Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Most pituitary region masses are pituitary adenomas; however, when other sellar region masses are encountered by clinicians or pathologists, the differential diagnosis can be broad. This chapter will focus on updates for pituitary adenomas, as well as discuss other sellar region entities that may mimic clinically nonfunctioning pituitary adenomas—including non-neuroendocrine tumors (pituicytoma, granular cell tumor of the neurohypophysis, and spindle cell oncocytoma), cystic lesions/tumors (Rathke cleft cyst and craniopharyngioma), and inflammatory processes (hypophysitis). Although the percentages differ from one practice to another, pituitary adenomas constitute the vast majority (at least 85%) of all sellar region masses, followed by craniopharyngioma (3%), Rathke cleft cyst (2%), sellar region meningioma (1%), and metastases (0.5%); other lesions such as hypophysitis, pituicytoma, spindle cell oncocytoma, and granular cell tumor of neurohypophysis are rare.
While the predominance of pituitary adenomas in the sellar region might suggest that pituitary pathology is relatively easy compared to neuropathology from other anatomic sites, in fact when the less common lesions occur, it is almost always the pathologist who makes the distinction, since clinically and on neuroimaging many of the other, less common sellar region masses mimic clinically nonfunctioning pituitary adenomas. Another challenge is that the biopsy materials may be small in size and can include fragments of normal anterior or posterior gland, which should not be mistaken for a neoplasm.
Thus, a review of normal histology of the gland is helpful for any surgical or autopsy pathologist who studies pituitary region tissues. A brief explanation of the very different embryologic origins of the two regions helps to explain not only their very different appearance and immunohistochemistry (IHC) profile, but also the occurrence of sellar region lesions such as Rathke cleft cyst and adamantinomatous craniopharyngioma, among others.
The embryologic origin of the anterior lobe (pars distalis, adenohypophysis) is from the Rathke pouch, a tube of tissue which invaginates from the fetal oral (stomodeal) ectoderm anterior to the roof of the primitive mouth, which forms by the fourth to fifth week of human gestation, migrates dorsally toward the base of the brain, and comes in contact with the diencephalon.
Six distinct major hormone-secreting cell types appear during anterior pituitary gland fetal development: the corticotroph (adrenocorticotropic hormone–secreting, ACTH), somatotroph (growth hormone–secreting, GH), lactotroph (prolactin-secreting, PRL), mammosomatotroph parent cell for GH and PRL, gonadotroph (follicle-stimulating hormone– and luteinizing hormone–secreting, FSH and LH, in a single cell), and thyrotroph (thytrotropin stimulating hormone, TSH).
The infundibular stalk and posterior gland (neurohypophysis) arise from the infundibular anlage in the floor of the third ventricle. Early in gestation, cells in the infundibular anlage and ependymal and subependymal cells of the fetal third ventricle manifest nuclear immunostaining for thyroid transcription factor-1 (TTF-1). The anterior pituitary cells are TTF-1 negative both in embryologic time periods and adulthood. In contrast, pituicytes of the posterior gland show nuclear TTF-1–positive IHC throughout pituitary development and maturity.
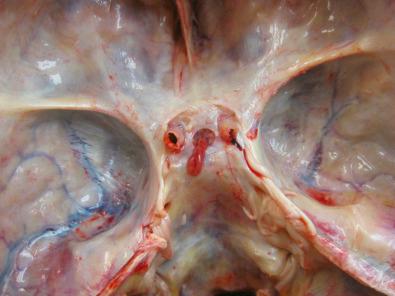
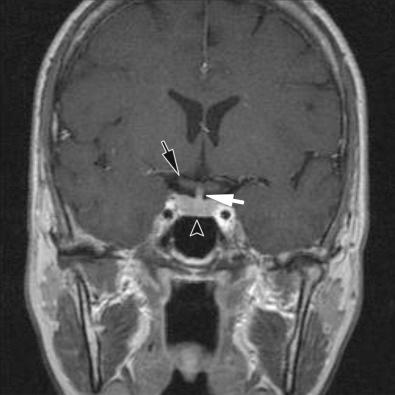
Anatomically, the autopsy pathologist can appreciate the intact diaphragma sella as well as the narrow opening in the diaphragma through which the infundibular stalk protrudes ( Fig. 20.1 ). The diaphragma, a fold in the dura, is often destroyed with sellar region masses such as large pituitary adenomas, but if not, it must be removed to see the sellar contents ( Fig. 20.2 ). The bony sellar region is also eroded with large adenomas. Anatomically, the close relationship between the infundibular stalk, suprasellar optic chiasm, and the gland is best seen on neuroimaging studies of normal gland ( Fig. 20.3 ), as is the proximity of the pituitary gland to the laterally located cavernous sinuses, which contain the cavernous carotid artery that forms a “curve,” allowing two cross-sectional profiles to be seen ( Fig. 20.3 ).
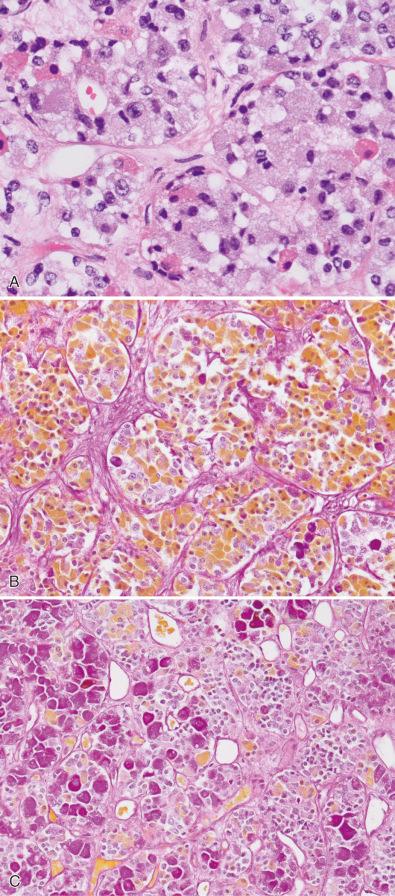
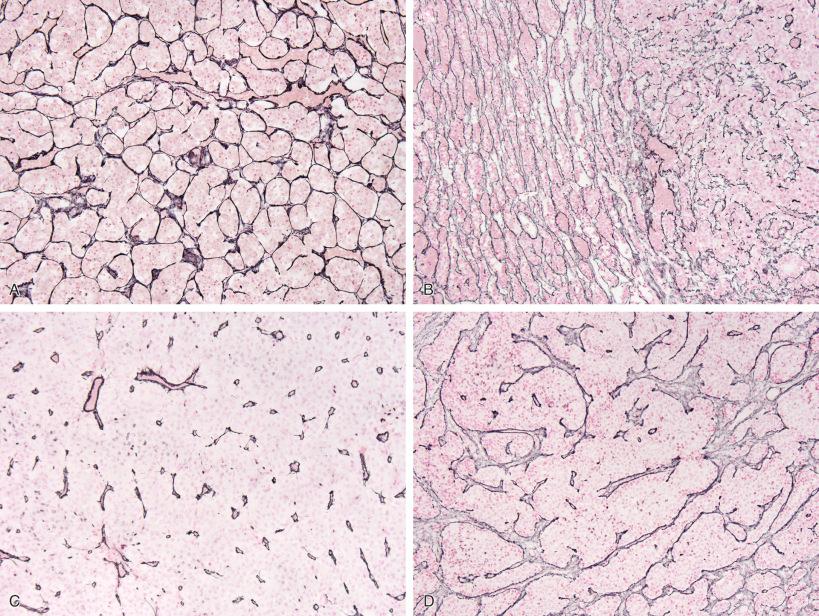
Due to these very different embryologic origins of the normal anterior and posterior glands, they are quite histologically distinct, even during fetal time periods ( Fig. 20.4A–D ). The anterior gland shows rich vascularity, as is typical of endocrine glands in general, but this is especially pronounced during fetal development ( Fig. 20.4A ). Anterior pituitary cells are arranged in clusters (“acini”), although this is more evident in adult gland than in fetal pituitary gland ( Fig. 20.4A and C ). Hematoxylin-eosin (H & E)–stained sections show an assortment of cell types within any one acinus in adult anterior pituitary gland ( Fig. 20.4C ), although this assortment of types is not discernable during much of fetal development ( Fig. 20.4A ).
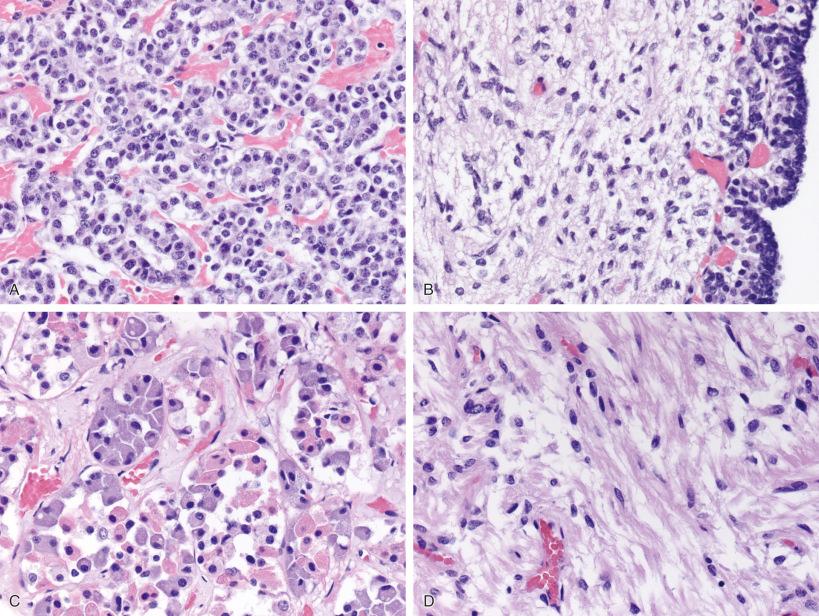
The posterior gland is composed of delicate fibrillary glial cells (“pituicytes”) and unmyelinated axons, manifesting a neuroglial appearance in both fetal ( Fig. 20.4B ) and adult counterparts ( Fig. 20.4D ). Small fragments of posterior gland may be included in both pituitary adenoma surgical resection specimens, and especially Rathke cleft cysts. Thus, it is important for the pathologist to be cognizant of its usual appearance. Although alarming to the surgical pathologist when these fragments of normal posterior gland are found within a surgical specimen, it is important to remember that the posterior gland is a storage site, not synthesis site, for the posterior gland hormones, oxytocin and vasopressin. Swollen axonal processes in normal posterior gland, known as Herring bodies, contain these hormone products as they are being conveyed intraaxonally and appear as rounded eosinophilic structures on routine histology. Interestingly, preoperative presentation of pituitary adenomas with diabetes insipidus is distinctly uncommon and postoperative posterior pituitary gland dysfunction after modern transsphenoidal resection is usually limited to transient diabetes insipidus that resolves after several days. Removal of these small fragments of posterior gland appears to have no long-lasting deleterious clinical effects in most patients.
Although the histochemical stains that illustrate the different tinctorial properties of cells within the anterior gland are less commonly used in daily practice now that IHCs for pituitary hormones and transcription factors are available, they (and routine H & E) allow distinction of acidophils, basophils, and chromophobes in the anterior gland. One example of such a histochemical stain is periodic acid–Schiff (PAS) combined with orange G. This stain can be very useful in showing that hormone cell types in the anterior gland are not equally distributed throughout the anterior lobe. While H & E does show a cell assortment in each acinus in normal gland ( Fig. 20.5A ), this is even better appreciated with PAS-orange G. Growth hormone cells are amongst the most numerous cell types in the anterior pituitary gland at all fetal and adult age time periods and are clustered in the lateral portions (lateral wings) of the gland ( Fig. 20.5B ). Prolactin hormone cells are mostly chromophobic but also predominate in the lateral wings. In contrast, the median areas (mucoid wedge) contain large numbers of basophils, which are predominantly corticotrophs, and to a lesser extent, thyrotroph cells ( Fig. 20.5C ). The single cell that produces both FSH and LH, the gonadotroph, shows more diffuse distribution throughout the anterior gland.
The large numbers of acidophils in the lateral wings ( Fig. 20.5B ) versus basophils in the mucoid wedge ( Fig. 20.5C ) can easily lead the unwary to incorrectly suspect a pituitary adenoma because of the prevalence of one cell type. The relative monotony of cell types in either of these areas is especially problematic on frozen sections or very small biopsies.
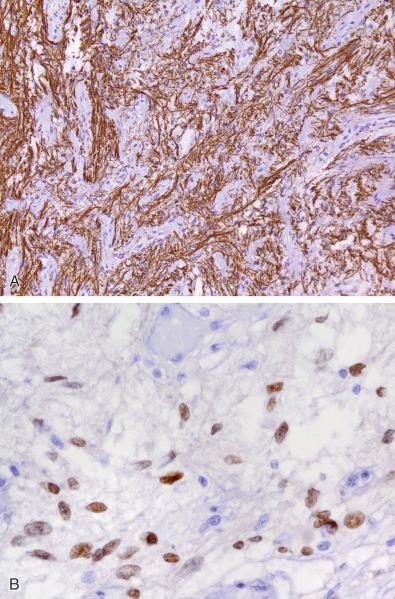
Posterior gland can be easily distinguished from a spindle cell tumor of the sellar region, such as schwannoma, fibrous meningioma, or pituicytoma, by the use of immunostaining for axons, usually with neurofilament IHC ( Fig. 20.6A ). While TTF-1 nuclear immunostaining would not be found in schwannoma or fibrous meningioma, it is present in several neoplasms of the posterior gland to be discussed later in the chapter (pituicytoma, spindle cell oncocytoma, and granular cell tumor of the neurohypophysis) and the normal adult posterior gland ( Fig. 20.6B ). Normal anterior gland and pituitary adenomas lack TTF-1 immunostaining.
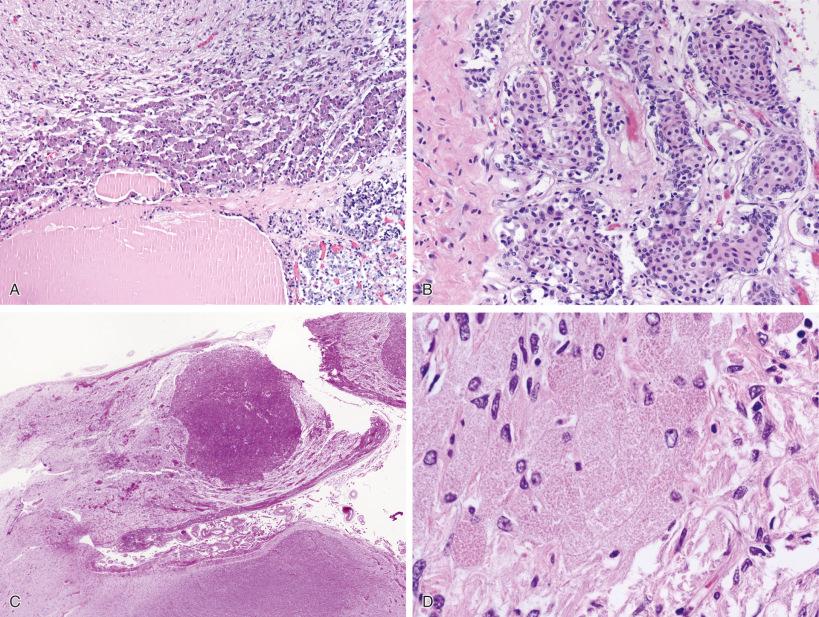
Several normal variations easily detected on H & E stains in both surgical and autopsy specimens can cause concern for pathologic conditions. One example seen with aging is the presence of basophils (ACTH immunohistochemistry positive) extending into the posterior gland near the junction with the anterior gland, so-called “basophil invasion” ( Fig. 20.7A ). The infundibular stalk is also composed of delicate fibrillary glial cells and is quite vascular. Another aging-associated finding can be seen in the thin lip of the anterior gland that extends rostrally along the infundibular stalk, the pars distalis. FSH/LH immunoreactive cells predominate in the pars distalis, and squamous metaplasia may be encountered in these cells ( Fig. 20.7B ). These should not be mistaken for metastatic squamous carcinoma or any other type of disease-related process. Much less common is the presence of an incidental granular cell tumor in the infundibular stalk ( Fig. 20.7C and D ).
Removal of the pituitary gland should be routine in any autopsy in which the cranial contents are examined. There is no need to further section the gland before formalin fixation.
For surgical specimens, the fragmentation and small size of the tissues usually sent to the pathology department leads to the inability to discern any details of invasion, and assessment of margins is not applicable for this tumor type.
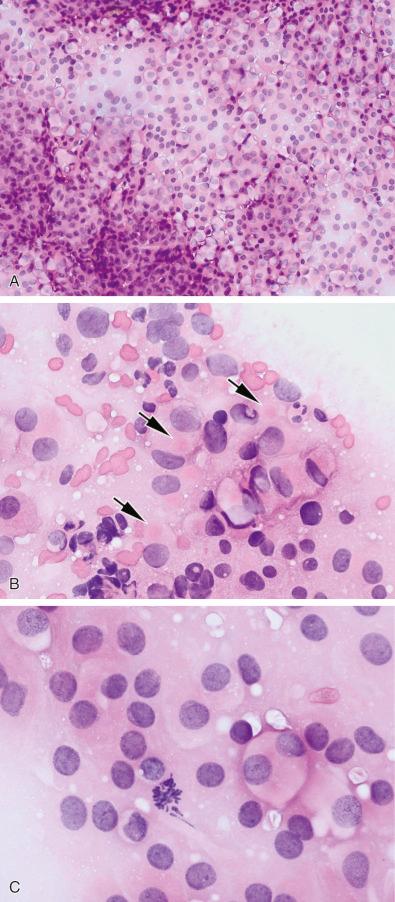
If an intraoperative consult is requested, touch preparation can be an excellent technique that allows the pathologist to appreciate exfoliation/shedding of adenoma cells onto a slide. Unlike normal gland where the intact acinar pattern usually results in a paucity of cells on touch preparation, in an adenoma where the acinar pattern is destroyed usually the number of cells shed onto the slide is directly proportional to the size of the tissue utilized for the preparation ( Fig. 20.8A ). An important caveat, however, is that if the neurosurgeon has extensively sectioned the gland intraoperatively while searching for a microadenoma, the normal acinar pattern will be destroyed and the number of shed cells may be increased. Conversely, if the adenoma manifests excessive fibrosis, a paucity of cells may be exfoliated. The second piece of information that a touch preparation provides is the ability to assess the monotony or variability of the exfoliated cells. Specifically, an H & E–stained touch preparation reveals the tinctorial properties of cellular cytoplasm, which should be relatively or completely uniform in an adenoma. The use of touch preparation further allows preservation of the specimen for permanent sectioning without exhausting the tissue; the latter is critical in very small adenomas. Subtyping clearly requires immunohistochemistry on permanent sections, but occasionally the very abundant amphophilic cytoplasm hints at gonadotroph adenoma ( Fig. 20.8A ) or very faint intracytoplasmic eosinophilic balls can be detected, as is seen in sparsely granulated growth hormone adenoma ( Fig. 20.8B ). Rarely, a mitotic figure can be found, but a mitotic figure on touch preparation is not required for diagnosis ( Fig. 20.8C ).
If the touch preparation is not fully informative, a frozen section can be performed; again, an H & E preparation is advised. Crush mechanical artifacts can be a problem, and recognition of preserved acinar pattern requires optimal staining. No special stains are practical at the time of frozen section. The specimen can then be placed in formalin as a fixative, with a small fragment set aside in glutaraldehyde for potential electron microscopy (EM) if desired. The use of the latter has become so infrequent that many practices no longer save a fragment for EM. EM is also possible, and often successful, on small tissue fragments fixed in routine formalin.
While PAS-orange G (or similar histochemical stains) is no longer routinely used in diagnostic practice, another histochemical stain remains a staple, namely reticulin. In the normal anterior gland, the intact acinar pattern is best outlined by a reticulin stain, which highlights basement membrane and reveals relatively uniform-sized clusters of cells ( Fig. 20.9A ).
Reticulin can therefore be useful in the workup of pituitary adenoma, since the basic definition of adenoma rests on the fact that there is disruption and loss of normal acinar architecture. Reticulin is an especially helpful tool in distinguishing small adenomas from normal gland ( Fig. 20.9B ). In small adenomas, fractured fragments of basement membrane may remain within the adenoma, but the normal pattern is nevertheless interrupted ( Fig. 20.9B ). In large adenomas, the acinar pattern is often completely lost, with reticulin staining only outlining the vasculature ( Fig. 20.9C ). Occasionally, a large adenoma, usually a gonadotroph adenoma, may manifest a “macronodular” pattern ( Fig. 20.9D ), although once again this architectural pattern is still clearly abnormal.
Even if the adenoma is quite large and easily recognizable as such on H & E stain, the use of reticulin may reveal normal anterior pituitary gland at the perimeter of resected adenoma fragments ( Fig. 20.10A ), sometimes appearing considerably compressed. Thus, the use of a reticulin can warn the pathologist that normal gland is included in the specimen and aid in the interpretation of IHC stains from adjacent levels. Immunostaining for several different hormone types in these junctional areas should not be misinterpreted as positivity in the adenoma itself (i.e., evidence for a plurihormonal adenoma). It should be noted that often a few normal, entrapped adenohypophyseal cells extend into the adenoma itself ( Fig. 20.10B–D ).
![Fig. 20.10, Reticulin staining is useful even if the adenoma is easily recognizable on H & E, since it may reveal normal anterior pituitary gland at the perimeter of resected adenoma fragments (A). Immunohistochemical (IHC) reactivity for several hormone types in these normal areas should not be misinterpreted as positivity in the adenoma itself (i.e., evidence for a plurihormonal adenoma), since often a few normal entrapped cells extend into the adenoma itself (see Fig. 20.3B–D : IHC for prolactin [B], growth hormone [C], and follicle stimulated hormone [D]). Fig. 20.10, Reticulin staining is useful even if the adenoma is easily recognizable on H & E, since it may reveal normal anterior pituitary gland at the perimeter of resected adenoma fragments (A). Immunohistochemical (IHC) reactivity for several hormone types in these normal areas should not be misinterpreted as positivity in the adenoma itself (i.e., evidence for a plurihormonal adenoma), since often a few normal entrapped cells extend into the adenoma itself (see Fig. 20.3B–D : IHC for prolactin [B], growth hormone [C], and follicle stimulated hormone [D]).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/HistologicFeaturesofPituitaryAdenomasandSellarRegionMasses/9_3s20B9780323449410000205.jpg)
Immunostains for anterior pituitary hormones are available in most laboratories that see moderate to large numbers of pituitary adenomas each year and are in reference laboratories for those departments that cannot keep these in house. Commercially available immunostains are directed against the corticotroph (ACTH secreting), somatotroph (GH secreting), lactotroph (PRL secreting), mammosomatotroph (GH and PRL in a single cell), gonadotroph (FSH and LH secreting, in a single cell), and TSH cell types. In addition, immunostains are available for the alpha subunit (α-SU) of gonadotroph hormones and TSH.
With better quality immunostains, few pituitary adenomas are truly hormone negative. Commercially available antibodies directed against transcription factors include (1) PIT1 for PRL, GH, and/or TSH adenomas, including plurihormonal PIT1-positive adenomas; and (2) SF1 for gonadotroph adenomas (i.e., FSH and/or LH+ adenomas). The third transcription factor, TPIT (T-box transcription factor) for corticotroph adenomas, has been used in research settings, but is not widely available. These transcription factors have provided insights into the origin of clinically silent/clinically nonfunctioning and hormone-negative pituitary adenomas. Only the adenomas that are negative for specific anterior pituitary hormones and transcription factors are designated “null cell adenomas” in modern series. The use of transcription factors further allows classification of adenoma by transcription factor family ( Table 20.1 ).
| PIT1 (Pituitary Transcription Factor-1) |
| Densely granulated growth hormone (somatotroph) adenoma Sparsely granulated growth hormone (somatotroph) adenoma |
| Mixed somatotroph-lactotroph adenoma Mammosomatotroph adenoma |
| Sparsely granulated prolactin secreting (lactotroph) adenoma Densely granulated prolactin secreting (lactotroph) adenoma Acidophil stem cell adenoma |
| Thyrotroph (TSH) adenoma |
| Plurihormonal PIT1-positive adenoma (previously called silent subtype 3 adenoma; all with PIT1, variable GH/PRL/TSH) |
| SF1 (Steroidogenic Factor) |
| Gonadotroph adenoma, with or without IHC(+) for FSH, LH, alpha subunit a |
| TPIT |
| Densely granulated ACTH (corticotroph) adenoma Sparsely granulated ACTH (corticotroph) adenoma Crooke cell adenoma Clinically silent ACTH adenoma (either densely granulated [i.e., subtype I], or sparsely granulated [i.e., subtype II]) |
| Transcription Factor Negative/Unknown |
| Null cell adenoma b |
| Plurihormonal Adenomas |
| Uncertain lineage (when excluding PIT1–driven GH+ PRL, +/− TSH, and/or gonadotrophic hormone adenomas—that is, GH + ACTH adenomas would be truly “plurihormonal”) |
a Presence of SF1 defines gonadotroph adenoma even if hormone negative.
b Increasingly defined as pituitary hormones IHC negative and SF1, PIT1, ACTH/TPIT IHC negative (TPIT IHC not as widely available).
Additional immunostains can be useful in select instances. Normal anterior gland shows variable immunostaining for keratins AE1/AE3 (mixed low- and high-weight keratins) and CAM5.2 (keratins 8/18). E-cadherin immunoreactivity is present. Synaptophysin and chromogranin immunostains are positive throughout the anterior gland, reflecting the presence of granules containing hormone product. Synaptophysin is diffusely positive in adenomas, no matter how sparse the hormone-containing granules. Chromogranin expression is even more specific, but is not positive in all adenomas, varying somewhat with antibody source and/or antigen retrieval methods. Immunostains for GFAP and S-100 highlight the folliculostellate cells. Posterior gland is also synaptophysin positive. Neurofilament IHC further aids the identification of posterior gland (neurohypophysis) because of the rich axonal content.
Immunostaining for keratins is also a necessity in the workup of pituitary adenomas since it serves to identify the characteristic fibrous bodies in sparsely granulated somatotroph adenomas, which behave more aggressively than densely granulated somatotroph adenomas (see section “Specific Subtypes of Pituitary Adenomas” on sparsely granulated growth hormone adenomas). Table 20.2 provides a list of histochemical and general immunohistochemical features of pituitary adenomas.
| Reticulin: disruption of acinar pattern in all adenomas, expansion in rare hyperplasia |
| Periodic acid–Schiff (PAS): strongly positive in densely granulated ACTH adenomas |
| Cytokeratin 7: negative or only rare positive cells in almost all adenomas |
| Cytokeratin 20: positive in most densely granulated ACTH adenomas, negative in sparsely granulated GH adenomas; positive in all sparsely granulated but not in any densely granulated GH adenomas; positive in nontumorous Crooke cells in patients with hypercortisolism of any etiology |
| CAM5.2 (keratin 8/18): extensive and diffuse in all densely granulated ACTH adenomas, most densely granulated GH adenomas in perinuclear pattern, all sparsely granulated GH adenomas in a fibrous body pattern, most mixed GH/PRL adenomas, half of all prolactinomas, gonadotroph adenomas, null cell adenomas |
| Synaptophysin: positive in normal anterior and posterior gland and all adenomas |
| Neurofilament: positive in axons in normal posterior pituitary gland |
| Glial fibrillary acidic protein (GFAP), S-100: focally present in posterior gland; identifies nonhormone containing folliculostellate cells in anterior pituitary gland |
| Chromogranin A: characteristic of gonadotroph adenomas; lacking in both PRL and ACTH functioning adenomas; poorly expressed in GH and GH/PRL functioning adenomas |
| Estrogen receptor: all prolactin-positive adenomas (sparsely, densely granulated lactotroph adenomas, acidophil stem cell, mixed GH/PRL, mammosomatotroph adenomas); gonadotroph adenomas (due to fixation issues can be focal and weakly positive) |
TTF-1 expression is found in pituicytoma, spindle cell oncocytoma, and granular cell tumor of the neurohypophysis, but is negative in adenomas, meningiomas, and craniopharyngiomas. GFAP, S-100, and epithelial membrane antigen (EMA) stains are often also utilized for the diagnosis and subclassification of TTF-1 immunoreactive tumors. IHC for mitochondria (antimitochondrial antibody) can be helpful in confirming rare mitochondrion-rich adenoma types, such as acidophil stem cell adenoma (see upcoming discussion). Although of great historical relevance in originally defining subtypes of adenoma, EM is now being used much less often. Nevertheless, the prior edition of this text can be utilized as a reference for EM images of sellar region masses, including adenomas.
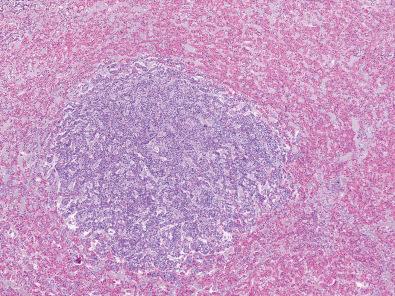
Pituitary adenomas predominantly affect adults although they also occasionally arise in children. The majority of clinically apparent cases occur in persons in the third through sixth decades, although it is worth noting that adenomas, usually minute in size, are among the most frequent incidental tumors at autopsy ( Fig. 20.11 ). Overall, there is no gender predilection, but corticotroph adenomas are found more frequently in women. Symptoms may also differ or be influenced by gender. For example, a prolactinoma (also known as lactotroph adenoma) is more likely to present with hormonally related symptoms such as amenorrhea and/or galactorrhea in premenopausal women than in postmenopausal women or men. A gonadotroph adenoma usually is clinically silent, but even small amounts of FSH or LH that reach the serum may be mistaken for normal physiologic end-organ failure in a postmenopausal woman, as opposed to an FSH/LH–producing adenoma. Finally, a corticotroph adenoma occurring around the time of puberty could produce facial plethora and weight gain that is otherwise clinically overlapping with normal pubescent changes.
The symptomatology engendered by pituitary adenomas is highly variable and may be complex. Basically, a large adenoma may cause hypersecretory endocrinopathy (usually no more than one to two hormones are sufficiently elevated to produce symptoms) or hypopituitarism due to compression and dysfunction of the normal anterior pituitary gland. As noted, significant disruption of posterior gland function, such as diabetes insipidus, is rare as a clinical preoperative symptom in pituitary adenomas of any size. Finally, a large pituitary adenoma may cause pressure on adjacent sellar structures—that is, “mass effect,” such as headache (due to traction on the dura), visual deficits (due to compression of the optic chiasm, the most classic of which is midline chiasmal compression with resultant bitemporal hemianopsia), or compression of cavernous sinus structures (most notably cranial nerves III, IV, and VI). Invasion of the cavernous carotid artery almost never occurs, but tumor growth into one or both cavernous sinuses is one of the major obstacles to neurosurgeons achieving gross total excision. Adenomas may grow straight “up” and compress optic chiasm and even third ventricle, anteriorly/inferiorly into the sphenoid sinuses (where they may be mistaken for a primary nasopharyngeal tumor), or laterally into the cavernous sinuses. Bone invasion of the sellar floor and destruction of the diaphragma sella as well as dural invasion are all frequent with large adenomas and can also occur with some other nonadenomatous sellar region masses. As discussed later in the chapter, metastasis of any sellar region tumor beyond the sella is exceedingly rare. The extent of growth of sellar region masses is best explained by the Hardy staging system, as explained later in the chapter.
Endocrinologic hypersecretion syndromes vary based on which hormone is secreted by the adenoma. Usually a single hormone is hypersecreted, with the exception of fairly frequent combinations of growth hormone and prolactin by the same tumor. What always needs to be considered, however, is the possibility that hyperprolactinemia in the patient with a large adenoma is a consequence of compression of the infundibular stalk and “stalk interruption.” Known as “stalk effect” clinically, it results from a physical block of dopamine, the major agonist of prolactin secretion produced in the nearby hypothalamus, from reaching the pituitary gland, resulting in loss of secretory inhibition and hyperprolactinemia.
Excess GH production by an adenoma leads to gigantism and/or acromegaly, the latter being the sole symptom after the epiphyseal bony plates close. Symptoms of acromegaly include acral enlargement, soft tissue swelling, hypertension, hyperglycemia, and sleep apnea. Excess prolactin production can yield amenorrhea and galactorrhea in premenopausal women and more subtle sexual dysfunction and infertility in males. Prolactinomas in older persons usually produce subtle hypogonadism, and, usually, symptoms of mass effect predominate. Excess secretion of ACTH causes Cushing disease (elevated cortisol levels due to a pituitary adenoma), the common signs and symptoms of which are central obesity, skin striae, hyperglycemia, osteoporosis, and hirsutism. Gonadotroph adenomas are most often clinically silent since the amounts of adenoma hormone product that reach the serum are usually low. The very rare thyrotroph cell adenoma can cause hyperthyroidism, although some may arise in a setting of hypothyroidism.
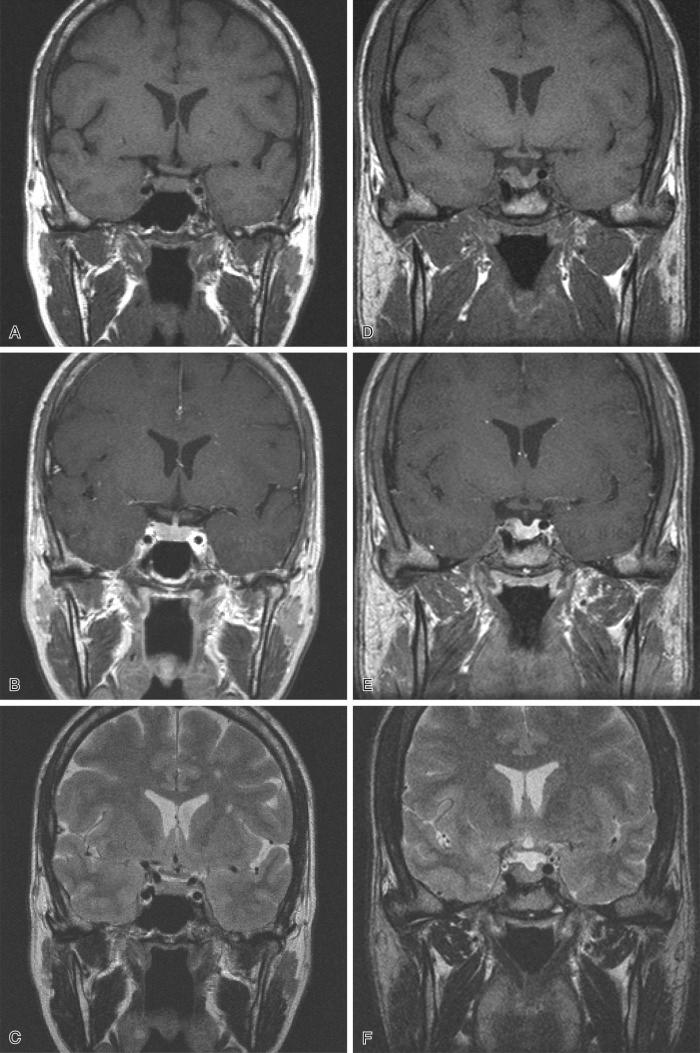
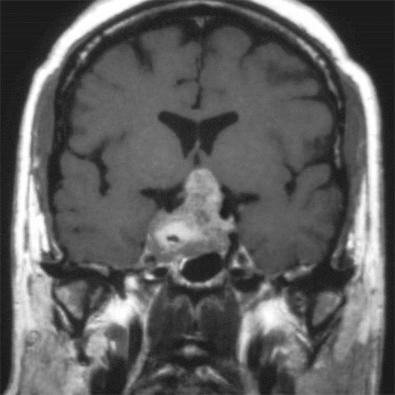
Adenomas are routinely diagnosed on magnetic resonance imaging (MRI) studies rather than plain skull films or computed tomography (CT) scans and, depending on the sequences utilized (T1 weighted, T1 weighted with gadolinium, T2 weighted), show varying details ( Fig. 20.12 ). Suffice it to say that detection of microadenomas requires knowledge about the appearance of normal gland on these sequences; these are illustrated for normal versus gland containing a very small adenoma in Fig. 20.12 . Larger adenomas are obvious masses ( Fig. 20.13 ); neuroimaging studies allow the pathologist to appreciate the extent of growth of some macroadenomas, particularly into the adjacent structures ( Fig. 20.13 ).
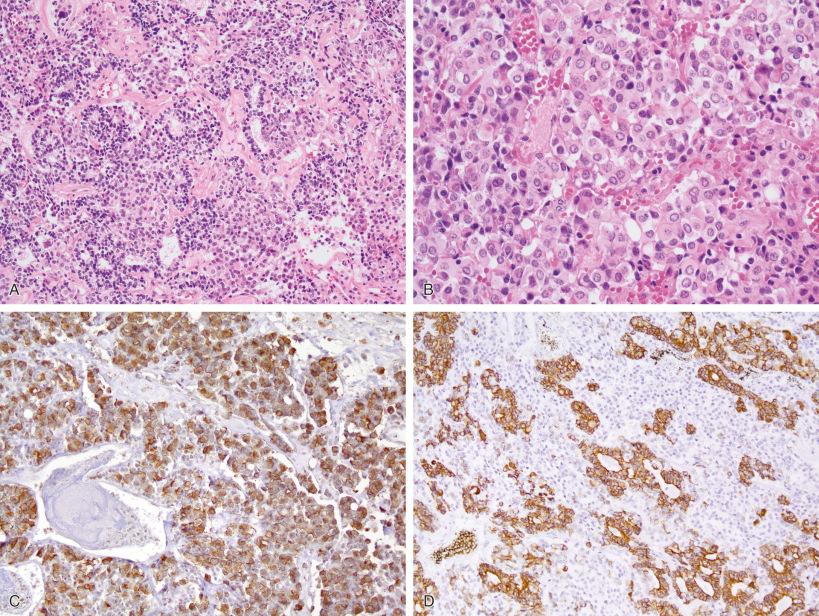
The great majority of pituitary adenomas arise in a sporadic manner and only a minority are part of hereditary or familial syndromes (see Chapter 22 ). Hereditary conditions associated with pituitary adenomas include the multiple endocrine neoplasia (MEN) syndromes MEN1 and MEN4, the Carney complex, the McCune-Albright syndrome, the hereditary pheochromocytoma/paraganglioma syndrome (related to succinate dehydrogenase [SDH] genes), the familial isolated pituitary adenoma (FIPA) syndrome, and the X-linked acrogigantism (XLAG) associated with GPR101 microduplication. Most tumors associated with these familial syndromes are somatotroph or lactotroph adenomas; rarely are they nonfunctioning adenomas. In addition, the rare primitive or embryonal-like pituitary blastoma is part of the DICER1 syndrome ( Fig. 20.14 ). These tumors occur in infants or very young children and often contain a fetal appearing tubular component ( Fig. 20.14A ; compare with Fig. 20.4A ), as well as more solid areas ( Fig. 20.14B ); they are invariably ACTH immunoreactive tumors ( Fig. 20.14C and D ).
The primary genetic defect in the majority of sporadic adenomas remains unknown. The most common genetic alteration in sporadic tumors is somatic mutations in the GNAS gene found in 40% of somatotroph adenomas resulting in upregulation of the cyclic adenosine monophosphate (cAMP) pathway. The GNAS gene mutation is rarely identified in other adenoma subtypes, occurring in only 10% of clinically nonfunctioning pituitary adenomas and in 5% of corticotroph adenomas. Nonsyndromic gigantism has been related to inactivating germline mutations on the aryl hydrocarbon receptor-interacting protein (AIP) gene and more recently to germline or somatic duplication of the GPR101 gene leading to familial XLAG.
Mutations in the USP8 (encoding ubiquitin-specific protease 8) gene, leading to an upregulated epidermal growth factor receptor (EGFR) pathway, have been identified in about 36% to 62% of sporadic corticotroph adenomas.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here