Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The head and neck is a unique anatomic region of the human body with specialized function. The sophisticated arrangement of multiple tissue types comprises unique physical traits from individual to individual. Regions of the head, face, and neck are responsible for multiple high functioning mechanisms, including mastication, swallowing, phonation, articulation, respiration, vision, periorbital protective mechanisms, audition, and olfaction, all of which have been characteristic in the evolutionary development of man. Moreover, interpersonal communication and recognition of social cues are all delivered and interpreted via facial contour and movement. The subtleties of facial anatomy contribute to individual identity, and although phenotypic differences are recognized among ethnicity, sex, and various individuals, there is an overarching commonality of features that permeates a sense of normal . Understanding these relationships, patient expectations, and the limits of reconstruction are certainly enhanced with surgeon experience in the treatment of craniofacial injury and deformity. However, a systematic approach to craniofacial reconstruction that abides by several tenets is essential in optimizing outcomes. Unlike other regions of the body, aesthetic outcomes may be just as important as functional outcomes in the head and neck due to the social implications of deformity.
The deformities of the head or neck are often the result of trauma, oncologic resection, infection, osteoradionecrosis, and congenital or developmental pathology. Multiple tissue types that compose the craniofacial region including bone, cartilage, nerve, fat, muscle, mucosa, and varying dermal and epidermal thickness, as well the inherently intricate contours of the craniofacial skeleton can complicate reconstruction. However, significant advances in microvascular and craniomaxillofacial surgery over the past 50 years have resulted in dramatically improved outcomes. The purpose of this chapter is to provide a methodological approach to microvascular reconstruction of the head and neck while optimizing aesthetic and functional outcomes, as well as developing an effective means of achieving reliable and replicable results. The workhorse flaps used in reconstructing the various regions of the head and neck are described with an emphasis on their application to the craniofacial segments, their advantages, and disadvantages.
As with all reconstructive procedures, meticulous assessment of wounds and careful reconstructive planning are essential to achieving successful outcomes. However, strict adherence to several critical principles specific to the demands of craniofacial surgery is paramount to addressing complex injury or deformity of the head and neck. These concepts are:
Aesthetic subunit appearance
Defect boundaries
Tissue requirements
Establishment of a skeletal buttress framework
Generous soft tissue volume
Early or immediate reconstruction
Local revisions through multi-stage planning
Consideration of these principles is essential in attaining a functional and aesthetic outcome that is predictable and reproducible. A methodical structure to plan and perform segmental or multi-segment craniomaxillofacial reconstruction of composite defects provides consistency in surgical approaches and optimizes flap selection. Furthermore, these principles mark a paradigm shift in head and neck reconstruction as they synthesize an amalgam of advancements from aesthetic surgery, craniofacial surgery, trauma surgery, and radiation, wound, and scar biology.
It is well established that replacing “like with like” is the gold standard of aesthetically pleasing reconstructions. However, this is often difficult to achieve because the face can be divided into discrete aesthetic subunits based on variations in skin texture, color, thickness, and histology. Injury can affect multiple subunits or portions of subunits that makes flap selection more challenging. Understanding the lines of demarcation between facial segments and appreciating the variation in skin thickness throughout the face can aid the surgeon in using skin grafts from areas with similar characteristics to the site being reconstructed. Pioneered by Mario González-Ulloa, these concepts permeate throughout multiple facets of plastic surgery but were lacking in early microvascular surgery of the head and neck. Perhaps, these principles were considered secondary because early microvascular flap failure rates were initially too high. Over time, advancements in technique and instrumentation have precluded the surgeon from simply trying to reconstruct a defect by “filling a hole,” and have permitted restoration of entire aesthetic subunits, even when removing healthy tissue may be required. This allows the surgeon to achieve superior reconstruction with concomitant consideration of cosmetic results.
It is imperative to recognize that aesthetic facial subunits are not just “skin deep.” Each facial region is comprised of vertical and horizontal skeletal buttresses that provide critical soft tissue support and shape. In addition, the subcutaneous fat is partitioned into discrete compartments of the face, a concept championed in cosmetic facial surgery. The perception of facial aesthetic subunit is actually dependent on multiple elements, representing an amalgam of interactions between composite soft and hard tissue. Therefore, a “defect-oriented approach,” in which a soft tissue-only flap is used to solve the immediate goal of wound closure will often result in suboptimal cosmesis. Reconstructing a composite tissue defect of an aesthetic subunit of the face with free tissue transfer requires the necessary elements that are absent in a wound including the underlying skeletal support with the coverage of soft tissue.
Rearrangement of defect boundaries affords the surgeon several benefits toward improving outcomes. First, the wound edges of various defects are prone to infectious complications and if not maintained in a pristine environment, the wound is prone to desiccation and breakdown. Expansion or rearrangement of wound borders allows for a decreased microbial burden and a healthy wound edge comprised of collagenous, elastic, and a well-vascularized environment facilitating better wound healing. Second, the dogmatic principles of aesthetic subunit reconstruction dictate that certain defects can be made larger in order to resurface the entire unit. This allows for incisions to be hidden in skin creases and behind or within specific structures. For defects that comprise >60% of an aesthetic subunit, resection of the entire subunit may be indicated since free flap reconstruction can reconstruct the expanded defect and achieve superior cosmetic results. Flap homogeneity throughout the entire subunit conforms to a more uniform distribution of color, thickness, and texture while disguising incisions at the junctions of subunits. This is depicted in Figure 14.1 , where a large forehead defect prompts extension of the borders of resection, making the forehead defect larger than initially encountered. Free tissue transfer allows for scars to be hidden in the pretrichial hairline and just superior to the eyebrows.
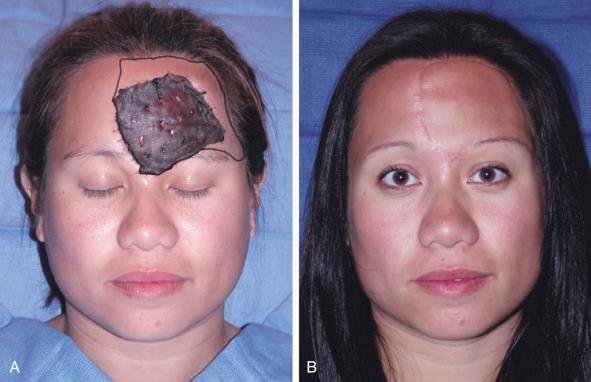
The concept of extending the boundary of defects when >60% deformity to an aesthetic subunit is present, is not absolute. The inherent characteristics of the subunit at hand must dictate the reconstructive effort because not all aesthetic subunits are as easy to re-create as others with free tissue transfer. For example, re-creation of the vermillion lip is challenging, since no autologous free flap options exist to re-create a satisfactory semblance of the subunit. In the case of a partial vermillion lip defect, resection and replacement of the entire unit would not result in a superior aesthetic outcome. When considering resection of additional local tissue the degree of resection may be dictated by plans for future revisionary procedures. In order to achieve total aesthetic subunit reconstruction, the option for potentially advancing local cutaneous tissue with successive revisionary procedures (or with tissue expansion) can lead to complete excision of the initial free flap skin paddle and reestablish the native skin of the original defect. This concept is further discussed in the revisionary procedures portion of this chapter.
Adhering to the principles of replacing missing components such as bone, soft tissue, or oral lining can guide the surgeon in planning and executing the replacement of “like with like” tissue to herald better results. The concept of considering the tissue types and establishing their continuity or obliterating an apparent discontinuity is particularly important when some form of “lining” tissue (e.g., conjunctiva, nasal mucosa, sinus mucosa, or oral mucosa) is deficient. Failure to provide an adequate lining can severely compromise results after free flap reconstruction, leading to contracture, fibrosis, and chronic infection. Meticulous preoperative planning and anticipation for such defects, although not initially obvious can help the surgeon to better predict which tissue types are deficient. Furthermore, reconstructions should be predicated on re-creation of the defect, especially in cases of delayed reconstruction where scarring and contracture may have obscured the initial defect, making it more difficult to appreciate a lining deficiency. The choice of free flap should not only include the adequate components required for skin coverage, soft tissue bulk, and skeletal support, but also a surplus of skin, fascia, or muscle that can be used to provide lining. This approach ensures that all lining deficiency is addressed. The importance of restoring tissue types and their subsequent continuity was stressed in the early years of plastic surgery by Gillies and Keegan, but despite the importance stressed on such structures by the pioneers of plastic surgery, the need for lining reconstruction is still often underestimated by contemporary microvascular surgeons.
It is imperative that the microsurgeon address the need for hard tissue reconstruction in free tissue transfer. When facing a large composite facial defect in which both bone and soft tissue are missing, selecting a flap consisting of abundant soft tissue rather than both bone and soft tissue is not recommended. This approach succeeds in “filling the hole,” but falls short of the fundamental reconstructive goal of replacing “like with like.” The importance of skeletal structure as a means of providing structural support and aesthetic guidance anchoring soft tissue has been well described by Rodriguez and colleagues by preserving craniofacial skeletal buttresses. Emphasis on skeletal reconstruction has largely been championed by craniofacial surgeons, but restoration of the deficient soft tissues may be overlooked when skeletal reconstruction is overemphasized. With the synthesis of microsurgery and craniofacial surgery, surgeons now recognize the importance of reconstructing both soft and hard tissue. The advent of prosthetic implants, fixation plates, and non-vascularized bone grafts has bridged the surgeon's understanding of the crucial relationship of bony stabilization and soft tissue re-draping. Options for reconstructive approaches may seem vast at times, but a means of providing an optimal relationship of hard and soft tissue, similar to the premorbid state, should drive the decision-making process.
Historically, non-vascularized bone grafts were the mainstay of bony reconstruction and still have a limited role in treatment of small skeletal defects. The disadvantage of non-vascularized bone grafts includes unpredictable resorption rates and the inability to replace large structural defects. The unfortunate result of excessively using non-vascularized bone grafts for inappropriately sized defects will necessitate further reconstructions over time. Additionally, bone grafts generally provide insufficient bone stock to allow placement of osseointegrated implant prostheses. In certain cases, craniofacial bone defects can be reconstructed with an alloplastic implant covered by a vascularized soft tissue flap. However, successful incorporation of alloplastic material requires wide debridement and clearance of marginal tissue edges to minimized dehiscence and infection. Osteoradionecrosis and prior chronic infections are not absolute contraindications for using alloplastic material, but the surgeon must be vigilant in ensuring the maximal removal of any infectious burden to the defect. Alloplastic materials are prone to late infection and tend to extrude over time, leading to implant exposure. Undoubtedly, situations will call for wider resection of the wound and acknowledging that sacrificing tissue cannot be afforded to certain defects may preclude the use of prosthetic materials for reconstruction. As with non-vascularized bone grafts, the long-term complications may require further surgical interventions later down the line. Vascularized bone obviates many of the unforeseen complications that are associated with non-vascularized bone grafts and alloplastic materials, and therefore should be used for hard tissue reconstruction whenever possible. Adequate soft tissue and bone can often be incorporated in the same free flap; however, multiple flaps may sometimes be required to provide sufficient quantities of both types of tissue.
The soft tissue volume included in a free flap should be in slight excess of the actual amount of tissue that is deficient. If postoperative radiation therapy is anticipated, the excess volume should be further increased, since significant soft tissue contraction can result from radiation exposure. The degree of soft tissue resorption and atrophy is unpredictable. Including excess soft tissue affords the surgeon insurance of coverage in the event of excessive contracture or atrophy. It is always easier to debulk excess soft tissue at a later stage than to add volume secondarily. The inclusion of ample volumes of well-vascularized fat in particular will minimize subsequent fat necrosis and soft tissue resorption. Healthy adipose tissue will also provide the necessary volume for future revisionary reshaping procedures as the transferred tissue settles. Options for debulking via direct excision and/or liposuction in a revisionary procedure allow the surgeon to focus on cosmesis after the initial healing period and after flap survival is assured. In contrast to adipose tissue, denervated muscle flaps tend to atrophy significantly over time, compromising long-term aesthetic outcome and failing to provide durable coverage for alloplastic implants. For example, latissimus dorsi muscle flaps have been considered the workhorse flap and traditional choice for scalp coverage of titanium mesh cranioplasties, but such flaps have shown to thin significantly over time, often resulting in tenuous coverage or ultimate exposure of the underlying mesh.
Selection of the donor site for free soft tissue transfer must be guided in a patient-centered, individualized basis. Patient-specific needs must be considered in the risk–benefit analysis of selecting a flap, especially in the elderly, with respect to donor site morbidity, those with physical disabilities, and in the actively growing young person. A color match and the compatibility between donor site soft tissue volume and recipient site soft tissue deficit then becomes the priority. For instance, the variability in subcutaneous fat among patients is apparent when considering an anterolateral thigh (ALT) flap. Although some advocate aggressive thinning of the ALT flap at the primary stage, this practice is not recommended for craniofacial reconstruction. It is preferable to err on the side of caution with a thicker flap, which can be debulked secondarily.
In the face of trauma or oncologic resection, the microsurgeon must not prolong the time to reconstruction. Reconstructing the injury acutely minimizes the cascade of scar formation associated with the initial injury and confines the deleterious effects to a single period of postoperative wound contracture. The advantages of initiating early free tissue transfer to avoid these effects are two-fold. First, dissection of the recipient site and its vessels is simplified when not operating in a scarred bed. Second, the functional and cosmetic results of the reconstruction are improved by reducing the overall scar burden of the soft tissues in that area.
In the approach to congenital deformity reconstruction, the timing for intervention differs since there is no scarring associated with creation of the initial defect. In these instances, delaying surgery to an extent may be more appropriate for the patient. Determining time to intervention is on an individualized basis because of multiple factors and comorbidity issues to contemplate. Ongoing child development and whether he or she will ultimately “outgrow” their free flap reconstruction is difficult to predict, which significantly contributes to the decision-making process of timing reconstruction. Additionally, the distribution of body surface area differs in the pediatric patient, as the head compromises a greater percentage of the total surface area. Ensuring ample tissue may be a challenge in the pediatric patient, especially when considering free tissue transfer to the pediatric craniofacial region. For example, a latissimus dorsi flap can easily cover a large adult scalp defect, but it may not provide sufficient size for coverage in the pediatric patient. A final concern that may affect timing of reconstruction is the practical consideration of vascular pedicle size. The microsurgeon must understand one's limitation when dealing with smaller-caliber vessel anastomoses. At this level of complexity, although feasible, successful outcomes may vary in each individual surgeon's hands.
Secondary revisions must not be considered failure of the initial surgery, but instead be incorporated in the overall reconstructive approach as a method of optimizing outcomes. There is often an impetus to minimize the number of reconstructive procedures a patient requires. As a result, microsurgeons attempt single-stage procedures aimed at definitive reconstruction. However, this approach will generally compromise long-term outcomes due to skin color mismatch and soft tissue contracture. Ambitious single-stage procedures do not capture all of the tools in the plastic surgeon's armamentarium in solving large craniofacial defects. With the understanding that secondary revisions are often inevitable in optimizing complex defects, the initial reconstructive procedure no longer assumes the burden of complete reconstruction but is the first of multiple approaches.
Secondary revisions are fundamental for achieving successful reconstruction of the facial aesthetic units since injury is likely to cross multiple subunits and resultant scar, contracture, and contour and color mismatch are much more pronounced to the lay observer. The aforementioned concept of aesthetic subunits and establishing homogeneity of skin characteristics is reiterated as the eventual need for revisionary procedures should be anticipated at the time of the initial free tissue transfer. In accordance with the concept of defect boundaries, if a defect comprises <60% of the unit, maximal preservation of local surrounding tissue is required, as it may be successively recruited with local tissue rearrangement during secondary procedures. If the previously mentioned concept of soft tissue is employed, the initial free flap should include excess soft tissue, which can later be debulked, shaped, and ultimately covered by local cutaneous advancement. For example, consider a cheek defect comprising 40% of the aesthetic unit being reconstructed with a free ALT flap. Excision of the entire unit during the first reconstruction is not ideal because this leaves an obvious area of color-mismatched and hair-bearing skin, demarcating a stark contrast of the cheek aesthetic unit. Recruiting distant tissue transfer serves as a base for local tissue advancement and resurfaced to provide a more favorable cutaneous texture and color match. The foresight of future revisions allows initial free tissue transfer to be planned and executed with more success. In this case, the free flap can be selectively de-epithelialized and the remaining dermis can serve as a substantial anchor from which to re-suspend advanced local skin.
Other secondary procedures may include dermabrasion, soft tissue re-suspension, excision of soft tissue, suction lipectomy, and fat grafting. At this juncture, skin excision from the free flap with full-thickness skin grafting from a donor site similar in color and texture to the facial subunit, remains an option.
The goal of craniofacial microsurgery is to reestablish a necessary structural foundation with hard and soft tissue, and the goal of subsequent revision surgery is to refine contour and volume while modifying the “unlike” flap skin with “like” local skin. Integration of the aforementioned concepts is crucial in replacing missing tissue, maximizing craniofacial function, and optimizing aesthetic results.
The application of microsurgery to craniofacial surgery has been successfully established, largely due to the synthesis of multiple disciplines and their respective collaborations in the treatment of complex disease processes. The indications for microvascular intervention in head and neck surgery are constantly evolving and must not be restricted to a certain measurement of defect, anatomic location, or disease process. Although the reconstructive ladder is dogmatic to the practice of many plastic surgeons, free tissue transfer has long been established as a form of “jumping” the “steps” of the ladder. Free tissue transfer to the head and neck is most frequently incorporated following oncologic resection, trauma, infection, osteoradionecrosis, or congenital deformity or malformation, or as a means of reconstructing a failed prior flap. However, indications for free tissue transfer to the craniofacial region must broadly be considered from an anatomic, functional, and aesthetic perspective, all while considering alternative options. Essentially, fixed indications are difficult to define as anatomic, functional, and aesthetic components vary considerably and a single component of the three can skew an evaluation to necessitate microsurgical intervention, rather than using other reconstructive methods.
To simplify the approach to numerous defects of the head and neck, and to facilitate a comprehensive understanding of workhorse flaps that have been developed, refined, and proven to yield successful outcomes throughout the last decade, surgical technique, advantages, disadvantages, pitfalls, and their application are reviewed below. These flaps include the ulnar forearm flap; anterolateral thigh (ALT) flap; latissimus dorsi flap; deep circumflex iliac artery (DCIA) flap; and free fibula flap. Each flap is dynamic and can be altered slightly to incorporate various characteristics necessary for specific craniofacial defects. Their application to a variety of anatomic locations, including the scalp, periorbital region, midface, and mandible, is now described with corresponding cases and figures.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here