Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Neonatal jaundice is a common phenomenon, noted in more than 80% of otherwise healthy, term newborns. , In the majority of cases, the jaundice is transient, usually resolving by the end of the first postnatal week, and serum total bilirubin (STB) concentrations are not harmful. In some infants, severe hyperbilirubinemia may develop with the potential for acute bilirubin encephalopathy. , Some of these cases may progress to the chronic athetoid form of cerebral palsy (kernicterus) or other forms of bilirubin neurotoxicity. These include deafness or mild neurologic sequelae attributed to high serum levels of bilirubin, called bilirubin-induced neurologic dysfunction (BIND) . Because of the wide range of clinical features resulting from bilirubin neurotoxicity, Le Pichon et al. suggested use of the term kernicterus spectrum disorders to include all above-mentioned forms of neurologic sequalae of bilirubin neurotoxicity.
In recent years it has become apparent that much of the mediation of bilirubin metabolism and the determination of whether STB concentrations remain within the physiologic range or increase to potentially harmful concentrations lies within genetic control. Indeed, one of the most important advances in our understanding of the genomics of bilirubin metabolism was the elucidation of the UGT1A1 gene encoding the bilirubin-conjugating enzyme, uridine diphosphate-glucuronosyltransferase 1A1 (UGT1A1). It is now apparent that a number of genes control both the production and elimination of bilirubin and that polymorphisms or mutations of these genes, sometimes acting in combination or synergistically, have the potential to cause extreme hyperbilirubinemia. Additionally, genetically inherited diseases due to mutations of genes not normally involved in the physiology of bilirubin are associated with increased hemolysis, sometimes to a marked degree. Select hereditary conditions with the potential to moderate bilirubin metabolism are listed in Box 91.1 . Furthermore, in addition to specific mutations and polymorphisms, there are racial and ethnic influences on bilirubin values and neonatal hyperbilirubinemia, as will be discussed. Finally, next-generation DNA sequencing, a modern diagnostic tool, has the potential to determine a specific genetic etiology in many newborns with extreme hyperbilirubinemia.
Increased heme catabolism
Heme oxygenase-1 ( HO-1 ) promoter polymorphisms
Diminished bilirubin formation
Bilirubin reductase polymorphisms
Diminished bilirubin hepatic uptake
Solute carrier organic anion transporter ( SLCO1B1 ) polymorphisms
Diminished bilirubin conjugation
Uridine diphosphate-glucuronosyltransferase 1A1 ( UGT1A1) promoter and coding area polymorphisms and mutations
As reviewed in the ensuing chapter, hyperbilirubinemia is not dependent on increased bilirubin production or diminished elimination as individual processes. Rather, these forces may combine and even interact to increase the STB at any point in time.
The STB at any point in time represents two cardinal contributing processes: bilirubin production and bilirubin elimination, the latter primarily by conjugation but also including bilirubin uptake into the hepatocyte and excretion of the conjugated product. In neonates, bowel reabsorption of bilirubin via the enterohepatic circulation may further add to the bilirubin pool.
In healthy adults and older children and infants, bilirubin production and elimination are in equilibrium, and the STB remains within normal limits. During the first postnatal days, however, physiologically increased heme formation and its catabolism result in increased bilirubin production, while diminished activity of the bilirubin-conjugating enzyme, UGT1A1, results in decreased bilirubin conjugation. Mild or moderate imbalance between these processes may pose little threat to an otherwise healthy, term infant. Almost all newborns have UGT1A1 enzyme immaturity; therefore, diminished bilirubin conjugation is universally present. In the immediate postnatal period, bilirubin production exceeds its elimination, resulting from a high intrauterine (and postnatal) hemoglobin concentration, a shortened red blood cell (RBC) life span, and elimination of transplacental clearance. Consequently, a physiologic increase in STB occurs during the first postnatal days. Given universally diminished bilirubin conjugation in the newborn and the delicate balance between production and excretion, increased hemolysis will be the mediating factor in the pathophysiology of hyperbilirubinemia in most cases.
Despite the significance of increased hemolysis, bilirubin accumulation in a hemolytic state should not always be assumed. Of prime importance is the concept of lack of equilibrium between bilirubin production and conjugation. Thus, in an infant who is hemolyzing but has efficient bilirubin-conjugating capacity, STB may not rise. On the other hand, in an infant with immature bilirubin-conjugating capacity, due, for example, to late prematurity or the presence of 7, rather than the wild-type 6, thymine adenine (TA) 7 repeats in the promoter region of the UGT1A1 gene (UGT1A1∗28) , associated with Gilbert syndrome, even minimally increased hemolysis may result in hyperbilirubinemia.
This lack of equilibrium between bilirubin production and conjugation in the pathophysiology of hyperbilirubinemia has been demonstrated mathematically. Kaplan and colleagues investigated the individual contributions of bilirubin production and conjugation to the STB concentration, and also the combined effects of these processes, in healthy, term neonates on the third postnatal day. Blood carboxyhemoglobin determinations corrected for ambient carbon monoxide (COHbc) were used as an index of heme catabolism, whereas bilirubin conjugation was reflected by serum total conjugated bilirubin expressed as a percentage of STB (TCB[%]). As expected, over the range of STB concentrations observed, STB increased in positive correlation with COHbc, and in inverse correlation with TCB(%) ( Fig. 91.1 ). An index or ratio, COHbc/TCB(%), termed the production-conjugation index , was constructed to reflect the combined forces of bilirubin production and conjugation. Correlation of this index with STB concentrations was higher than those for either COHbc or TCB(%) independently ( Fig. 91.2 ), confirming the importance of imbalance between bilirubin production and conjugation, rather than the independence of these processes, in the physiologic rise of serum bilirubin. Although the relationship between the index and STB tended to plateau with increasing index values, at the lower end of the index scale, small increases in the index were associated with large increases in STB.
![Fig. 91.1, (A) Regression analysis between total serum bilirubin (STB) and carboxyhemoglobin corrected for ambient carbon monoxide (COHbc) values. Increasing STB values correlated positively with COHbc ( r = 0.38; s = 46.1; y = 9.36 + 323.5 x – 378.4 x 2 + 172.5 x 3 ). (B) Regression analysis between STB values and total conjugated bilirubin (TCB) , expressed as a percentage of STB (TCB[%]). Increasing STB values were inversely proportional to TCB(%) ratio ( r = 0.40; s = 45.8; y = 136.5 – 27.0 x +1.3 x 2 ). Fig. 91.1, (A) Regression analysis between total serum bilirubin (STB) and carboxyhemoglobin corrected for ambient carbon monoxide (COHbc) values. Increasing STB values correlated positively with COHbc ( r = 0.38; s = 46.1; y = 9.36 + 323.5 x – 378.4 x 2 + 172.5 x 3 ). (B) Regression analysis between STB values and total conjugated bilirubin (TCB) , expressed as a percentage of STB (TCB[%]). Increasing STB values were inversely proportional to TCB(%) ratio ( r = 0.40; s = 45.8; y = 136.5 – 27.0 x +1.3 x 2 ).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/HereditaryContributionstoNeonatalHyperbilirubinemia/0_3s20B9780323712842000914.jpg)
![Fig. 91.2, Curvilinear regression analysis between total serum bilirubin (STB) values and the combined effect of bilirubin production and conjugation, reflected by the bilirubin production/conjugation index COHbc /( TCB [%]). Increasing values of STB correlated positively to this index ( r = 0.61; s = 39.1; y = 32.1 + 132.1 x – 45.8 x 2 + 4.6 x 3 ). Fig. 91.2, Curvilinear regression analysis between total serum bilirubin (STB) values and the combined effect of bilirubin production and conjugation, reflected by the bilirubin production/conjugation index COHbc /( TCB [%]). Increasing values of STB correlated positively to this index ( r = 0.61; s = 39.1; y = 32.1 + 132.1 x – 45.8 x 2 + 4.6 x 3 ).](https://storage.googleapis.com/dl.dentistrykey.com/clinical/HereditaryContributionstoNeonatalHyperbilirubinemia/1_3s20B9780323712842000914.jpg)
Heme oxygenase-1 (HO-1) is the rate-limiting enzyme in the heme degradation pathway. Biliverdin, which is formed as a result of this reaction, is subsequently converted to bilirubin. It is logical, therefore, to expect that increased activity of HO-1 should lead to increased heme catabolism and thereby enhanced bilirubin production. The gene encoding HO-1, the inducible isoform of HO, has a polymorphic dinucleotide guanine thymine repeat (GT) n in the promoter region. , This regulatory region modulates gene transcription. Expression of HO-1 is enhanced by a lower number of (GT) n repeats. Therefore, short HO-1 promoter sequences should be associated with increased HO activity and lead to increased bilirubin production.
(GT) n promoter repeat lengths range from 12 to 40 repeats with a bimodal distribution at or around 23 and 30 repeats ( Fig. 91.3 ). , This has allowed the identification of three subclasses according to the number of (GT) n repeats: fewer than 25 repeats (short); between 25 and 30 repeats (medium); and more than 30 repeats (long). The impact of various (GT) n repeat lengths on the transcriptional activity of the HO-1 promoter has been studied. Compared with constructs with greater than 25 repeats, those with fewer than 25 repeats showed increased HO-1 promoter activity, , , HO-1 messenger RNA (mRNA) expression, and enzyme activity. HO-1 (GT) n allele length combinations have been used to designate promoter genotypes including short/short, short/medium, short/long, medium/medium, medium/long, and long/long.
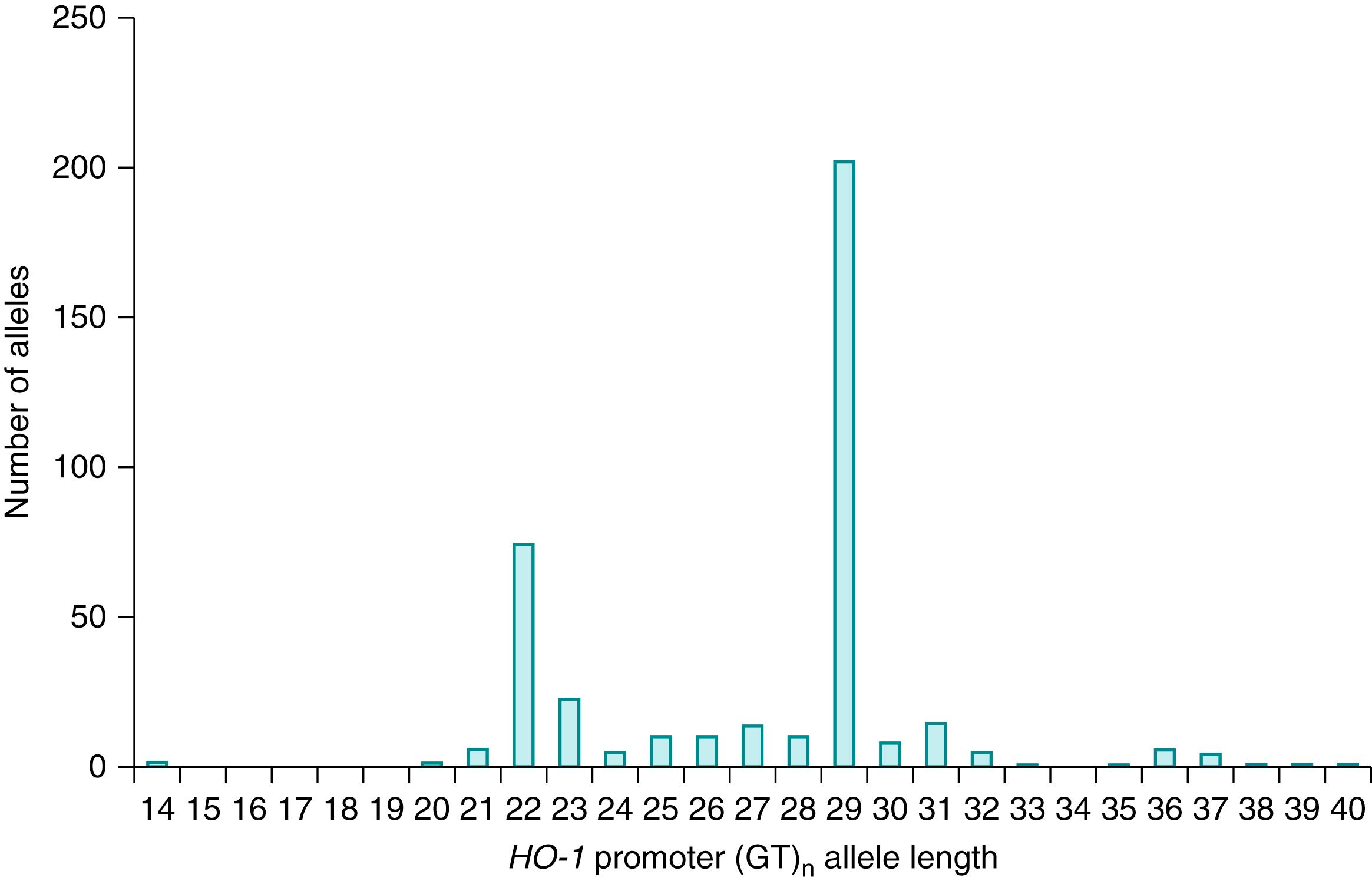
Variations in the distributions of the short, medium, and long promoter length sequences may be due to ethnicity or classification differences. Thus the distribution of short, medium, and long promoter alleles differed significantly in three separate studies. , , However, the data are not entirely clear because the exact cutoff points defining short, medium, and long (GT) n alleles are somewhat arbitrary. This is a key issue because only the short allele is likely to affect heme catabolism. It is unlikely that variation in the cutoff point between the medium and long alleles would have a significant effect on the variation in bilirubin levels between studies.
In adults in the steady state, some studies have demonstrated increased STB values in carriers of the short HO-1 allele, compared with those without any short allele. Differences in STB values between groups were often minor but did reach statistical significance; it is not clear, however, whether these variations had any clinical significance. ,
Variations in STB values among varying HO-1 (GT) n lengths are not necessarily consistent and may demonstrate ethnic distinction. Lin and colleagues studied three distinct Asian ethnic populations, finding an association between STB values and HO-1 (GT) n polymorphisms in Uyghurs, but not in the Han or Kazak populations. It is possible that during hemolysis the large amounts of heme released may induce HO-1 expression. This is consistent with a report of a boy with exceptionally elevated STB values during an episode of autoimmune hemolytic anemia, who was found to be homozygous for short HO-1 (GT) n promoter alleles.
The case cited above and adult studies suggest the potential for HO-1 (GT) n promoter polymorphisms to modulate the severity of neonatal hyperbilirubinemia. However, few studies have investigated the relationship of short promoter polymorphism and neonatal hyperbilirubinemia. Kanai and colleagues studied HO-1 (GT) n promoter sequences in Japanese infants who had undergone phototherapy and found no relationship between these polymorphisms and neonatal hyperbilirubinemia. Similarly, Bozkaya and colleagues did not detect significant differences in HO-1 promoter allele lengths between newborns with STB values greater than 12.9 mg/dL and those in whom the STB value did not exceed this level. They did, however, find a correlation between alleles with fewer than 24 (GT) n repeats and prolonged hyperbilirubinemia.
In a study by Kaplan and colleagues, in which both glucose 6-phosphate dehydrogenase (G6PD)–normal and –deficient newborns were analyzed, no significant correlation was found between the frequency of HO-1 (GT) n promoter polymorphism genotypes, or the presence of any short HO-1 promoter allele, and STB concentrations of 15.0 mg/dL or greater. There was a trend, however, for the G6PD–normal, but not G6PD–deficient, newborns with COHbc values exceeding the 75th percentile to have a higher percentage of short HO-1 promoter alleles, suggesting a possible role of short HO-1 alleles in modulating heme catabolism. Absence of this pattern among the G6PD–deficient newborns excludes, at least in the steady state, any potential role of the short HO-1 allele in modulating heme degradation in this subgroup. These findings do not, however, exclude a role for short HO-1 promoter expression in exacerbating hyperbilirubinemia in acute hemolytic episodes associated with G6PD deficiency or other hemolytic conditions, in which the large amounts of heme released may induce HO-1 expression.
In contrast to the above studies, Tiwari and colleagues did find a significant association between homozygosity for the short (GT) n promoter genotype and STB levels exceeding the 95th percentile on the hour-specific bilirubin nomogram in North Indian newborns. Also, in a report from Japan, a higher proportion of allele frequencies in class S (small alleles) was found in hyperbilirubinemic infants than in non-hyperbilirubinemic neonates, while homozygous or heterozygous S allele carrier individuals were encountered more frequently in hyperbilirubinemic neonates than in non-hyperbilirubinemic counterparts.
Biliverdin reductase A reduces biliverdin to bilirubin, and the potential exists for polymorphisms of the gene encoding this enzyme to affect neonatal hyperbilirubinemia. However, the biliverdin reductase variant studied in adults is not associated with elevated STB levels, and there are no studies of these polymorphisms in neonates.
Hepatic cellular uptake of bilirubin is mediated by the solute carrier organic anion transporter polypeptide 1 enzyme (OATP1B1). This enzyme and the gene encoding it, solute carrier organic anion transporter (SLCO1B1) , therefore play an important role in modulating serum bilirubin levels. Variations in the SLCO1B1 gene include 388 G>A, 521 T>C, and 463 C>A. Laboratory expression studies have demonstrated that 388 G>A is associated with reduced transport activity of OATP1B1 and that transient hyperbilirubinemia may result from transporter inhibitors such as rifampin. , It is therefore logical to suspect that in populations with a high frequency of SLCO1B1 388 G>A, such as Asians, who also have a high incidence of neonatal hyperbilirubinemia, this polymorphism may play a role in the pathogenesis of jaundice. In a meta-analysis of studies of neonatal hyperbilirubinemia, Liu and colleagues found no statistically significant differences in the overall incidence of hyperbilirubinemia in association with SCLO1B1 388 G>A. When focusing on the specific ethnic subgroups included in this analysis, it did become apparent that this polymorphism was associated with the development of hyperbilirubinemia in Chinese neonates (odds ratio [OR], 1.39; 95% confidence interval [CI], 1.07 to 1.28), but not in other ethnic subgroups, which included whites, Asians, Thai, Brazilians, and Malaysians. Overall SCLO1B1 521 T>C was not associated with increased risk for hyperbilirubinemia and was actually associated with diminished risk in Chinese newborns (OR, 0.60; 95% CI, 0.40 to 0.92). The C>A substitution at nucleotide 463 was detected in only one study from the United States in which no statistical association was determined between this polymorphism and the development of neonatal hyperbilirubinemia.
In a study that included 18 SLCO1B1 variants, no significant differences were observed in the frequency of these variants between those with STB values exceeding the 95th percentile on the hour-specific bilirubin nomogram (hyperbilirubinemia) versus those with STB values not exceeding the 40th percentile. There was, however, an effect of SLCO1B1 variants when they occurred in combination with additional genetic factors (see below). Huang and colleagues found an association between the SCLO1B1 388 G>A variant and the development of neonatal hyperbilirubinemia (OR, 2.10; 95% CI, 1.02 to 4.30), but not for the other variants studied. Coexpression of this polymorphism with additional risk factors, not necessarily genetic, such as breast-feeding, was instrumental in increasing the risk for hyperbilirubinemia.
The UGT gene is a superfamily of genes whose function is to encode the conjugation of glucuronic acid to a variety of substrates and to facilitate their elimination from the body. The UGT1A1 gene isoform, which belongs to the UGT1 gene family, plays an important role in the conjugation and, therefore, elimination of bilirubin. The gene isoform has been mapped to chromosome 2q37 and was cloned by Ritter and colleagues in 1991. UGT1A1 is a major locus influencing bilirubin levels. Multiple genome-wide association studies have identified variants in UGT1A1 to be associated with STB levels in European, East-Asian, and African American populations.
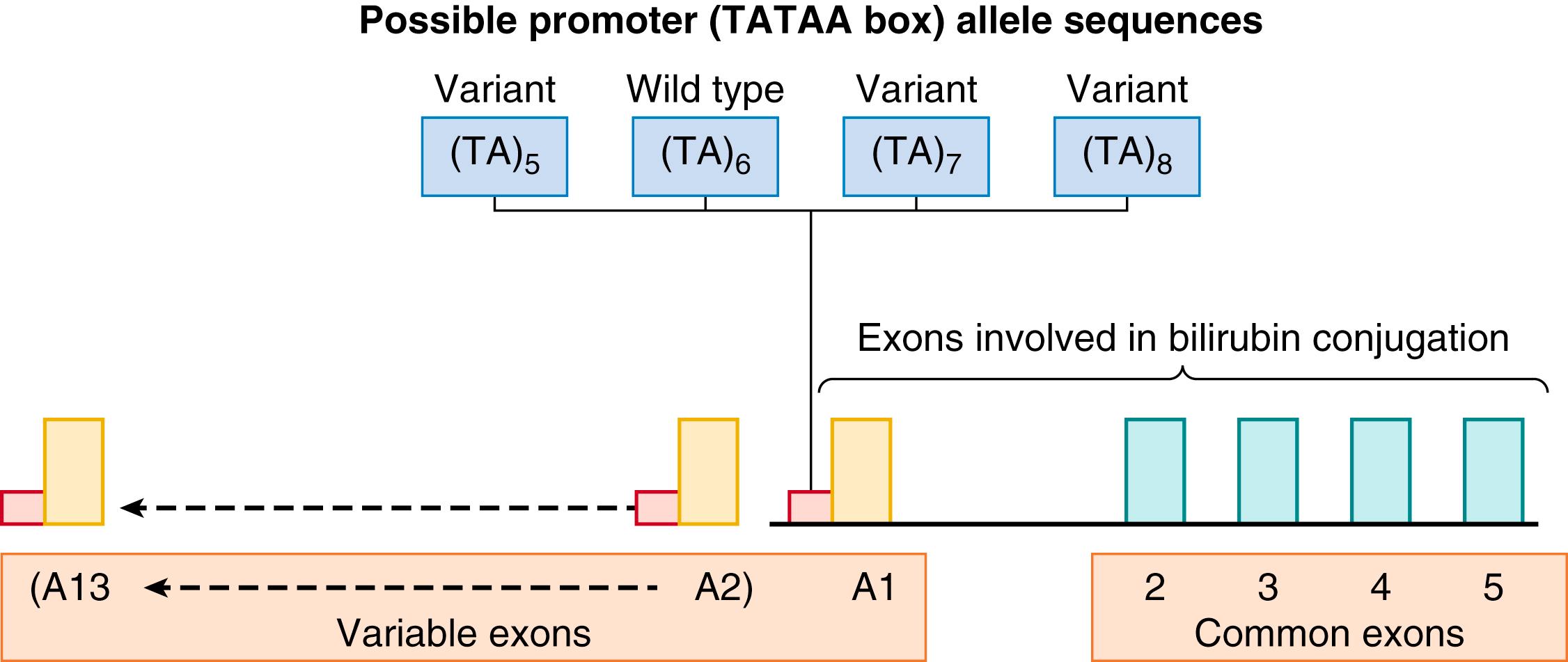
As seen in Fig. 91.4 , the gene-coding area comprises four common exons (exons 2, 3, 4, and 5) and 13 variable exons. However, only one of the variable exons, A1, is of importance with regard to bilirubin conjugation. Variable exon A1 functions in conjunction with common exons 2 to 5 to splice mRNA from the variable exon to the common exons. This process provides a template for the synthesis of an individual enzyme isoform. Upstream of each variable exon is a regulatory noncoding promoter that contains a TATAA box sequence of nucleic acids. Coding-area mutations of variable exon A1 or the common exons 2 to 5 may result in deficiencies of bilirubin conjugation due to structural alterations in the UGT1A1 enzyme, such as in Crigler-Najjar syndrome. Polymorphisms of the noncoding promoter area affect bilirubin conjugation by diminishing expression of a normally structured enzyme. The wild-type promoter contains six sequences of TA nucleotides (TA) 6 . Shorter TATAA box sequences (TA) 5 , although rare, are associated with enhanced UGT1A1 expression and, therefore, more effective bilirubin conjugation. Longer TATAA polymorphisms, such as the not infrequent (TA) 7 or the rare (TA) 8 , are associated with diminished expression and, therefore, less effective bilirubin conjugation. Using a reporter gene, Beutler and colleagues demonstrated an inverse relationship between the number of (TA) repeats and the activity of the promoter through the range of five to eight (TA) repeats. Allele frequency of the (TA) 7 promoter varies among populations (0.35 in Sephardic Jews in Israel, 0.39 in Caucasians, 0.16 in Asians, and 0.43 in individuals of African descent ), while in Nigerian neonates the distribution of (TA) 6 and (TA) 7 was almost equal. Homozygosity for (TA) 7 , also known as UGT1A1∗28 , is associated with Gilbert syndrome in adults and with hyperbilirubinemia in neonates, primarily when in combination with additional icterogenic factors such as G6PD deficiency. The high frequency of (TA) 7 in Nigerians and individuals of African descent may contribute to the high incidence of kernicterus associated with G6PD deficiency in these ethnic groups. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here