Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Several hereditary disorders of the skin primarily manifest as disorders of the dermis. Clinical manifestations range from laxity of skin to infiltrated papules and from rigidity to thinning of the dermis. Disorders of mucopolysaccharides are reviewed in Chapter 24 .
Ehlers–Danlos syndrome (EDS) consists of a group of six inherited disorders of collagen characterized by increased cutaneous elasticity, hyperextensibility of the joints, and fragility of the skin, sometimes with the formation of pseudotumors and large gaping scars. The 1997 Villefranche classification was refined in the 2017 International Classification ( Table 6.1 ). For all subtypes other than the hypermobile type, confirmation of subtype relies on molecular genetic analysis.
| Type | Mutations (Protein Products) | Skin Findings | Joint Changes | Inheritance | Other Comments |
|---|---|---|---|---|---|
|
COL5A1, COL5A2 (type V collagen) | Hyperextensibility, bruising, velvety skin, widened atrophic scars, molluscoid pseudotumors, spheroids | Hypermobility and its complications, joint dislocations | AD (usually) |
|
| CLASSICAL VARIANT | |||||
| EDS/OI overlap | COL1A1 (type I collagen) | Classic EDS features | AD |
|
|
|
TNXB (tenascin X) | Hyperextensibility, marked hypermobility, severe bruising, velvety skin, no scarring tendency | Hypermobility | AR |
|
|
COL1A2 (type I collagen alpha 2 chain) | Classic EDS features | AR |
|
|
|
COL3A1 (type III collagen) | Thin, translucent skin; bruising; early varicosities; acrogeria | Small joint hypermobility | AD |
|
|
Unknown; some may have COL5A1 mutations | Mild hyperextensibility, scarring, textural change | Hypermobility, chronic joint pain, recurrent dislocations | AD |
|
|
Exon 6 deletion of COL1A1 or COL1A2 | Hyperextensible, soft skin with or without abnormal scarring | Marked hypermobility with recurrent subluxations | AD |
|
|
ADAMTS2 (type I collagen N-peptidase) | Severe fragility Sagging, redundant skin |
AR |
|
|
|
PLOD1 (lysyl hydroxylase) FKBP14 (FKBP prolyl isomerase 14 or FKBP22) | Soft, hyperextensible, bruising, atrophic scars | Hypermobility | AR |
|
|
ZNF469 (zinc finger protein 469) PRDM5 (PR domain zinc finger protein 5) | Skin hypermobility | Joint hypermobility | AR |
|
|
SLC39A13 (ZIP13 zinc transporter) β4GALT7 or β3GalT6 (galactosyltransferase I or II, key enzymes in glycosaminoglycan synthesis) |
Soft, hyperextensible, bruising, atrophic scars | Hypermobility | AR |
|
|
CHST14 (dermatan 4-O-sulfotransferase) or DSE (dermatan sulfate epimerase) | Fragile, hyperextensible skin with atrophic scars and delayed wound healing | Hypermobility | AR |
|
|
COL12A1 (type 12 collagen alpha 1 chain) | AD or AR |
|
||
|
C1R or C1S (complement components C1r and C1s) | Can have classic EDS features | Can have hypermobility | AD |
|
Most forms of EDS are linked to mutations in genes encoding fibrillar collagens, enzymes involved in posttranslational modification of these proteins, or extracellular matrix proteins. Mutations in type V and type III collagen cause classic and vascular EDS, respectively, whereas mutations involving the processing of type I collagen are involved in the arthrochalasis and dermatosparaxis types and of collagen crosslinking in the kyphoscoliotic type of EDS. In addition to the clinical classification of EDS, a grouping according to pathogenetic mechanism has also been proposed ( Table 6.2 ). The underlying cause for most individuals with the hypermobile type remains unknown.
| Group A: Disorders of Collagen Primary Structure and Collagen Processing |
| Classical type: COL5A1, COL5A2 , rarely COL1A1 (selected genotypes) Cardiac-valvular type: COL1A2 Vascular type: COL3A1 , rarely COL1A1 (selected genotypes) Arthrochalasia type: COL1A1, COL1A2 Dermatosparaxis type: ADAMTS2 |
| Group B: Disorders of Collagen Folding and Crosslinking |
| Kyphoscoliotic type: PLOD1, FKBP14 |
| Group C: Disorders of Structure and Function of the Interface Between the ECM and Muscle ("Myomatrix") |
| Classic-like type: TNXB Myopathic type: COL12A1 |
| Group D: Disorders of Glycosaminoglycan Biosynthesis |
| Spondylodysplastic type: B4GALT7, B3GALT6 Musculocontractural type: CHST14, DSE |
| Group E: Disorders of Complement Pathway |
| Periodontal type: C1R, C1S |
| Group F: Disorders of Intracellular Processes |
| Spondylodysplastic type: SLC39A13 Brittle cornea syndrome: ZNF469, PRDM5 |
The prevalence of EDS, including the mild forms, may be as high as 1:5000 individuals. The spectrum of severity of EDS ranges from almost imperceptible findings to severe, debilitating disease. The most common type with skin manifestations is the classical type ( Box 6.1 ). The vascular type tends to be most devastating. The kyphoscoliosis, arthrochalasis, and dermatosparaxis types are considerably less common than other forms. The effect on quality of life can be considerable, and three-quarters of affected individuals report fatigue that may be affected by pain, sleep disturbance, difficulty with concentration, and diminished social functioning.
Major criteria
Skin hyperextensibility and atrophic scarring
Generalized joint hypermobility
Minor criteria
Soft, doughy skin
Easy bruisability
Skin fragility with trauma
Molluscoid pseudotumors
Subcutaneous spheroids
Hernia
Epicanthal folds (especially during childhood)
Complications of joint hypermobility (pain, flexible pes planus, sprains, luxation/subluxation
Family history of a first-degree relative with Ehlers–Danlos syndrome (EDS) per clinical criteria
Minimal criteria to suggest classical EDS: Skin hyperextensibility and atrophic scarring plus either the other major criterion (joint hypermobility) or at least three minor criteria
Molecular testing is necessary to confirm the diagnosis
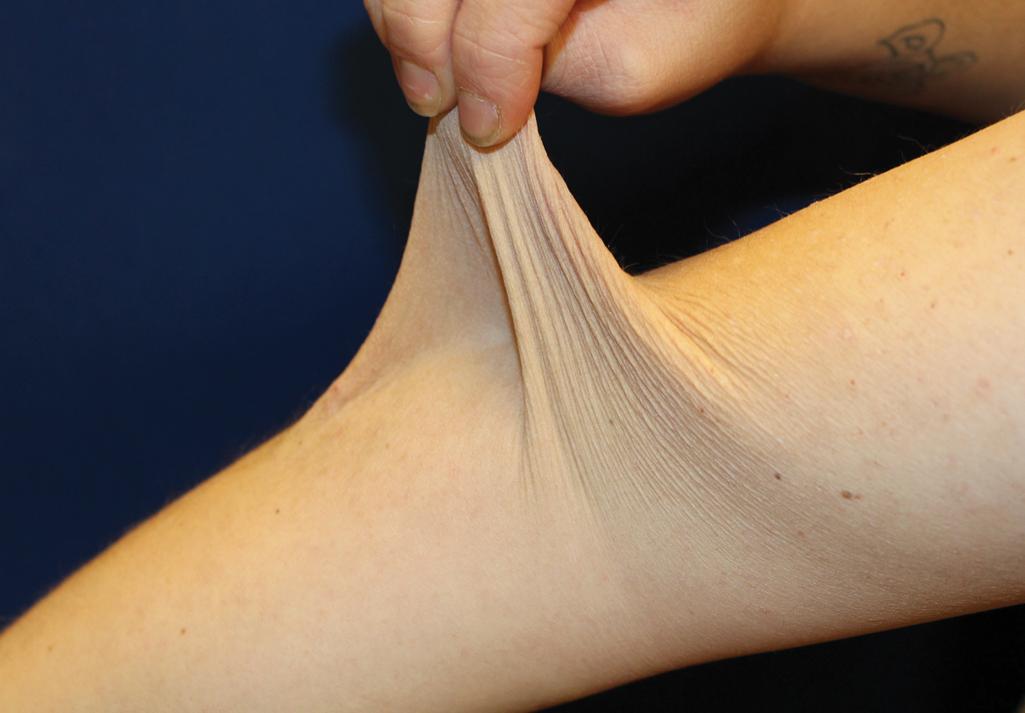
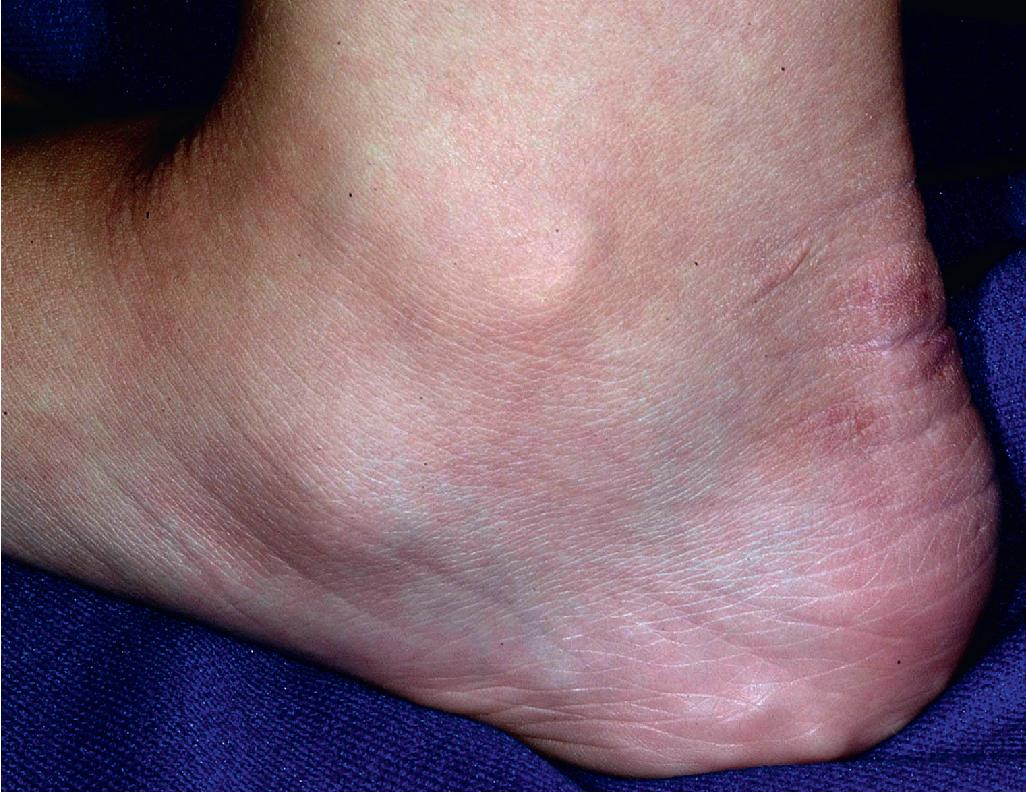
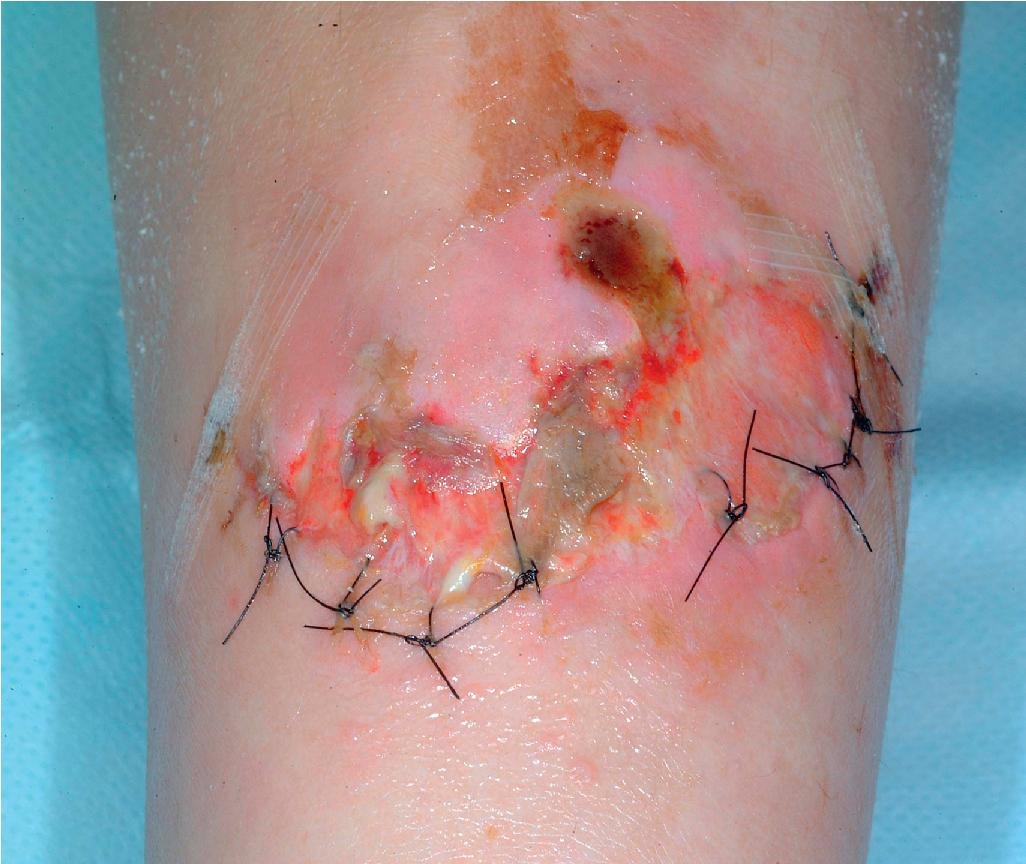
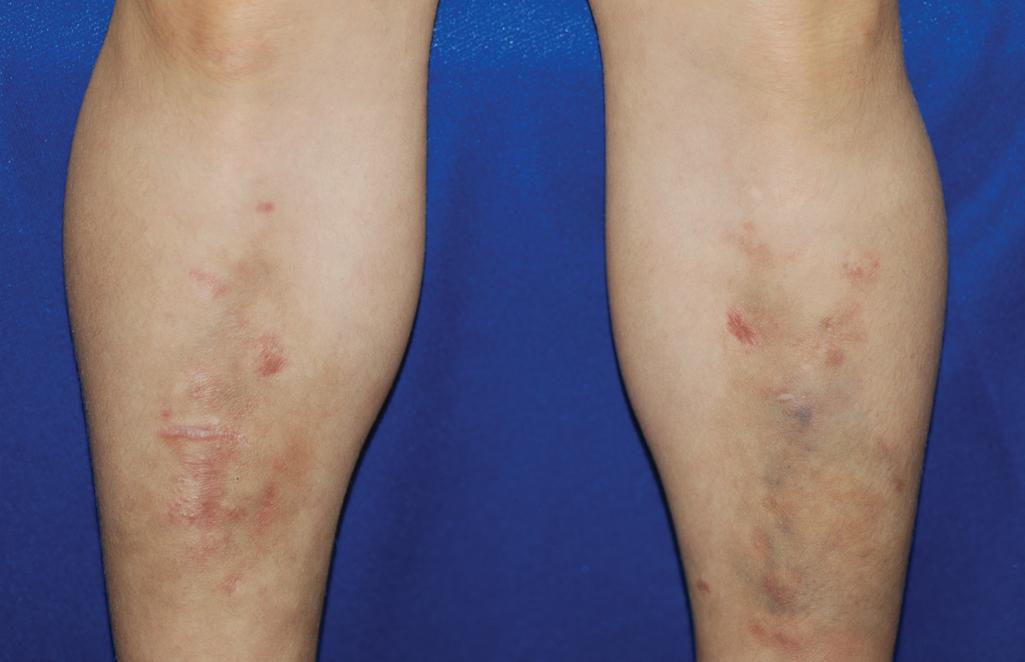
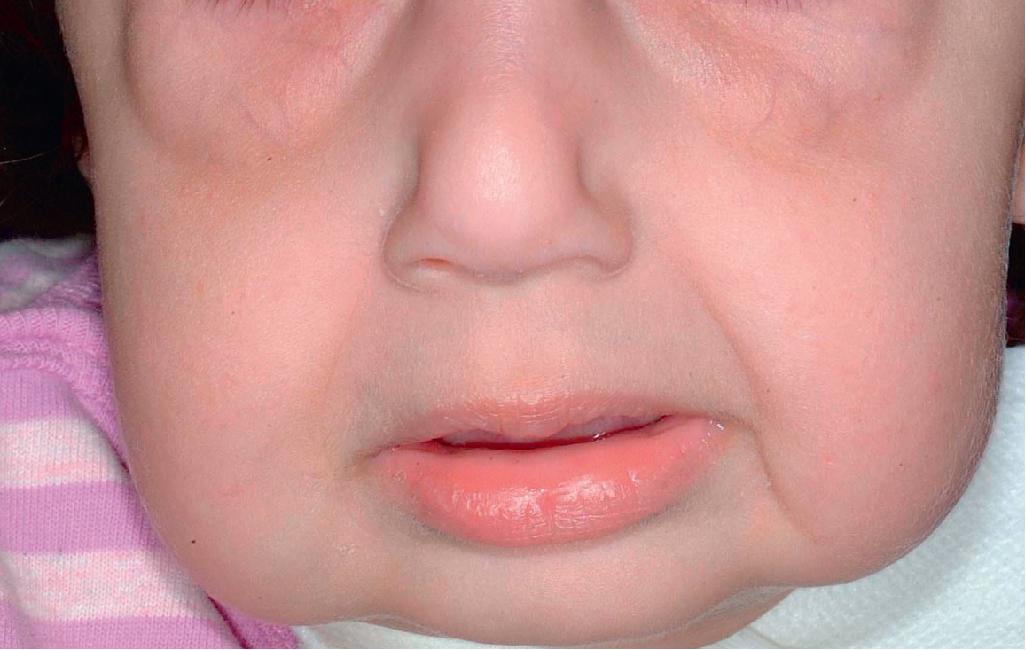
The skin of individuals with the classical type (cEDS) is velvety, soft, and has a doughy consistency. After being stretched, it returns to its normal position as soon as released, in contrast to the lax skin of cutis laxa. Skin hyperextensibility ( Fig. 6.1 ) should be tested at a site not subjected to mechanical forces or scarring, such as the volar surface of the forearm. In contrast to 10% of apparently normal individuals, approximately 50% of patients with EDS can touch the tip of their nose with their tongue (Gorlin sign) ( Fig. 6.2 ). In addition, the skin of the hands, feet, and at times the elbows tends to be lax and redundant, thus resulting in a loose-fitting glove- or moccasin-like appearance. A number of patients have pressure-induced herniation of subcutaneous fat on the wrists or on the medial or lateral aspect of the heels, evident when the patient is standing (piezogenic pedal papules) ( Fig. 6.3 ). In addition to abnormal elasticity, the skin of patients with the classical (but not the hypermobile) form of EDS is extremely fragile, and minor trauma may produce gaping “fishmouth” wounds ( Fig. 6.4 ). It has poor tensile strength and cannot hold sutures properly. This leads to dehiscence, poor healing, and the formation of wide, papyraceous, wrinkled hernia-like scars, particularly over areas of trauma (such as the knees, forehead, pretibial areas, and elbows). Blood vessels are fragile, resulting in hematomas and a chronic bruise-like appearance, particularly on the anterior aspect of the lower extremities ( Fig. 6.5 ). The resolution of hematomas is accompanied by fibrosis, which produces soft subcutaneous nodules (molluscoid pseudotumors, especially over joints such as the elbows and fingers) and small calcified subcutaneous nodules, especially on the shins and forearms (spheroids). Facial dysmorphism of the midface and orbital areas and infraorbital creases (epicanthal folds) are common, especially in children.
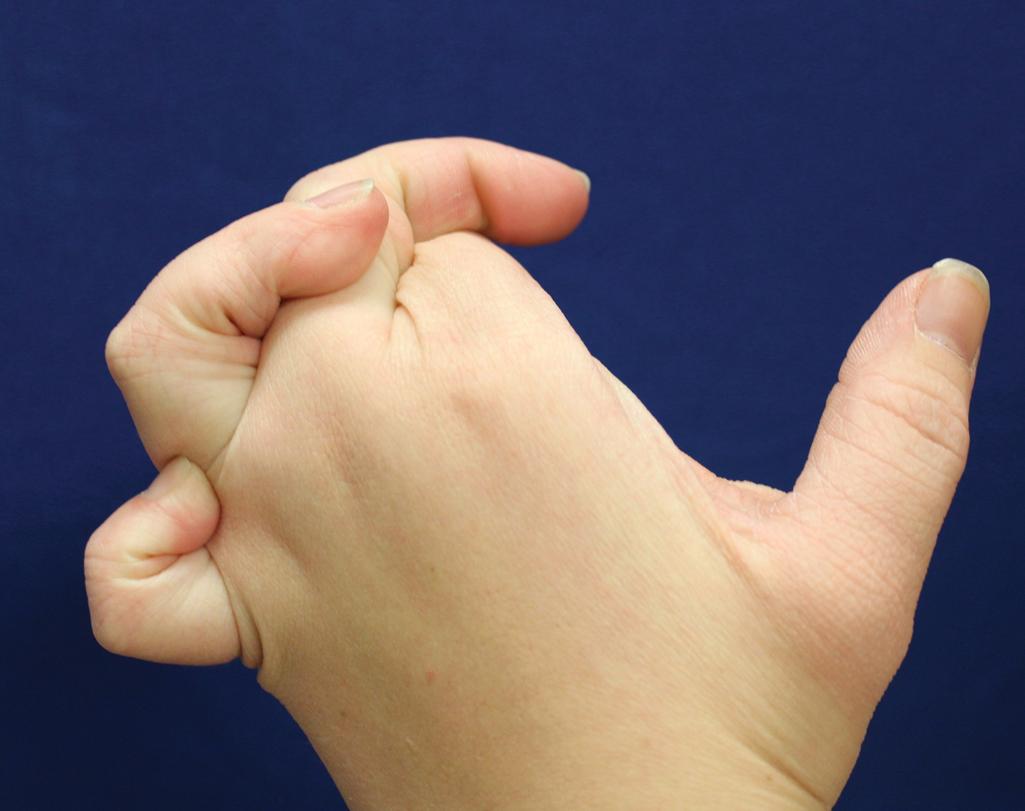
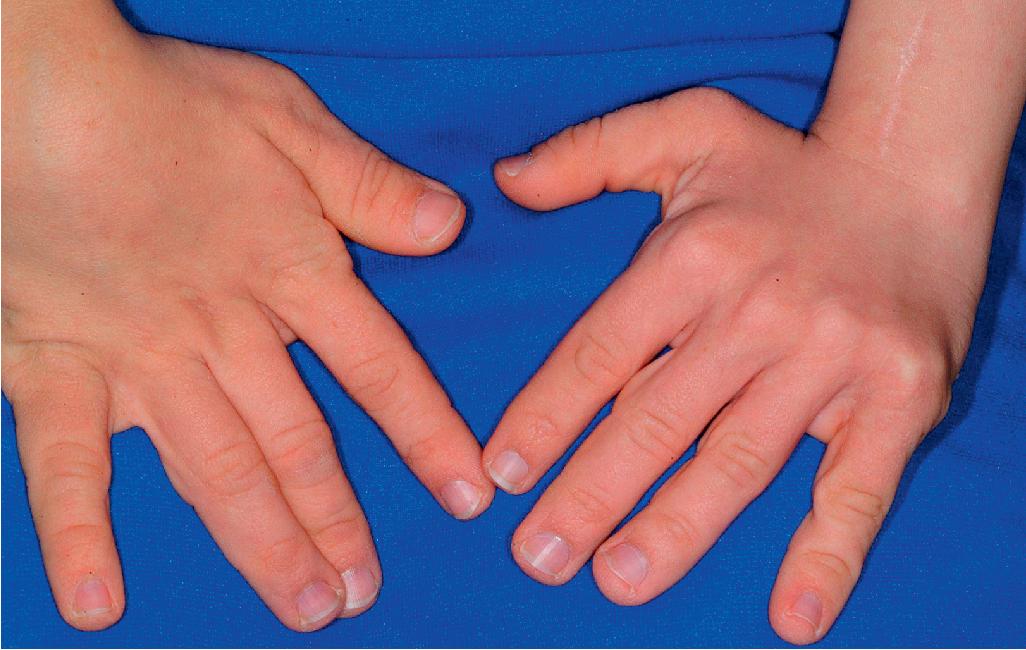
Hyperextensible joints may result in “double-jointed” fingers or subluxation of larger joints ( Fig. 6.6 ). This may occur spontaneously or follow slight trauma. Generalized hypermobility is determined by a score of 5 or higher in the 9-point scale established by Beighton and colleagues ( Fig. 6.8 ; Table 6.3 ). Sprains, dislocations or subluxations, and pes planus occur as complications, and patients complain of chronic joint and limb pain despite normal skeletal radiographs. , Muscle hypotonia and delayed gross motor development have been described. Hiatal hernia, postoperative hernias, and anal prolapse have been noted as manifestations of the tissue hyperextensibility and fragility. Anomalies of the heart and dissecting aortic aneurysms have rarely been described in the classical form, , in contrast to the arterial form. Mitral valve prolapse is a common manifestation, but aortic root dilatation is uncommon; both can be assessed by echocardiography, computed tomography (CT), or magnetic resonance imaging (MRI) examinations.

| Finding | Negative | One Side | Both Sides |
|---|---|---|---|
| Passive flexion of thumb to forearm | 0 | 1 | 2 |
| Passive dorsiflexion of fifth finger >90 degrees | 0 | 1 | 2 |
| Hyperextension of elbows >10 degrees | 0 | 1 | 2 |
| Hyperextension of knees † >10 degrees | 0 | 1 | 2 |
| Hyperextension of knees is done by being able to touch the floor with palms It is 0 if no It is 1 if yes |
0 | 1 | 2 |
† Touching the floor with palms open and knees fully extended.
The diagnosis of classical EDS is primarily clinical. Routine histopathologic examination of skin from patients with EDS is usually normal but may show loose collagen and irregular fibroblasts ; electron microscopy demonstrates disorganized collagen fibers and fibrils with variable cross-sectional diameters. Studies of platelet function and coagulation are usually normal despite a tendency for bruising and increased bleeding, further implicating the defective structural integrity of skin and blood vessels.
The underlying molecular defect for 93% of patients with the classical form of EDS is mutations in the α1 (COL5A1) or α2 (COL5A2) chain of type V collagen, a minor fibrillar collagen that regulates collagen fibril diameter. These mutations typically lead to haploinsufficiency for COL5A1 and are dominant negative for COL5A2 . , , Autosomal recessive type I EDS has been described from homozygous variants in exon 64b of COL5A1, an alternatively spliced isoform that accounts for only 30% of COLA5A1 . Patients with mutations in COL1A1 at a nonglycine site (mutations at glycine sites lead to osteogenesis imperfecta [OI]) may exhibit features of classic EDS alone or with subtle features of OI (blue sclera, relatively short stature, osteopenia, or fractures). Some children with nonglycine substitutions in type I collagen have classic EDS but develop spontaneous arterial rupture during adulthood.
The “classic-like” autosomal recessive form of EDS (clEDS), caused by a deficiency of the extracellular matrix protein tenascin-X, shows typical EDS (hyperextensibility, bruising) but normal wound healing and without atrophic scarring. Serum levels of tenascin-X are undetectable. Heterozygous carriers may exhibit only joint hypermobility. A contiguous gene syndrome caused by a 6p deletion can present with tenascin-X deficiency, as well as congenital adrenal hyperplasia (deletion in CYP21A2 ) with or without ovarian failure (deletion in MSH5 ). ,
The cardiac-valvular type (cvEDS) results from a total absence of the proα2-chain of type I collagen. Severe progressive cardiac valve issues (especially aortic and mitral) predominate in adults, but may not be present in the first decade. The typical features of cEDS are commonly seen.
The vascular type of EDS (vEDS) is characterized by extensive bruising and often thin, translucent skin that is not hyperextensible. , Joint hypermobility is usually limited to the digits, and only 17% of patients meet Beighton criteria. The facial appearance is often typical, with sunken eyes, a thin upper lip, and decreased facial fat. Spontaneous rupture of arteries, particularly midsized arteries, may occur during childhood, although its peak age of incidence is the third or fourth decade of life , ; the diagnosis of vascular EDS may be missed until a devastating vascular complication occurs. Arterial or intestinal rupture often presents as acute abdominal or flank pain (including from gastrointestinal [GI] bleeding or bowel perforation), and intracranial aneurysms may be associated with cerebrovascular accidents. Arterial rupture is the most common cause of death. Pregnancies may be complicated by prepartum and postpartum arterial bleeding and by intrapartum uterine rupture. Vaginal and perineal tears from the delivery heal poorly, and pneumothorax occurs in 11% of affected individuals. In one series, 94% of patients with vEDS showed blue sclerae. Given the autosomal dominant nature of vEDS, the diagnosis should be considered if there is a family history of arterial dissection or rupture under 40 years of age, or if spontaneous pneumothorax or unexplained sigmoid colon rupture with other vEDS features exists.
The vascular form of EDS has been associated with mutations in type III collagen (COL3A1) , resulting in a reduced amount of type III collagen in dermis, vessels, and viscera. The mean lifespan is reduced to 51 years of age. However, the prognosis is related to the underlying COL3A1 mutation, with individuals with null mutations having a better prognosis than those with missense or exon-skipping mutations.
vEDS may be confused with periodontal EDS and Loeys–Dietz syndrome (LDS). The latter is an autosomal dominant disorder characterized by joint hypermobility with dislocations; soft, velvety skin; arterial tortuosity; widespread vascular aneurysm and dissection ; and an increased risk of atopy. The disorder has been divided into LDS type 1 and type 2. Patients with type 1 disease have a marfanoid habitus (but do not fulfill Ghent criteria for Marfan syndrome [MFS] including lack of ectopia lentis), craniofacial abnormalities, and mutations in TGFBR1 (transforming growth factor [TGF]-β1 receptor), whereas patients with type 2 have mutations in TGFB2R and no craniofacial or skeletal anomalies.
The most common form of EDS, the hypermobile type (hEDS), is associated with extensive joint hypermobility and dislocations, particularly of the shoulder, patella, and temporomandibular joints. The skin is often soft and velvety but heals well and only occasionally shows hyperextensibility. Children often receive a diagnosis of chronic pain syndrome because no specific underlying cause is identified. Up to 40% of school-aged children may show a score of 5 or higher using the 9-point Beighton criteria, leading to overdiagnosis and making diagnosis of hEDS difficult. It is also hard to distinguish from benign joint hypermobility syndrome, and the term joint hypermobility syndrome has been suggested to encompass any child with symptomatic joint hypermobility, , including those with hypermobility-type EDS. The autonomic burden of hypermobility-type EDS is as high as in fibromyalgia and is primarily characterized by orthostatic issues (dizziness or even transient loss of consciousness with postural change or standing), palpitations, shortness of breath, and GI complaints. Involvement during infancy may present as unexplained fractures, with parents accused of child abuse and neglect, and an affected parent unaware of the EDS diagnosis.
Patients with the arthrochalasis type (aEDS) show generalized joint hypermobility with severe recurrent subluxations and congenital bilateral hip dislocation. The skin may be hyperextensible, fragile, and easy to bruise. Early and significant muscle hypotonia, kyphoscoliosis, and mild osteopenia have been described. Mutations in COLA1A1 or COL1A2 that result in skipping of exon 6 lead to defective collagen synthesis.
The dermatosparaxis form of EDS (dEDS) is characterized by severe skin fragility with sagging, redundant skin ; wound healing leads to wide atrophic scars, especially past infancy. The skin may be soft and doughy in texture with easy bruisability. Characteristic facies, gingival hyperplasia, large umbilical and inguinal hernias, delayed closure of the fontanels, postnatal growth failure, and premature rupture of the fetal membranes may be seen. Electrophoretic demonstration of procollagen α1(I) or α2(I) chains from collagen or cultured fibroblasts is seen with the arthrochalasis and dermatosparaxis forms.
The kyphoscoliotic type of EDS (kEDS) is characterized by generalized joint laxity with severe muscle hypotonia at birth, which leads to gross motor delay and congenital progressive scoliosis. Severe hypotonia is predominant during infancy, and patients are thought to have congenital muscular dystrophy or myopathies. By the second or third decade of life, patients tend to lose the ability to ambulate. The skin may be fragile and heals with atrophic scars. Easy bruisability and arterial rupture have been described. Patients often show a marfanoid habitus, osteopenia, and scleral fragility with rupture of the ocular globe. Because of PLOD1 mutations, this form shows a deficiency of collagen lysyl hydroxylase. The diagnosis can be confirmed by finding a high ratio of lysyl pyridinoline (LP) to hydroxylysyl pyridinoline (HP) crosslinks (LP/HP) in the urine by high-performance liquid chromatography (HPLC). , Mutations in FKBP14, , which catalyzes the folding of type III collagen, are an unusual cause of kEDS and are associated with sensorineural healing loss and a higher risk of serious vascular abnormalities in adulthood than with PLOD1 mutations.
The brittle cornea syndrome (BCS), an autosomal recessive condition caused by mutations in either ZNF469 or PRDM5, is characterized by thin, brittle cornea; ocular fragility; blue sclera; and keratoconus, as well as associated skin and joint hypermobility and kyphoscoliosis.
The spondylocheirodysplastic form of EDS (spEDS) resembles the kyphoscoliotic form but lacks the severe muscular hypotonia from birth and progressive kyphoscoliosis. spEDS shows moderate and progressive short stature, variable hypotonia, and bowing of the limbs, with osteopenia and pes planus; delayed motor and cognitive development; and translucent, doughy, hyperextensible skin. Biallelic mutations in SLC39A13, which encodes the zinc transporter protein ZIP13 , important for intracellular zinc homeostasis, are causative. ,
The progeroid forms of EDS are now classified as subsets of spondylodysplastic EDS. They are caused by biallelic mutations in either galactosyltransferase I (B4GALT7) or II (B3GalT6) , key enzymes in initiating glycosaminoglycan (GAG) synthesis. In addition to the typical features of spEDS from SLC39A13 mutations, both have joint hypermobility and blue sclerae. Each has its own gene-specific minor criteria for diagnosis, including gene-specific craniofacial features. For example, spEDS from B4GALT7 mutations is also characterized by craniofacial features, elbow contractures, clouded cornea, and characteristic radiographic features, whereas spEDS from B4GALT6 mutations is associated with joint contractures (typically of the hands), peculiar fingers, talipes equinovarus, and dental issues. ,
Musculocontractural EDS (mcEDS) is a variant with characteristic facial features, multiple contractures, a hemorrhagic diathesis with large subcutaneous hematomas, and several other systemic complications; this variant has been linked to dysfunction of dermatan sulfate from mutations in both alleles of genes encoding dermatan 4-O-sulfotransferase (CHST14) , or dermatan sulfate epimerase (DSE).
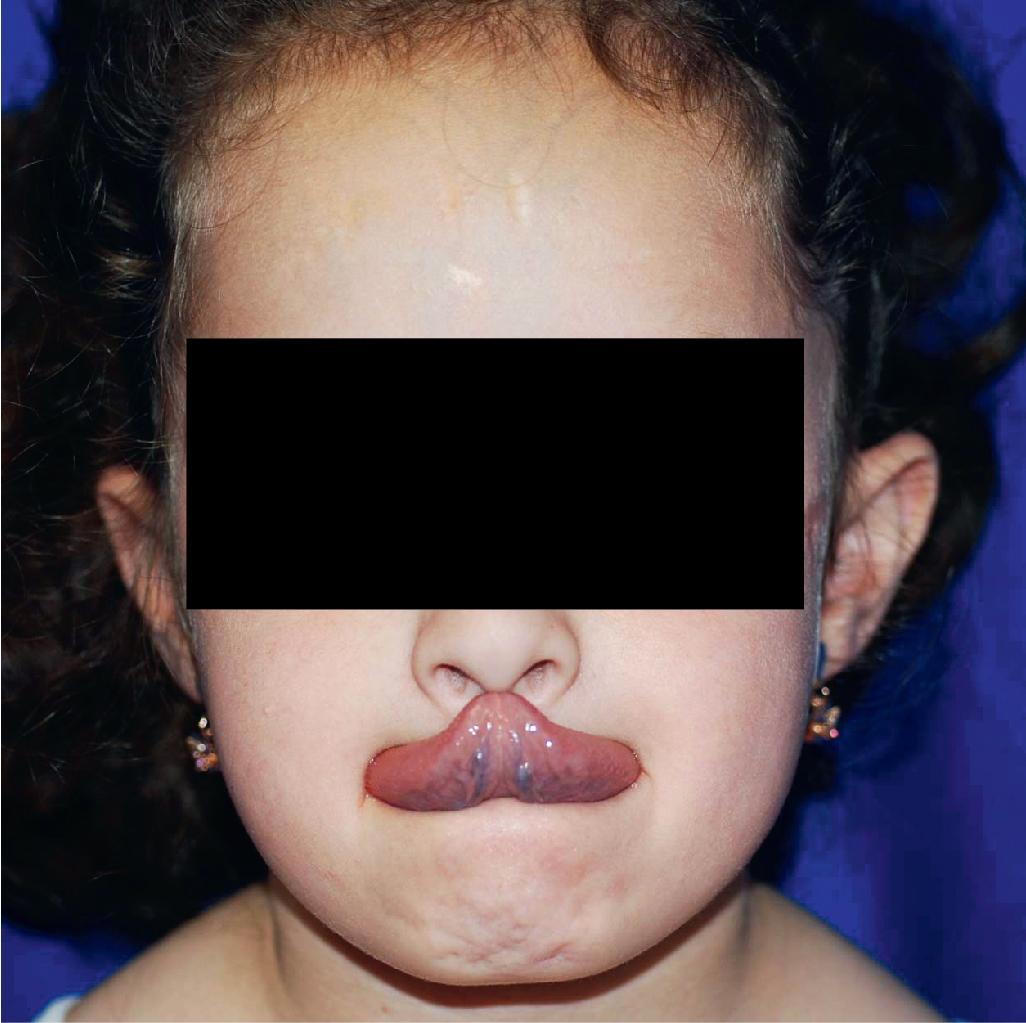
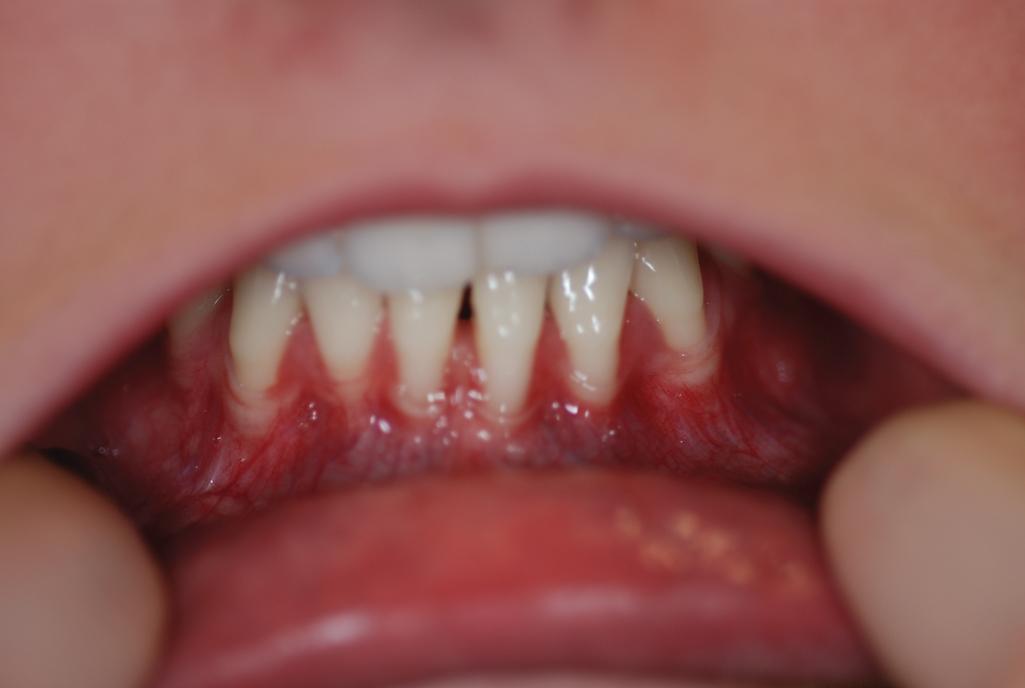
The myopathic form (mEDS) can be autosomal dominant or recessive, but the congenital muscle hypotonia or atrophy, proximal joint contractures, and distal joint hypermobility are major criteria. Many patients have soft, doughy skin and atrophic scarring. The periodontal type (pEDS; formerly type VIII EDS) is a dominant form of EDS and shows variable interfamilial and intrafamilial clinical manifestations (including patients without skin or joint abnormalities). The periodontal type is associated with early gingival regression and periodontitis, joint hypermobility, mild skin findings, and often a marfanoid body habitus, as well as prominent eyes, a short philtrum, pretibial hyperpigmentation, and hoarseness ( Fig. 6.7 ). Acrogeria and prominent vasculature are often associated. It results from heterozygous variants in C1R and C1S, encoding complement subcomponents C1r and C1s.
Management is mainly supportive, but cardiovascular workup, physiotherapy, pain management, and psychological and genetic counseling are important. Wearing a medical bracelet warning of EDS and carrying information for teachers and healthcare workers can be helpful. The Ehlers–Danlos National Foundation ( www.ednf.org ) is a national support group. Children with skin fragility should use protective pads on the shins, knees, and forehead to decrease the risk of laceration and bruising. Low-impact sports (such as swimming) are preferable to contact sports and weight training in patients with hypermobility. Physical and occupational therapists or rheumatologists can suggest devices (such as splints and braces) to make movement more comfortable. Children with significant hypotonia or motor developmental delay should be referred for appropriate physical therapy to improve muscular strength and coordination. Pain medication should be tailored to individual needs. Psychological counseling to help develop coping strategies and treat depression can be important, especially in patients with hypermobility and chronic pain.
A baseline echocardiogram should be performed in all patients with EDS to evaluate aortic root diameter and cardiac valves. Echocardiograms should be repeated in children and adolescents approximately every 3 years if the results are normal and annually if results are abnormal. Patients who have mitral valve prolapse and regurgitation should be given prophylactic antibiotics to prevent bacterial endocarditis.
Surgical procedures present problems, because tissues are friable and difficult to suture. Therefore edges of wounds should be approximated without tension by closely spaced sutures in two layers (absorbable and retained); adhesive reinforcement (e.g., Steri-Strips) to minimize scar spread; and pressure bandages to aid healing, diminish scarring, and lower the risk of hematoma and pseudotumor formation. Sutures should be kept in place for at least twice as long as for normal skin, and use of absorbable suture material that does not get removed has been proposed. Prophylactic antibiotics after injury and close monitoring for postoperative infection are important to minimize postoperative complications.
Ascorbate is a cofactor that stimulates prolyl hydroxylase and lysyl hydroxylase, enzymes that catalyze the formation of hydroxyproline and hydroxylysine to promote collagen deposition. Doses of 2 to 4 g/day have been administered to patients with the kyphoscoliosis type to improve wound healing and decrease the bleeding tendency. Ascorbic acid sometimes decreases the bleeding tendency in other types. Antiinflammatory drugs may improve the musculoskeletal pain associated with EDS, but those interfering with platelet function and prolonging bleeding (acetylsalicylic acids, nonsteroidal antiinflammatory drugs [NSAIDs] other than celecoxib family) should be avoided in patients with significant bruising. Dental procedures require extra precautions as well, because of increased bleeding. More than half of patients who undergo posterior spinal fusion for scoliosis develop various complications.
In patients with the vascular type of EDS, blood pressure should be maintained in the normal range, and annual surveillance of the aortic vasculature is important. Invasive vascular procedures (such as arteriography and catheterization) increase the risk of vascular rupture and should be replaced by Doppler ultrasound and/or magnetic resonance angiography (MRA). Surgery should be avoided if possible but may be required for arterial or bowel complications. Manipulation of vascular and other tissues should be minimized if surgery is required. Celiprolol, a long-acting β 1 antagonist and partial β 2 agonist, decreased by threefold the incidence of arterial rupture or dissection in patients with vascular EDS ; no other β-blockers have been rigorously tested. Pregnancy for women with EDS is considered high risk, but particularly with the vascular type of EDS (up to 12% die from uterine rupture or peripartum arterial rupture). Planned caesarean section may be preferable to vaginal delivery. Infants with EDS are prone to premature birth because of early rupture of membranes. Preterm delivery occurs in 40% of pregnancies with affected neonates and 21% of pregnancies of affected mothers.
MFS is an autosomal dominant disorder that occurs in approximately 1:5000 persons and affects primarily the skeletal, ocular, and cardiovascular systems. Mutations primarily occur in the FBN1 gene that encodes fibrillin-1 (66% to 91%) and rarely in TGFBR1 or TGFBR2, the genes also associated with LDS (see Ehlers–Danlos Syndrome section). Fibrillin-1 is secreted into the extracellular matrix and polymerizes to form microfibrils of the zonular fibers of the lens, which are associated with elastic fibers in the aorta and skin. , The microfibrils stabilize latent TGF-β-binding proteins (LTBPs), which bind to and maintain TGF-β in an inactive state. Deficiency of fibrillin-1 thus leads to abnormal TGF-β signaling. About 27% of cases occur by spontaneous mutation; recurrence as a result of parental germline mosaicism has been described. ,
Diagnosis is best made by clinical examination (including careful cardiac evaluation and measurement of body proportions) ( Box 6.2 ), echocardiography, slit-lamp ophthalmologic examination, and radiographs or imaging as needed to find criteria. Cardiac examinations should be repeated every year until adulthood. , Early diagnosis is critical because of the potentially fatal complications, but the clinical features evolve with advancing age, and diagnosis is difficult. Family history is also important, but manifestations may not be evident until adolescence, and expressivity is variable, so generations appear to be skipped. The Ghent diagnostic criteria ( Box 6.3 ) were simplified in 2010, eliminating major and minor criteria and giving more weight to ectopia lentis and aortic root aneurysm/dissection. Other organ involvement contributes to a systemic score to guide diagnosis if aortic disease, but not ectopia lentis, is present. A more important role is assigned to genotyping FBN1 and other relevant genes. Criteria now enable diagnosis of ectopia lentis and distinction from MASS syndrome ( m itral valve prolapse and m yopia, mild and nonprogressive a ortic root dilatation, marfanoid s keletal changes, and s kin features, especially striae) and mitral valve prolapse syndrome. Criteria are still less reliable in children, especially because several of the more specific features are age dependent (e.g., aortic dilatation, ectopia lentis, dural ectasia, protrusion acetabuli), , , and care should still be made to avoid branding children unnecessarily with MFS, given the potential stigma and lifestyle restrictions.
Autosomal dominant disorder
Present in ≥75% of individuals with Marfan syndrome (MFS):
Positive wrist and thumb sign
Present in ≥50% and <75% of individuals with MFS:
Dolichostenomelia
Pes planus
Joint hypermobility
Aortic root dilatation
Mitral valve prolapse
High arched palate with crowded dentition
Typical facial features
Lens displacement (ectopia lentis)
Present in ≥25% and <50% of individuals with MFS:
Pectus excavatum or carinatum requiring surgery
Myopia
Scoliosis
Striae
* Dolichostenomelia refers to a habitus in which the limbs are unusually long, as is typical of MFS ( dolichos, long; steno, narrow or close; melia, of the limbs).
Without a family history of Marfan syndrome (MFS), any of these combinations allow diagnosis:
Aortic diameter (Z score * ≥2) AND ectopia lentis
Aortic diameter (Z ≥2) AND fibrillin-1 (FBN1) mutation
Aortic diameter (Z ≥2) AND systemic criteria (≥7 points; see criteria later)
Ectopia lentis AND fibrillin-1 (FBN1) mutation with known aortic diameter (Z ≥2)
With a positive family history, these features allow the diagnosis:
Ectopia lentis
Systemic criteria (≥7 points) (see criteria later)
Aortic diameter (Z ≥2 above 20 years old, ≥3 below 20 years old)
Alternative diagnoses (no family history of MFS):
Ectopia lentis syndrome: ectopia lentis with or without systemic criteria AND: (1) with an FBN1 mutation but one not known to be associated with aortic diameter (Z ≥2) OR (2) no FBN1 mutation
MASS: aortic diameter (Z <2) AND systemic criteria (≥5 with at least one skeletal feature) but without ectopia lentis
MVP syndrome: MVP AND aortic diameter (Z <2) AND systemic criteria (<5) without ectopia lentis
Systemic criteria (maximum total: 20 points; score .7 indicates systemic involvement):
Wrist AND thumb sign = 3 (wrist OR thumb sign = 1)
Pectus carinatum deformity = 2 (pectus excavatum or chest asymmetry = 1)
Hindfoot deformity = 2 (plain pes planus = 1)
Pneumothorax = 2
Dural ectasia = 2
Protrusio acetabuli = 2
Reduced US/LS AND increased arm/height AND no severe scoliosis = 1
Scoliosis or thoracolumbar kyphosis = 1
Reduced elbow extension = 1
Facial features (3/5) = 1 (dolichocephaly, enophthalmos, downslanting palpebral fissures, malar hypoplasia, retrognathia)
Skin striae = 1
Myopia >3 diopters = 1
MVP (all types) = 1
*Z score is the standard deviation from normal means of the inner to inner-edge diameter of the aortic root at the sinus of Valsalva, normalized for the subject’s body surface area and age.
Patients are often tall with long extremities (“marfanoid habitus”; see Fig. 6.8 ). The arm span characteristically is greater than the height and, after puberty, the upper segment (vertex to pubis)–to–lower segment (pubis to sole) ratio is less than 0.86. Arachnodactyly, kyphoscoliosis, pectus carinatum or excavatum, and a hindfoot deformity or flat feet are commonly seen in patients with this disorder. Joint laxity from capsular, ligamentous, and tendinous involvement may cause hyperextensibility and/or dislocation. Patellar dislocation is not uncommon; dislocation of the hip, often detected during the newborn period, may be the first sign of MFS. The thumb sign (thumb extends well beyond the ulnar border of the hand when overlapped by fingers) and wrist sign (thumb overlaps the fifth finger as they grasp the opposite wrist) are screening tests for the joint hypermobility of MFS. Dural ectasia is occasionally seen in children, and imaging is performed largely to consider an additional Ghent criterion for diagnosis, but its presence is nonspecific. Specific facial features and pneumothorax are additional criteria (see Box 6.3 ).
Lack of subcutaneous fat and the presence of striae, most prominent on the upper chest, arms, thighs, and abdomen, are the most common cutaneous manifestations of MFS. In addition, elastosis perforans serpiginosa (EPS; see Elastosis Perforans Serpiginosa section) occurs with increased incidence in patients with MFS.
The most common ocular abnormalities are lens displacement (ectopia lentis; the hallmark of ocular involvement, seen in at least 55% of affected children and usually bilateral) and myopia (almost half of children). Retinal detachment, cataracts, or glaucoma may impair vision and cause blindness.
Cardiovascular abnormalities occur in almost 70% of children with MFS. Dilatation of the aorta is the most common defect and is generally greatest at the sinuses of Valsalva, diffuse dilatation of the proximal segment of the ascending aorta with aortic regurgitation often occurs, and dilatation of the pulmonary artery has been detected in 8% of affected children. Mitral valve prolapse occurs in approximately 60% of affected children and adolescents. Left ventricular dilatation may predispose patients to alterations of repolarization and fatal ventricular arrhythmias. The most severely affected children, those with neonatal MFS, almost always have aortic dilatation and severely impaired valves, leading to congestive heart failure, pulmonary hypertension, and a risk of early death. In a child diagnosed with MFS, serial echocardiography at 6- to 12-month intervals is recommended, with the frequency depending on the aortic diameter in relation to the body surface area and the rate of increase. Pregnancy has been associated with significant cardiac complications (aortic dissection, arrhythmia), postpartum hemorrhage, thromboembolism, premature babies, and babies who are small for gestational age.
Conditions most often considered when patients have cutaneous and other system concerns are vEDS (aortic root dilatation or dissection) (see Ehlers–Danlos Syndrome and Table 6.1 ), the MASS phenotype, LDS (aortic root dilatation or dissection), Shprintzen–Goldberg syndrome, homocystinuria, and methylmalonic aciduria with homocystinuria ( Table 6.4 ). Patients with MFS do not have the translucent and velvety skin or easy bruising of EDS or LDS. Patients with homocystinuria have ectopia lentis and a marfanoid habitus, but mental retardation is a distinguishing feature. Individuals with an inherited deficiency in processing cobalamin C/vitamin B 12 similarly may have marfanoid features in addition to macrocytic anemia and neurodevelopmental issues. Perhaps hardest to distinguish is MASS, a marfanoid disorder caused by FBN1 mutations but with a better prognosis. Striae are a prominent feature, and atrophic skin patches with abnormal elastic fibers may be present. Pectus excavatum and marfanoid skeletal features are most common. Among the distinguishing features are a stable and mild aortic root dilatation and lack of ectopia lentis, but distinguishing MASS from MFS can be difficult in children and adolescents.
| Disorder | Subtype | Gene | Distinguishing Features |
|---|---|---|---|
| Marfan syndrome (MFS) | — | FBN1 | —- |
| MASS syndrome | — | FBN1 |
|
| Loeys–Dietz syndrome (LDS) |
|
|
|
| Shprintzen–Goldberg syndrome (SGS) | — | SKI |
|
| Homocystinuria | CBS |
|
|
| Cobalamin C (CblC) defect | MMACHC | Marfanoid features, arachnodactyly, joint laxity, and scoliosis, macrocytic anemia, developmental delay, behavioral issues |
The prognosis of MFS depends on the extent and severity of cardiovascular defects. Death usually occurs in adulthood, but occasionally during childhood, as a result of cardiovascular sequelae, especially due to complications related to dilatation of the aortic root. , A rare neonatal form of MFS features the marfanoid body disproportion, lax skin, emphysema, ocular abnormalities, joint contractures, kyphoscoliosis, adducted thumbs, crumpled ears, micrognathia, muscle hypoplasia, and deficient subcutaneous fat over joints. Severe cardiac valve insufficiency and aortic dilatation result in death during the first 2 years of life. A neonatal progeroid variant of MFS with prematurity, congenital lipodystrophy, and frameshift mutations at the 3′ end of FBN1 has been described. In the differential diagnosis of progeria, the marfanoid habitus with accelerated linear growth, severe myopia, and dilatation of the aortic bulb allow correct diagnosis. ,
Children with MFS but without serious cardiac issues can participation in some sports but should avoid potentially harmful exertion, particularly contact sports (to protect the aorta and lens), sports with bursts of activity (such as sprinting), and isometric exercises (such as weightlifting), which might lead to further aortic root dilatation, aortic rupture, or congestive heart failure. Scuba diving should be avoided because of the risk of pneumothorax.
Systemic pharmacologic therapy has been directed at limiting aortic root dilatation. , The largest study in children, the Pediatric Heart Network trial, showed that atenolol, a β-blocker, and losartan, an angiotensin receptor blocker that reduced TGF-β pathway aberrant signaling, each reduced the rate of aortic dilatation. , When atenolol is used, the dose should be individualized and uptitrated on the basis of hemodynamic response to achieve maximal β-blockade. Doses of 1.4 mg/kg per day losartan (100 to 150 mg per day in adult-sized adolescents) are recommended. For patients with severe or progressive aortic root dilatation, the combination of β-blocker and angiotensin receptor blocker therapy should be considered. Given that the greatest improvement was noted in younger children, medical therapy should be prescribed even in young children with evidence of early aortic root dilatation. There is limited evidence to support the routine use of angiotensin-converting enzyme inhibitors or calcium channel blockers for prophylactic treatment of aortic enlargement. Not all patients respond, and those who experience progressive aortic root dilatation with its ongoing risk for aortic dissection should be considered for prophylactic aortic root surgery. In one study, striae together with elevated TGF-β and matrix metalloproteinase (MMP)-3 serum levels correlated with a higher risk of aortic dissection. Aneurysmal and valvular heart defects may require prosthetic replacement, but this should be postponed as long as possible to avoid recurrent prosthesis replacement, particularly in growing children.
Early and regular ophthalmologic examinations are required to detect correctable amblyopia and retinal detachment. Ectopia lentis and even complete luxation may be tolerated for decades, but lens extraction may be required to treat diplopia, glaucoma, cataracts, or retinal detachment. Repair of the pectus excavatum is indicated if cardiopulmonary compromise occurs, but should be delayed until skeletal maturation is nearly complete to prevent recurrence and should employ internal stabilization. Scoliosis may be lessened in adolescent girls by estrogen therapy, but this therapy may also produce an overall decrease in height. Bracing, physical therapy, and vertebral fusion may all be necessary to prevent severe scoliosis. The website for the National Marfan Foundation is www.marfan.org .
OI refers to a group of inherited disorders of bone fragility in which the skin may be thin, atrophic, and somewhat translucent. The easy bruisability and bone fractures may raise the possibility of child abuse. Wound healing may be normal, but scars are commonly atrophic or hypertrophic. Patients often show hyperlaxity of ligaments and hypermobility of joints, but joint dislocation does not occur. Blue sclerae are distinctive features in approximately 90% of patients but can also be seen in MFS and EDS; increased electron-dense granular material between scleral fibers permits scattering of light by normal pigment within the orbit. Otosclerosis with hearing loss may begin during adolescence (50% of the common type I by adulthood), and fragile, discolored teeth are particularly common in the more unusual forms (dentinogenesis imperfecta [DI]). Patients may show mitral and aortic valve dilatation and regurgitation and cystic medionecrosis of the aorta.
More than one classification scheme has recently been proposed; the newer Sillence classification includes types 1 through 5 and the more-newly recognized, less common, and largely autosomal recessive forms based on clinical features. The classification described by Marini et al. separates types of OI based on genetics and extends the five Sillence classification subtypes to 12 subtypes (I through XII) and an unclassified/other category ( Table 6.5 ). OI has also been classified by severity of clinical features (mild to extremely severe), fracture frequency, bone densitometry, and level of mobility. Changes in 16 genes may be classified under OI, presenting variable clinical features, severity, and prognosis. More than 85% of affected individuals have an autosomal dominant form of OI (AD OI), of which most are type I OI, the mildest form.
Patients with AD OI almost all have mutations in type I collagen, which is composed of two α1 (COLIA1) and one α2 (COLIA2) polypeptide chains, forming a triple helical structure. Dominant OI can also result from mutations in IFITM5, leading to abnormalities in matrix mineralization. Overall, 10% to 15% of cases are autosomal recessive and result from a mutation in one of at least 11 genes involved in posttranslational processing of type I collagen and bone formation (see Table 6.4 ). Some of these encode proteins involved in a complex required for proline (P986) hydroxylation of type I collagen (CRTAP, P3H1, CYPB) . Others are involved in collagen mineralization ( BRIL and PEDF ), collagen crosslinking, folding and chaperoning (FKBP65 and HSP47) , and osteoblast development (osterix, OASIS, TRICB, and WNT1 ) (see Table 6.4 ).
Supportive orthopedic therapy includes physical therapy to prevent contractures and immobility-induced bone loss, orthoses to protect the lower limbs, and, in more severe cases, bisphosphonates. Cyclical treatment with intravenous bisphosphonates is the mainstay of medical therapy for children with moderate to severe OI and has been shown to increase bone mass density, improve muscle strength and mobility, and decrease fractures and bone pain. ,
Cutis laxa (generalized elastolysis) results from mutations in one of 14 genes that disrupt the elastic tissue network in skin , ( Box 6.4 ; Table 6.6 ). In general, the products of these genes are either (1) proteins involved in the assembly of elastic fibers and upregulation of TGF-β signaling (e.g., ELN, FBLN4, FBLN5, and LTBP4 ); (2) Golgi proteins that facilitate secretion of elastic network components (e.g., ATP6V0A2, RIN2, and GORAB ); or (3) mitochondrial proteins (e.g., PYCR1, ALDH18A1, and SLC2A10 ). Some affect protein glycosylation (ATP6V0A2 , ATP6V1E1, and ATP6V1A) . ,
Autosomal dominant or autosomal recessive forms
Autosomal recessive forms subdivided into those with cardiovascular features (ARCL-1); those with developmental delay (ARCL-2); and those with developmental delay, athetosis, and corneal clouding (ARCL-3). A variety of underlying molecular bases for each group have been discovered (see Table 6.5 )
| Type | Frequency/Inheritance | Gene; Protein | Clinical Features | Effect of Gene Alteration |
|---|---|---|---|---|
| I | AD (>50%) | COL1A1, COL1A2 ; type I collagen |
|
Collagen I quantity |
| II | AD * | COL1A1, COL1A2 glycine substitutions; type I collagen |
|
Collagen I structure |
| III | AD | COL1A1, COL1A2 glycine substitutions; type I collagen |
|
Collagen I structure |
| IV | AD | COL1A1, COL1A2 glycine substitutions; type I collagen |
|
Collagen I structure |
| V | AD (4% to 5%) | IFITM5 ; BRIL |
|
Matrix mineralization |
| VI | AR | SERPINF1 ; PEDF |
|
Matrix mineralization |
| VII | AR | CRTAP ; cartilage-associated protein |
|
3-hydroxylation of collagen |
| VIII | AR | LEPRE1 ; prolyl-3-hydroxylase-1/P3H1 |
|
3-hydroxylation of collagen |
| IX | AR | PPIB ; peptidyl-prolyl isomerase B/CyPB |
|
3-hydroxylation of collagen |
| X | AR | SERPINH1 ; HSP47 |
|
Collagen chaperone |
| XI | AR | FKBP10 ; FKBP10 (also called FKBP65) |
|
Telopeptide hydroxylation for crosslinking |
| XII | AR | BMP1 ; BMP1/mTLD | Clinically type III | Collagen processing (cleaves C-propeptidase) |
| OTHERS | ||||
| AR | CREB3L1 ; OASIS | Clinically type III | Osteoblast development | |
| AR | SP7/OSX ; SP7/osterix | Clinically type II | Osteoblast development | |
| AR | TMEM38B ; TRICB | Clinically type III | Osteoblast development | |
| AD/AR | WNT1 ; WNT1 | Clinically type II (AD) or type III (AR) | Osteoblast development | |
| AR | PLOD2 ; PLOD2 | Clinically type III; can have congenital joint contractures | Telopeptide hydroxylation | |
| XLR | PLS3 ; plastin 3 | Clinically closest to type IV; no features beyond childhood fractures; manifests in hemizygous males and variable in heterozygote females | Bone formation and remodeling | |
* Usually new mutation; parental germline mosaicism in 2% to 6% of cases.
Skin
Loose, inelastic skin
Most common other features:
Facial dysmorphism
Aortic dilatation
Pulmonary artery stenosis
Pulmonary emphysema
Diverticulae: gastrointestinal, genitourinary
Uterine or rectal prolapse
Ventral, hiatal, inguinal hernias
| Disorder | ADCL | ARCL-1A | ARCL-1B | ARCL1C/URDS | ARCL-2A | ARCL-2B | ARCL-2C | ARCL-2D | ARCL-3/De Barsy | GO | XLCL | MACS | ATS | Lens-Majewski |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gene | ELN | FBLN4/EFEMP2 | FBLN5 | LTBP4 | ATP6V0A2 | PYCR1 | ATP6V1E1 | ATP6V1A | ALDH18A1; PYCR1 | GORAB | ATP7A | RIN2 | SLC2A10 | PTDSS1 |
| Protein | Elastin | Fibulin-4 | Fibulin-5 | LTBP4 | ATP6V0A2 | PYCR1 | ATP6V1E1 | ATP6V1A | P5CS; PYCR1 | GORAB | ATP7A | RIN2 | GLUT10 | PSS1 |
| Lax skin | +++ * | ++ | +++ | +++ | +++ | +++ | +++ | +++ | +++ | ++ | +++ | ++ | +++ | ++ |
| Facial features | — | |||||||||||||
| Prominent ears | +++ | + | +++ | + | — | — | + | + | — | — | ++ | + | ++ | |
| Hypertelorism | — | +++ | — | +++ | — | — | ++ | ++ | — | — | — | ++ | ++ | |
| Retrognathia | — | +++ | — | +++ | +++ | +++ | ++ | ++ | — | — | — | +++ | +++ | |
| Other dysmorphism | — | ++ | — | ++ | — | ++ | ++ | ++ | ++ | +++ | ++ | ++ | +++ | |
| Emphysema | ++ | ++ | +++ | +++ | — | — | __ | __ | — | — | — | — | — | __ |
| Supravalvular aortic stenosis | — | — | +++ | — | — | — | __ | __ | — | — | — | — | — | __ |
| Aortic aneurysm | ++ | +++ | — | — | + | — | + | + | — | — | — | — | ++ | __ |
| Arterial tortuosity | — | +++ | + | — | — | — | __ | __ | ++ | — | ++ | — | +++ | __ |
| Hernias | ++ | +++ | +++ | +++ | +++ | ++ | + | + | ++ | ++ | +++ | + | ++ | __ |
| Bladder diverticula | — | — | + | +++ | + | — | __ | __ | — | — | +++ | — | — | __ |
| Delayed motor development | — | + | + | — | +++ | — | __ | + | +++ | +++ | +++ | — | — | __ |
| Mental retardation | — | — | — | — | +++ | +++ | __ | __ | +++ | ++ | +++ | — | — | ++ |
| Growth delay | — | ++ | + | +++ | +++ | +++ | __ | __ | +++ | +++ | + | — | — | ++ |
| IUGR | — | — | — | + | +++ | +++ | __ | __ | +++ | +++ | — | — | — | |
| Lax joints | — | + | — | ++ | +++ | +++ | + | + | ++ | +++ | ++ | +++ | +++ | ++ |
| Hypotonia | — | + | — | +++ | +++ | — | ++ | ++ | ++ | ++ | ++ | — | — | __ |
| Congenital hip dislocation | — | + | — | — | + | +++ | + | + | ++ | ++ | — | — | — | ++ |
| Patent anterior fontanel | — | — | — | ++ | +++ | — | __ | __ | ++ | ++ | ++ | — | — | ++ |
| Occipital horns | — | — | — | — | — | — | __ | __ | — | — | +++ | — | — | __ |
| Osteoporosis | — | — | — | — | — | ++ | __ | __ | — | +++ | ++ | + | — | __ |
| Scoliosis | — | — | — | — | ++ | + | ++ | __ | ++ | +++ | ++ | +++ | ++ | +++ |
| Macrocephaly | — | — | — | — | — | — | __ | __ | — | — | — | +++ | — | __ |
| Microcephaly | — | — | — | — | — | + | __ | __ | ++ | ++ | — | — | — | __ |
| Gingival hyperplasia | — | — | — | — | — | — | __ | __ | — | — | — | +++ | — | __ |
| Corneal opacity | — | — | — | — | — | + | __ | __ | +++ | — | — | — | — | __ |
| Athetoid motion | — | — | — | — | — | + | __ | __ | +++ | — | — | — | — | __ |
| Alopecia | — | — | — | — | — | — | — | — | — | — | +++ | — | + |
* Incidence of finding range: —, not reported to +++, common. Of note, some of these disorders have been reported in a very small number of patients, and thus the incidence range may be inaccurate at this time.
Cutis laxa can be acquired or congenital, with the genetic forms inherited as either autosomal dominant or autosomal recessive forms, the latter usually being more severe. An X-linked form (X-linked cutis laxa, or occipital horn syndrome; formerly Ehlers–Danlos syndrome type IX) is now classified as a copper transport disease caused by a mutation in the copper transporting adenosine triphosphatase (ATPase), α-polypeptide ATP7A.
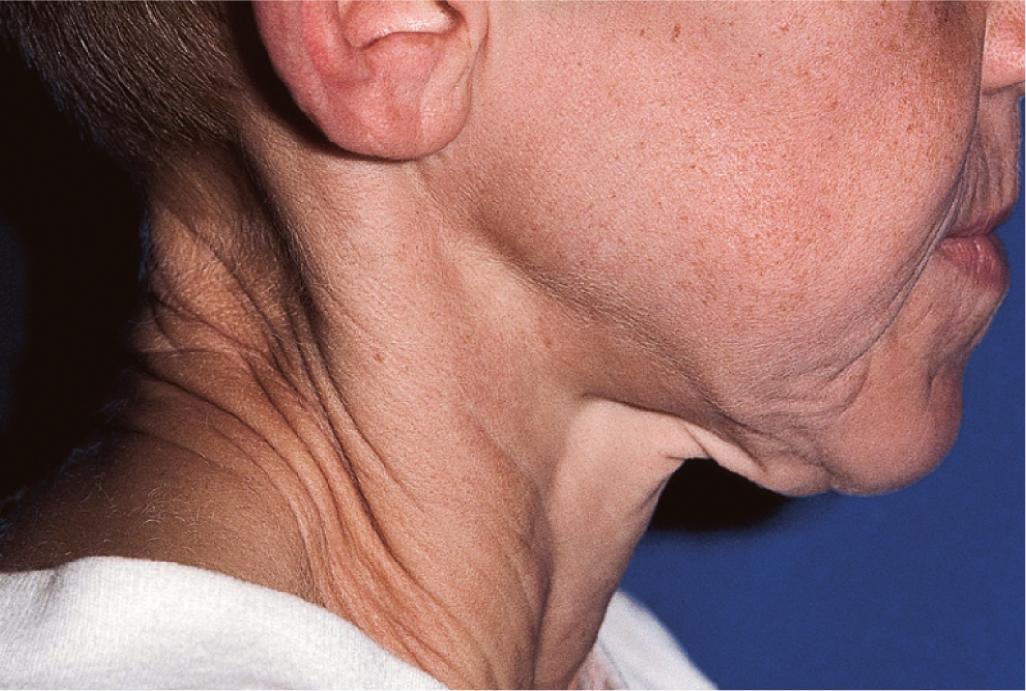
Patients with cutis laxa present a striking picture of loose, inelastic, redundant skin that sags and hangs in pendulous folds, as if it were too large for the body. The drooping and ectropion of the eyelids, together with the sagging facial skin and accentuation of the nasal, labial, and other facial folds, help produce the “bloodhound” or aged appearance ( Fig. 6.9 ).
Most dominant cases result from heterozygous frameshift mutations in the gene encoding elastin. The autosomal dominant form may appear at any age and presents as a cosmetic problem with few systemic changes. The skin in cutis laxa is extensible but, in contrast to that of EDS, does not spring back to place on release of tension. Special stains for elastic tissue (Verhoeff–van Gieson stain) of skin biopsy specimens demonstrate significantly decreased or absent dermal elastic fibers.
Systemic manifestations caused by weakened supportive tissue include aortic root dilatation (55%), hernias (51%; ventral, hiatal, or inguinal), pulmonary emphysema (37%), pulmonary artery stenosis, diverticulae of the GI tract or urinary bladder, and uterine or rectal prolapse. In patients with the severe autosomal recessively inherited disease, the disorder is gradually progressive, and death from pulmonary complications related to emphysema may occur early in infancy or in many instances in the second to fourth decades of life.
The autosomal recessive forms have traditionally been divided into three subgroups based on clinical characteristics: autosomal recessive cutis laxa (ARCL)-1, -2, and -3 (see Table 6.6 ). Most genes mutated in ARCL have been identified, and the group is quite heterogeneous. All patients tend to have cutis laxa and facial dysmorphism. Hernias (umbilical, inguinal, diaphragmatic) are common. ARCL-1 is characterized by severe cardiopulmonary lesions, including infantile emphysema and cardiac defects, and bladder diverticulae. Fibulin-5 deficiency has been associated with supravalvular aortic stenosis. Fibulin-4 deficiency (mutations in FBLN4, also called EFEMP2 ) may be associated with mild to moderate cutis laxa and has been linked to arachnodactyly, tortuous vessels with aneurysm, joint laxity, microcephaly, bone fractures, and pulmonary artery occlusion ( Fig. 6.10 ). Fibulins-4 and -5 are critical in elastic fiber assembly in the skin and other elastic tissues. More recently, LTBP4 mutations (ARCL type 1C) have been found in patients with emphysema and severe GI and genitourinary (GU) tract involvement but no vascular abnormalities.
Patients with ARCL-2 (which includes “wrinkly skin syndrome”) have mild cutis laxa with growth, distinct facial features, hip dysplasia, and often developmental delay without prominent vascular lesions or emphysema, in contrast to ARCL-1. Patients with ARCL-2A may show pretibial pseudoecchymotic skin lesions that resemble those found in EDS, and ARCL-2B is associated with osteoporosis and fractures. The most recently described ARCL-2C and D are characterized by generalized skin wrinkling with sparse subcutaneous fat and often large skin folds with abnormal fat distribution; some have congenital heart defects and/or seizures. ARCL-3, or De Barsy syndrome, can be distinguished from ARCL-2 by athetoid movements and corneal opacities, but otherwise there is overlap between ARCL-2 and ARCL-3. , , , Gerodermia osteodysplastica, resulting from mutations in GORAB , shows features of ARCL-2A, ARCL-2B, and ARCL-3 (De Barsy syndrome) but has no associated corneal opacities or athetoid movement and characteristically has osteopenia with fractures (as in ARCL-2B). In fact, more severe cases of ARCL-2B show juvenile cataract and dystonic movement, as in ARCL-3. Cutis laxa is a major component for children with Lenz–Majewski syndrome in association with generalized hyperostosis, proximal symphalangism, syndactyly, brachydactyly, mental retardation, hypertelorism, and enamel hypoplasia. Lenz–Majewski syndrome has recently been linked to gain-of-function mutations in phosphatidylserine synthase 1 (PTDSS1) , which affect phospholipid synthesis. Neonatal brachydactyly with later severe dwarfism, intellectual disability, and hyperostotic skeletal dysplasia are distinguishing features.
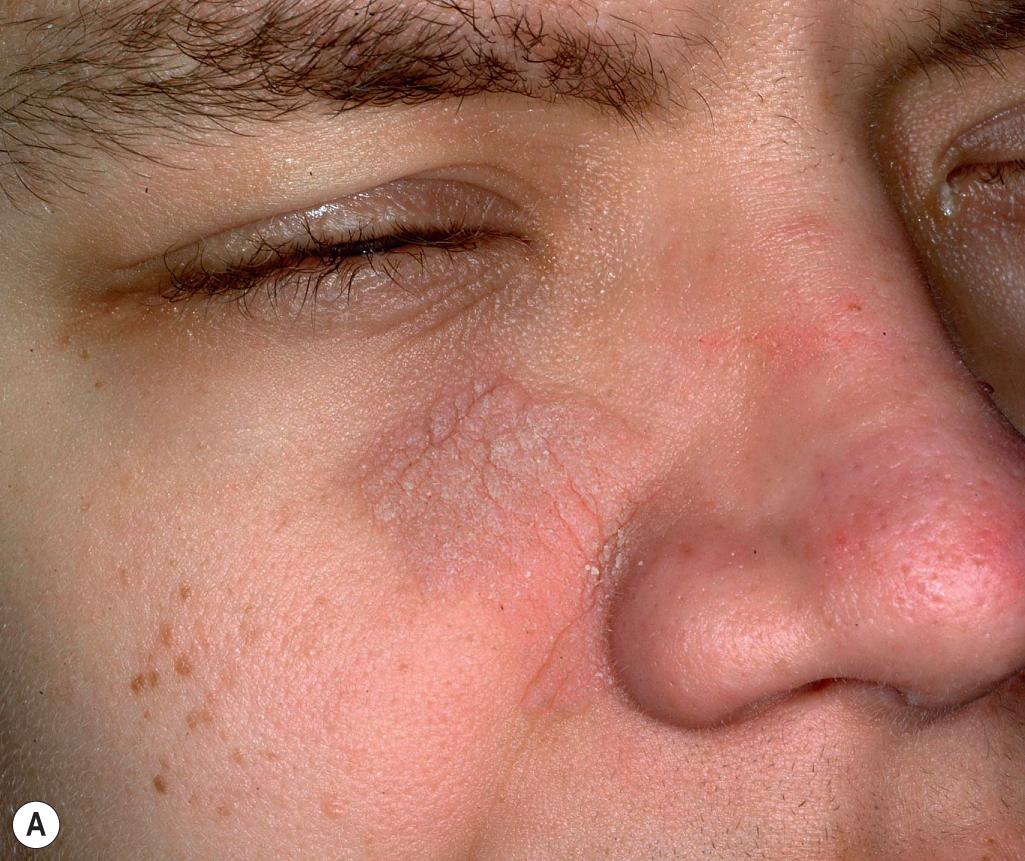
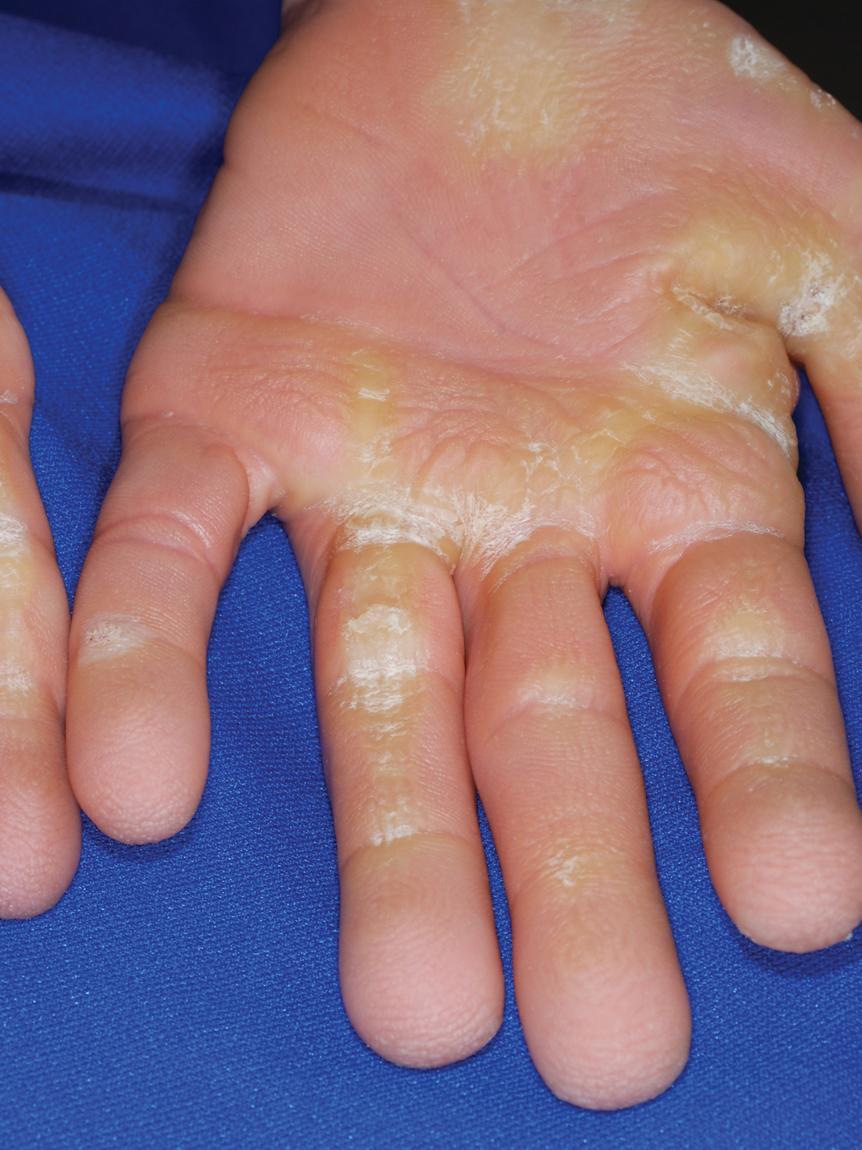
Cutis laxa is also a component of several additional syndromes ( Box 6.5 ). Cutis laxa is a component of macrocephaly, alopecia, cutis laxa, and scoliosis (MACS) syndrome, an autosomal recessive disorder that results from deficiency of RIN2, leading to decreased fibulin-5 and microfibrils. Arterial tortuosity syndrome (ATS) features facial dysmorphism, vessel tortuosity, and joint laxity with scoliosis. Costello syndrome is an autosomal dominant disorder characterized by soft, loose skin of the neck, hands, and feet with excessive wrinkling and deep creases resembling cutis laxa. , , The digits tend to be hyperextensible, with loose skin and characteristic ulnar deviation at the wrist ( Fig. 6.11 ). Affected children develop papillomatous plaques ( Fig. 6.12 , A ) and/or papules ( Fig. 6.12 , B ) around the nares, mouth, and anal areas; acanthosis nigricans; and sometimes striate palmoplantar keratoderma ( Fig. 6.13 ). Although prenatal overgrowth and polyhydramnios occur, patients tend to have postnatal failure to thrive and a distinctive appearance with craniofacial findings that resemble those of lysosomal storage disorders. The facies are coarse with thick lips, macroglossia, and relative macrocephaly. Severe short stature, mental retardation, curly hair, palmoplantar keratoderma (sometimes striate), and hypertrophic cardiomyopathy are other associated manifestations. Costello syndrome results from activating mutations in HRAS and is associated with a 15% lifetime risk for malignancy, most commonly rhabdomyosarcoma. Deficiency of transaldolase (TALDO) presents with hepatosplenomegaly, liver failure, and cirrhosis in association with dysmorphic features, hypertrichosis, cutis laxa, congenital heart disease, hemolytic anemia, thrombocytopenia, and GU malformations , ; patients may die in utero with hydrops fetalis. Cutis laxa has also been described in a child with typical Kabuki makeup syndrome.
Ablepharon–macrostomia syndrome
Barber–Say syndrome
Cantú syndrome
Costello syndrome
GAPO syndrome
Kabuki makeup syndrome
Menkes syndrome
Progeria
Patterson pseudoleprechanism
SCARF syndrome
Transaldolase deficiency
Williams syndrome
Cutis laxa can be an acquired condition ( Box 6.6 ), although it occurs less commonly in children than in adults. It can be localized, but in its generalized form ill-defined areas of loose skin appear insidiously but progressively, usually first on the face and then progressing in a cephalocaudal direction. The development of cutis laxa is often preceded by an urticarial eruption. Emphysema, aortic aneurysms with subsequent rupture, and GI and GU diverticulae have been associated. Although most affected children have not had systemic evidence of acquired cutis laxa, aortic dilatation, emphysema, and severe tracheobronchomegaly have led to the demise of children with acquired cutis laxa. Children may develop cutis laxa after drug exposure to penicillin, D-penicillamine, or isoniazid. EPS may be related (see Elastosis Perforans Serpiginosa section). Marshall syndrome (also called type II acquired cutis laxa ) is a postinflammatory elastolysis characterized by well-demarcated, nonpruritic erythematous plaques that appear in crops during a period of days to weeks, often in association with fever, malaise, and peripheral eosinophilia. Several children have developed cutis laxa after Sweet syndrome, especially at sites of previous inflammation. Acquired cutis laxa has been described in a child with extensive cutaneous mastocytosis. Systemic involvement is rare, but fatal aortitis has been described. Interestingly, a child with acquired cutis laxa after Toxocara infestation was found to have compound heterozygous mutations in elastin and a fibulin-5 mutation on one allele, suggesting that environmental factors may trigger acquired cutis laxa in genetically predisposed individual.
Infections: Toxocara canis (cat scratch); Treponema pallidum (syphilis); Borrelia burgdorferi (Lyme disease); Onchocerca volvulus (onchocerciasis)
Medications: isoniazid; penicillins; D-penicillamine
Inflammatory diseases: celiac disease; sarcoidosis; dermatitis herpetiformis, Sweet syndrome
Rheumatic disorders: systemic lupus erythematosus; rheumatoid arthritis
Others: α1-antitrypsin deficiency; mastocytosis; nephrotic syndrome; amyloidosis; malignancy
Therapy for cutis laxa is limited. Surgery can correct diverticulae, rectal prolapse, or hernias, and plastic surgery can make a dramatic improvement in patients, inevitably with important psychological benefit. Unlike patients with EDS, patients with cutis laxa have no vascular fragility and heal well, enabling repeated facelift procedures. Pulmonary function studies may aid in the early detection of emphysema.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here