where it remains latent until reactivation at a later time.
Herpes zoster appears upon reactivation of latent VZV, which may occur spontaneously or be induced by stress, fever, radiation therapy, local trauma, or immunosuppression. During a herpes zoster outbreak, the virus continues to replicate in the affected dorsal root ganglion and produces a painful ganglionitis. Neuronal inflammation and necrosis can result in a severe neuralgia that intensifies as the virus spreads down the sensory nerve. Fluid from herpes zoster vesicles can transmit VZV to seronegative individuals, leading to varicella but not herpes zoster. The transmission rate to susceptible household contacts is ~15% for zoster, compared to 80–90% for varicella.
A prodrome of mild fever, malaise, and myalgia may occur, especially in adults. This is followed by an eruption of pruritic, erythematous macules and papules, which starts on the scalp and face, and then spreads to the trunk and extremities ( Fig. 80.13 ). Lesions rapidly evolve over ~12 hours into 1–3 mm clear vesicles surrounded by narrow red halos (“dew drops on a rose petal”). The number of vesicles varies from only a few to several hundred, and there is often involvement of the oral mucosa (see Fig. 80.13D ). Sparing of the distal and lower extremities is common. Older vesicles evolve to form pustules and crusts, with individual lesions healing within 7–10 days. The presence of lesions in all stages of development is a hallmark of varicella.
The disease course is usually self-limited and benign in healthy children. However, prior to introduction of the varicella vaccine, complications resulted in 11 000 hospitalizations annually in the US. Secondary bacterial infection of the skin with subsequent scarring is the most common complication. CNS sequelae are uncommon (<1 per 1000 cases) and may include encephalitis and acute cerebellar ataxia; Reye syndrome which consists of encephalitis plus fatty liver is now rare due to avoidance of aspirin in children with varicella. Varicella in adolescents and adults is often more severe than in children, with an increased number of skin lesions (see Fig. 80.13A ) and more frequent development of pneumonia, which has a 10–30% mortality rate if untreated . Occasional complications include thrombocytopenia, hepatitis, glomerulonephritis, optic neuritis, keratitis, arthritis, myocarditis, pancreatitis, orchitis, and vasculitis.
Maternal varicella during the first 20 weeks of pregnancy is associated with a ~2% risk of congenital varicella syndrome (varicella embryopathy). Possible congenital defects include a low birth weight, cutaneous scarring, ocular abnormalities, cortical atrophy, psychomotor retardation, and hypoplastic limbs. Children whose mothers had varicella during pregnancy may also develop zoster early in life without ever having extrauterine varicella. Severe neonatal varicella, in which amelioration by maternal antibodies is lacking, can occur when maternal varicella develops between 5 days before and 2 days after delivery.
In immunocompromised individuals, varicella may lead to severe complications and even death. These patients frequently have a more extensive and atypical cutaneous eruption, often with hemorrhagic or purpuric lesions . Involvement of the lungs, liver, and CNS is common.
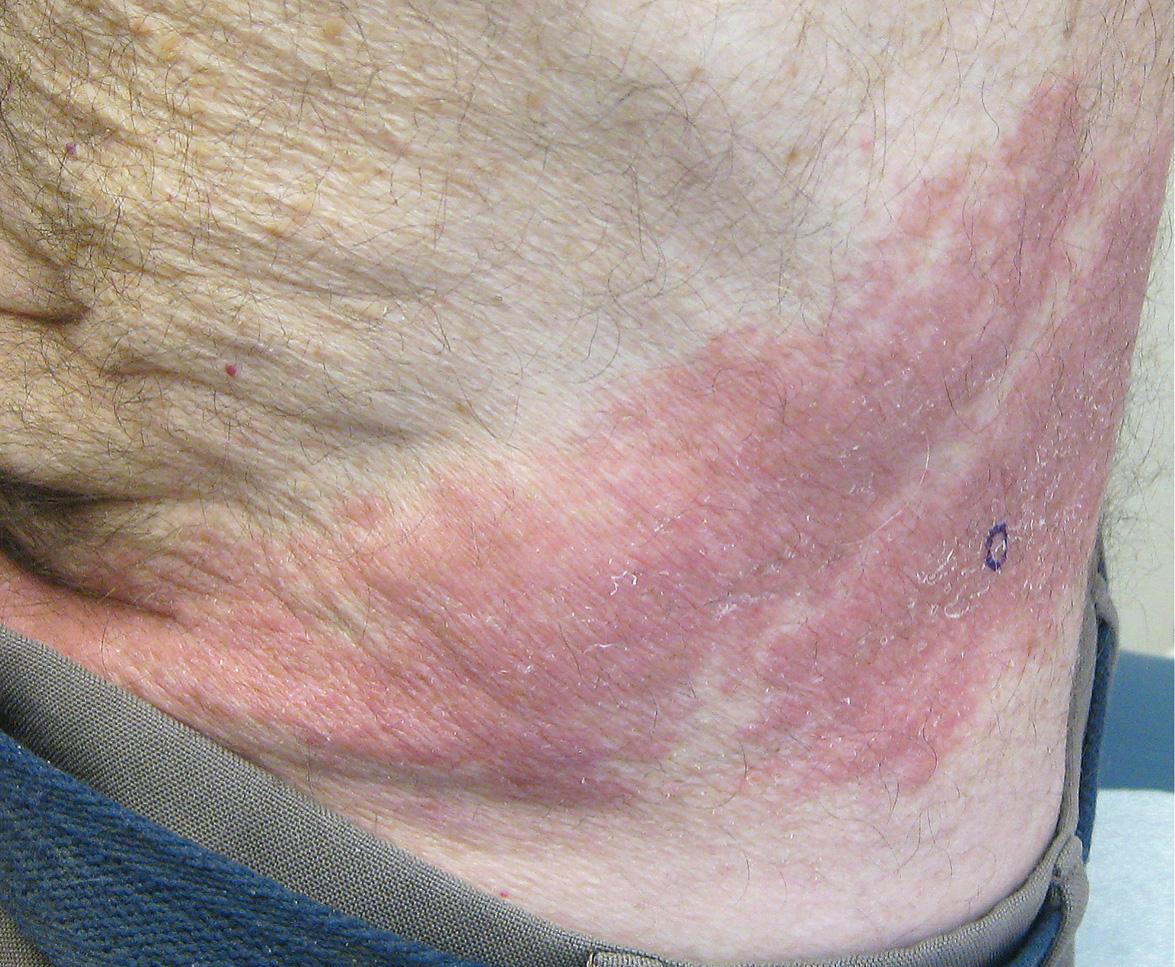
Reactivation of VZV may occur at any time after the development of varicella. In >90% of patients, herpes zoster begins with a prodrome of pruritus, tingling, tenderness, hyperesthesia, and/or intense pain, which can mimic a myocardial infarction, surgical abdomen, or toothache. These symptoms occasionally occur without subsequent skin lesions, a phenomenon referred to as “zoster sine herpete”. However, most patients develop a painful eruption of grouped vesicles on an erythematous base in a dermatomal distribution ( Figs 80.14–80.16 ); the lesions can involve more than one contiguous dermatome and occasionally cross the midline. Edematous papules and plaques may precede the appearance of vesicles, and progression to pustules or bullae can occur (see Fig. 80.15 ). Any cutaneous site may be affected, with the trunk representing the most common location, followed by the face. VZV DNA can often be detected in the peripheral blood and saliva of patients with herpes zoster, even after initiation of antiviral treatment .
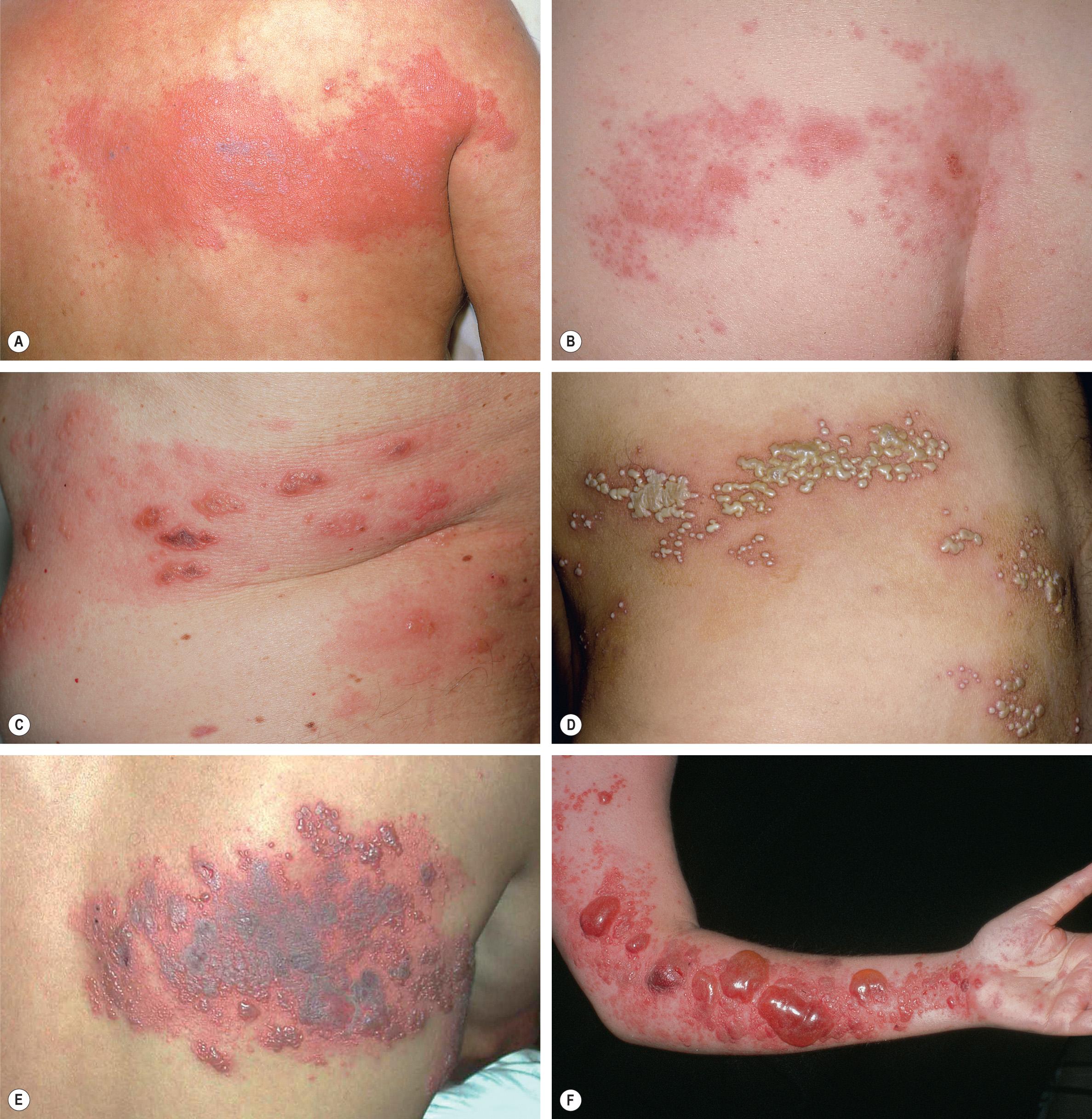
Herpes zoster usually resolves without sequelae in immunocompetent children and young adults. However, the pain, cutaneous lesions, and complications of herpes zoster become more severe with increasing age and immune compromise. The most common complication is postherpetic neuralgia, which is characterized by dysesthetic pain (e.g. burning or stabbing sensation, allodynia) that persists after the skin lesions have healed. This affects 10–20% of all herpes zoster patients and increases in both incidence and severity with age . Complications of herpes zoster related to the dermatome that is affected are presented in Table 80.6 . Other complications can include secondary bacterial infection, scarring, meningoencephalitis, pneumonitis, and hepatitis. Cutaneous disorders that can develop within sites of previous herpes zoster, including granulomatous ( Fig. 80.17 ) and pseudolymphomatous lesions, are listed in Table 80.7 .
| CLINICAL FINDINGS RELATED TO THE DERMATOME INVOLVED IN HERPES ZOSTER | ||
|---|---|---|
| Dermatome/nerve involved | Cutaneous findings | Extracutaneous findings |
| Ophthalmic division of the trigeminal nerve (V 1 ; ~10–15% of zoster patients) | Hutchinson's sign refers to increased risk of ocular involvement if skin lesions are in the distribution of the nasociliary branch, which supplies the nasal tip, dorsum and root of the nose, and medial canthus as well as the cornea |
|
| Geniculate ganglion of the facial nerve – Ramsay Hunt syndrome | Lesions affecting the external auditory canal, tympanic membrane, anterior two-thirds of the tongue, and/or hard palate |
|
| Cervical dermatome | Lesions in cervical dermatome |
|
| Thoracic dermatome | Lesions in thoracic dermatome |
|
| Lumbar dermatome | Lesions in lumbar dermatome |
|
| CUTANEOUS DISORDERS THAT CAN DEVELOP WITHIN SITES OF HERPES ZOSTER |
|
* “Giant cell lichenoid dermatitis” has also been described within herpes zoster scars in an allogeneic bone marrow transplant recipient.
† In patients with chronic lymphocytic leukemia, a diagnosis of “pseudolymphoma” should be made only if leukemia cutis has been excluded.
** Some solid organ metastases may show a zosteriform arrangement of the lesions without history of previous herpes zoster.
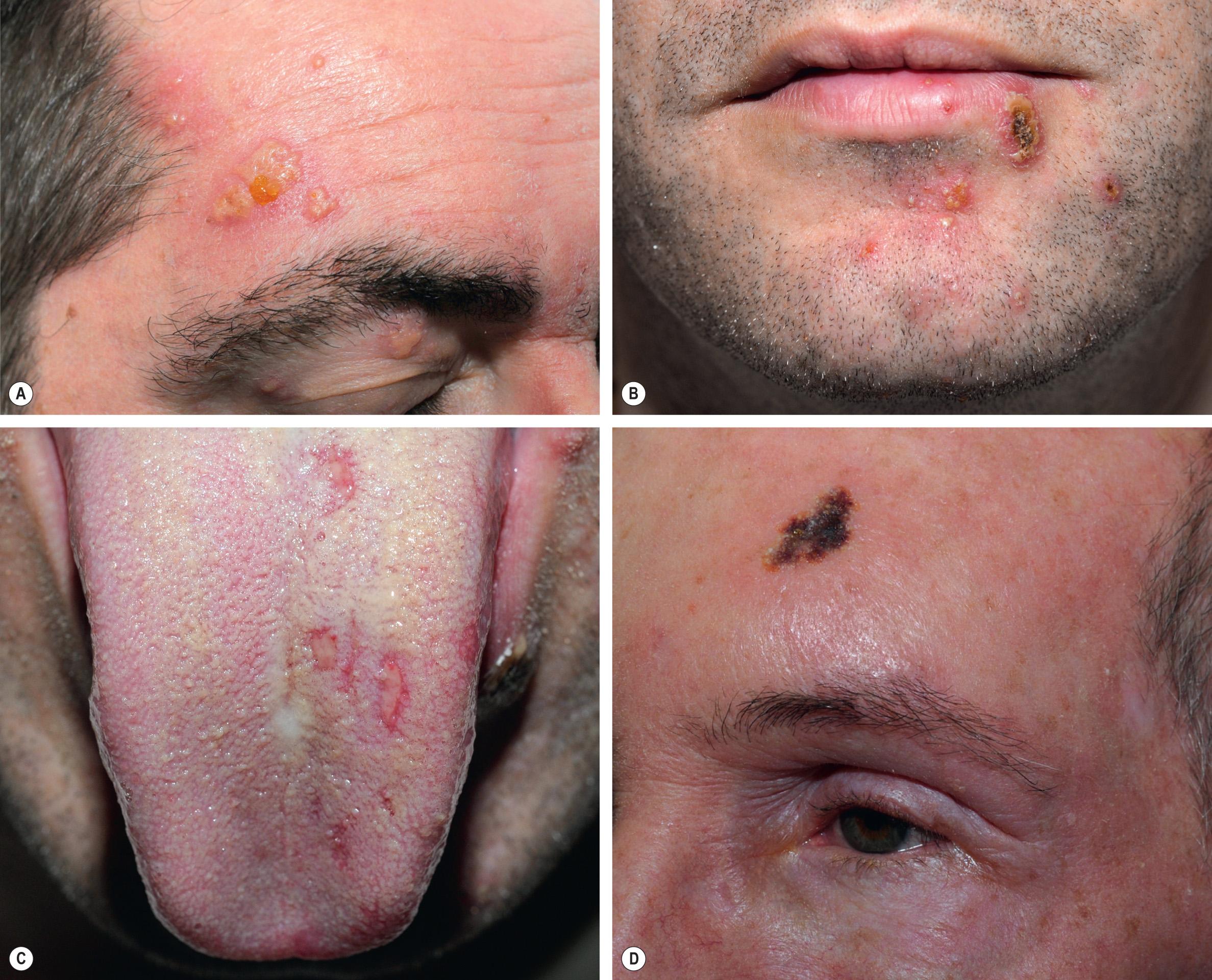
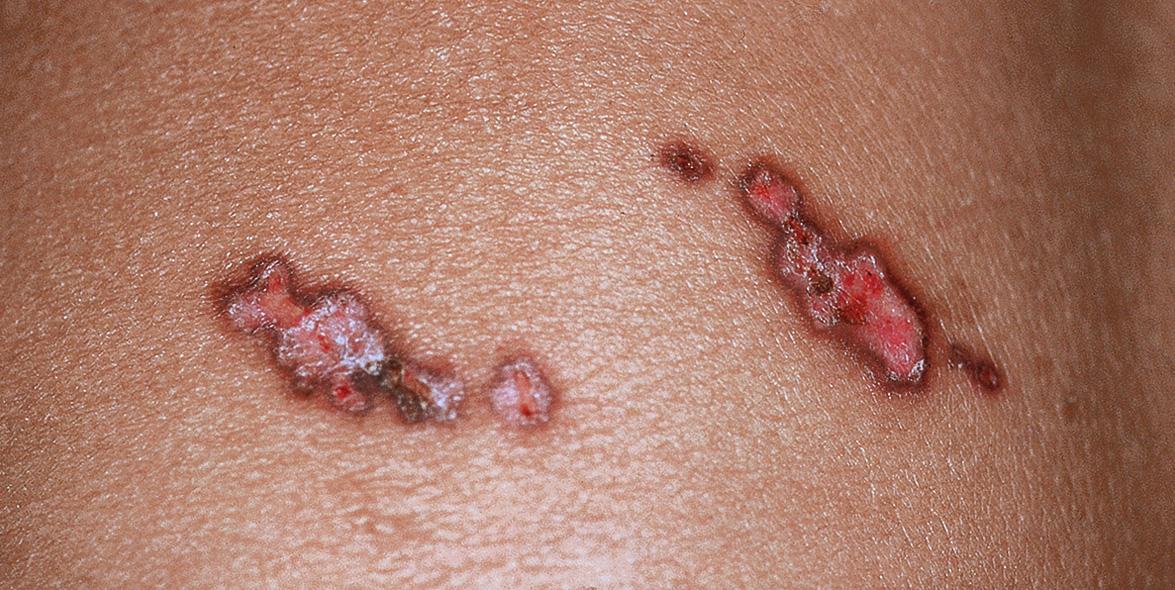
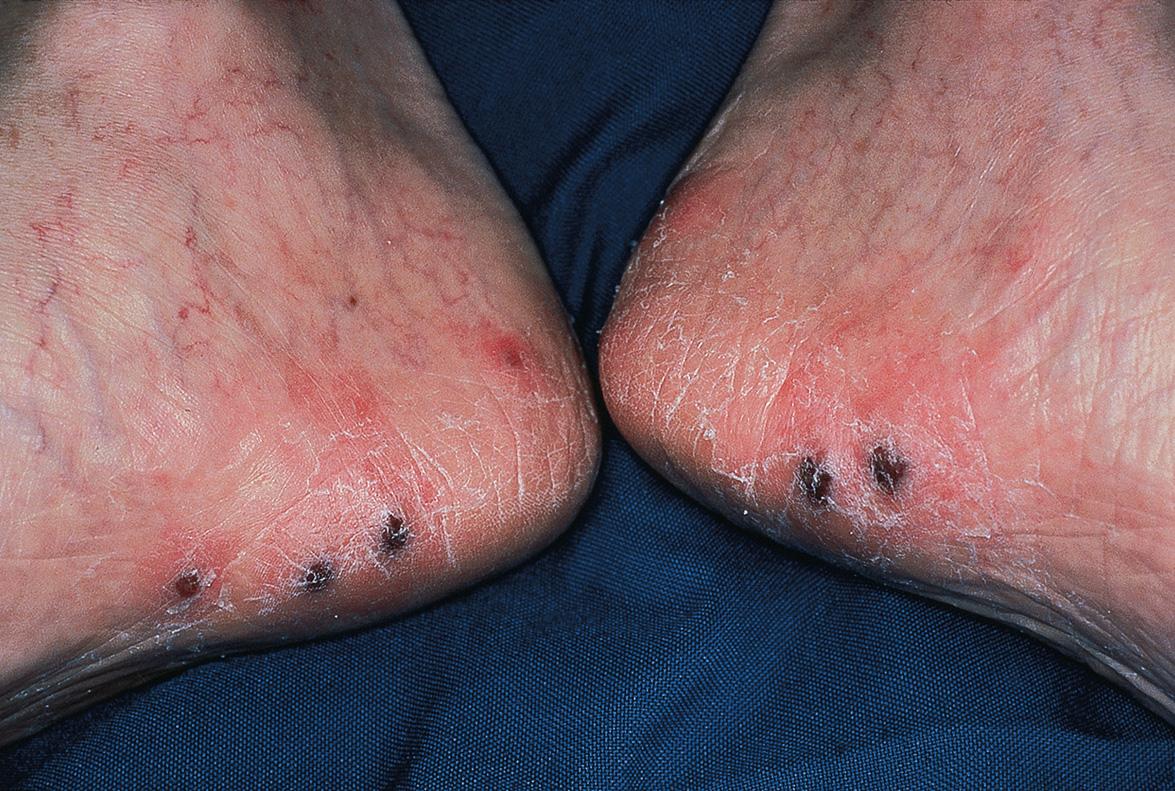
In immunocompromised individuals, herpes zoster may be more severe and can have unusual clinical presentations, e.g. persistent crusted, verrucous lesions in HIV-infected patients ( Fig. 80.18 ) or postherpetic hyperhidrosis . Disseminated cutaneous disease, defined as more than 20 vesicles outside the area of primary or adjacent dermatomes, and/or visceral involvement occurs in ~10% of immunocompromised persons with zoster ( Fig. 80.19 ) .
A clinical diagnosis can usually be made based upon the history, including previous varicella or vaccine administration, and physical examination. A Tzanck smear, PCR or DFA can assist in quickly confirming the diagnosis (see above and Figs 80.10 & 80.11 ). The latter two (but not the Tzanck smear) can differentiate between HSV and VZV. Varicella and herpes zoster have the same histologic findings as HSV infections (see above and Fig. 80.12 ), but immunohistochemical staining can distinguish between the two viruses.
Additional laboratory tests include viral culture and serology. Viral culture is a very specific test; however, it is not very sensitive and results may not be available for >1 week. Serologic assays are diagnostic of VZV if the convalescent serum has at least a fourfold increase in the VZV titer relative to the acute serum. As a result, serology is only useful in retrospect.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here