Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The hereditary disorders of cornification, or the ichthyoses, are a group of disorders characterized by scaling, skin thickening, and usually underlying inflammation, , leading to a significant impact on quality of life. In many patients, itch is also an issue. Ichthyoses are largely distinguished by their clinical and, in some cases, histologic and ultrastructural features. During the past 2 decades, next-generation sequencing has facilitated the discovery of the underlying molecular basis of most of the forms of ichthyosis, further refining classification , and facilitating our understanding of the role and interactions among epidermal proteins and lipids in normal epidermal function. Most variants have been identified in exomes by whole-exome sequencing, but pathogenic variants have been discovered in intronic regions and require more comprehensive investigations, including whole-genome sequencing.
Desquamation is the end result of proteolytic degradation of corneodesmosomes (the intercellular junctions within the stratum corneum) and is abetted by friction and cell hydration. Desquamation requires normal epidermal differentiation and depends on a gradient of pH, the presence of protease inhibitors, and the generation of hygroscopic molecules within the stratum corneum cells. Abnormalities in desquamation and differentiation usually result from abnormal corneocyte shedding (retention hyperkeratosis) or from increased epidermal cell proliferation (epidermal hyperplasia).
In addition to the stratum granulosum–based tight junction, the stratum corneum plays an important role in the epidermal barrier. The stratum corneum is composed of stacked corneocytes and surrounding highly hydrophobic lipid layers (lamellae) formed by the secretion of lamellar body contents at the interface of the stratum granulosum and the stratum corneum. Most of the mutations that lead to ichthyosis affect lipid metabolism or epidermal proteins, leading to barrier dysfunction and resulting in increased transepidermal water loss and decreased water-holding capacity. , Many of the features of ichthyosis are thought to be continuous compensatory attempts to restore the barrier (e.g., upregulated epidermal lipid synthesis, epidermal hyperproliferation, and inflammation with increased expression of Th17 cytokines). This homeostatic response likely allows survival of affected individuals but may also contribute to the ichthyosis phenotype.
Four major forms of nonsyndromic ichthyosis have been delineated, based largely on clinical and genetic characteristics:
Ichthyosis vulgaris, the most common ichthyosis, transmitted as an autosomal semi-dominant trait (meaning it can manifest when one allele is abnormal and to a greater extent when both alleles have a pathogenic gene variant)
Recessive X-linked ichthyosis (RXLI), expressed only in males and transmitted as an X-linked recessive trait
Keratinopathic ichthyosis, an autosomal dominant trait (most commonly epidermolytic ichthyosis [EI])
Autosomal recessive congenital ichthyosis (ARCI) (most collodion babies; exhibits a spectrum of characteristics from lamellar ichthyosis [LI] to congenital ichthyosiform erythroderma [CIE]).
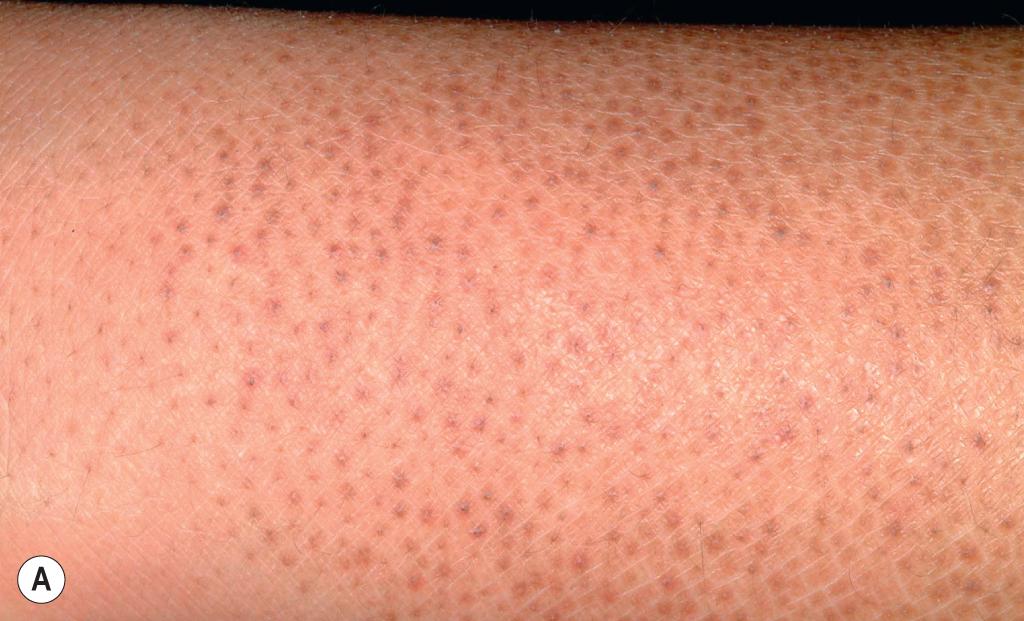
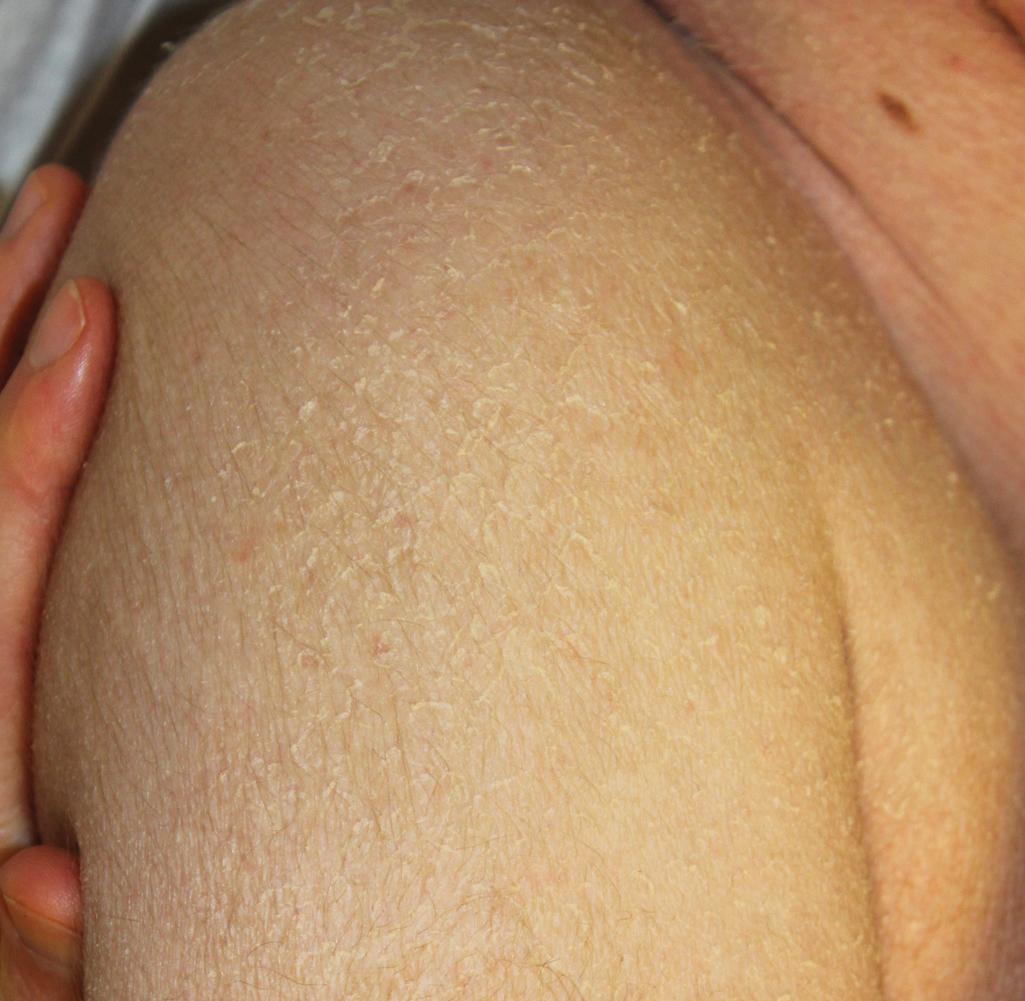
Ichthyosis vulgaris is by far the most common genetic form of ichthyosis, and the majority of individuals affected by the disorder are undiagnosed ( Table 5.1 ), either because of the minimal clinical manifestations (which are affected by climate) or because the condition is dismissed as dry skin. This disorder, which is not present at birth, may be noted after the first 2 to 3 months of life and often not until later in childhood. Ichthyosis vulgaris generally improves with age, in summer, and in warm moist environments. Scales are most prominent on the extensor surfaces of the extremities and are most severe in cold and dry weather. Scales on the pretibial and lateral aspects of the lower leg are large and plate-like, resembling fish scales ( Fig. 5.1 , A ); the flexural areas are characteristically spared. In other areas, small, white, bran-like scales may be seen. Scales tend to be darker on dark-skinned individuals. Scaling of the forehead and cheeks, common during childhood, generally diminishes and clears with age, but hyperlinearity and mild to moderate thickening of the palms and soles are characteristic ( Fig. 5.1 , B ). Discrete hyperkeratosis may occur on the elbows, knees, and ankles.
| Ichthyosis Vulgaris | X-Linked Recessive Ichthyosis | Keratinopathic Ichthyosis: Epidermolytic Ichthyosis | ARCI: Classic Lamellar Ichthyosis | ARCI: Classic Congenital Ichthyosiform Erythroderma | |
|---|---|---|---|---|---|
| Prevalence | 1:30 (1:12 with mutation) | 1:1500 males | 1:300,000 | 1:300,000 | 1:300,000 |
| Inheritance | Autosomal semi-dominant | X-linked recessive | Usually autosomal dominant | Usually autosomal recessive | Autosomal recessive |
| Onset | 2 months and beyond | 17% at birth; 83% by 1 year | Birth, with superficial blistering | Birth, as collodion baby | Birth, as collodion baby |
| Character of scales | Fine, white to larger scales, esp. on legs | Large, brown | Verrucous scale, superficial blisters | Large, plate-like scales | Fine white scaling overlying erythema |
| Localization | Can be generalized; relative sparing of flexures; hyperlinear palms | Accentuation at neck and behind ears; relative sparing of flexures | Generalized; especially at flexures and overlying joints | Generalized; ectropion, occasional alopecia, nail dystrophy | Generalized; ectropion; occasional alopecia |
| Distinct histologic features | Often shows decreased granular layer | None | Epidermolytic hyperkeratosis | Massive orthokeratosis; moderate acanthosis | More acanthosis |
| Molecular basis | Mutations in profilaggin (FLG) ; worse if both alleles | Deletions in ARSC1 (arylsulfatase C) | Mutations in KRT1 and KRT10 ; superficial form with mutations in KRT2 | Most commonly mutations in TGM1 ; rarely other types (see CIE) | Mutations in TGM1 ; ALOXE3; ALOX12B; NIPAL4; ABCA12; CYP4F22; PNPLA1; LIPN; CerS3; SDR9C7; SLC27A4 |
| Comments | Increased risk of atopic dermatitis and keratosis pilaris | Accumulation of cholesterol sulfate; FISH analysis to detect deletion; genital abnormalities rare; asymptomatic corneal opacities; contiguous gene deletion: with anosomia, retardation (Kallmann syndrome), and/or chondrodysplasia punctata | Superficial form shows more superficial blistering and much less thickening; secondary Staphylococcus aureus infection; palm/sole thickening predominantly with KRT1 mutation | Transglutaminase activity can be measured in skin samples | May be associated with neurologic abnormalities |
A reduced or absent granular layer in skin sections may help to differentiate ichthyosis vulgaris from other forms of ichthyosis. Given that filaggrin is the major protein of the granular layer, it is not surprising that mutations in FLG , the gene encoding for profilaggrin, the precursor of filaggrin, are responsible. In fact, FLG mutations are observed in 7.7% of Europeans and 3.0% of Asians, although not all manifest with ichthyosis vulgaris, given the dependence of its phenotype on environmental factors. Ichthyosis vulgaris is now known to be a semi-dominant condition, in contrast to the previous assumption of dominant inheritance, i.e., manifestations are seen with a mutation on one allele in 1:12 Europeans but are worse if null mutations are on both alleles (probability, 1:576). The cleaved product of profilaggrin, filaggrin, plays an important role in linking a protein (involucrin) and lipids (ceramides) in the corneocyte envelope (see Chapter 3 ). In addition, filaggrin breaks down to amino acid metabolites that increase skin hydration (a natural moisturizing factor). Without filaggrin, the epidermis cannot provide normal barrier function; transepidermal water loss is increased, leading to xerosis; and the ingress of foreign substances (such as allergens and pathogens) occurs more readily, thereby increasing the risk of exposure to triggers of atopy.
Patients with ichthyosis vulgaris often have an atopic background with a tendency toward atopic dermatitis (AD) (see Chapter 3 ). , The risk is highest in individuals with two mutated alleles. Overall, up to 30% of patients of a Northern European background with AD and more than 20% of Japanese with AD , have FLG mutations. FLG mutations are uncommon in darker-skinned populations, even with ichthyosis vulgaris, although FLG2 variants have been correlated with more persistent AD in Blacks. Filaggrin-2 is expressed in the stratum granulosum and is essential for proper epidermal cornification (see Peeling Skin Syndrome later). Signs of AD may be seen before other clinical signs of ichthyosis vulgaris, and most individuals with the condition never show evidence of AD. Individuals with AD and a FLG mutation tend to have more severe and more persistent AD, particularly if FLG mutations occur on both alleles. The occurrence of other forms of atopy (asthma, allergic rhinitis) typically follows the onset of AD in children with FLG mutations, suggesting that the barrier defect of ichthyosis and the occurrence of AD are a driving force toward the development of other atopic conditions. Mechanistic studies have shown increased penetration of allergens and chemicals in filaggrin-deficient skin, and epidemiologic studies have found higher levels of hand eczema, irritant contact dermatitis, and nickel sensitization in association with FLG variants.
The diagnosis of ichthyosis vulgaris should be considered in patients with AD who show large scales, particularly on the extensor aspects of the extremities; examination of the palms and soles shows the hyperlinearity. Keratosis pilaris, which is commonly associated with ichthyosis vulgaris and atopy (see Chapter 3 and Chapter 7 , Figs. 7.25 and 7.26 ), is most predominant on the upper arms, buttocks, and thighs.
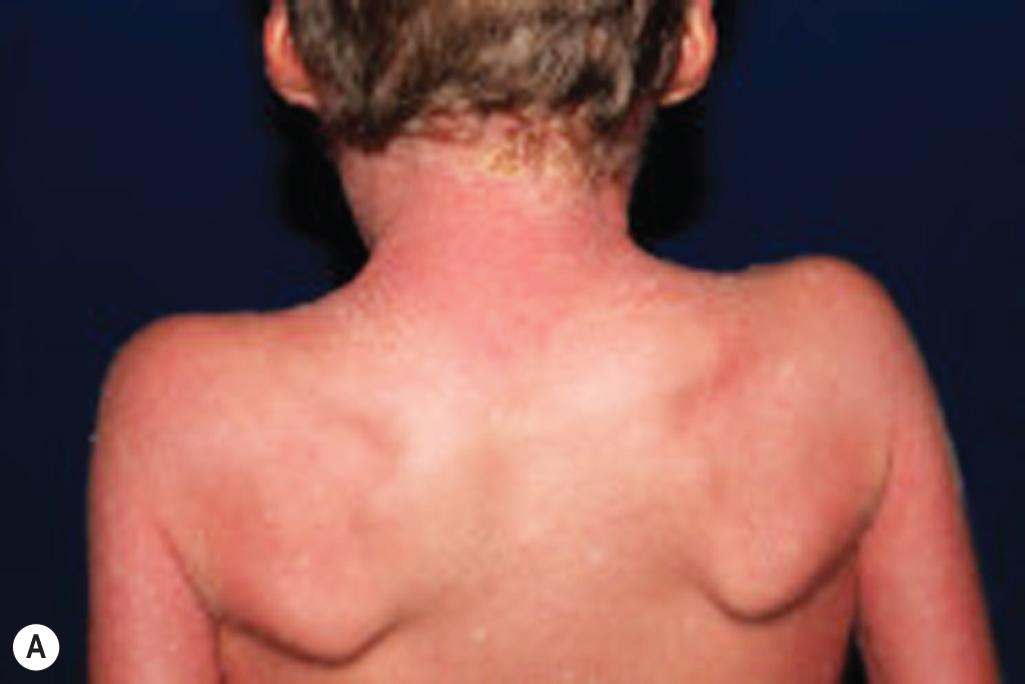
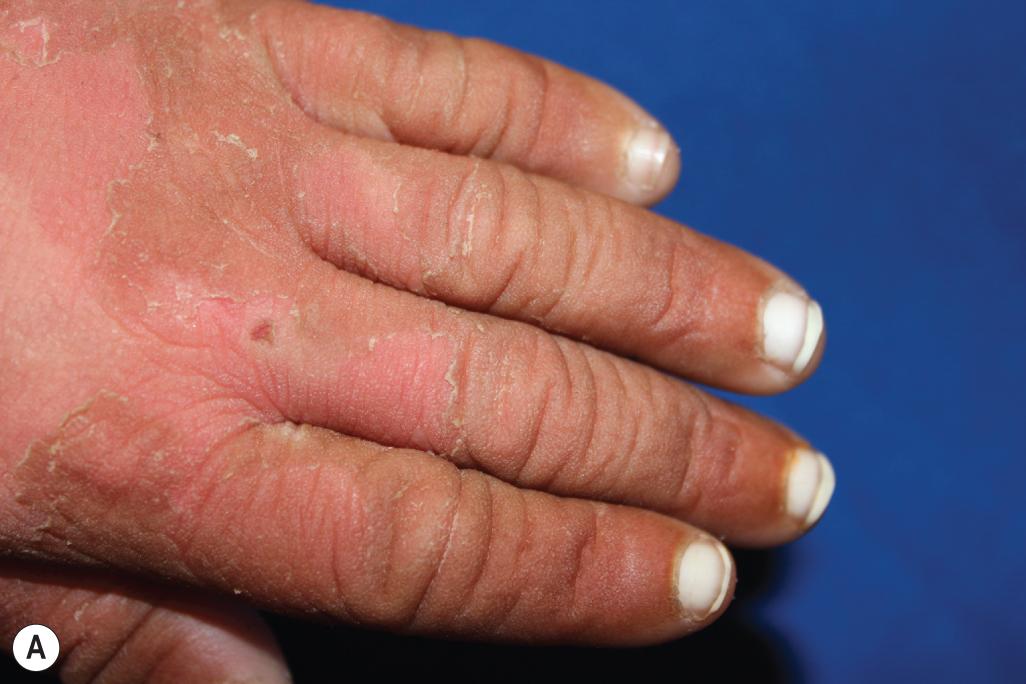
An autosomal recessive form of ichthyosis vulgaris can result from biallelic mutations in CASP14 , which encodes caspase 14, the enzyme that degrades filaggrin monomers in the stratum corneum to hygroscopic amino acids. In addition, mutations in ASPRV1 , which encodes skin aspartic protease (SASPase), lead to a severe ichthyosis vulgaris–like disorder with large, plate-like scales ( Fig. 5.2 , A ) and hyperlinear thickened palms ( Fig. 5.2 , B ) that has recently been described in humans. SASPase enables profilaggrin-to-filaggrin processing, and the described missense mutations reduce this processing.
Emollients may improve dry skin but do not normalize the epidermal gene expression profile of filaggrin. Excessive exposure to factors that decrease skin barrier functions and increase the risk of AD should be avoided. Individuals with ichthyosis vulgaris should be protected against neonatal exposure to cats to prevent AD and should abstain from smoking to prevent asthma.
Although rare in children, “acquired” ichthyotic scaling has been described in patients with nutritional disorders such as hypovitaminosis or hypervitaminosis A, hypothyroidism, sarcoidosis, dermatomyositis, leprosy, tuberculosis, human immunodeficiency virus (HIV) infection, and neoplastic disorders, particularly lymphomas. It should be noted that these forms were described before the availability of testing for FLG /profilaggrin mutations; given the many undiagnosed patients, it is possible that the underlying condition increased the risk of manifesting the ichthyosis in an individual with a FLG mutation. Pityriasis rotunda, a rare variant of acquired ichthyosis, is characterized by asymptomatic, circular or oval, brown scaly patches on the trunk or extremities. Seen primarily in individuals of Japanese, African, and West Indian origin, its occurrence in Whites is extremely rare. The condition may at times be associated with an underlying disorder, may follow pregnancy, or may be familial. In contrast to ichthyosis vulgaris, pityriasis rotunda is chronic, is resistant to treatment, and tends to improve only when the underlying disorder is treated.
RXLI occurs in 1:1500 males and results from a mutation (deletion of the entire gene in 90%) in the ARSC1 gene that encodes steroid sulfatase (STS), also known as arylsulfatase C (see Table 5.1 ). The disorder has rarely been described in females who have Turner syndrome or carry the mutation on both alleles. Female carriers do not tend to show ichthyosis because the affected gene is located at the distal tip of the X chromosome, a location that escapes X-inactivation. Thus rather than having random inactivation of one of the X chromosomes, as dictated by the Lyon hypothesis, both of the alleles are expressed in every cell, providing sufficient enzyme. STS normally is concentrated in lamellar bodies and secreted into the spaces between stratum corneum cells. It degrades cholesterol sulfate, generating cholesterol for the epidermal barrier. Cholesterol sulfate itself is an epidermal protease inhibitor, so STS deficiency prevents normal degradation of the stratum corneum desmosomes and leads to corneocyte retention. ,
Low placental production of estrogens and elevated sulfated steroid levels have been described in the urine of mothers of boys with RXLI, associated with a difficult or prolonged labor and failure to have cervical dilation. Deletion of both ARSC1 and a contiguous gene (up to 10% of patients) results in both ichthyosis and Kallmann syndrome (associated with mental retardation, hypogonadism, and anosmia; deletion of KALI gene), X-linked recessive chondrodysplasia punctata (bone dysplasia with stippled epiphyses; deletion of arylsulfatase E/ ARSE ), and/or retardation with autism (neuroligin 4/ NLGN4 ). , RXLI is commonly predicted prenatally by chromosomal microarrays, which have a high sensitivity for the detection of microdeletions. The presentation can be highly variable, and in one study, most patients had “dry” skin or “eczema” rather than recognized ichthyosis. Fluorescent in situ hybridization (FISH) analysis for the STS gene is another way to show the gene deletion. RXLI can be confirmed by reduced arylsulfatase C activity in leukocytes or by elevated blood levels of cholesterol sulfate and increased mobility of β-lipoproteins.
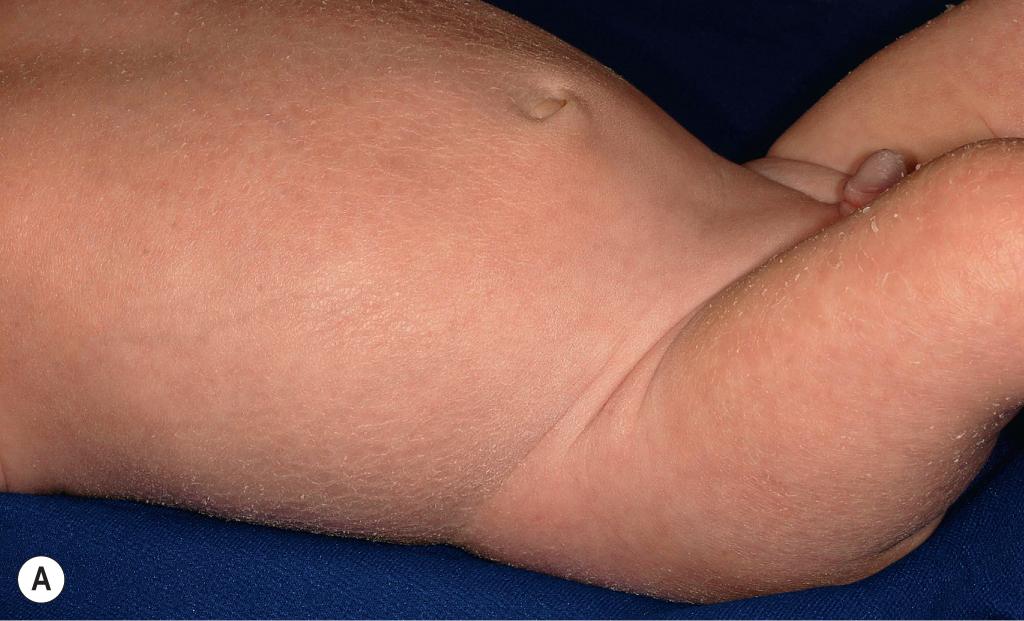
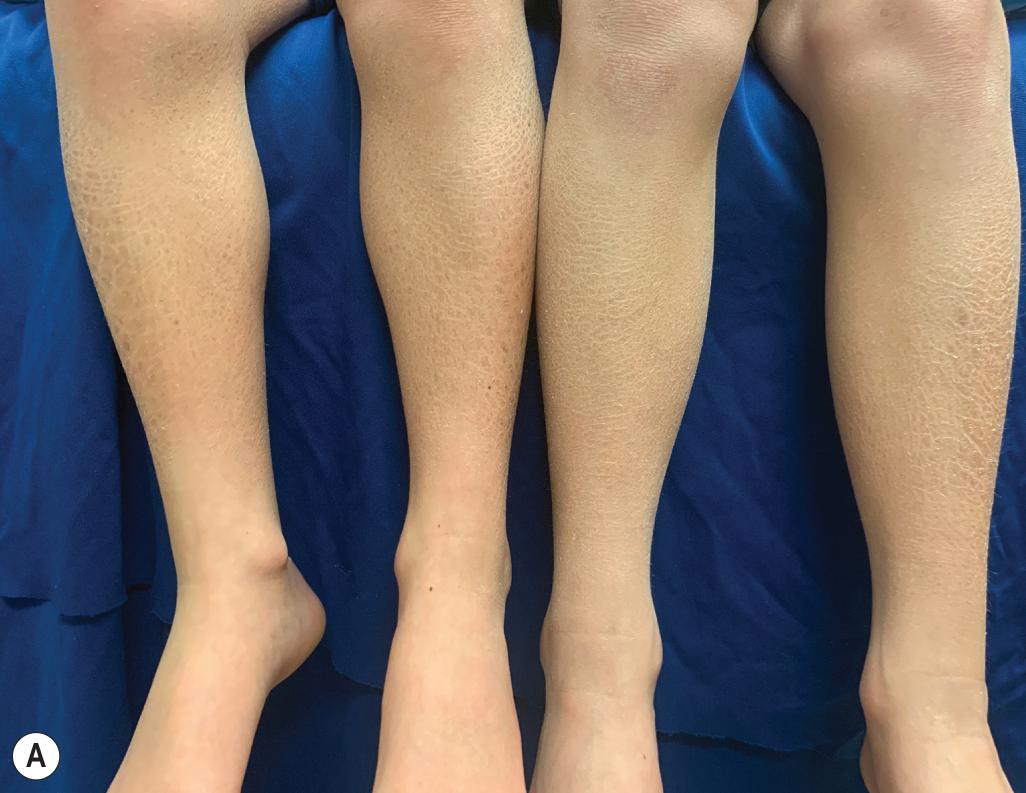
RXLI typically manifests within the first 3 months of life. Approximately 17% of affected individuals show scaling at birth, often in the form of a mild collodion-like membrane. Most develop scaling during the first 6 months of life ( Fig. 5.3 ). The severity of the scaling can range from mild to severe. This form of ichthyosis generally involves the entire body, with accentuation on the scalp, neck, abdomen, back, front of the legs, and feet but spares the palms, soles, central face, and flexural areas ( Fig. 5.4 , A ). Scales may be small to large and tend to be brown in coloration and darker in darker-skinned patients. The sides of the neck often appear dark and unwashed ( Fig. 5.4 , B ). Patients may shed or molt their scales episodically, particularly in the spring and fall. RXLI can be difficult to distinguish from ichthyosis vulgaris and may occur concurrently; RXLI is more severe with concomitant FLG mutation, and palmar hyperlinearity is a diagnostic clue.
Boys with X-linked ichthyosis rarely have hypogonadism and/or cryptorchidism; testicular cancer has been described in one patient. Deep corneal opacities may be found in approximately 50% of affected adult males and less often in female carriers of this disorder. The opacities, easily detectable by slit-lamp examination, are discrete and diffusely located near the Descemet membrane or deep in the corneal stroma. Although they are a marker for the disorder in older patients, these opacities do not affect vision.
The features of RXLI may be seen in patients with autosomal recessive multiple sulfatase deficiency ( Fig. 5.5 ), a syndromic form of ichthyosis characterized by features of mucopolysaccharidoses, metachromatic leukodystrophy, X-linked recessive chondrodysplasia punctata, and RXLI. The disorder results from mutations in sulfatase-modifying factor 1 (SUMF1) , which encodes the α-formylglycine–generating enzyme required for posttranslation modification of sulfatases. , Progressive neurologic deterioration, a feature of the metachromatic leukodystrophy, usually leads to death during infancy.
Keratinopathic ichthyosis includes the epidermolytic forms of ichthyosis associated with keratin gene mutations . The major subgroups are EI (formerly called bullous CIE, Brocq type ) and superficial EI (formerly called ichthyosis bullosa of Siemen s). The designation epidermolytic hyperkeratosis is a histologic description that is not specific to this group of disorders, although it traditionally referred to EI.
Mutations are usually point mutations that lead to an abnormal, but full-length keratin that incorporates into the keratin filament. The resultant keratin network functions poorly, leading to skin cell collapse and clinical blistering, especially in response to trauma. The thickening of skin is thought to be compensatory to protect against blistering, but has also been linked to abnormal lamellar-body secretion. Virtually all forms are inherited in an autosomal dominant manner, although EI may rarely be autosomal recessive. EI results from mutations in KRT1 or its partner in intermediate filament formation, KRT10 ; both are expressed throughout the suprabasal layers of epidermis. Superficial EI is caused by mutations in KRT2 , which is only expressed in more superficial epidermis and also partners with keratin 10.
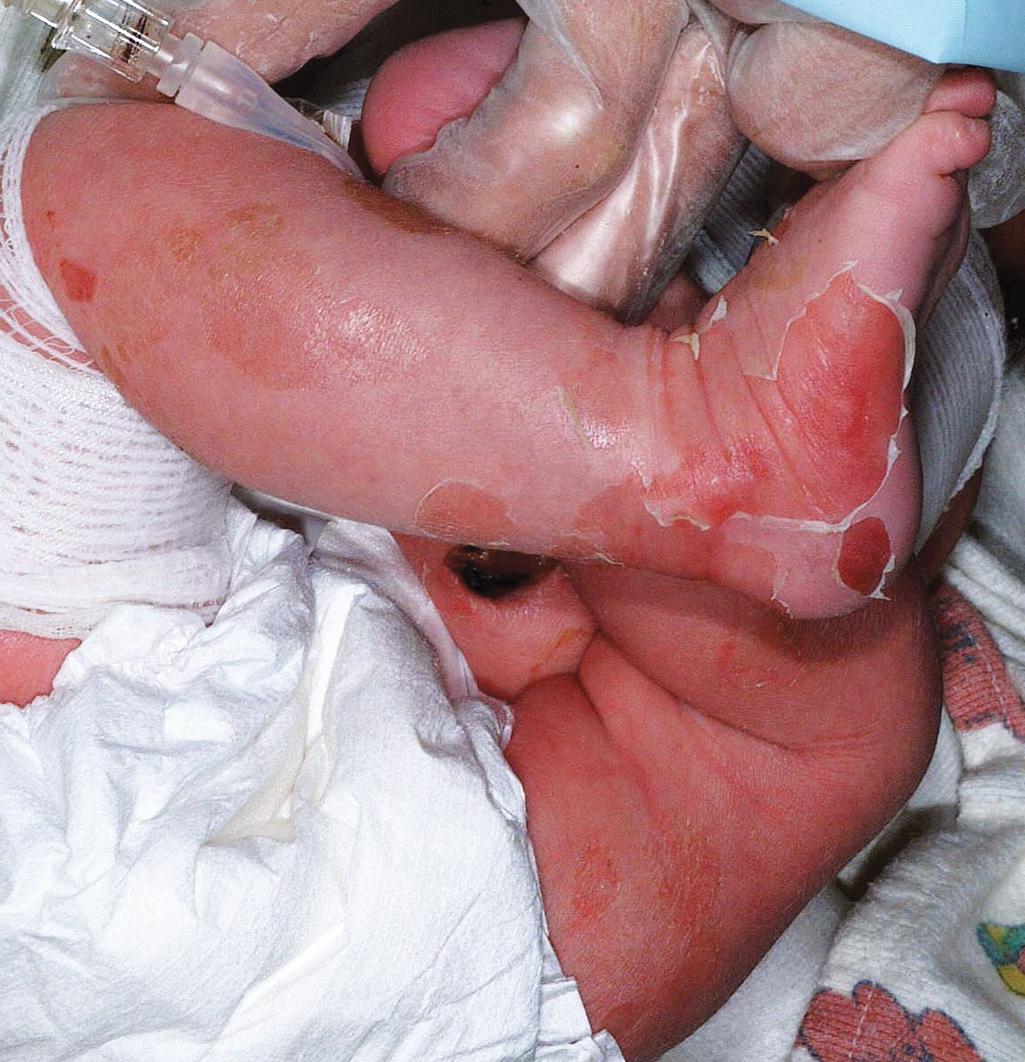
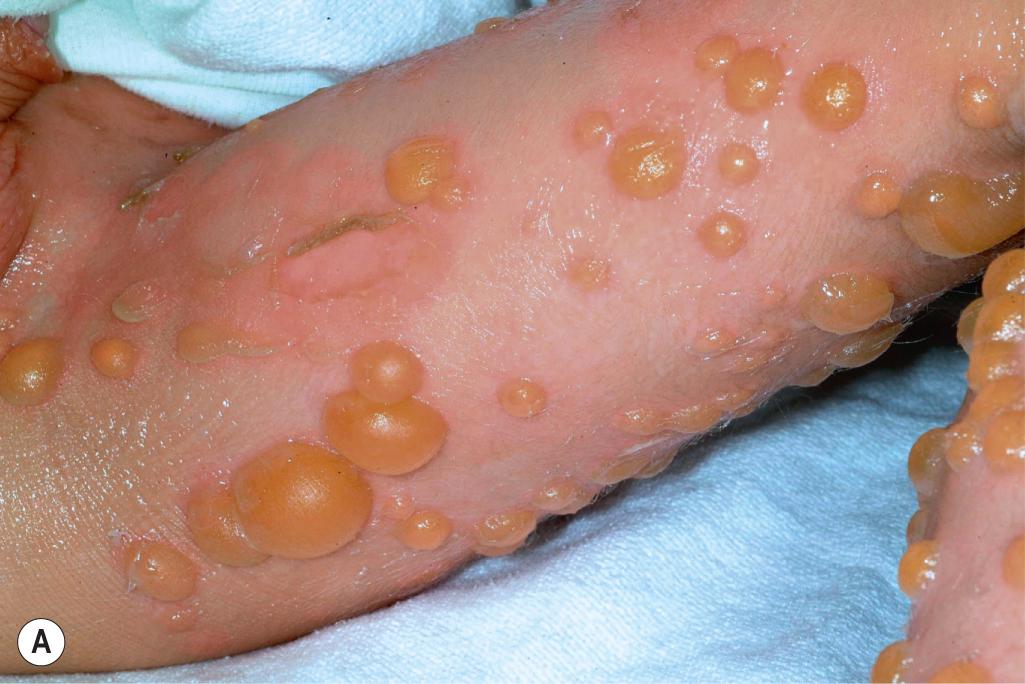
EI affects approximately 1 in 300,000 individuals, and 50% of patients have new mutations. The skin is red and may be tender at birth. Superficial bullae generally appear within the first week of life (often within a few hours after delivery; Fig. 5.6 ) and may be confused with those of epidermolysis bullosa (see Chapter 13 ) or with staphylococcal scalded skin syndrome (see Chapter 14 ). Skin thickening may be present at birth, sometimes even with the typical scale of EI, but often appears during the months after birth ( Fig. 5.7 ) with only subtle thickening during the neonatal period, especially on the elbows and knees. The blisters are primarily seen in areas of trauma and can occur in crops. They vary from 0.5 cm to several centimeters in diameter. They tend to heal quickly, consistent with their superficial location. When ruptured, they discharge clear fluid and leave raw denuded areas. Secondary bacterial infection, especially with Staphylococcus aureus , is commonly associated with this disorder.
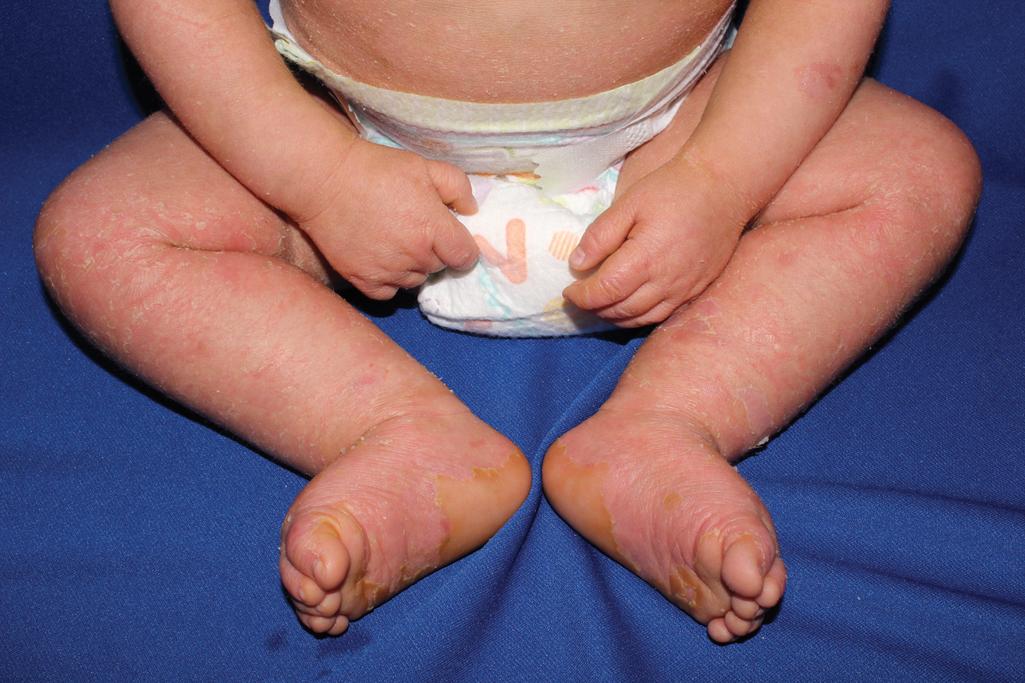
Verruciform grayish-brown scales eventually cover most of the skin surface; the flexural creases and intertriginous areas show particularly marked involvement ( Figs. 5.8 and 5.9 ). Palms and soles have varying degrees of thickening and scaling ( Fig. 5.10 ), but much more marked involvement often occurs in individuals with mutations in KRT1 , because keratin 9 expression in the palms and soles can compensate for abnormal keratin 10 expression, but expression of KRT1 (which is the partner for both keratins 9 and 10) remains critical. Facial involvement may occur, but ectropion does not. Although scalp involvement may result in nit-like encasement of hair shafts, the hair, eyes, teeth, and nails are normal. A disagreeable body odor is commonly associated with severe forms of this disorder due to the thick, macerated scale and overgrowth of bacteria and, less often, candida. Skin biopsy specimens show marked hyperkeratosis with lysis of the epidermal cells above the basal cell layer (epidermolytic hyperkeratosis) leading to the bullae. Keratolytic agents are often poorly tolerated in keratinopathic forms of ichthyosis and can increase skin fragility.
The epidermolytic hyperkeratotic form of epidermal nevus shows a histologic appearance identical to that of epidermolytic ichthyosis, but is not associated with skin fragility or a blistering tendency (see Chapter 9 ). This form of nevus represents a mosaic condition in which the affected skin, but not the normal intervening skin, carries a mutation in KRT1 or KRT10 ( Fig. 5.10 ). Individuals with more extensive forms of the epidermolytic hyperkeratotic form of epidermal nevus can have offspring with generalized epidermolytic ichthyosis, reflecting a germline mutation. Prenatal diagnosis can be performed in at-risk families.
Mutations in regions of KRT1 and KRT10 that are less critical than the α-helical rod domain can lead to milder forms of EI with thickening limited to the fold areas. Mutations in KRT1 and KRT10 can also cause an annular variant (annular EI), , in which annular erythematous polycyclic scaling plaques on the trunk and extremities slowly enlarge, resolve, and later recur.
Ichthyosis Curth–Macklin (formerly called ichthyosis hystrix ) has its onset of manifestations during early childhood as progressively worsening diffuse or striate palmoplantar keratoderma (PPK) that can be associated with deep fissuring, flexural contracture, and digital constriction. Affected individuals develop characteristic “porcupine quill–like” verrucous yellow-brown scaling, especially on the hands and feet and overlying the large joints ( Fig. 5.11 ). Binuclear cells and pathognomonic concentric perinuclear “shells” of aberrant keratin are characteristic ultrastructural findings. KRT1 mutations have been described in some, , but not all, affected patients.
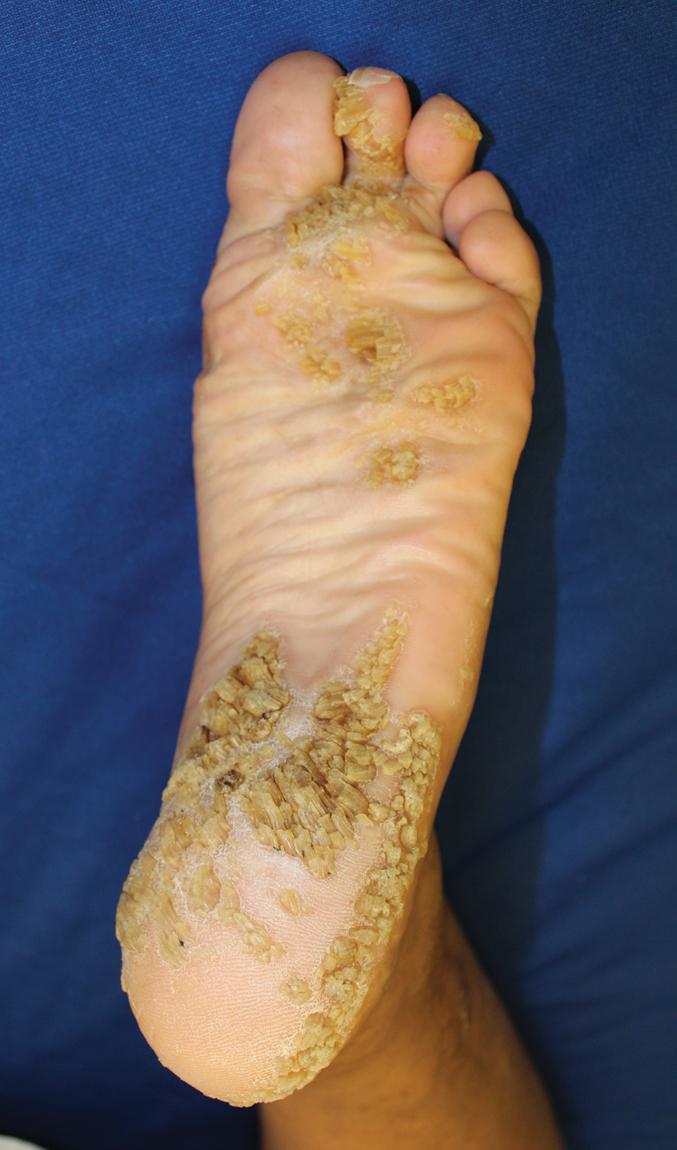
Superficial EI shows milder thickening and more superficial blistering. The palms and soles are minimally thickened, if at all. Although large, tense bullae can occur ( Fig. 5.12 , A ), in general, the appearance of blisters has been likened to molting (mauserung phenomenon) and can appear as subtle peeling skin, especially by adolescence and adulthood ( Fig. 5.12 , B ), because of the superficial location of the cleavage plane. Accordingly, less hyperkeratosis is seen in sections of skin biopsies, and the lysed areas of epidermis only begin halfway up the stratum spinosum. Affected individuals may be misdiagnosed as mild EI clinically, but the limited localization of the epidermolysis histologically and the finding of mutations in KRT2 by molecular genetic testing can distinguish the disorders. ,
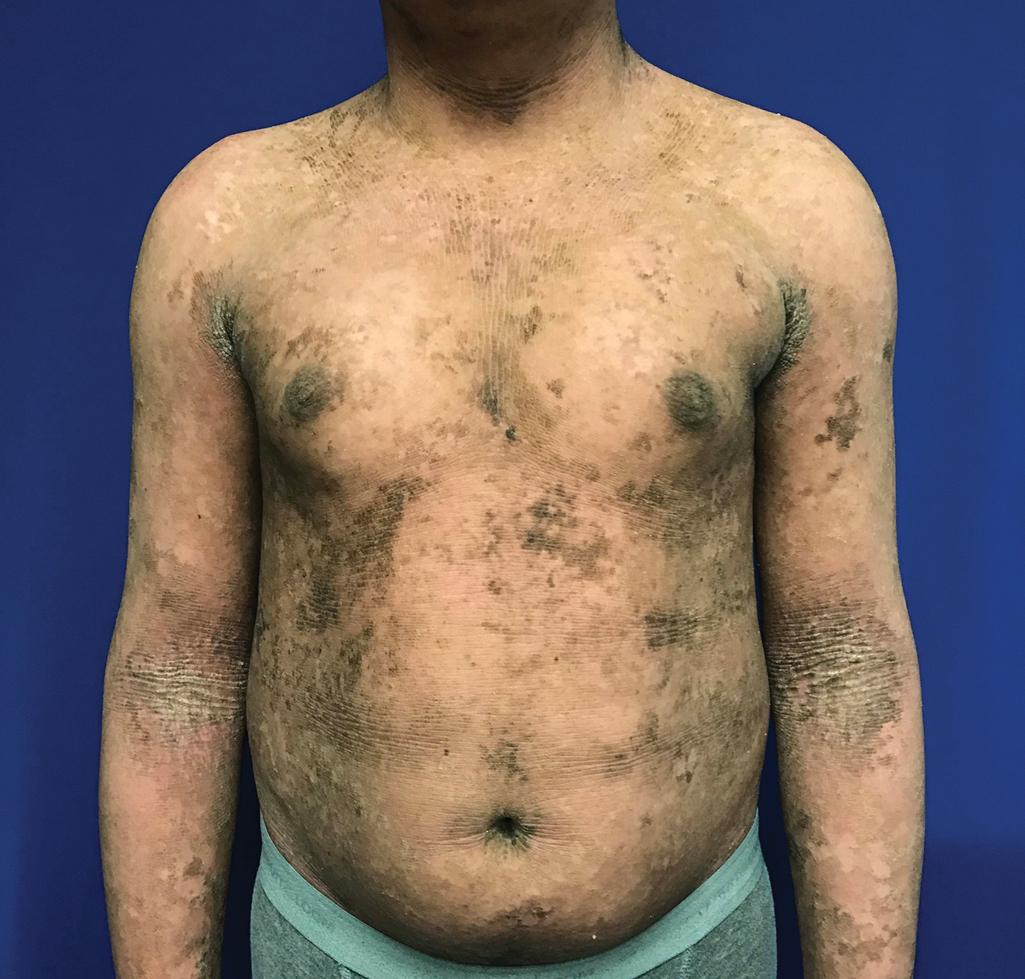
Ichthyosis with confetti (IWC), also called congenital reticular ichthyosiform erythroderma or ichthyosis variegata, is an autosomal dominant keratinopathy characterized by generalized erythroderma and lack of blistering, and this is often mistaken for CIE. IWC is distinguished by its progressive reversion of affected areas to normal-appearing skin in a “confetti” pattern of macules. These specific mutations, typically frameshift mutations in exon 6 or 7 of KRT1 or KRT10, predispose to postzygotic translocations that allow protein expression of the mutated allele and correct the phenotype. In general, KRT1 mutations are associated with the “confetti” first appearing during infancy, whereas KRT10 mutations tend to manifest the cleared areas beginning during childhood or even young adulthood ( Fig. 5.13 , A and B ). Other major features include malformation of ears, hypoplasia of mammillae, and dorsal acral hypertrichosis. Severe cases of pityriasis rubra pilaris from CARD14 mutations may resemble IWC because of the extensive erythroderma with areas of sparing (see Chapter 4 ); severe manifestations of CARD14 mutations can include ectropion and PPK, requiring differentiation from CIE (see later).
Collodion baby is a descriptive term for an infant who is born with a membrane-like covering resembling collodion ( Fig. 5.14 ). , The collodion baby is not a specific disease entity, but rather a phenotype common to several forms of ichthyosis. At least 65% of collodion babies have ARCI (see Autosomal Recessive Congenital Ichthyosis), a group of genetically distinct forms of ichthyosis with overlapping clinical features ; some infants in this group (5% to 6%) shed their collodion membranes and show apparently normal skin (self-healing collodion baby [SHCB]). , Having a collodion membrane at birth, however, is not a necessary feature for ARCI. Less often, collodion babies shed their membranes and show features of syndromic forms of ichthyosis (Conradi–Hünermann–Happle [CHH] syndrome, trichothiodystrophy [see Chapter 7 ], or RXLI). There is one case of a collodion membrane associated with medication use in a mother who received infliximab before and throughout the course of the pregnancy; although the collodion membrane shed and the skin appeared normal 1 year after birth, no genotype analysis was performed to consider a mild variant of ichthyosis rather than resulting from medication use during pregnancy.
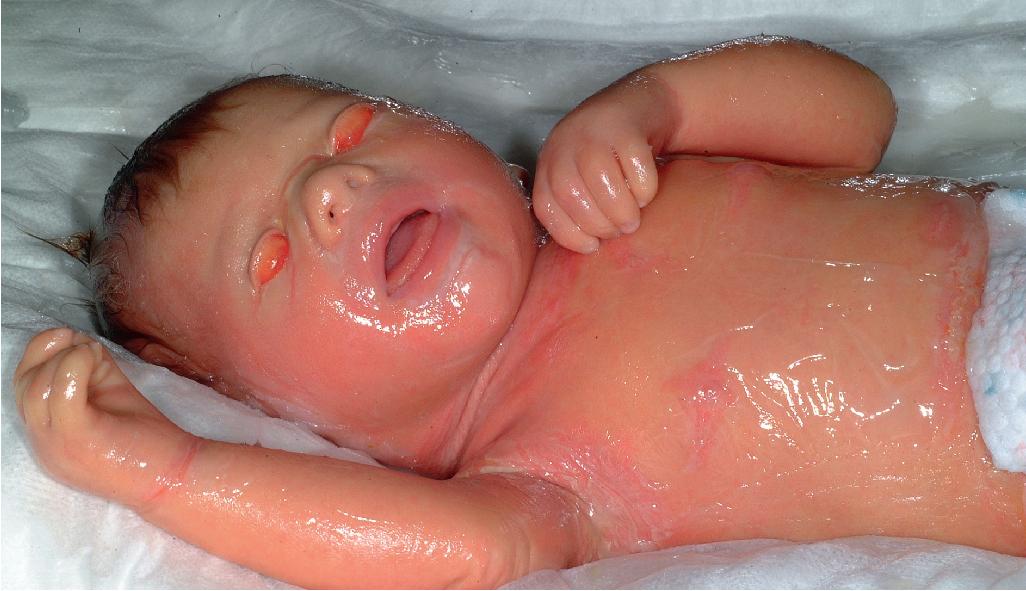
At birth, collodion babies are completely covered by a cellophane- or oiled parchment–like “membrane” that, due to its tautness, may distort the facial features and extremities. Thus peripheral edema with digital constriction, flattened ears, and bilateral eversion of the eyelids, lips, and at times the vulva often cause affected infants to resemble one another during the first few days of life. Among the problems facing these infants are an inability to suck properly, respiratory difficulty because of restriction of chest expansion by the thick membrane, cutaneous and systemic infection, and aspiration pneumonia. Despite the thickening of the stratum corneum, this membrane is a poor barrier, leading to excessive transcutaneous fluid and electrolyte loss, hypernatremic dehydration, increased metabolic requirements, and temperature instability.
Collodion babies are often born prematurely. Patients born as collodion babies may develop neurologic abnormalities. These should be interpreted in the context of history. Although most likely related to premature birth, syndromic disorders should also be considered. For example, patients with Sjögren–Larsson syndrome are very occasionally born with a collodion membrane and may resemble LI during childhood but tend to progressively develop neurologic features.
Supportive care is of primary importance in the management of collodion babies. , They are traditionally managed in a humidified incubator with special attention given to the prevention of temperature instability, sepsis, and fluid and electrolyte imbalance. Systemic antibiotic therapy should be initiated if infection is detected, but not prophylactically. Desquamation is encouraged by the application of emollients rather than manual debridement; given the poor cutaneous barrier and potential toxicity, use of keratolytic agents should be avoided during the first several months of life.
ARCI encompasses a wide range of clinical phenotypes that range from classic harlequin ichthyosis (HI) and nonbullous CIE to classic LI. The same individual may show a range of overlapping clinical features; for example, retinoid treatment may decrease the lamellar scaling but increase visible erythroderma. Failure to thrive may be a feature of ARCI, especially if severe, and short stature may ensue. Overall, ARCI occurs in approximately 1 in 100,000 to 300,000 live births. The diagnosis is based on clinical findings; biopsy is not helpful unless needed to exclude alternative diagnoses. The features of the ARCI group of disorders usually persist throughout the affected individual’s lifetime. Many affected individuals complain of associated pruritus. Severely affected patients tend to experience hypohidrosis, which may lead to heat intolerance, with hyperpyrexia and heat exhaustion during periods of hot, humid weather and vigorous physical exercise. Although the hypohidrosis may reflect obstruction of eccrine glands by hyperkeratosis, persistence with ameliorative retinoid therapy suggests an eccrine gland/duct developmental abnormality in many affected with ichthyosis. Squamous cell carcinomas have been reported at a younger age, but are rare.
Mutations in 12 known genes have been shown to result in the ARCI phenotype, and most of these mutated genes have been related to the CIE phenotype (see Table 5.1 ). A few cases of autosomal dominant LI have been described. The discovery of the underlying genetic basis in families with ARCI has facilitated the prenatal diagnosis based on genotyping rather than the riskier diagnosis by fetal skin biopsy.
Overall, 34% to 55% of individuals with ARCI have mutations in the gene encoding transglutaminase 1 (TGM1) , and these patients typically feature the LI phenotype. TGM1 crosslinks several proteins to form the cornified envelope surrounding corneocytes. In patients missing transglutaminase, transglutaminase activity is undetectable in frozen skin specimens.
The 11 genes linked to ARCI are largely involved in synthesis and metabolism of the intracellular lipids of the stratum corneum, including the critical epidermal ω-O-acylceramides from N-acyl fatty acids ( Table 5.2 ). HI is the most severe form of CIE and has been shown to result from nonsense mutations in the gene encoding the ABCA12 transporter. , Missense mutations in ABCA12 cause a milder CIE phenotype than null type mutations. A deficiency of ABCA12 perturbs lipid transport, formation of lamellar bodies, keratinocyte terminal differentiation, and transfer of proteolytic enzymes required for normal desquamation. In addition, genes encoding lipoxygenases, ALOX12B (encoding 12(R)-lipoxygenase) and ALOXE3 (encoding lipoxygenase-3), , are often mutated. Other genes with biallelic mutations leading to ARCI are LIPN , CYP4F22, PNPLA1, , SLC27A4, NIPAL4, and CERS3 , Studies in PNPLA1-deficient mice and keratinocytes suggest that topical application of ω-O-acylceramides could be a replacement therapy for these forms of CIE. Other genes found to be mutated in ARCI include SDR9C7 , and SULTB1 (see Table 5.2 ). Although biallelic mutations in CAPN12 (encoding calpain 12) and CASP14 (encoding caspase 14) also are autosomal recessive and congenital, these disorders are not associated with collodion membranes and more closely resemble exfoliative ichthyosis (superficial exfoliation overlying intense erythroderma) and ichthyosis vulgaris (fine, white scaling without erythroderma), respectively, and thus are not included here as part of ARCI.
| Gene (% affected) | Gene Product | Function | Comments |
|---|---|---|---|
| ABCA12 (9%) | ABCA12 transporter | Lipid transport for formation of lamellar bodies and provision of intracellular lipids of the stratum corneum; deficiency also leads to premature terminal differentiation and normal transfer of proteolytic enzymes for desquamation | Nonsense mutations: harlequin ichthyosis; missense mutations: more severe CIE |
| ALOX12B (7%) | 12(R)-lipoxygenase | Oxidizes acylglucosylceramide (to be a substrate for ω-O-acylceramide) | |
| NIPAL4 (6%) | Ichthyin | Needed to convert ω-O-free fatty acids to ω-O-fatty acid-CoA in the formation of ω-O-acylceramides from N-acyl fatty acids | |
| ALOXE3 (3%) | Lipoxygenase-3 | Oxidizes acylglucosylceramide (to be a substrate for ω-O-acylceramide) | |
| PNPLA1 (3%) | Patatin-like phospholipase domain containing 1 | Esterification of ω-hydroxyceramides to acylceramides | Some patients with PNPLA1 mutations have a phenotype that could be confused with EKV and is not generalized |
| CYP4F22 (2%) | Cytochrome P450 family 4 subfamily F member 22 | Epidermal ω-hydroxylase that converts longer chain (C28-C34) N-acyl fatty acids to ω-hydroxy-free fatty acids | |
| LIPN (<1%) | Lipase N | Adds a ω-hydroxyl group to oxidized acylglucosylceramide to make ω-acylglucosylceramide | Ichthyotic changes may begin during early childhood |
| SLC27A4 (<1%) | Fatty acid transport protein 4/FATP4 | Fatty acid transporter that associates with ichthyin (for triglyceride and ω-hydroxy-fatty acid-CoA synthesis) | Prematurity, polyhydramnios, perinatal respiratory distress, and thick white vernix-like scaling at birth that can be confused with KID syndrome; atopy diathesis |
| CERS3 (<1%) | Ceramide synthase 3 | Synthesis of ω-hydroxy-ceramide from ω-hydroxy-fatty acid-CoA | |
| SDR9C7 (<1%) | Short chain dehydrogenase/reductase family 9C member 7 | Converts retinal to retinol; affects vitamin A metabolism and terminal differentiation | Usually are not born with collodion membrane |
| SULTB1 (<1%) | Sulfotransferase family 2B member 1 | Cytosolic sulfotransferase important for making cholesterol sulfate from cholesterol |
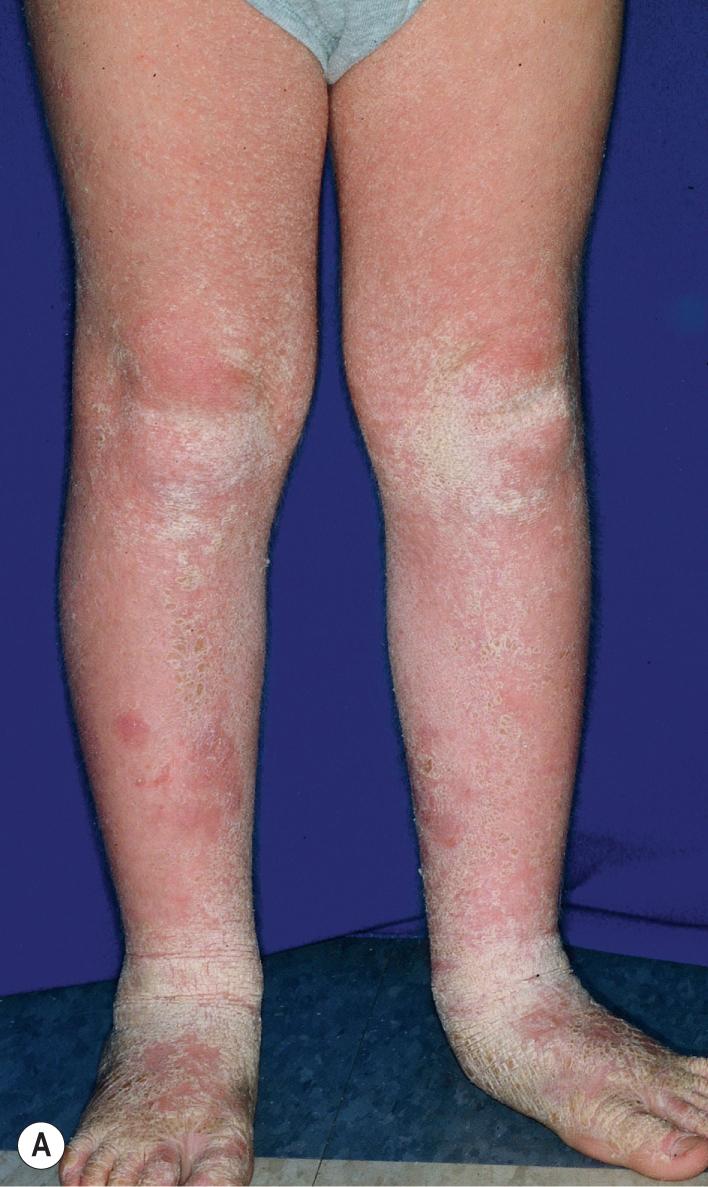
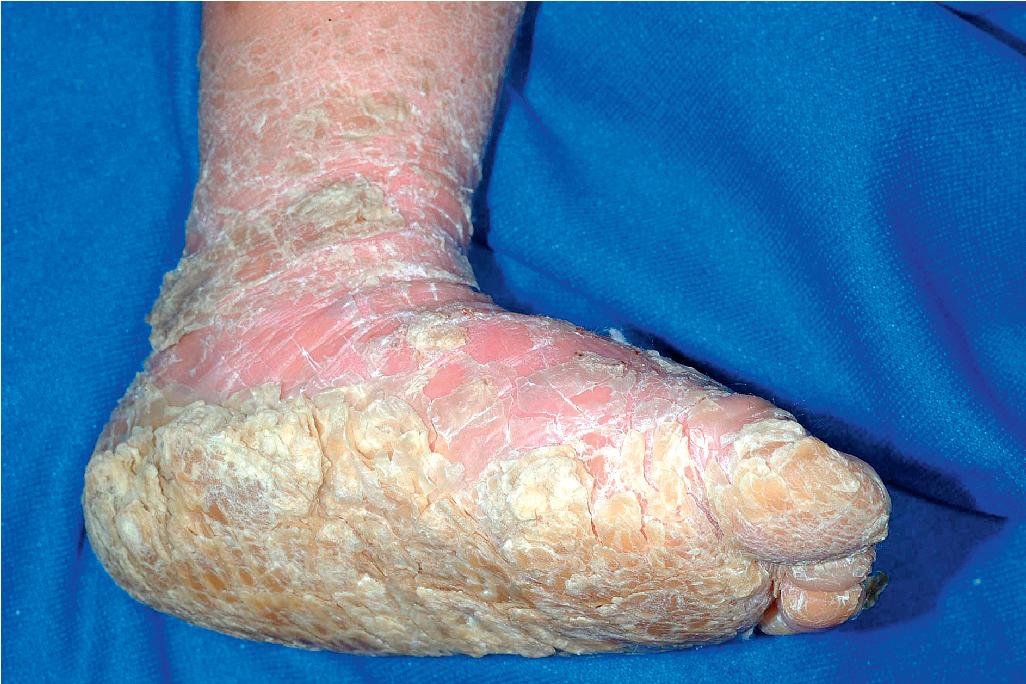
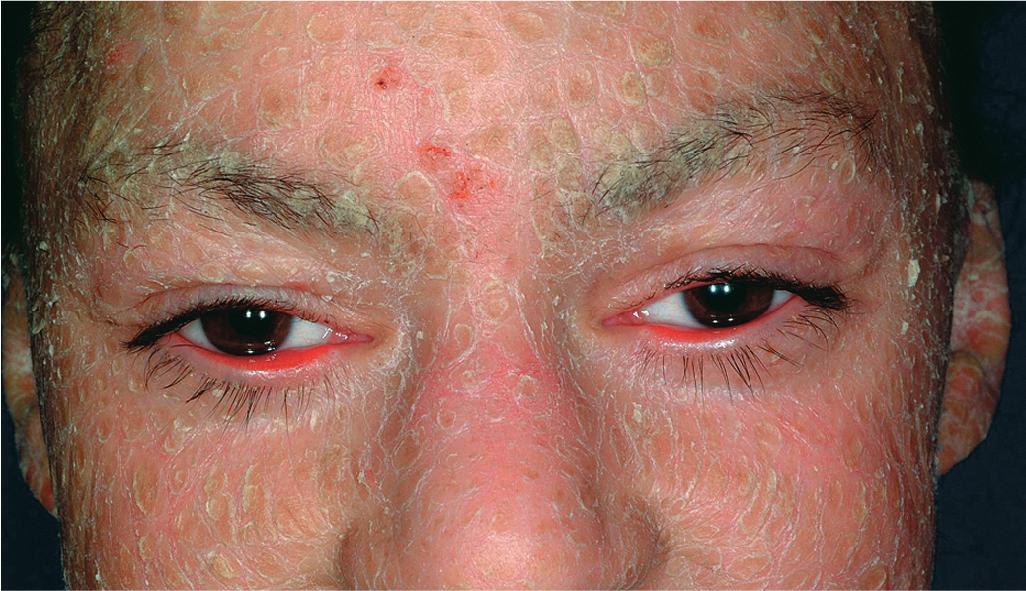
The clinical features of the classic LI phenotype may range from very mild to severe. Individuals who show the greatest severity of LI have large, lamellar, plate-like scales with relatively mild underlying erythroderma; ectropion (eversion of an edge or margin of the eyelid resulting in exposure of the palpebral conjunctiva) ( Fig. 5.15 ); and mild eclabium (eversion of the lips) (see Table 5.1 ). Lamellar scales are large, quadrangular, yellow to brown-black, often thick, and centrally adherent with raised edges resembling armor plates (hence the term lamellar ) ( Fig. 5.16 ). Scales are most prominent over the face, trunk, and extremities, with a predilection for the flexor areas. Cheeks are often red, taut, and shiny; more scales appear on the forehead than on the lower portion of the face. The palms and soles are almost always affected in LI; severity varies from increased palmar markings to a thick keratoderma with fissuring. Digital involvement may lead to pseudoainhum (constricting bands of the digits) and autoamputation. The scalp is scaly ( Fig. 5.17 ) with variable alopecia, which can be extensive and scarring. Involvement of the nails is variable. They may be stippled, pitted, ridged, or thickened, often with marked subungual hyperkeratosis.
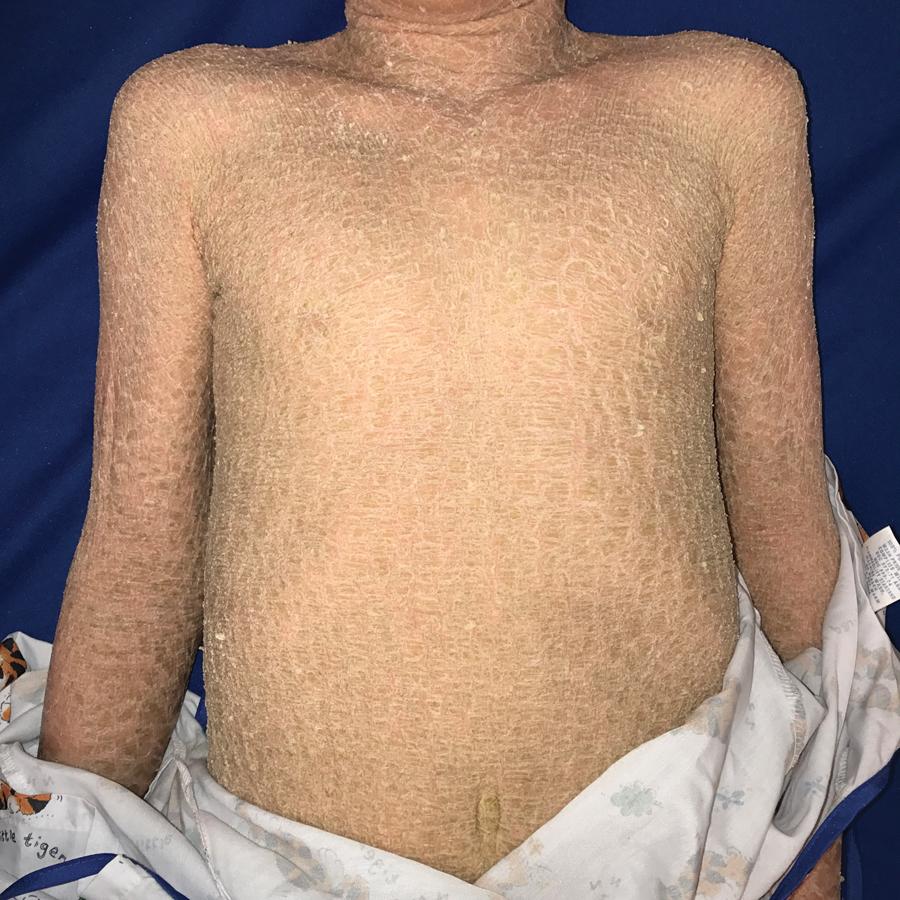
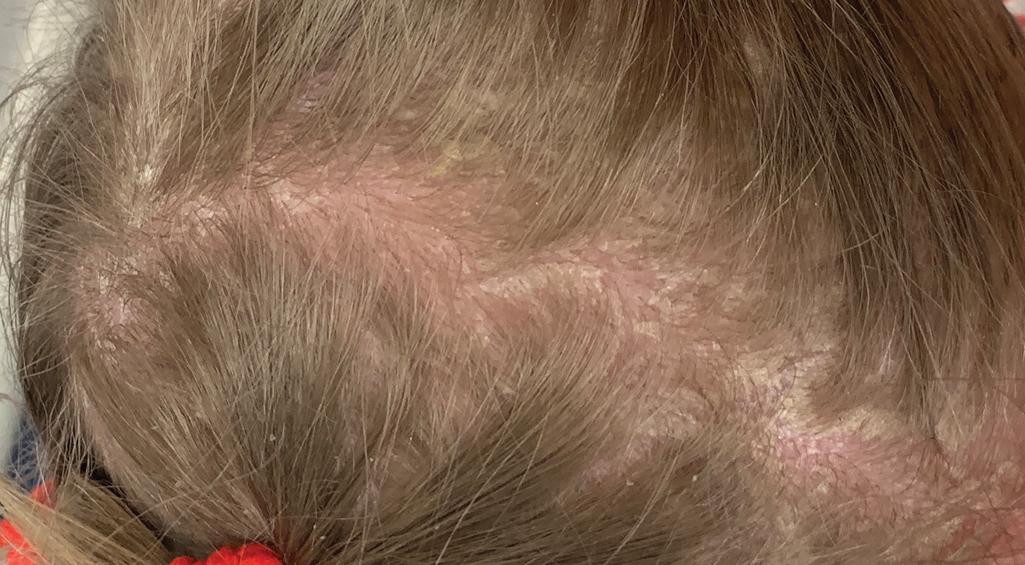
Variant forms of LI from mutations in TGM1 manifest in a more limited distribution of lesional skin. Patients with bathing suit ichthyosis (BSI) are born with a full collodion membrane and transition to LI, but within the first months of life, the scaling on the extremities clears ( Fig. 5.18 ). The residual LI on warmer skin areas (e.g., axillae, trunk, scalp, and neck) has been linked to temperature-sensitive mutations in TGM1 . Mutations in TGM1 that encode proteins sensitive to hydrostatic pressure can result in SHCB. Affected neonates show either a generalized or acral collodion membrane at birth that clears entirely as the baby transitions to the dry environment postnatally. The TGM1 mutations in both BSI and SHCB phenotypes are missense mutations that are predominantly in the catalytic core domains. ALOX12B and ALOX3B mutations have also been described with SHCB. ,
Mutations in SDR9C7 lead to early keratoderma of the extensor elbows and knees with PPK. Affected individuals usually are not born with a collodion membrane and tend to have generalized scaling with variable erythema. Patients with biallelic mutations in SULT2B1 either are collodion babies or have dry, scaling, erythematous skin at birth and thereafter have larger, dark scales that resemble RXLI or LI (although scales are not as large). The skin is erythematous and thickened, with accentuation overlying the joints.
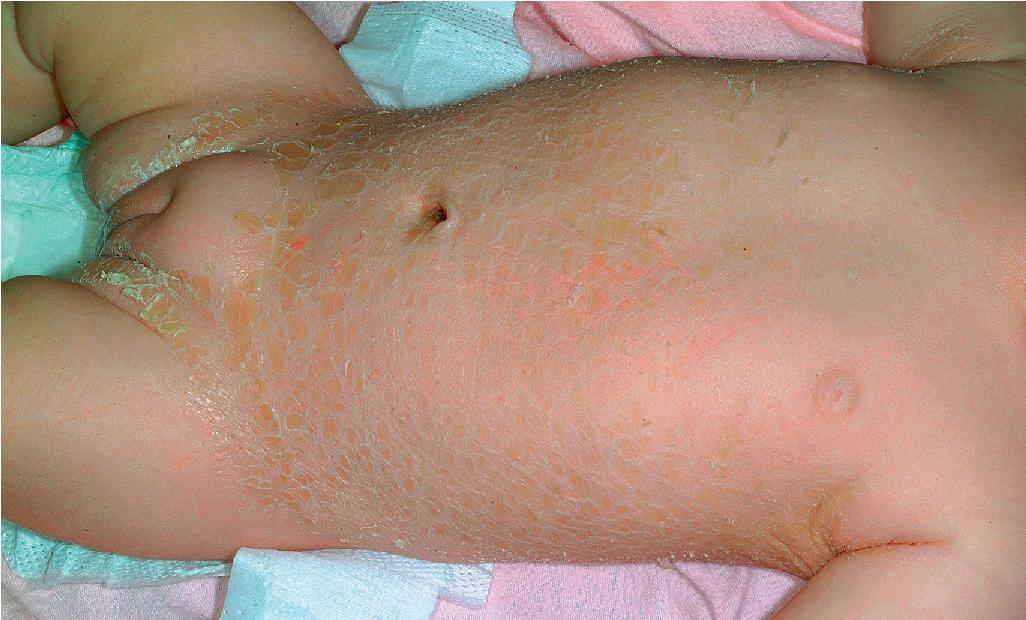
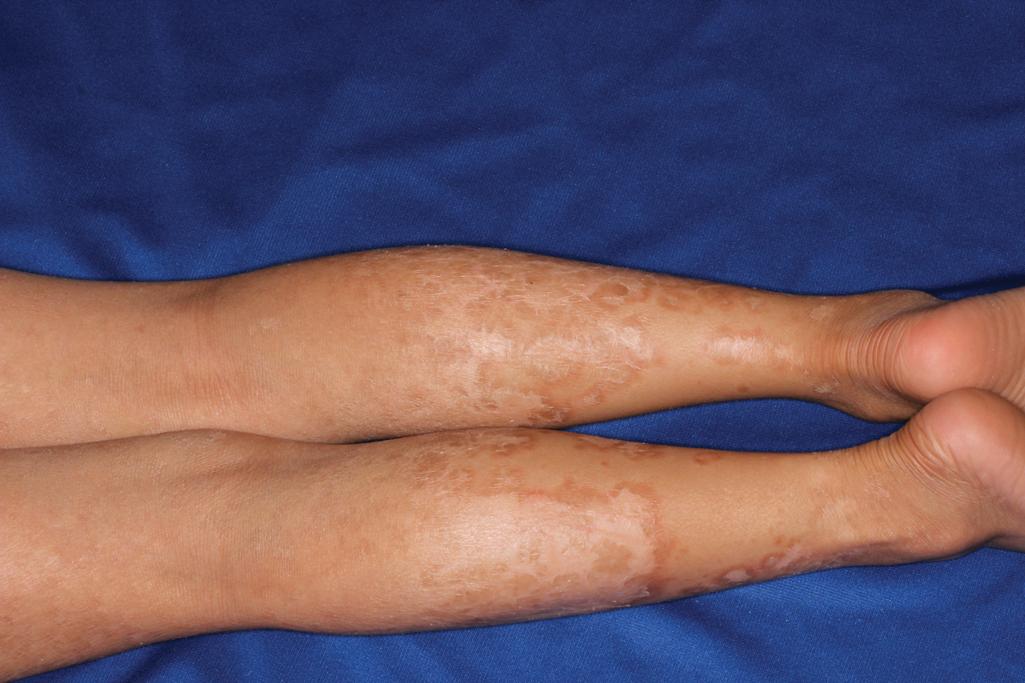
Classic nonbullous CIE is characterized by a much more prominent erythrodermic component that may first become apparent as the collodion membrane (or HI scale) is shed; some patients show CIE at birth without a classic collodion membrane. Affected individuals show fine, white scales on the face, scalp, and trunk, although scaling may consist of more plate-like scales on the extensor surfaces of the legs ( Fig. 5.19 ). The degree of ectropion is variable but often milder than with LI and there is less PPK. Cicatricial alopecia is possible (but not as frequent as in severe LI), and nails may show thickening and ridging. Some patients with CIE show intrauterine growth retardation and/or failure to thrive, although nutritional deficiency and gastrointestinal abnormalities are uncommon. In general, phenotypes of CIE among the various gene-linked subsets are not specific enough to pinpoint a particular gene without genotyping, although LIPN mutations have been reported to have a later onset (early childhood). “Skip areas” without scaling, resembling erythrokeratodermia variabilis, have been described in association with variants in PNPLA1 ( Fig. 5.20 ).
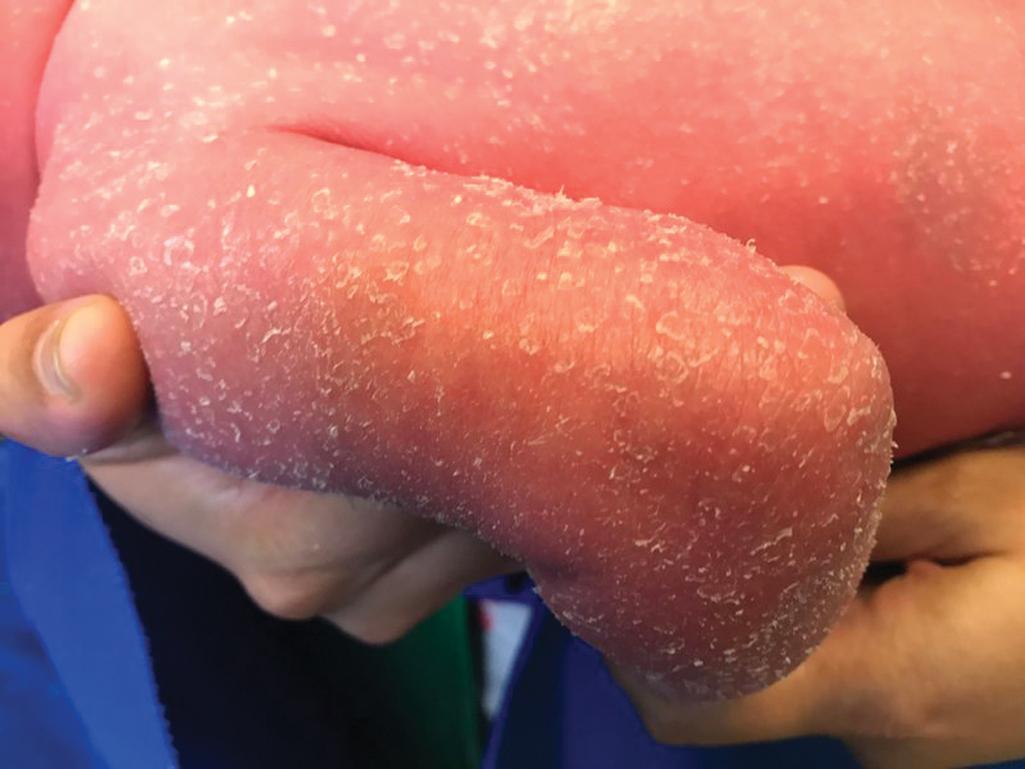
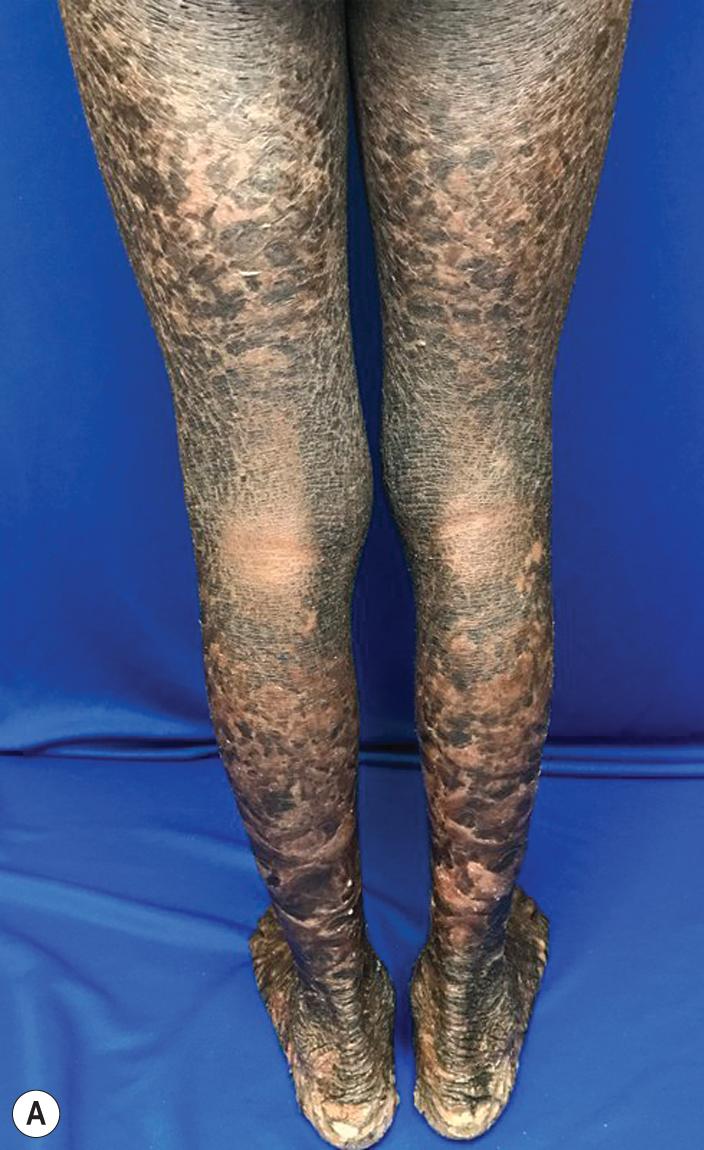
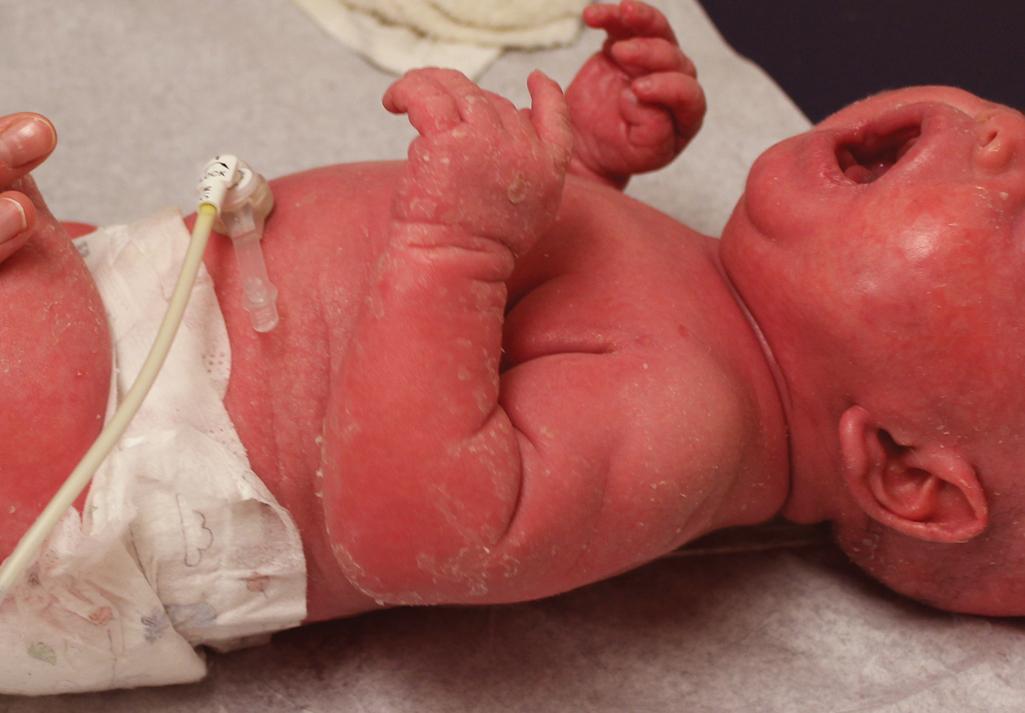
Among the many different subtypes of CIE, HI is the most distinctive and severe form of ARCI. HI manifests at birth as profoundly thickened, armor-like skin that is fissured into polygonal, triangular, or diamond-shaped plaques that simulate the traditional costume of a harlequin ( Fig. 5.21 ). Rigidity of the skin results in marked ectropion; everted O-shaped lips with a gaping fish-mouth deformity; and a distorted, flattened, and undeveloped appearance to the nose and ears. The skin rigidity can restrict respiratory movements, sucking, and swallowing. The hands and feet are ischemic, hard, and waxy, often with poorly developed digits and an associated rigid and claw-like appearance. Flexion deformity of the limb joints is common, and the nails may be hypoplastic or absent. Restrictive dermopathy (see Chapter 6 ) shows congenital contractures, tight skin, ectropion, and intrauterine growth retardation and can thus sometimes be confused with HI, but it shows no hyperkeratosis or scaling.
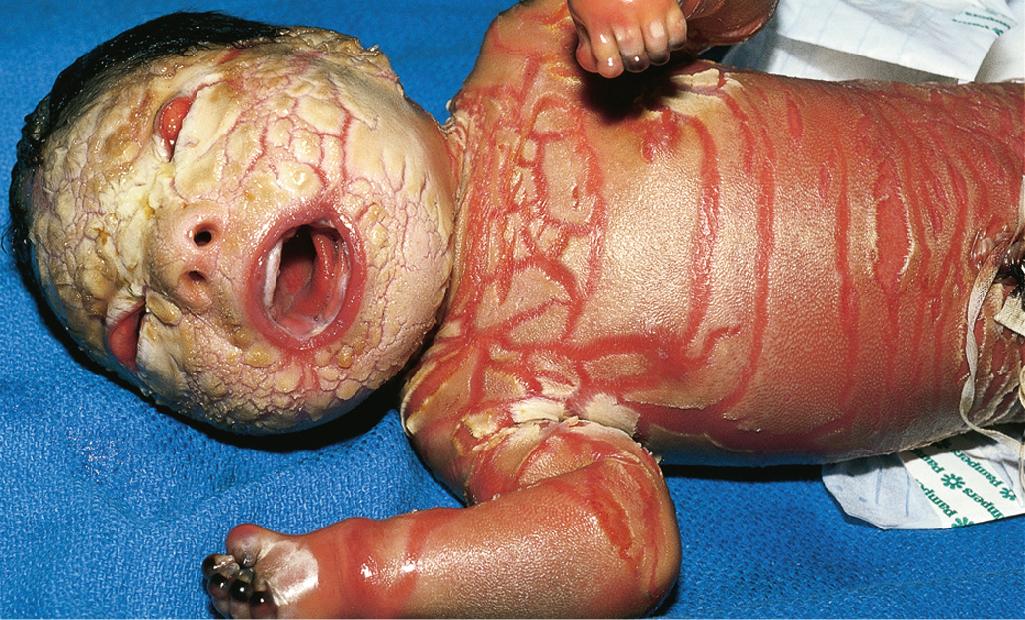
Death is usually associated with prematurity, pulmonary infection (in part associated with hypoventilation caused by thoracic rigidity), poor feeding, excessive fluid loss, poor temperature regulation, or sepsis as a result of cutaneous infection. The severity may be variable, however, and prolonged survival has been achieved by intensive supportive measures, emollients, and in some cases, oral administration of systemic retinoids. Initiation of aggressive intervention for babies with HI is controversial. Even with administration of systemic retinoids or spontaneous clearance of the armor-like scaling, the optimal outcome resembles that of severe CIE ( Fig. 5.22 ). Physicians often reserve initiation for babies who survive the first few weeks, because most infants are stillborn or die during the neonatal period (usually during the first few hours or days of life); however, it is unclear whether the use of systemic retinoids leads to a better outcome than excellent supportive care, especially during the neonatal period of therapy. In addition to initiation of systemic retinoids, topical application of tazarotene or surgical lysis has been used to quickly reduce the encasing bands in HI, which can lead to digital and limb constriction.
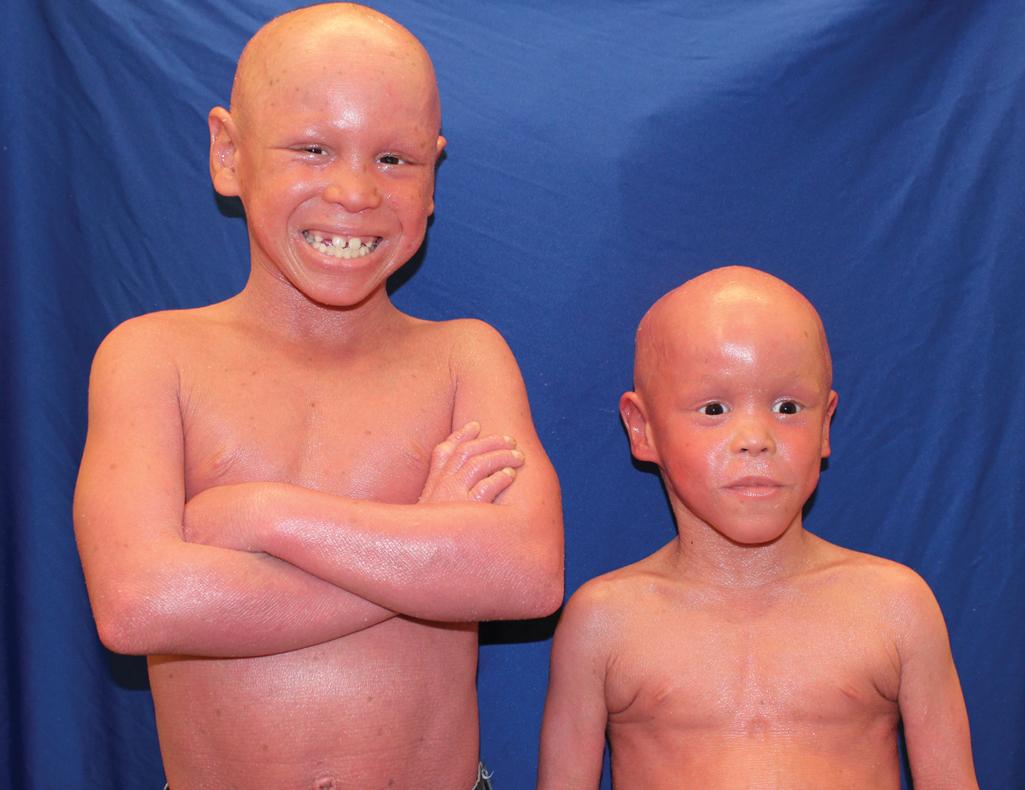
HI babies who survive the neonatal period have an increased risk of developing arthritis during childhood, perhaps associated with the high frequency of T cells expressing interleukin-17A or interleukin-22 (IL-17A/IL-22), which is highest in HI among the forms of CIE. Pulmonary issues can be ongoing in infants and young children. Prenatal diagnosis of HI has been suspected based on ultrasound-based discovery of distal arthrogryposis and can be performed definitively by molecular analysis.
Another form of ARCI, ichthyosis prematurity syndrome (IPS) , is distinctive in its neonatal appearance and course. Resulting from biallelic mutations in SLC27A4/FATP4, affected babies are characteristically born more than 6 weeks premature in association with polyhydramnios and opaque amniotic fluid because of the extensive shedding of epidermal cells. , Affected neonates have generalized thick, spongy white, desquamating skin that resembles vernix caseosa, accentuated on the scalp and eyebrows, which can be confused with the neonatal thick, white scaling at birth in keratitis-ichthyosis-deafness (KID) syndrome. , Neonates often have perinatal respiratory distress and sometimes life-threatening neonatal asphyxia of amniotic keratin debris. During the neonatal period, the scaling may evolve to resemble cobblestones overlying moderate erythroderma. Although the marked thickening clears within weeks, xerosis with follicular keratosis and pruritus persist, and patients often show AD, dermographism, asthma, and eosinophilia (which tends to classify IPS as a syndromic form). More than 70% develop respiratory and/or food allergy.
Loricrin keratoderma (also called Camisa disease, a variant of Vohwinkel keratoderma) is an autosomal dominant disorder with ichthyosis and PPK. Mutations occur in the gene encoding loricrin, a protein that is linked by transglutaminase-1 to involucrin and other proteins of the corneocyte envelope, thereby participating in barrier function and normal epidermal maturation. , Affected individuals may be born with a collodion membrane and later show a mild, minimally erythrodermic, generalized ichthyosis with flexural accentuation. The PPK is initially noted during the first weeks of age, is often transgrediens, and usually shows a honeycomb pattern that resembles that of Vohwinkel syndrome (connexin defect). Pseudoainhum may occur but usually not until adolescence or even adulthood; the starfish-shaped keratoses of Vohwinkel syndrome are not seen with loricrin keratoderma. Alopecia and knuckle pads are occasionally seen, but not other ectodermal abnormalities or hearing impairment. Parakeratosis on routine skin biopsy is a characteristic histologic feature, but termination mutations in the C-terminus have been linked to milder PPK without pseudoainhum and without parakeratosis on affected skin biopsy.
Erythrokeratodermias are a group of predominantly autosomal dominant ichthyoses. Erythrokeratodermia variabilis (EKV) is characterized by two distinct types of lesions: (1) sharply marginated, pruritic, or burning areas of erythema with finer scaling that are often figurate in configuration and undergo changes in size, shape, and distribution during a period of days to weeks ( Fig. 5.23 ); and (2) hyperkeratotic plaques with thick, yellow-brown scales that usually overlie erythema ( Fig. 5.24 ). Lesions are most often symmetrically distributed on the limbs, trunk, and buttock with relative sparing of the face, scalp, and flexures. In contrast to the chronic but remitting appearance of these plaques and figurate lesions, plaques on the knees, elbows, Achilles tendons, and soles of the feet are often persistent. PPK has been described in 50% of affected families.
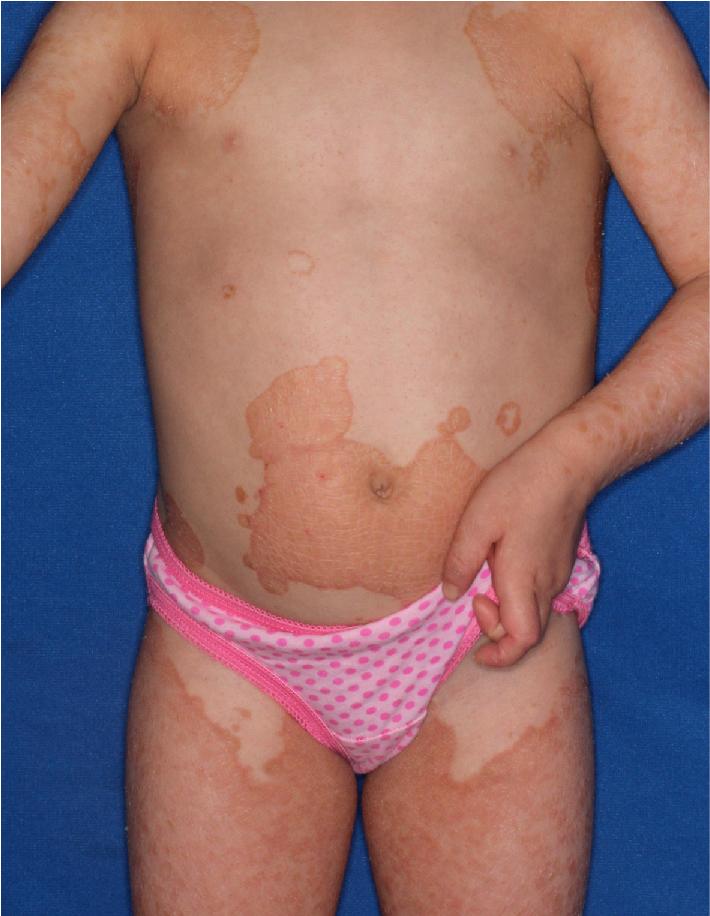
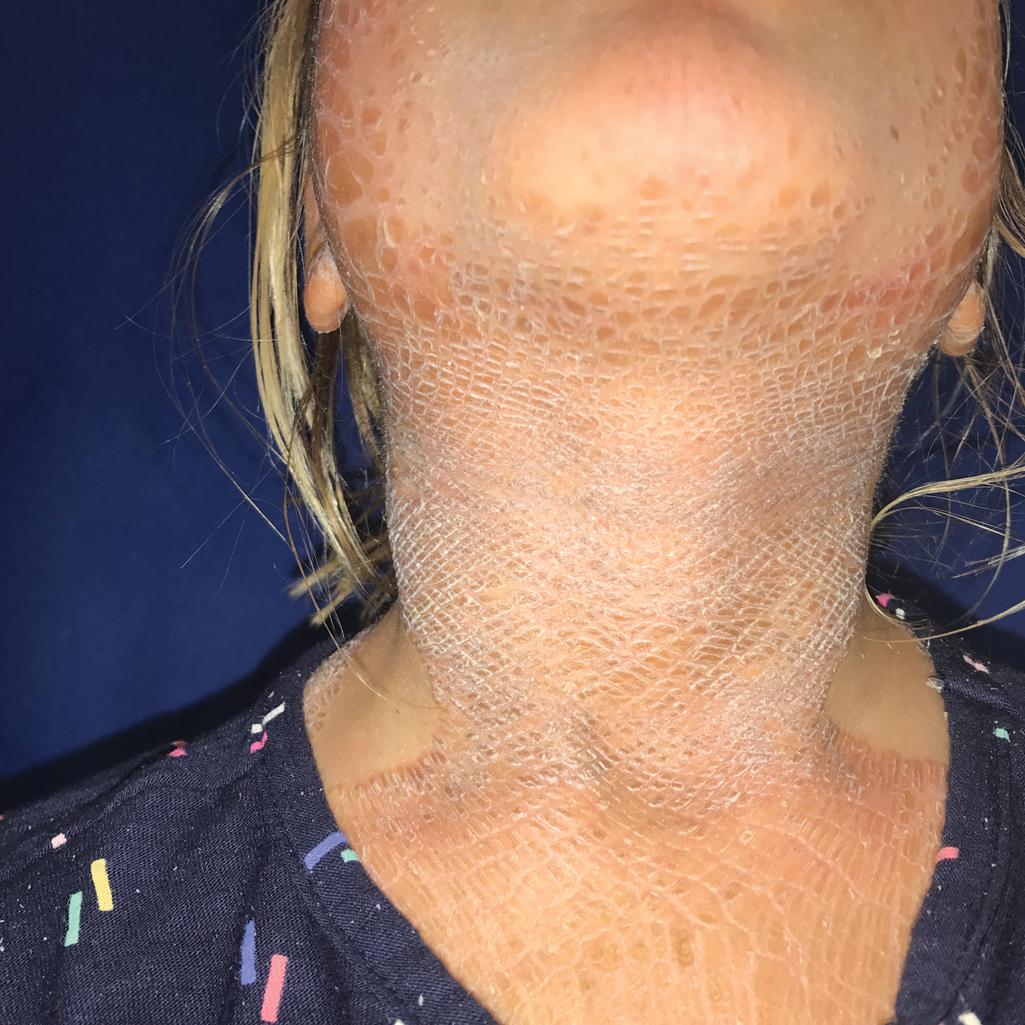
Although lesions are usually noted at birth or shortly thereafter during the first year of life, in a few individuals the onset has been noted during late childhood or early adulthood. The disorder may partially regress at puberty and tends to improve in summer. Patients usually tend to respond well to systemically administered retinoids. EKV has been known to result from mutations in genes that both map to chromosome 1p35.1 and encode interacting connexins: the GJB3 encoding connexin 31 and GJB4 gene encoding connexin 30.3. Well-demarcated plaques of mild erythema, thickening, and scaling, rather than generalized CIE-like involvement, have been described with mutations in PNPLA1 (see CIE). Migrating plaques have also been described in neutral lipid storage disease (Chanarin–Dorfman syndrome) from variants in ABHD5 .
Mutations in GJA1 , encoding connexin 43, are associated with plaques of progressive erythrokeratoderma that often begin during infancy with areas of hyperpigmentation, particularly periorally and over the joints. Associated features are PPK, leukonychia, and often profound hyperpigmentation, even in light-skinned individuals ( Fig. 5.25 ). ,
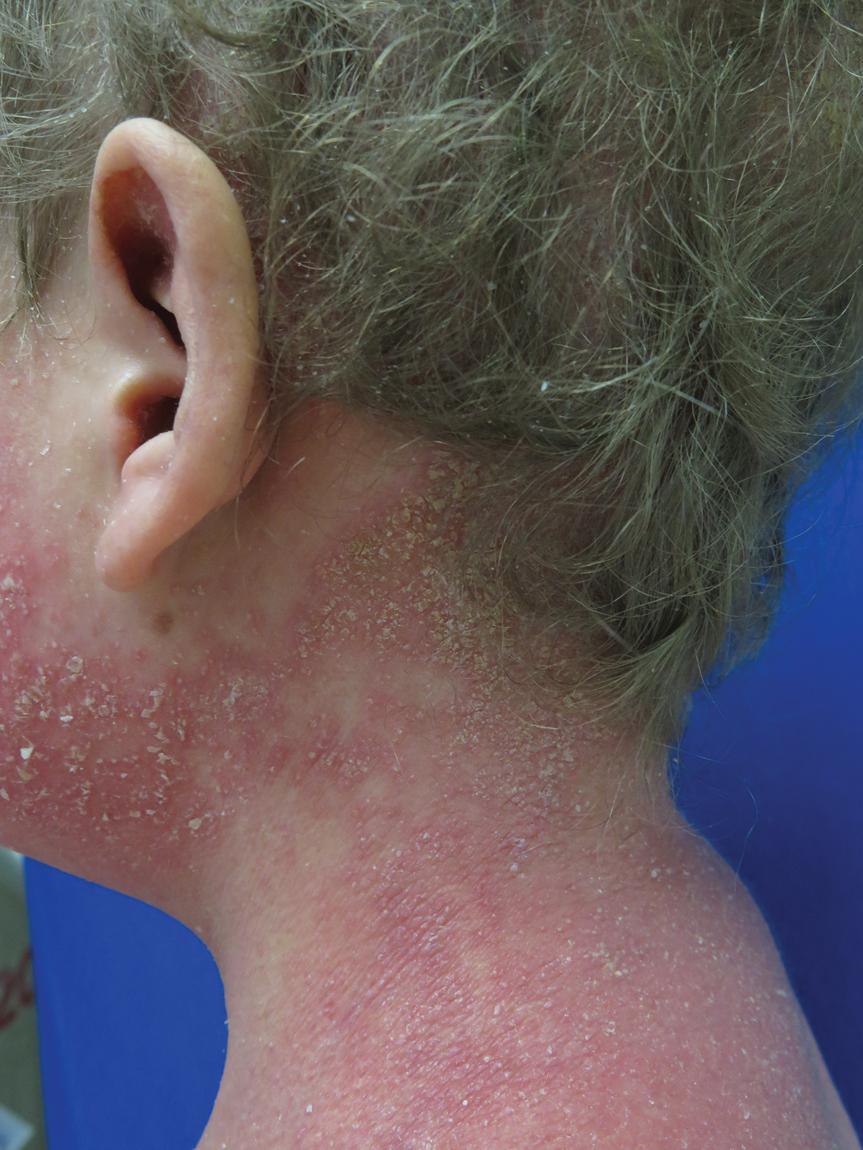
Erythrokeratodermia with cardiomyopathy (EKC) is an autosomal dominant disorder characterized by erythrokeratodermia that tends to begin during infancy, early failure to thrive, woolly hair with alopecia, and nail and variable dental abnormalities ( Fig. 5.26 ). Signs of cardiomyopathy tend to develop by the end of the first decade and may require transplantation. The disorder results from missense mutations, all leading to proline substitution and clustered within the same region-terminal region. Two children with DSP mutations and a similar phenotype responded dramatically to treatment with ustekinumab after failure to respond to broader immunosuppressants or retinoids, one of whom was demonstrated to have high expression of Th1/Th17 pathway genes in the skin and increases in IL-17-/IL-22-expressing cells in the blood before initiating therapy.
Individuals with symmetrical EKV lesions during early infancy that clear by 25 years of age may develop progressive spinocerebellar ataxia in adulthood in association with heterozygous mutations in ELOVL4. Patients with biallelic ELOVL4 mutations or heterozygous mutations in ELOVL1 have ichthyosis in association with spastic diplegia and severe neurodevelopmental delay and must be distinguished from Sjögren–Larsson syndrome (SLS; see Sjögren–Larsson Syndrome). Mutations in ELOVL1 also lead to sensorineural deafness. ELOVL1 and ELOVL4 encode elongases needed for synthesis of very long-chain fatty acids in skin, eyes, and the neurologic system.
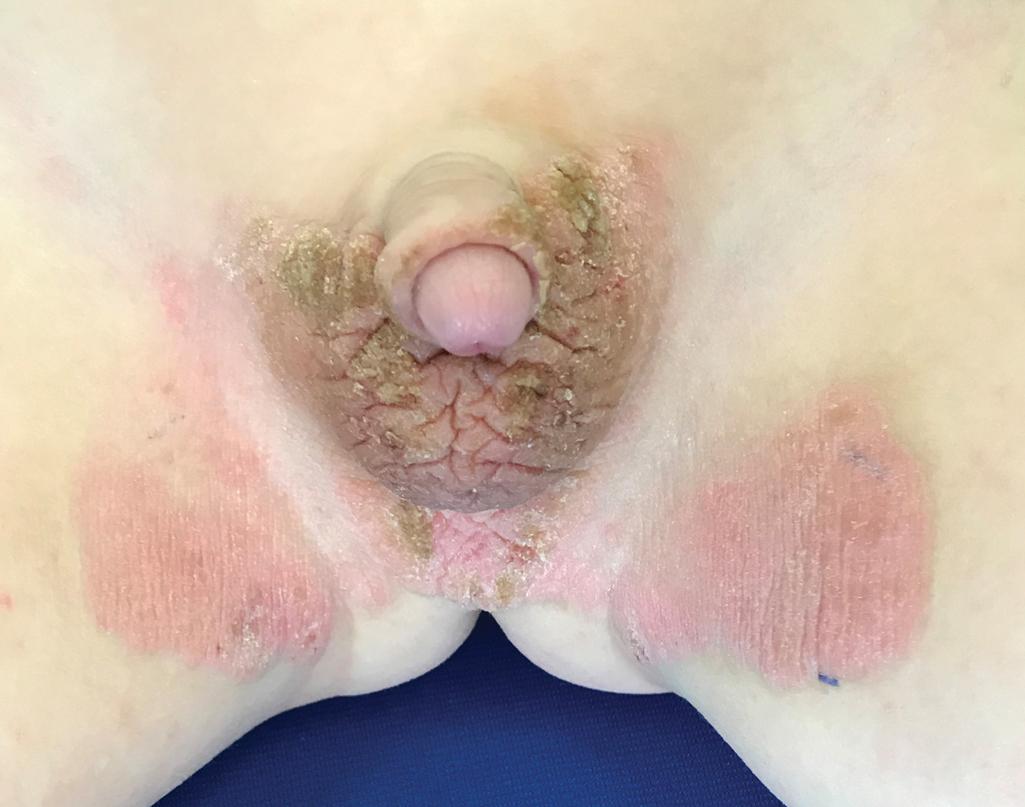
Progressive symmetric erythrokeratodermia (PSEK; Darier–Gottron syndrome) tends to have its onset during infancy, stabilizes after 1 or 2 years, and may partially regress at puberty. PSEK has been distinguished from EKV by the absence of the migrating red patches; the typical sparing of the chest and abdomen with scaling, often verrucous plaques limited to the extremities, genitals/buttocks ( Fig. 5.27 ), and face; and a higher incidence of PPK (;50% of cases). PSEK has recently been shown to result from biallelic mutations in KDSR , encoding 3-ketodihydrosphingosine reductase, an enzyme required for ceramide synthesis. Affected individuals tend to respond well to retinoids, perhaps because retinoids can induce an alternative pathway for ceramide generation. Some cases have associated thrombocytopenia. Biallelic mutations in KRT83 (a gene linked with monilethrix with heterozygous mutations) have also been found to cause a form of PSEK without hair or nail abnormalities. Transgrediens PPK with nail dystrophy and extension onto the extremities, periorificial (cheilitis, perianal, groin up to the abdomen, buttocks) keratotic plaques, and associated woolly hair and scant eyebrows have been described in patients with heterozygous or biallelic mutations in PERP , encoding a p53/p63 tetraspan membrane component of desmosomes. Signs begin during the first months of life and may be confused with Olmsted syndrome.
Peeling skin syndrome (PSS; also called exfoliative ichthyosis or keratolysis exfoliativa congenita ) encompasses a group of unusual autosomal recessive diseases characterized by lifelong, spontaneous superficial peeling of the skin that may be persistent or periodic. The Nikolsky sign tends to be positive. Seasonal variation with worsening during summer months has been described. PSS must be distinguished from other causes of peeling skin, such as staphylococcal scalded skin syndrome in infants (generalized) or localized epidermolysis bullosa simplex (acral forms). In general, proteases are activated in these peeling skin disorders.
Generalized PSS may be noninflammatory (type A) or inflammatory (type B). Mutations in CHST8, which encodes a Golgi transmembrane N-acetylgalactosamine-4-O-sulfotransferase (GalNAc4-ST1), cause exfoliative ichthyosis (PSS3), an asymptomatic form of noninflammatory generalized peeling skin that usually begins by 6 years of age, but may be congenital. Skin biopsy shows a thickened stratum corneum with an intracorneal or subcorneal separation.
Superficial peeling skin without inflammation has also been described in association with moderate diffuse PPK (which is easily removed without pain) in families with biallelic loss-of-function mutations in SERPINB8 , encoding the protease inhibitor SERPIN8, and leading to disadhesion of keratinocytes in the basal and suprabasal epidermal layers. In contrast to mutations in SERPIN7 (Nagashima-type PPK), water exposure does not alter the appearance of the keratoderma. Homozygous mutations in FLG2 , encoding filaggrin 2, have been found to cause generalized PSS (PSS6). Superficial trauma leads to focal areas of denudement in association with chronic hyperkeratotic plaques on joint areas. The condition characteristically improves with advancing age. Generalized peeling in association with leukonychia, acral punctate keratosis, cheilitis, and knuckle pads are the features of PLACK syndrome, a form of PSS due to loss-of-function mutations in CAST , the gene encoding calpastatin, inhibitor of the protease calpain. In addition, biallelic deleterious mutations in CAPN12 , encoding calpain 12, have led to intense erythroderma and exfoliative scaling in association with hypotrichosis and severe nail dystrophy in an infant who also had biallelic mutations in ABCA12 but no features of HI (e.g., harlequin scaling at birth, total alopecia) other than the intense erythroderma. Calpain 12 is a protease that plays an important role in epidermal differentiation and hair follicle cycling.
Inflammatory generalized skin peeling results from mutations in CDSN and loss of corneodesmin, found in the stratum corneum and hair follicle (PSS1). This form always begins at birth. Patients show erythematous migratory peeling patches and complain of associated pruritus or burning. Short stature and easily removable anagen hairs may be associated. Biopsy specimens reveal psoriasiform thickening of the epidermis and a subcorneal or intracorneal split. The differential diagnosis includes Netherton syndrome (NS) and SAM syndrome. SAM syndrome is characterized by severe dermatitis, multiple allergies, and metabolic wasting due to biallelic mutations in DSG1 (encoding desmoglein 1 ) ( Fig. 5.28 ). However, there is wide phenotypic variability, ranging from dermatitis and PPK without systemic manifestations to the food allergies with limited skin involvement. Parents may demonstrate PPK.
An acral or localized form of hereditary PSS is characterized by lifelong painless peeling of predominantly the dorsal aspect of the hands and feet in superficial sheets and exacerbation during summer months with increased sweating. , A facial variant has been described. This form is caused by mutations in TGM5 encoding transglutaminase-5, , which crosslinks epidermal proteins (PSS2), or CSTA , encoding cystatin A, a protease of the cornified cell envelope. ,
Keratosis linearis–ichthyosis congenital–keratoderma (KLICK) is a rare, autosomal recessive disorder characterized by distinctive striate hyperkeratosis in the flexures (perpendicular to the fold) and PPK. , It results from a single-nucleotide deletion in the 5′ untranslated regions (UTRs) of the proteasome maturation protein (POMP) gene, which reduces levels of POMP and leads to proteasome insufficiency, increased endoplasmic reticulum (ER) stress, and perturbed processing of profilaggrin in differentiating keratinocytes. ,
Syndromic forms of ichthyosis may be associated with a variety of extracutaneous abnormalities, most commonly involving the hair (e.g., Netherton; ichthyosis follicularis, alopecia, and photophobia [IFAP] and ichthyosis, hypotrichosis, and sclerosing cholangitis [IHSC] syndromes and ichthyosis with hypotrichosis; trichothiodystrophy) and neurologic system (e.g., Sjögren–Larsson; Refsum; cerebral dysgenesis, neuropathy, ichthyosis, and palmoplantar keratoderma [CEDNIK]; and mental retardation, enteropathy, deafness, neuropathy, ichthyosis, and keratoderma [MEDNIK] syndromes). Some syndromic disorders are often lethal in the neonate (e.g., Neu–Laxova syndrome), infant (e.g., Gaucher disease type 2; arthrogryposis, renal dysfunction, and cholestasis [ARC] syndrome; multiple sulfatase deficiency), or child (e.g., CEDNIK syndrome).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here