Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Drs. Jennifer T. Wells and Robert Perrillo contributed to this chapter in previous editions of the book.
An estimated 260 million persons in the world today are chronically infected with HBV. The majority of these individuals will not experience complications, but 15% to 40% will have serious sequelae such as cirrhosis or HCC, and many will die prematurely. , It is estimated that chronic HBV infection results in over 887,000 deaths per year globally. The number of HBV-related deaths due to cirrhosis and liver cancer has increased by 33% from 1990 to 2013. In the USA, the rate of ALF attributable to hepatitis B has been declining, as has the number of cases listed for LT for chronic liver failure. This decline is most likely due to broader vaccination and use of antiviral therapy. Unfortunately, these favorable trends are counterbalanced by a continuing increase in new cases of chronic hepatitis B and HCC in the Western world, mainly due to immigration from high endemic regions. ,
Effective vaccines against HBV have been available since the early 1980s, but perinatal and early life exposures continue to be major sources of infection in much of the developing world because of limited resources that preclude a policy of universal vaccination for newborns. From a global perspective, widespread implementation of early-life vaccination programs in high- and intermediate-risk countries will ultimately have the greatest impact on liver disease-related mortality in future generations. Promiscuous sexual contact and injection drug use account for most new cases of hepatitis B in adults in low-prevalence areas such as the USA and Western Europe. Even in these areas, however, a further reduction in the incidence of acute infections will remain a challenge in the future because persons in these risk groups greatly underutilize vaccination. The WHO has implemented a strategy to eliminate hepatitis B and C infection globally through increased vaccination, screening, and treatment.
The prevalence of HBV infection varies markedly around the world. In highly endemic regions such as Southeast Asia (excluding Japan), China, and much of Africa, 6% or more of the population are chronic HBV carriers, and the lifetime risk of infection ranges from 60% to 80%. In these high-risk areas, perinatal transmission and horizontal spread among children are the major means of transmission. Approximately 60% of the world’spopulation resides where HBV is highly endemic. Regions of intermediate risk include parts of southern and Eastern Europe, the Middle East, Japan, the Indian subcontinent, much of the former Soviet Union, and northern Africa. In these areas, the lifetime risk of infection is between 20% and 60%. Horizontal transmission occurs among a broad age range, but neonatal exposure is also presumed to be common. Areas of low prevalence include North America, Western Europe, certain parts of South America, and Australia, where the lifetime risk of HBV infection is less than 20% and transmission is primarily horizontal between young adults. Sexual transmission is the main mode of transmission in Europe and North America, but injecting drug use continues to be a major contributor to new cases as well.
Perinatal transmission accounts for most new infections in the world and is believed to account for at least half of all hepatitis B surface antigen (HBsAg)-positive carriers. Sixty percent to 90% of hepatitis B e antigen (HBeAg)-positive mothers typically have a high level of viremia (“viral load”) and transmit the infection to their offspring, whereas mothers who are positive for antibody to HBeAg (anti-HBe) transmit the disease less frequently (5% to 15%) (see later). Fortunately, the incidence of new infections and childhood HCC has diminished greatly in countries such as Taiwan, where universal vaccination has been in place for decades.
HBV is transmitted efficiently by percutaneous and mucous membrane exposure to infectious body fluids. The virus is 50 to 100 times as infectious as HIV and 10 times as infectious as HCV. A high viral load indicates a higher risk not only of transmission from mother to child, but also after needlestick exposure and in the setting of household contact. HBV DNA has been detected by sensitive techniques such as PCR in most body fluids, except for stool that has not been contaminated with blood. Although HBV replicates primarily in hepatocytes, the presence of replicative intermediates and virally encoded proteins in other sites, such as the adrenal gland, testis, colon, nerve ganglia, and skin, suggests that a vast extrahepatic reservoir for infectious virus exists. Small amounts of HBV DNA have been demonstrated in peripheral mononuclear cells and liver tissue years after apparent resolution of chronic infection. , Extrahepatic localization of low levels of replicating virus may also explain the relatively high rate of HBV transmission from organ donors positive for antibody to hepatitis B core antigen (anti-HBc) (see later).
In several highly endemic countries, the prevalence of hepatitis B is decreasing due to improved socioeconomic status, universal vaccination programs, and, possibly, effective antiviral therapy. Estimates of the prevalence of chronic HBV infection in the USA vary from 875,000 to 2.2 million persons. , Of all recent immigrants to the USA, an estimated 60% were born in regions of intermediate or high HBV prevalence. This finding explains why most chronic HBV infections in the USA are imported ( Fig. 79.1 ). ,
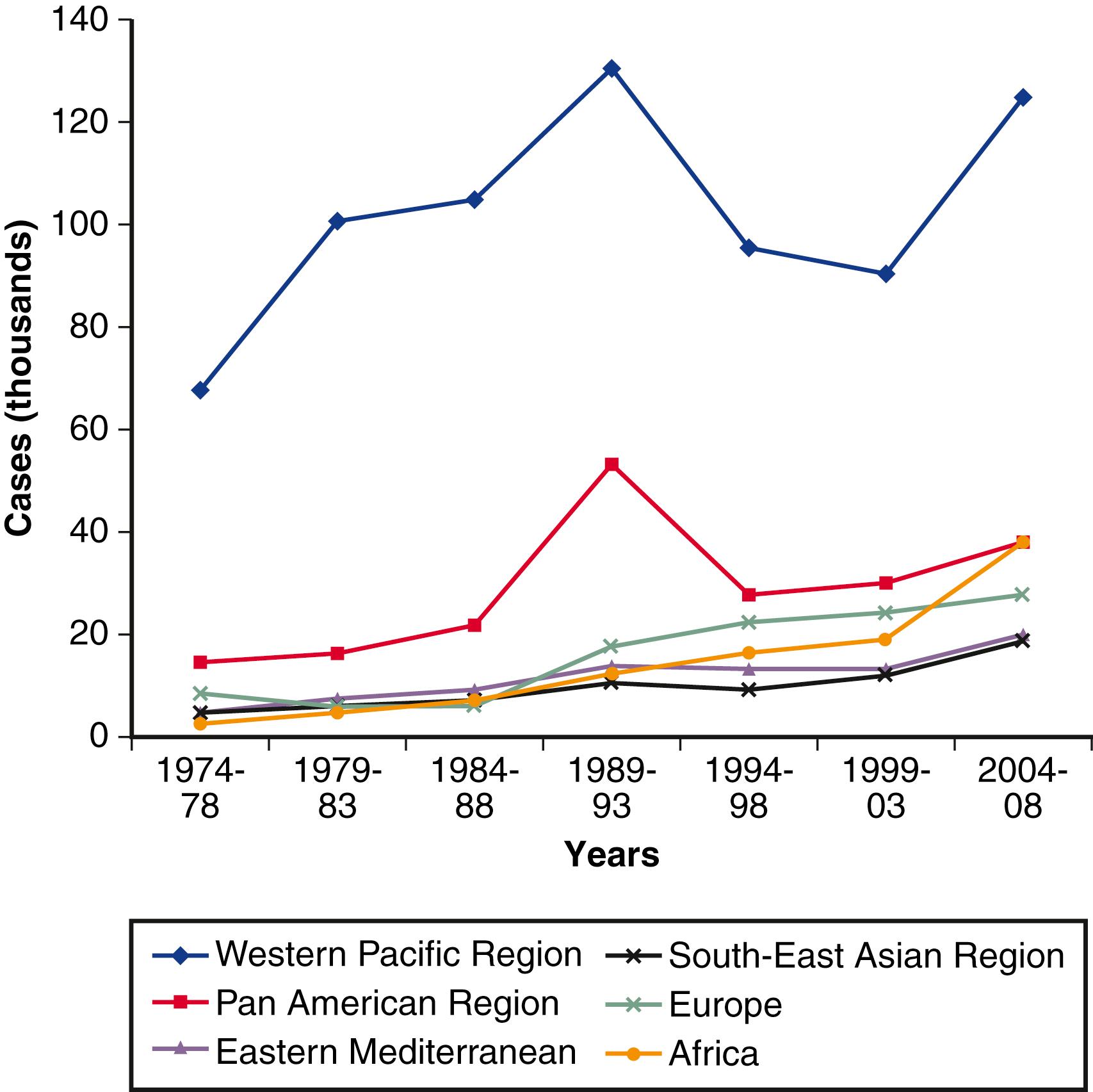
In contrast to the growth of new cases of chronic hepatitis B, a decline in acute cases of hepatitis B since the 1990s has been the result of universal vaccination of newborns, adult vaccination programs for high-risk persons, changes in sexual lifestyle, refinements in blood screening procedures, and the availability of virus-inactivated blood components. Health care workers have experienced a striking decline in HBV infection owing to high rates of vaccination.
In much of the developed world, the highest incidence of acute cases continues to be in sexually active young adults. Since 1995, most cases of acute hepatitis B reported to the Centers for Disease Control and Prevention (CDC) were caused by intimate contact among heterosexuals, followed by injection drug use and sex between men. In the USA, the most common risk factor among people with new HBV infections is injection drug use related to the opioid abuse epidemic. Rates of new HBV infection are highest among adults 30 to 49 years of age, reflecting low HBV vaccination coverage among adults at risk. Cases of hepatitis B continue to result from hemodialysis, acupuncture, artificial insemination, and, rarely, blood transfusion, but these cases account for a small contribution to the overall number of newly established acute infections.
Data from the CDC indicate that over 95% of pregnant women in the USA are tested for HBsAg, and infant vaccine coverage levels are up to 93%. Despite these encouraging figures, however, the CDC also estimates that approximately 1000 new cases of hepatitis B in newborns each year result from a missed birth dose or failure to complete the vaccine schedule (see later).
The age at which a person becomes infected with HBV is the principal determinant of the clinical outcome. Perinatal exposure leads to the chronic HBV carrier state in as many as 95% of persons because of immunologic tolerance to the virus (see later). By contrast, children exposed during the first 5 years of life have a 30% chance of developing chronic HBV. Only 2% to 5% of adults with an intact immune system become chronically infected.
Two thirds of patients with acute hepatitis B have an asymptomatic or subclinical illness that goes unrecognized. In the other third, acute hepatitis, ranging from mild to moderate in severity, develops, with ALF occurring in 1%. Although uncommon, hepatitis B accounts for 7% of all cases of ALF (see Chapter 95 ). Rapid viral elimination may result in clearance of HBsAg from serum by the time of initial presentation. In these cases, the accurate diagnosis of acute hepatitis B may require testing with immunoglobulin M (IgM) anti-HBc (see later).
The rate of spontaneous survival in ALF caused by HBV is only approximately 20%. LT has resulted in survival rates of 50% to 60%. Recurrent disease in the allograft is uncommon because of the successful application of hepatitis B immune globulin (HBIG) and antiviral agents (see later and Chapter 97 ).
Progressive liver disease (including cirrhosis and HCC) can be expected to develop in one quarter to one third of people who acquire infection in the first few years of life. An estimated 15% to 25% of predominantly middle-aged or older men who acquired infection early in life ultimately die of liver-related causes. Outcomes are related to host (age, male gender, genetic background, immune status) and viral (serum HBV DNA level, HBV genotype, mutation patterns) factors. HCC is 4 times as likely to develop in males as in females.
The presence of active viral replication and long-standing necroinflammatory liver disease caused by HBV strongly influences the rate of progression to cirrhosis. The major determinant of survival is the severity of liver disease when the patient first comes to medical attention. Cirrhosis is associated with decreased survival and an increased frequency of HCC. Prior to the advent of antiviral therapy, 5- and 20-year survival rates of 55% and 25%, respectively, were reported in patients with HBV-related cirrhosis, compared with 97% and 63%, respectively, for those with mild (noncirrhotic) disease. In one study, an 84% 5-year survival rate was reported for patients with compensated HBV-related cirrhosis, compared with 14% for patients with cirrhosis complicated by ascites, jaundice, encephalopathy, or a history of variceal bleeding (see Chapter 74, Chapter 92 ). Multivariate analyses in several large cohort studies have identified age, ascites, hyperbilirubinemia, and renal dysfunction as correlating independently with survival in patients with HBV-related cirrhosis. Therefore, early hepatic decompensation is an immediate indication for antiviral therapy as well as evaluation for LT (see later).
Clearance of HBsAg from serum in patients with early HBV-related cirrhosis has been associated with an excellent prognosis, including improvement in liver histology and function, a decreased risk of viral reactivation, and improved long-term survival. HBsAg clearance, however, is not an absolute safeguard against the future development of HCC in persons who have preexisting cirrhosis.
HBV is a small DNA virus that belongs to the Hepadnaviridae family. Other members of this family are HBV-like agents that infect the woodchuck, ground and tree squirrels, woolly monkey, crane, heron, Ross goose, and duck. HBV is a small (3.2-kilobase [kb]) virus with a DNA genome that has a relaxed, circular, partially double-stranded configuration ( Fig. 79.2 ). The genome is composed of 4 open reading frames (ORFs) and has a compact design in which several genes overlap and use the same DNA to encode different viral proteins. The 4 viral genes components include the core, surface, X, and polymerase genes. The core gene encodes the core nucleocapsid protein, which is important in viral packaging and production of HBeAg. The surface gene encodes the pre-S1, pre-S2, and S proteins (comprising the large [L], middle [M], and small [S] surface proteins). The X gene encodes the X protein, which has transactivating properties and may be important in hepatic carcinogenesis. The polymerase gene has a large ORF (≈800 amino acids) and overlaps the entire length of the surface ORF. It encodes a large protein with functions that are critical for packaging and DNA replication (including priming, RNA- and DNA-dependent DNA polymerase, and RNase H activities).
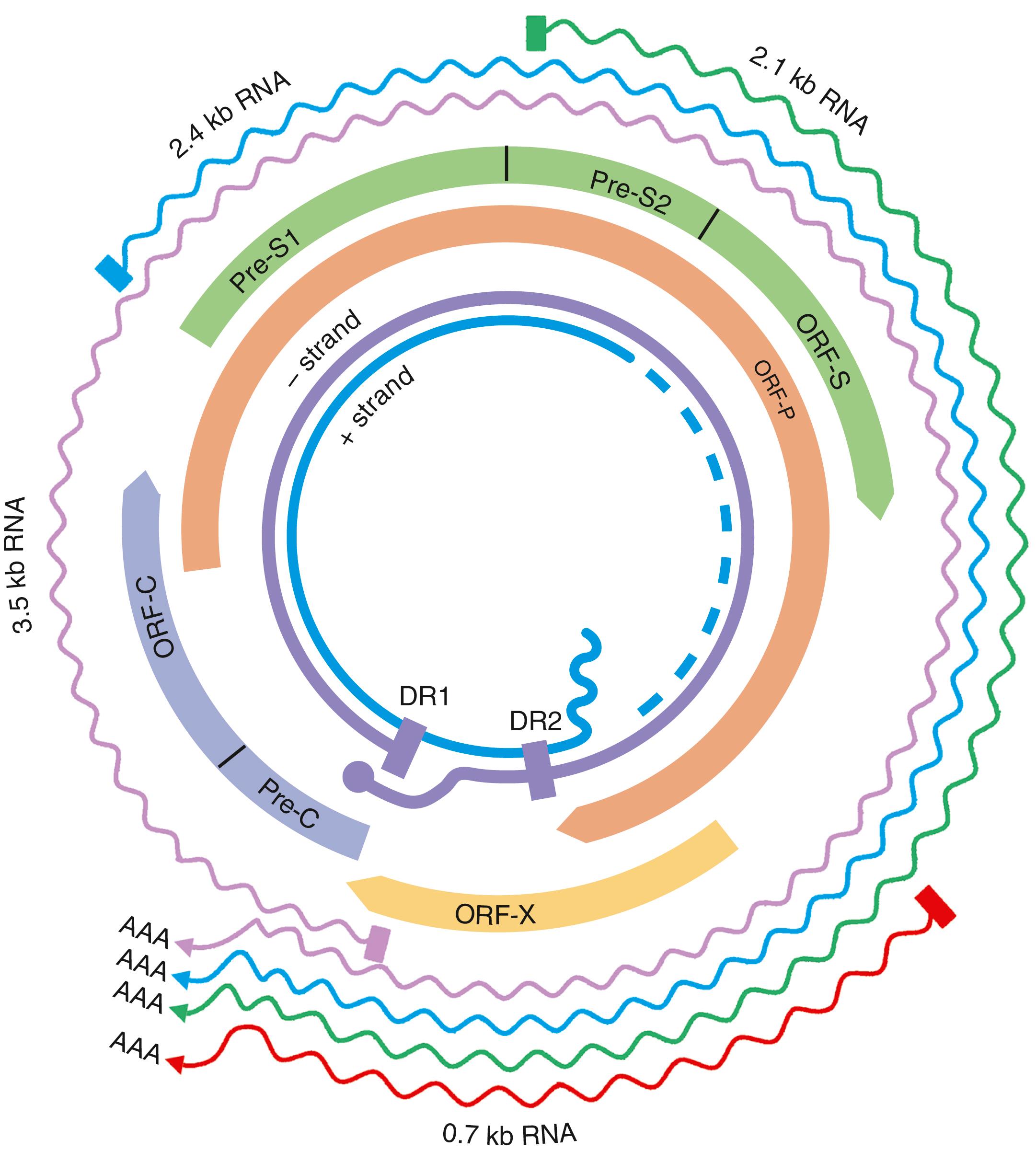
Although HBV is a DNA virus, replication occurs through an RNA intermediate and requires an active viral reverse transcriptase/polymerase enzyme ( Fig. 79.3 ). The mutation rate is higher for HBV than for other DNA viruses (with an estimated 10 13 to 10 15 point mutations per day). Complete HBV genomic sequencing has identified a large number of mutations within the HBV genome, many of which are silent or do not alter the amino acid sequence of encoded proteins. Because of genomic overlap, however, some of the silent mutations in one ORF (e.g., the polymerase gene) may result in an amino acid substitution in an overlapping ORF (surface gene), although with uncertain clinical implications.

HBV replication begins with encapsidation of the pregenomic RNA through complex interactions between host and viral proteins. HBV DNA polymerase reverse transcribes the pregenomic RNA into a negative-strand HBV DNA, which in turn serves as the template for positive-strand synthesis to form a partially double-stranded genome. Concurrent with HBV DNA synthesis, the nucleocapsid undergoes maturation and, through an incompletely understood mechanism, interacts with the S protein to initiate viral assembly in the endoplasmic reticulum. S protein is synthesized in the endoplasmic reticulum, where monomer aggregates that exclude host membrane proteins subsequently bud into the lumen as subviral particles. When formed, HBsAg undergoes glycosylation in the endoplasmic reticulum and the Golgi apparatus. Noninfectious subviral particles (spherical and filamentous forms of HBsAg) are secreted in great abundance when compared with mature virions. These subviral HBsAg particles exceed virions in number by a variable factor of 10 2 to 10 5 and can accumulate up to concentrations of several 100 μg/mL in serum.
A genetic classification based on comparisons of complete genomes has demonstrated 10 genotypes (designated A through J) and numerous subtypes of HBV ( Box 79.1 ). These classifications are defined as a divergence in the entire HBV genomic sequence of 8% or more. Genotype A is the predominant genotype in northern Europe and the USA. Genotypes B and C are confined to populations in eastern Asia and the Far East, but changes in immigration patterns have resulted in an influx of Asian HBV carriers with these genotypes into the USA. Genotype D is found worldwide but is especially prevalent in the Mediterranean area, Middle East, and southern Asia. Genotype E is indigenous to western areas of sub-Saharan Africa, and genotype F prevails in Central America. Cases of genotype G have been reported in the USA and France. Genotype H has been described in Mexico. Genotypes I and J are the most recently discovered and have been observed in Vietnam and the Ryukyu Islands in Japan, respectively.
A: Northwestern Europe, North America, Central Africa
B: Southeast Asia, including China, Japan, and Taiwan (prevalence is increasing in North America)
C: Southeast Asia (prevalence is increasing in North America)
D: Southern Europe, Middle East, India
E: West Africa
F: Central and South America, USA (Native Americans), Polynesia
G: USA, France, Germany
H: Central and South America
I: Vietnam and Laos
J: Ryuku, Japan
Shorter time to HBeAg seroconversion and higher probability of HBsAg loss: B > C
Response to treatment with interferon-α: A > B ≥ C > D
Precore/core promoter mutant frequency: B and D > A and C
Active liver disease activity and risk of progression: C > B
Evolution to chronic liver disease: non-A > A
HCC risk: B > C in younger age group in Taiwan, but C > B in older age group in Japan
Clinical associations appear to exist with the various genotypes (see Box 79.1 ). The strongest clinical associations appear to be that (1) HBeAg seroconversion occurs earlier in patients with HBV genotype B than in those with genotype C, and (2) the response to therapy with interferon (IFN) is better with genotypes A and B than with C and D (see later). The viral genotype also has implications for the frequency of precore and core mutations (see later) and may have an effect on the frequency of HCC. There is no compelling evidence that genotypes affect the HBV DNA response to nucleoside analogs (see later).
The clinical associations with the various genotypes have become increasingly clear but have not led to specific recommendations for routine testing because genotype classification does not generally lead to a difference in management. One exception to this rule, however, occurs when a patient is being considered for pegylated IFN (PegIFN) therapy. In patients who are suitable candidates based on age and other factors (see later), genotype testing may have clinical value because genotypes A and B are associated with higher rates of a sustained virologic response and HBsAg clearance.
Most mutations in the HBV genome that are identified by comparing nucleotide sequences with those of wild-type HBV are silent or do not alter the amino acid sequence in a particular ORF. Some mutations have potentially important disease associations, however, and are described later.
HBsAg gene mutants result from a primary mutation in the HBsAg gene or a mutation in the overlapping DNA polymerase gene arising during nucleoside antiviral therapy (see later). Once the mutation appears, mutated virions can become selected immunologically as the dominant form of the virus.
Mutations in the HBsAg gene between amino acid positions 124 to 147 are potentially important because this region of the HBsAg gene includes the major “a” epitope that binds to neutralizing antibody to HBsAg (anti-HBs). The mutation can lead to failure to detect HBsAg by commercial assays, which depend on binding to anti-HBs, and to failure of neutralization by HBIG or of vaccination.
Infection with HBsAg gene-mutant HBV is accompanied by detection of anti-HBc. Serum HBV DNA levels can vary to the same extent seen in HBsAg carriers (see later). These mutants need to be distinguished from cases of “occult” hepatitis B, which has been linked to cryptogenic cirrhosis and an increased risk of HCC. , In occult HBV infection, HBsAg-negative persons have detectable HBV DNA in serum. Some of these persons may lack evidence of other serologic markers of infection (e.g., anti-HBc). Occult HBV infection is thought to result from active suppression of viral replication by the host immune system; as a result, when HBV DNA is detectable in serum, it is present in low levels (<200 IU/mL).
Large-scale vaccination programs in regions endemic for HBV have revealed a 2% to 3% frequency of vaccine-escape HBsAg mutants. It remains controversial whether additional mutants will be further selected in the future and increase the risk for widespread vaccine failure, but most available evidence does not support this scenario. The importance of HBsAg gene mutants for HBIG failure after LT is less controversial.
Mutations in the precore and basal core promoter regions of the HBV genome can influence the production of HBeAg. A precore mutation results in a stop codon at nucleotide 1896 that abolishes the synthesis of HBeAg, whereas mutations in the basal core promoter at nucleotides 1762 and 1764 decrease HBeAg synthesis by approximately 70% while maintaining pregenomic RNA levels. Both types of mutations have been observed in cases of severe hepatitis, which has been attributed to the loss of the immune-tolerizing effects of HBeAg (see later). The presence of core promoter mutations has been linked to an increased risk of HCC, and a higher frequency has been found in patients infected with HBV genotype C. Precore and basal core promoter mutants have been described in the same patients and are particularly common in Asian and South European patients harboring HBV genotypes B, C, and D. A large serosurvey of HBV carriers residing in the USA has found that precore and core promoter mutations are common (frequencies of 27% and 44%, respectively). Both mutant forms of HBV were observed to occur far more commonly in HBeAg-negative patients (precore mutation in 38% of HBeAg-negative vs. 9% of HBeAg-positive patients; core promoter mutation in 51% vs. 36%). In addition to these mutations, upstream mutations in the core gene can influence immunologic responses to HBV. Core gene mutations have been shown to block recognition of HBV by cytotoxic T lymphocytes (CTLs), a key mode of viral clearance. Therefore, the mutations contribute to HBV immune escape and possibly influence the response to IFN. , Core gene mutations within the immunodominant epitopes of the HBV nucleocapsid also can affect CD4 + T-cell reactivity.
In patients with perinatally acquired chronic hepatitis B, a prolonged immune tolerant phase with minimal to absent hepatic necroinflammatory activity is typically seen for the first 20 to 30 years of HBV infection. Sequencing studies have demonstrated stable core gene sequences during this phase. Precore mutations are also uncommon during this phase. Core gene mutations become more common as patients pass from the immune active phase and undergo HBeAg seroconversion, at which time a growing number of mutations are observed in the region of the core gene that includes many B- and T-cell epitopes. Both precore stop codon mutants and core gene mutants have been associated with a poor response to IFN therapy.
The polymerase gene encodes a DNA polymerase enzyme needed for encapsidation of viral RNA into core particles, conversion of the pregenomic viral RNA into a negative strand of viral DNA (reverse transcription), and conversion of this first HBV DNA strand into a second DNA strand of positive polarity. In general, the HBV reverse transcriptase function of the polymerase gene is highly conserved because major mutations that impair the efficiency of viral replication lead to selection pressure against such variants. As indicated earlier, HBV has low replication fidelity, however, meaning that it has a propensity to mispair nucleotide bases when it reverse transcribes viral RNA to DNA. HBV DNA polymerase also lacks any proofreading activity, so it cannot repair its mistakes. Therefore, when a nucleotide base is misplaced, it remains in the growing viral DNA strand as a base mutation, and the new HBV DNA genome has a different sequence from the original (wild-type) genome. The overall error rate of HBV DNA polymerase is estimated to be 1 per 10,000 nucleotides copied, which translates to the potential for 10 million base-pair errors per day in an infected person. All possible single-base mutations can be produced in a 24-hour period, although many such mutations will yield nonviable viruses.
Single or double nucleotide substitutions alter the amino acid sequence in the reverse transcriptase domain of the HBV DNA polymerase enzyme, thereby decreasing binding of drugs to its active site. Mutations in the HBV polymerase gene can lead to clinically apparent resistance to nucleoside analog therapy whenever there is both decreased susceptibility to the antiviral drug and sufficient replication fitness to allow continued propagation in the expanding viral population (“quasispecies”).
High levels of viral replication and high mutability of the virus allow the emergence of single and even double polymerase mutants as a minor component of the viral quasispecies even before antiviral therapy is begun. Because of the limitations in sensitivity of currently available molecular assays (e.g., the line probe assay), these mutants are not detectable until they constitute at least 5% to 10% of the entire viral population. Ultradeep pyrosequencing is a technique with the ability to detect HBV mutants that constitute less than 1% of the total population. It is expected that the advent of next-generation sequencing will further dissect viral genome variability, thereby opening avenues to individualized precision medicine. Persistent infection with drug-resistant HBV has been associated with progression of disease and blunting of hepatic histologic improvement with antiviral therapy. Severe flares of hepatitis have also been reported in conjunction with the emergence of drug-resistant mutants, and acquisition of these mutants may lead to rapidly progressive liver disease after LT. Horizontal transmission of these mutants also has been described.
HBV is generally not a cytopathic virus, and the severity of HBV-associated liver disease is considered related to the intensity of the host immunologic response to the virus. Whereas both cellular and humoral immune responses are needed for effective clearance and long-term protection against reinfection, the cellular immune response appears to be the arm principally involved in the pathogenesis of disease. The immunologic response to HBV encompasses both an innate, or nonantigen-specific, response (e.g., natural killer cells and IFNs) and an adaptive immune response, including antibodies to viral antigens, HLA class II-restricted CD4 + T cells, and HLA class I-restricted CD8 + CTLs. Induction of the antigen-specific T-cell response is thought to occur in lymphoid organs, where the host T cells encounter viral peptide antigens (or epitopes) that are presented by antigen-presenting cells such as dendritic cells, B cells, and macrophages. This process results in the maturation and expansion of T cells that are specific for these viral epitopes and is followed by their migration to the liver, where they perform their effector function.
During acute HBV infection, HBV spreads through the liver without causing clinically overt liver inflammation until it reaches a logarithmic phase of replication. At this point, HBV-specific T cells are detectable and most HBV DNA molecules are cleared from the liver via noncytopathic mechanisms mediated by cytokines that are released by liver-infiltrating HBV-specific CD8 + cells. Cell-mediated immune responses are efficient in self-limited infection because the responses are vigorous, multispecific, and oriented toward type 1 helper T-cell functions. In contrast, persons with chronic HBV infection exhibit infrequent, narrowly focused, and weak HBV-specific T-cell responses that fail to clear HBV from infected hepatocytes. In chronic hepatitis B, the majority of mononuclear cells in liver infiltrates of patients at any given time are non-antigen-specific.
CD8 + CTLs are the primary mediator of HBV clearance and higher frequencies of HBV-specific T cells correlate with better control of HBV replication in chronically infected patients. , However, the antiviral function of CD8+ CTLs can also contribute to immunopathology by inducing chemokines that drive nonspecific mononuclear inflammatory infiltrates observed during hepatitis. To be recognized by the CD8 + CTLs, targeted hepatocytes must present viral epitopes as short peptides that have been endogenously processed and fit within the peptide-binding groove of the class I MHC molecules. The binding of the CTL T-cell receptor to the peptide-MHC complex on the hepatocyte surface can then result in the direct killing of the infected cell and release of potent antiviral cytokines by the activated CTL. Although hepatocytes express MHC-I, there is little evidence that they express MHC-II or are directly recognized by MHC class II-restricted CD4 + helper T cells. CD4 + T cell activation requires the appropriate presentation of viral peptides in the context of class II MHC molecules, which may occur in lymphoid tissues or liver-resident antigen-presenting cells such as dendritic cells, Kupffer cells, and endothelial cells. The CD4 + cells produce antiviral cytokines and provide help in stimulating neutralizing antibody production. Antibody neutralization limits intrahepatic spread of virus during primary infection and serves an important role in preventing reinfection.
Five phases of chronic hepatitis B have been described: (1) HBeAg-positive chronic HBV infection (immune tolerance), (2) HBeAg-positive chronic hepatitis B (immune clearance), (3) HBeAg-negative chronic HBV infection (inactive carrier state), (4) HBeAg-negative chronic hepatitis B (reactivation), and (5) the HBsAg-negative phase ( Fig. 79.4 ). These consecutive phases are much more likely to be apparent in patients with acquisition of chronic hepatitis B early in life. The newer nomenclature is based on the description of the 2 main characteristics of chronicity: infection (no inflammation) versus hepatitis (inflammation).
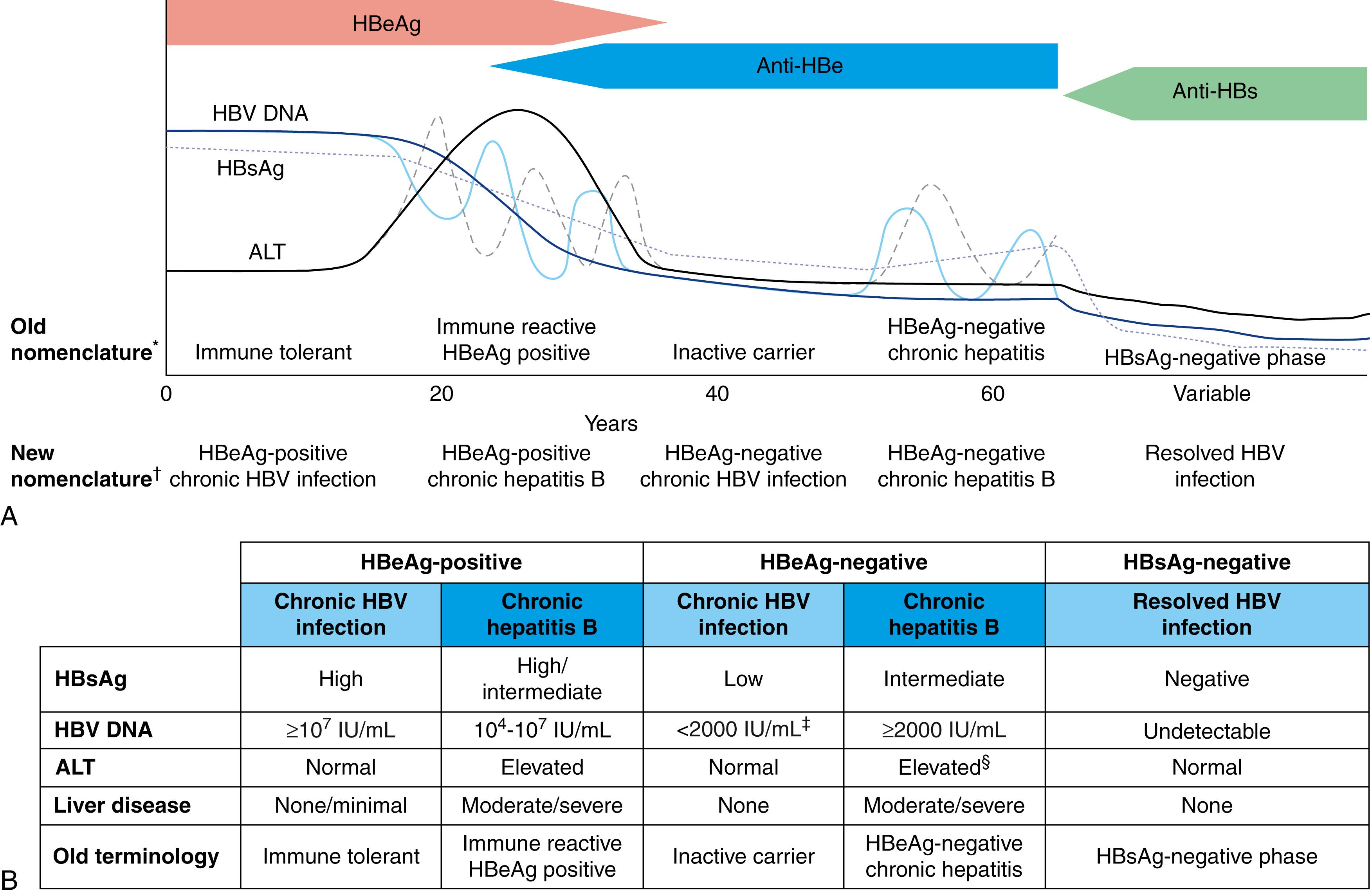
HBeAg-positive chronic HBV infection is often the earliest phase to be recognized when there is a history of infection at birth or the first few years of life. It is characterized by HBeAg positivity, high levels of HBV DNA (≥10 7 IU/mL), low or normal levels of serum aminotransferases, and minimal or no necroinflammation or fibrosis in the liver. During this phase, the rates of HBeAg loss are low. Experiments in transgenic mice suggest that HBeAg induces a state of immunologic tolerance to HBV in neonates. Perinatal transmission of HBeAg is considered one of several potential mechanisms underlying the immune-tolerance phase.
HBeAg-positive chronic hepatitis B often begins after several decades of HBV infection and is characterized by elevated serum aminotransferase levels, lower HBV DNA levels than in HBeAg-positive chronic HBV infection, and histologic evidence of chronic hepatitis. The trigger mechanisms for this apparent immunologic activation against HBV are poorly understood, but CD8 + CTL-mediated lysis of infected hepatocytes has been shown to occur. The duration of this phase varies and frequently lasts many years. Continued pressure by the host immune system against the virus may result in HBeAg seroconversion (loss of HBeAg with the development of anti-HBe in serum). The mean annual rate of spontaneous HBeAg seroconversion generally ranges from 8% to 15% in older children or adults with elevated serum ALT levels; however, the rate is considerably lower among Asian children and immunocompromised persons.
HBeAg-negative chronic HBV infection is the third phase and occurs in most patients who undergo HBeAg seroconversion. This phase is characterized by normalization of serum ALT and low (<2000 IU/mL) or nondetectable serum HBV DNA levels (see Fig. 79.4 ). Over time, hepatic necroinflammation and fibrosis subside. The inactive phase may last a lifetime.
HBeAg-negative chronic hepatitis B develops after reactivation, which may occur spontaneously because of a loss of immunologic control over viral replication or may be due to immunosuppressive drug therapy (see later). Reactivation is defined by the reappearance of high levels of HBV DNA in serum and often a noticeable rise in serum ALT levels. Therefore, HBeAg seroconversion does not always indicate quiescent disease. As many as 30% of persons who undergo HBeAg seroconversion enter into a subsequent phase of active disease that is caused by the selection of HBeAg-negative mutants (precore mutation, core promoter mutation, or a combination of both). At least 50% of these persons exhibit fluctuations in HBV DNA and aminotransferase levels, and recognition of active disease and exclusion of the inactive HBsAg carrier state (see later) may require serial assessment of both serum HBV DNA, quantitative HBsAg, and aminotransferase levels.
The HBsAg-negative phase is the last phase and is characterized by a negative test for HBsAg and a positive test for antibody to HBcAg (anti-HBc), with or without detectable antibody to HBsAg (anti-HBs). In rare cases, the absence of HBsAg could be related to the sensitivity of the assay used for detection. Patients in this phase have normal ALT values and almost always undetectable serum HBV DNA. Covalently closed circular HBV DNA (cccDNA) can still be detected in the liver (see later).
If the active hepatitis phases remain untreated, cirrhosis can be anticipated to develop in at least 20% of cases. Various factors have been determined to increase the risk of cirrhosis, and, of these, older age, male gender, the stage of fibrosis at presentation, and ongoing HBV replication are perhaps the most important clinically. Combined infection with HDV (see Chapter 81 ), HCV (see Chapter 80 ), or HIV, as well as concomitant alcohol abuse, has also been linked to a higher rate of development of cirrhosis and HCC.
When cirrhosis develops, 2 major complications may occur: hepatic decompensation and HCC. The estimated annual frequency of developing hepatic decompensation in HBV-associated cirrhosis is 5% to 8%; the annual rate of HCC is 2% to 4%. Factors associated with an increased risk of HCC include male gender, age 45 years or greater, having a first-degree relative with HCC, the presence of cirrhosis, HBeAg positivity, reversion from anti-HBe to HBeAg positivity, and increased HBV DNA levels regardless of the HBeAg state. Many of these risk factors are part of the risk scores that have been developed for predicting the occurrence of HCC in persons with chronic hepatitis B. HCC can still develop in HBsAg-positive persons with none of the identified risk factors, but less frequently. In addition, HCC can occur in cirrhotic patients who have undergone HBsAg seroconversion, and all patients with cirrhosis need continued surveillance ( Box 79.2 [see Chapter 96 ]).
African American carriers over 20 years of age
Asian female carriers over 50 years of age
Asian male carriers over 40 years of age
Carriers who have a family history of HCC
Persons who are coinfected with HDV, HCV, or HIV
Persons with HBV cirrhosis (at any age)
Persons with persistent active infection (high serum levels of HBV DNA and evidence of ongoing liver injury)
The serum ALT level has been used conventionally as a measure of disease activity in patients with chronic hepatitis B. A serum ALT level within the normal laboratory reference range, however, has been shown to be an imperfect surrogate marker for lack of disease activity. Clinical laboratories base their range of normal values on blood donors without known liver disease, but this population may include persons who are obese, consume alcohol, and have diabetes mellitus, each of which tends to increase the apparent upper limit of normal (ULN). For purposes of guiding management of chronic hepatitis B, a ULN for serum ALT of 35 U/L for males and 25 U/L for females is recommended. Studies in Asia and the USA have shown that as many as 20% to 30% of Asian HBV carriers with persistently normal serum ALT levels and serum HBV DNA levels over 2000 IU/mL (roughly equivalent to 10,000 copies/mL) have grade 2 or greater inflammation and stage 2 or greater fibrosis (on scales of 0 to 4) on a liver biopsy specimen. HBeAg-negative Asian HBV carriers with high-normal serum ALT levels by standard reference ranges tend to be older, have a greater frequency of serum HBV DNA levels in excess of 2000 IU/mL, and have a higher frequency of basal core promoter HBV mutations—all features that can be associated with adverse long-term outcomes. Therefore, liver biopsy or a noninvasive determination of hepatic fibrosis can be a useful tool to ensure that the severity of underlying liver disease is not underestimated in such persons (see Chapter 73, Chapter 74, Chapter 80 ).
Population-based Asian cohort studies have established that the serum HBV DNA level is the single best predictor of future progression to cirrhosis and HCC in HBV-infected persons. , In the prospective REVEAL-HBV natural history cohort study, over 3600 untreated HBV (HBsAg-positive) carriers from Taiwan were followed for more than 11 years. Of these, 60% were male, 40% were older than age 50, 85% were HBeAg negative, and 95% had normal serum ALT levels using standard reference ranges. The calculated relative risks for cirrhosis and HCC were shown to correlate with the level of HBV DNA on entry into the study when compared with a reference population of HBV carriers with undetectable serum HBV DNA. Even serum HBV DNA levels as low as 10,000 copies/mL (equivalent to 2000 IU/mL) were associated with a higher relative risk of cirrhosis and HCC. The relative risk was highest (hazard ratio of 10) in persons with a serum HBV DNA level that was greater than 20,000 IU/mL and intermediate (hazard ratio of 3.8) in persons in whom the serum HBV DNA level decreased spontaneously from greater than 100,000 copies/mL at the time of enrollment to less than 2000 IU/mL at the last point of follow-up. These data can be interpreted to mean that both the duration and level of viremia are important risk factors for the development of HCC. The data also suggest that suppression of serum HBV DNA levels, whether spontaneous or induced by antiviral therapy, lowers the risk of HCC.
Some authorities recommend that Asian men 50 years of age or older with serum HBV DNA levels 20,000 IU/mL or greater receive long-term therapy with a nucleoside (or nucleotide) analog to prevent HCC, even if serum ALT values are normal. Additional support for this recommendation can be found in a landmark study in which more than 600 Asian patients with advanced fibrosis and a serum HBV DNA level greater than 20,000 IU/mL were randomized in a ratio of 2:1 to active treatment with the nucleoside analog lamivudine or placebo. Disease progression and HCC occurred significantly less frequently in the group of patients randomized to lamivudine.
The incubation period of acute hepatitis B varies from a few weeks to 6 months (average, 60 to 90 days), depending on the amount of replicating virus in the inoculum. In many patients, particularlB978-0-323-60962-3.00074-6y children, acute hepatitis B remains asymptomatic. The disease may be more severe in patients coinfected with other hepatitis viruses and in those with established underlying liver disease. Acute infections are heralded by malaise, nausea, vomiting, and, in 10% to 20% of patients, a prodrome of fever, arthralgias or arthritis, and rash, which is most commonly maculopapular or urticarial. This prodrome results from circulating HBsAg-anti-HBs complexes that activate complement and are deposited in the synovium and walls of cutaneous blood vessels. These features generally abate before the manifestations of liver disease and peak serum aminotransferase elevations are observed. Jaundice develops in only about 30% of patients.
Clinical symptoms and jaundice generally disappear after 1 to 3 months. In general, elevated serum ALT levels and serum HBsAg titers decline and disappear together, and in approximately 80% of cases, HBsAg disappears by 12 weeks after the onset of illness. IgM anti-HBc is the best viral biomarker to confirm acute hepatitis B and to distinguish it from chronic infection. Persistence of HBsAg after 6 months implies development of a chronic infection state, with only a small likelihood of recovery during the next 6 to 12 months.
Serum aminotransferase levels of 1000 to 2000U/L are typical during acute hepatitis B, with the ALT higher than the AST level. In patients with icteric hepatitis, the rise in serum bilirubin levels often lags behind the rise in ALT levels. The peak ALT level does not correlate with prognosis; the prothrombin time (INR) is the best indicator of prognosis. If ALF develops, patients usually present within 4 weeks of the onset of symptoms and have associated multiorgan dysfunction, coagulopathy, encephalopathy, and high mortality rates if they are not treated with prompt antiviral therapy and LT. Patients older than 40 years of age appear to be more susceptible than younger persons to “late-onset” liver failure, which occurs several months after the onset of acute symptoms and is associated with encephalopathy and renal dysfunction. The pathogenic mechanisms of this severe form of HBV-related hepatitis are poorly understood but are presumed to involve massive immune-mediated lysis of infected hepatocytes and possibly impaired regeneration of new hepatocytes (see Chapter 95 ).
A history of acute or symptomatic hepatitis is often lacking in patients with chronic HBV infection. When symptoms are present, fatigue tends to predominate over other constitutional symptoms, such as poor appetite and malaise. Patients may remain asymptomatic even during periods of reactivated hepatitis. In other instances, particularly when superimposed on cirrhosis, reactivation of HBV infection may be associated with frank jaundice and signs of liver failure.
Physical examination may be normal, or hepatosplenomegaly may be found. In decompensated cirrhosis, jaundice, ascites, and peripheral edema are common. During exacerbations of disease, serum ALT levels may be as high as 1000 U/L or more, and the clinical and laboratory picture is indistinguishable from that of acute hepatitis B, including the presence in serum of IgM anti-HBc in some cases. Progression to cirrhosis should be suspected whenever hypersplenism, hypoalbuminemia (in the absence of nephropathy), or a high INR is found. The serum AST level is often higher than the serum ALT level in patients with advanced cirrhosis.
Although uncommon, extrahepatic syndromes can occur with chronic and acute hepatitis B. They are important to recognize because they may occur without clinically apparent liver disease and can be mistaken for independent disease processes in other organ systems. The pathogenesis is not completely understood but likely involves an aberrant immunologic response to extrahepatic viral proteins. Many of the extrahepatic manifestations are observed in association with circulating immune complexes that activate serum complement. Serum complement levels are generally low, and antiviral therapy may be beneficial in reducing the amount of immunologically activating viral antigens.
The arthritis-dermatitis prodromal manifestations of acute hepatitis B must be distinguished from inflammatory forms of arthritis, because glucocorticoid therapy, if mistakenly given to these patients, can lead to enhanced HBV replication, and abrupt withdrawal of these agents may be associated with a potentially severe flare in disease activity.
As many as 30% of patients with polyarteritis nodosa are infected with HBV, but the disorder develops in less than 1% of patients with chronic HBV infection. This association has been reported predominantly in North America and Europe but less so in Asia, where HBV is acquired perinatally. Typical features include arthralgias, fever, rash, abdominal pain, renal disease, hypertension, mononeuritis multiplex, and CNS abnormalities. Plasmapheresis may be useful, but the best therapeutic responses have also been observed with antiviral agents, given alone or in combination with plasmapheresis or immunosuppressive therapy.
Several types of glomerular lesions have been described in patients with chronic HBV infection; membranous glomerulonephritis and membranoproliferative glomerulonephritis are the most common. Renal biopsy specimens have demonstrated immune-complex deposition and cytoplasmic inclusions in the glomerular basement membrane. Nephrotic syndrome is the most common presentation of HBV-associated glomerulonephritis. The diagnosis requires the presence of immune-complex glomerulonephritis in a renal biopsy specimen and the demonstration of glomerular deposits of one or more HBV antigens, such as HBsAg, HBcAg, or HBeAg, by immunohistochemistry. The renal disease typically resolves in months to several years in children. Resolution may occur after HBeAg seroconversion. The natural history of HBV-related glomerulonephritis in adults has not been well defined, but several reports suggest that glomerular disease is often slowly and relentlessly progressive. Successful treatment has been accomplished with IFN-α and has been linked to long-term control of HBV replication. Therapy with nucleoside (or nucleotide) analogs has also resulted in improved renal function and diminished proteinuria.
Type II cryoglobulins consist of a polyclonal IgG and monoclonal IgM with rheumatoid factor activity, whereas type III cryoglobulins contain polyclonal IgG and IgM. Type II and type III cryoglobulinemia have been associated with hepatitis B, but the association, unlike that with hepatitis C, is uncommon. Cryoglobulinemia may be associated with systemic vasculitis (purpura, arthralgias, peripheral neuropathy, and glomerulonephritis) but is often asymptomatic. Nucleos(t)ide analog therapy has been used successfully to treat symptomatic cryoglobulinemia.
Chronic HBV infection is characterized by mononuclear cell infiltration in the portal tracts. Periportal inflammation often leads to the disruption of the limiting plate of hepatocytes (interface hepatitis), and inflammatory cells often can be seen at the interface between collagenous extensions from the portal tracts and liver parenchyma (referred to as active septa ). During reactivated hepatitis B, lobular inflammation is more intense and reminiscent of that seen in acute viral hepatitis. Steatosis is not a feature of chronic hepatitis B, as it is of chronic hepatitis C.
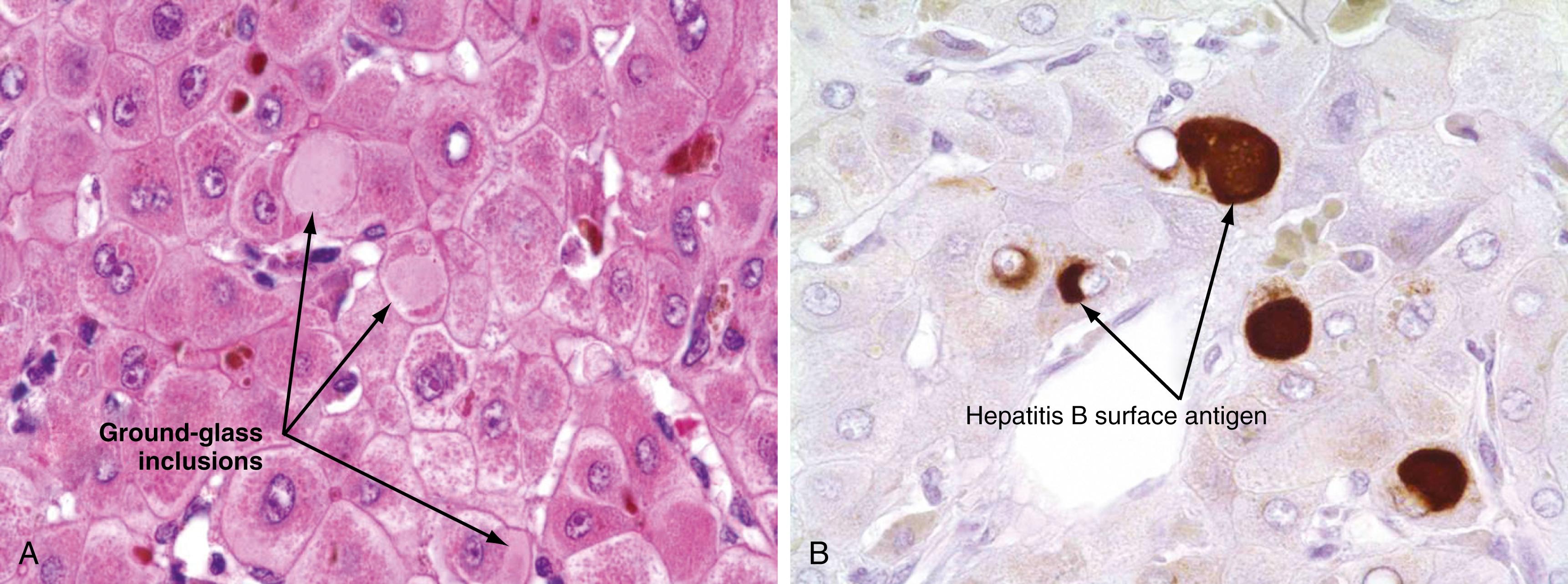
The only histologic feature noted on routine light microscopy that is specific for chronic hepatitis B is the presence of ground-glass hepatocytes ( Fig. 79.5 ). This morphologic finding results from accumulation of HBsAg particles (20 to 30 nm in diameter) in the dilated endoplasmic reticulum. Because of high levels of cysteine in HBsAg, ground-glass cells have a high affinity for certain dyes, such as orcein, Victoria blue, and aldehyde fuchsin. Ground-glass hepatocytes may also be seen in HBV carriers, in whom they may be detected in up to 5% of cells. When present in abundance, ground-glass hepatocytes often reflect a high level of viral replication. Immunofluorescence and electron microscopic studies have shown HBcAg inside the hepatocyte nuclei of affected cells. During periods of intense hepatitis activity, cytoplasmic core antigen staining is generally observed. After successful treatment of HBV infection with a nucleos(t)ide analog, the cytoplasmic core antigen staining often disappears, but nuclear core antigen staining due to persistence of the HBV cccDNA transcriptional template may remain.
Chronic hepatitis B is often punctuated by sudden flares of disease activity that are characterized by a precipitous increase in serum aminotransferase levels. Although a uniform biochemical definition of a flare is lacking, it has frequently been described as an increase in serum ALT levels to at least 2 to 3 times the baseline value and at least 100 IU/mL. Flares are an important part of the natural history of hepatitis B because they can lead to histologic progression when they occur repeatedly and are moderate or severe. Acute flares in chronic hepatitis B occur in association with a number of circumstances ( Table 79.1 ). Most flares are preceded by an increase in viral replication, which stimulates an enhanced cellular immune response that targets virus-infected hepatocytes. The mechanisms behind the increase in viral replication are unknown in many instances and are presumed to be due to the weakening of immune control over viral replication or to the emergence replication-fit viral mutants such as core promoter mutants or drug-resistant mutant HBV (see earlier). Flares following viral relapse after nucleos(t)ide analogs are stopped have been well described. Irrespective of the cause of the increased viral replication, however, the biochemical abnormalities usually occur coincident to or immediately after an increase in serum HBV DNA levels.
| Cause of Flare | Comment |
|---|---|
| Spontaneous | Factors that precipitate viral replication and loss of immune control are unclear |
| Immunosuppressive therapy | Flares are often observed during or shortly after withdrawal of the agent; preemptive antiviral therapy is required |
|
Flares may be observed within the first 3 mo of initiating therapy in 30% of patients and may herald HBeAg seroconversion in some patients |
|
– |
|
Reinforce patient education to improve medication adherence |
|
Mainly an issue for agents that have a low genetic barrier to resistance such as lamivudine and telbivudine Confirm genotypic resistance with resistance testing |
|
Flares indicate clinical relapse (re-elevation of serum ALT levels andre-appearance of HBV DNA in serum in those previously virally suppressed) |
| HIV coinfection | Flares can occur as a result of the direct toxicity of ART or with immune reconstitution; HBV increases the risk of antiretroviral drug hepatotoxicity |
| Other liver diseases | Alcohol overuse Autoimmune liver disease Drug and toxin-induced liver injury NAFLD |
| Precore and core promoter mutants | Fluctuations in serum ALT levels are common in HBeAg-negative patients who harbor these variants |
| Coinfection with HCV | HBV may be suppressed in HCV-coinfected patients Beware of HBV reactivation in coinfected patients undergoing DAA therapy for HCV infection |
| Coinfection with HDV | HBV is typically suppressed in HDV coinfected patients Higher risk of liver disease progression requires close monitoring |
Spontaneous flares have been observed in patients with HBeAg-positive chronic hepatitis B, in whom they occur in 5% to 10% of patients annually, and in those with HBeAg-negative chronic hepatitis B, in whom fluctuations of both serum HBV DNA and ALT levels are common. It is not clear if severe physical or emotional stress can weaken the immune system and lead to a secondary increase in viral replication.
In persons who acquire HBV infection early in life, flares become more common during adulthood. In this situation, the flares are almost certainly host-driven rather than virally mediated, and although poorly understood, they are most likely the result of a change in the regulation of viral antigen-specific T cells.
Reactivation of hepatitis B with flares of serum aminotransferase levels is a well-recognized complication of cytotoxic or immunosuppressive therapy, including conventional cancer chemotherapy and potent biologic response modifiers that are used to treat rheumatic, gastrointestinal, and skin disorders. , Although many drugs have been reported to induce HBV reactivation (HBV-r), they tend to fall into one of several classes of agents ( Table 79.2 ). Suppression of the normal immunologic responses to HBV during therapy leads to enhanced viral replication and is thought to result in widespread infection of hepatocytes. In general, the more potent the immunosuppression, the higher the level of viral replication and, thus, the greater the potential for serious clinical consequences. The literature provides ample evidence for HBV-r leading to severe hepatitis, death from ALF, and delay or inability to continue treatment for the underlying disease. When reactivation occurs in the setting of cancer chemotherapy or systemic treatment for a severe autoimmune disorder, the patient may not be eligible for salvage LT. A growing body of evidence shows benefit to screening all patients in need of immunosuppressive drug therapy for HBsAg and anti-HBc and prophylactically treating HBsAg-positive patients with antiviral therapy. Reactivation of hepatitis B is discussed later in the chapter.
| Risk ∗ | ||
|---|---|---|
| Medication | HBsAg-positive Patient | HBsAg-negative/anti-HBc-positive Patient |
| Anti-CD20 B cell-depleting agents (rituximab, ofatumumab) | High | High † |
| Hematopoietic stem cell transplantation | High | Moderate † |
| Anthracycline (doxorubicin, epirubicin) | High | Moderate ‡ |
| High-dose prednisone (>20 mg daily) for >4wk | High | Moderate ‡ |
| Anti-CD 52 (alemtuzumab) | High | Low |
| Anti-TNF (infliximab, adalimumab) | Moderate | Moderate ‡ |
| Cytokine or integrin inhibitors (abatacept, natalizumab, ustekinumab, vedolizumab) | Moderate | Moderate |
| Tyrosine kinase inhibitor (imatinib, nilotinib, sorafenib) | Moderate § | Moderate § |
| Cytotoxic chemotherapy | Moderate | Low |
| Low-dose prednisone (<10 mg daily) for >4 wk | Moderate | Low |
| Antirejection regimens for solid organ transplant | Moderate | Low |
| Traditional immunosuppressive therapy: azathioprine, methotrexate, 6-mercaptopurine | Low | Low |
| Any dose of glucocorticoid for <1 wk | Low | Low |
| Intra-articular glucocorticoids | Low | Low |
∗ See Box 79.3 .
† AASLD classifies risk as moderate.
Antiviral treatment of chronic hepatitis B can be associated with ALT increases and flares of hepatitis in several circumstances. Flares may occur during IFN or nucleos(t)ide analog therapy, after withdrawal of nucleos(t)ide analogs or glucocorticoid therapy, and in association with the emergence of lamivudine-, adefovir-, entecavir-, or telbivudine-resistant mutants.
IFN-induced flares of chronic hepatitis B occur in approximately one third of treated patients and result from the immunostimulatory properties of the drug. Flares occur with conventional and pegylated formulations of IFN (see later and Chapter 80 ) and have been reported to occur more frequently in patients infected with HBV genotype A than with other genotypes. This finding may explain the higher rate of sustained virologic response and HBsAg clearance seen in this group of patients. Serum ALT flares have been shown to be a predictor of sustained virologic response and may be especially important in achieving a sustained virologic remission in patients with a high level of viremia. , Flares that occur in patients with advanced liver fibrosis have been associated with clinical deterioration, and, as a result, IFN should be used cautiously in patients with cirrhosis and should not be used in patients with decompensated cirrhosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here