Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Because the female genital tract comprises both cutaneous and mucosal surfaces in contact with the external environment, exposure to a significant number of infectious agents, either commensal or pathogenic, is the norm. And because the lower genital tract is in communication with the normally sterile peritoneal cavity, ascending infections can be treacherous. In addition, close physical contact with sexual partners can lead to infectious sexually transmitted disease (STD), especially if the woman is exposed to multiple sexual partners. Since the first edition of this book, knowledge of the mirobiome of the female genital tract has greatly expanded by use of molecular techniques; the work has been supported by the National Institutes of Health Human Microbiome Project (HMP). The work has demonstrated the significant microbial diversity among women. The external genitalia and the lower female genital tract are the sites of the majority of infections, which are often transitory and without serious sequelae. Most of these infections are recognized clinically without need of biopsy and are treated on the basis of the clinical presentation. However, biopsy is warranted if the prescribed treatment is not effective or if the presentation is atypical. In addition, routine cervical cytology specimens (the Pap smear), used to screen for malignant and premalignant lesions, regularly identify a limited number of potential pathogens.
The upper genital tract can be the site of silent infections with serious secondary effects, such as infertility. In the case of suspected infection, the endometrium is readily amenable to biopsy, but the fallopian tubes and ovaries are usually examined only if significant disease is present.
Immunocompromised patients often experience more severe infections or infections due to organisms not previously associated with disease. The patients most affected include transplant recipients, oncology patients, and patients with acquired immunodeficiency syndrome (AIDS).
Most clinical reviews of infections of the female genital tract emphasize the anatomic site and clinical manifestations, with an added emphasis on the clinical appearance of lesions arising in the lower genital tract and the appearance on radiologic imaging for infections in the upper genital tract. For example, herpes infection of the cervix often causes small vesicles that evolve into shallow ulcers. Where appropriate, the clinical or radiologic appearance of the lesion is mentioned in the text here.
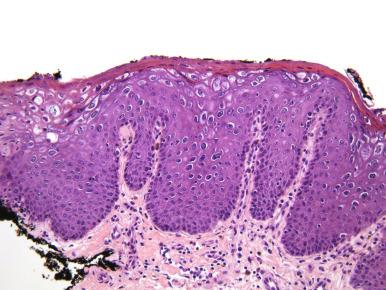
Genital infection by human papillomavirus (HPV) is the most common sexually transmitted infection in the United States. More than 100 HPV types have been identified, at least 40 of which infect the genital tract. The HPV genotypes are divided into low-risk and high-risk types depending on their oncogenic potential. All HPV types are epitheliotropic obligate intracellular viruses that promote cell proliferation in normally quiescent, more differentiated epithelial cells to produce a hyperplastic epithelial lesion. Low-risk HPV, most often types 6 and 11, produce exophytic genital warts or condyloma acuminata, usually found on the perineum and labia and, with less frequency, on the vagina, cervix, and anal region. High-risk HPV, such as HPV-16 and HPV-18, cause preneoplastic intraepithelial lesions in the lower genital tract, most often in the cervix and vagina; the oncogenic risk is highest in persistent infections. On the external genitalia, condyloma acuminata appear as exophytic, smooth to somewhat rough, flesh-colored papules, either single or multiple, that are not usually biopsied before treatment. However, because cervical or vaginal lesions often accompany condyloma acuminata of the external genitalia, a careful vaginal and cervical examination should be done, usually with a speculum examination after application of acetic acid, which causes an acetowhite appearance of the HPV-induced lesions due to coagulation of cytokeratins of infected squamous cells. If the condyloma acuminatum is biopsied, the histologic findings include acanthosis, parakeratosis, and hyperkeratosis, which may be associated with a branching papillomatous architecture with fibrovascular cores ( Fig. 17.1 ), but the hallmark is the HPV viral cytopathic effect, known as koilocytosis, which corresponds to a cavitation of the cytoplasm of the infected cell along with nuclear hyperchromasia and enlargement with membrane irregularity. Koilocytosis occurs as a result of the vegetative cycle of the HPV and is due to the accumulation of infective virions in the nucleus with disruption of cytoplasmic keratin in infected squamous cells. Koilocytosis is less pronounced in lesions of the perineum, vulva, and vagina than in lesions of the cervix. Diagnosis of HPV infection can be confirmed on tissue samples by molecular testing for HPV DNA or RNA; the HPV cannot be cultured and serologic testing is not routinely performed.
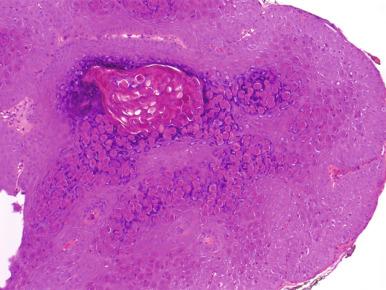
Molluscum contagiosum is caused by a chronic, localized infection with poxvirus that causes flesh-colored, rounded, raised papules with an umbilicated center. Diagnosis is usually based on the clinical appearance, but if a biopsy is performed to rule out other clinically similar lesions, such as those caused by cryptococcosis or histoplasmosis, the keratinocytes are found to contain large, eosinophilic cytoplasmic viral inclusions ( Fig. 17.2 ).
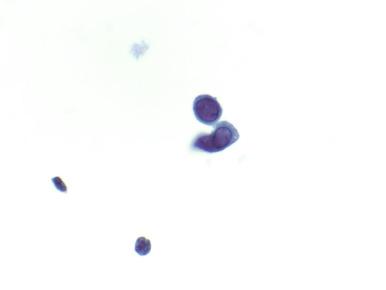
Herpes simplex virus (HSV) is common and often subclinical (manifested only by the serum antibody response), so the incidence of disease is difficult to determine. Both HSV type 1 and type 2 cause genital HSV infections. HSV infections are categorized as primary if the patient has no preexisting antibodies to HSV-1 or HSV-2; as nonprimary if a new infection arises in a patient with antibody evidence of the other HSV type; or as recurrent if there is serologic evidence of the same type as found in the reactivated infection. In a survey of adults in New York City, 28% were found to be infected with HSV-2 but only 12% were aware that they were infected. The most severe symptoms tend to occur during primary infection and include genital vesicles with subsequent ulcer formation, fever, and regional lymphadenopathy. Clinical diagnosis of HSV infection is confirmed by viral culture, polymerase chain reaction (PCR), direct fluorescence antibody, and type-specific serologic tests. PCR and serology are more sensitive than culture. If need be, a genital ulcer can be scraped to obtain material for cytology (Tzanck preparation) or biopsied. The Tzanck smear, reported to have a low sensitivity and specificity for HSV cytopathic effect, is prepared by scraping the base of a fresh ulcer ( Fig. 17.3 ). The slide may be air dried and stained with Wright-Giemsa stain, or the cells may be fixed with alcohol and stained with Papanicolaou stain. HSV-infected epithelial cells show either a homogenization of the chromatin (ground-glass appearance) or clumping of the chromatin on the nuclear membrane and in the center of the nucleus. The infected cells are often multinucleated. Biopsy is most informative if an intact vesicle is sampled. The intraepidermal vesicle shows degenerating keratinocytes; intranuclear viral cytopathic changes are most easily found on the margin of the vesicle. With ulceration, the changes are less specific and include chronic inflammation and granulation tissue ( Fig. 17.4 ).
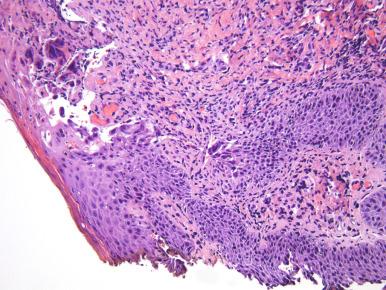
HSV-2 infection in HIV-infected women may produce hypertrophic vulvar lesions that may mimic a neoplasm. Biopsies to demonstrate viral cytopathic effect and culture are necessary to diagnose this uncommon condition.
Rarely, primary infection with Epstein-Barr virus (EBV) can be accompanied by genital ulcers that clinically mimic the more common ulceration caused by HSV ( Table 17.1 ). Ulceration due to EBV should be considered in young patients who have painful ulcers, lymphadenopathy in a region distant from the ulcer, and systemic symptoms. An EBV etiology should be considered if bacterial and viral cultures, as well as the results of syphilis serology studies, are negative.
| Organism (Disease) | Gross Characteristics |
|---|---|
| HSV | Shallow painful ulcers, initially pruritic focus evolves to blister |
| EBV | Ulcer similar to herpes |
| CMV | Ulcer similar to herpes |
| Treponema palladium (chancre of primary syphilis) | Painless ulcers |
| Chlamydia trachomatis (LVG) | Shallow, painless ulcer |
| Haemophilus ducreyi (chancroid) | Begins as nodule, evolves to pustule, then to painful ulcer |
| Klebsiella granulomatis (granuloma inguinale) | Painless ulcers evolve to hidradenitis |
| Escherichia coli (malakoplakia) | Papule or plaque evolving to ulcer |
Cytomegalovirus (CMV) infection of the vulva manifests with ulceration. Characteristic viral intranuclear and intracytoplasmic inclusions are identified in epithelial cells and endothelial cells ( Fig. 17.5 ).
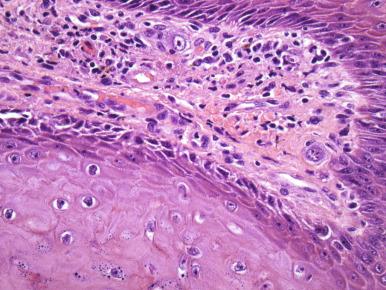
Erythrasma, an uncommon infection of the vulva caused by Corynebacterium minutissimum , is usually diagnosed by fluorescence of the bacteria under the Wood lamp. Rarely, Wood lamp examination is negative, but the diagnosis can be made histologically. The clinical presentation is similar to vulvar candidiasis, but, on biopsy, filamentous bacteria are present in the stratum corneum.
The chancre, the primary lesion of syphilis caused by the spirochete Treponema pallidum , is a nontender ulcer with a clean base. T. pallidum cannot be cultured routinely. Either serology or direct visualization of the organism in clinical specimens are the traditional testing methods. Darkfield microscopy, nucleic acid amplification (PCR), or direct fluorescent antibody is used on scrapings from the ulcer bed to diagnosis primary infection. On biopsy, the epidermis is acanthotic along the rim and thinned or eroded in the center of the lesion. A dense lymphoplasmacytic infiltrate is found in the dermis surrounding blood vessels with swollen endothelial cells. Silver stains, such as Warthin-Starry stain, demonstrate many 8- to 15-µm spirochetes within the epidermis and around blood vessels. Immunohistochemical staining for T. pallidum has been reported to be more sensitive than the silver stain.
Condyloma lata, the lesions of secondary syphilis that arise 1 to 6 months after healing of the primary chancre, appear clinically raised, somewhat flattened, and velvety. On biopsy, condyloma lata show epidermal hyperplasia and lymphoplasmacytic infiltration of dermal vessels with endothelial hyperplasia, dilatation of the vascular lumens, and thickening of their walls. Interface dermatitis and scattered epithelioid granulomas may be seen. Silver stain highlights spirochetes in a minority of cases, usually around blood vessels and within the epidermis.
Bacillary angiomatosis of the vulva and cervix has been reported in a patient with AIDS; it manifested as red-purple nodules that microscopically comprised a lobular epithelioid vascular proliferation with neutrophils and aggregates of bacterial organisms ( Table 17.2 ). The responsible gram-negative bacterium, Bartonella henselae , can be demonstrated by blood cultures and histologically with Warthin-Starry stain. The differential diagnosis in an immunocompromised patient is Kaposi sarcoma secondary to human herpesvirus 8 (HHV-8) infection.
| Organism (Disease) | Characteristics |
|---|---|
| HPV | Smooth to rough, flesh-colored papules |
| Molluscum contagiosum | Nodule with central crater |
| Cryptococcus neoformans | Nodules can evolve to painless ulcer |
| Histoplasma capsulatum | Nodules can evolve to ulcers |
| Treponema pallidum (condyloma lata of secondary syphilis) | — |
| Bartonella henselae (bacillary angiomatosis) | Red-purple papules |
| HHV-8 (Kaposi sarcoma) | Red-purple papules |
| Mycobacterium tuberculosis | Dermal or lamina propria nodules |
| Trichophyton spp. (Majocchi granuloma) | Erythematous raised plaque with prominent border |
| Myiasis | Dome-shaped lesion with central pore |
Acute inflammation or folliculitis of the hair follicles of the vulva produces an erythematous papule that evolves into a pustule, then a carbuncle through coalescence of several pustules, and even a furuncle or boil if the abscess extends into the subcutaneous tissue. The most common causative species is Staphylococcus aureus , increasingly, methicillin-resistant S. aureus (MRSA) but other bacteria may be the cause, such as Pseudomonas aeruginosa secondary to hot tub or swimming pool contamination. Less common causative agents include nontuberculous mycobacteria, Klebsiella oxytoca , and Acinetobacter baumannii .
Recurrent abscess formation leading to scarring and sinus formation is termed hidradenitis suppurativa; it is a relatively common condition, occurring in 1% to 4% of the population. It is probably caused by obstruction of a hair follicle followed by secondary bacterial infection. The condition affects more women than men and primarily the intertriginous skin, including the perianal and perineal regions. Clinically, hidradenitis suppurativa is characterized by recurrent deep, painful nodules that ultimately drain malodorous, purulent material and that often lead to scarring and sinus formation or even fistulas to contiguous organs, such as the bladder. Central necrosis, characteristic of a furuncle, is not a feature of hidradenitis suppurativa. The lesion is excised if malignancy is suspected or to remove scarred tissue harboring sinuses. The differential diagnosis includes Crohn disease of the vulva and chronic granuloma inguinale, which is distinguished by involvement of the vagina and cervix, as well as the external genitalia.
Chancroid, although rarely encountered in the United States, had been a common cause of genital ulcers in sub-Saharan Africa, Latin America, and Asia but has been reported to be in steep decline due to increased use of condoms and effective therapy. The cause is Haemophilus ducreyi , a small, gram-negative rod that requires technically challenging culture conditions to grow. Gram staining of the culture shows a parallel array of bacteria resembling a school of fish. This sexually transmitted infection produces a papule that progresses to a pustule and finally to a painful ulcer with a gray, purulent base, usually associated with painful inguinal lymph node swelling. Biopsy of the lesion shows acute inflammation in the epidermis and a perivascular mononuclear infiltrate or granulomatous response within the dermis; gram-negative bacteria can sometimes be found in the tissue with special staining techniques. Clinical diagnosis and Gram staining of a sample of the exudate from the bed of the ulcer have low sensitivity; culture is more sensitive, but the culture medium is not widely available. The most sensitive method of diagnosis is multiplex PCR. Of note, although chancroid is on the decline, leg ulcers due to H. ducreyi in children and adults in Pacific and African countries are on the rise.
Lymphogranuloma venereum (LVG), until recently confined mainly to the tropics, is increasingly identified in temperate climates. It is caused by the L serovars of Chlamydia trachomatis , which cause a painless, shallow, solitary ulcer on the vulva, vagina, or perianal region. The ulcer, which spontaneously heals in 3 to 12 days, is often missed. If it is left untreated, painful regional lymphadenopathy of the secondary, or inguinal, stage ensues. If the disease continues to progress, fistulas, labial scarring, and even labial elephantiasis can develop. Diagnosis is primarily serologic, but nucleic acid detection techniques are more sensitive and specific. Biopsy findings show nonspecific, chronic inflammation or granulomatous inflammation with necrosis; Brown-Hopp and Warthin-Starry stains highlight the short rods within vacuoles of macrophages.
Granuloma inguinale or donovanosis, rarely encountered in temperate climates except in travelers, is common in the tropical regions of Pacific countries, Asia, and South America. Granuloma inguinale, or donovanosis, is caused by the gram-negative rod, Klebsiella granulomatis (formerly called Donovania granulomatis or Calymmatobacterium granulomatis but reclassified by means of 16S ribosomal RNA analysis). Initially a papule appears on the vulva, vagina, or cervix. The papule progresses to a red ulcer that bleeds easily; if left untreated, granuloma inguinale can lead to scarring similar to hidradenitis suppurativa. Rarely the upper genital tract may be involved. K. granulomatis can be cultured only with difficulty, so diagnosis relies on demonstration of the bacteria on cytology or histology by special stain. Romanowsky-stained air-dried smears from the ulcer bed demonstrate numerous intracytoplasmic, short bacilli within macrophages. Rarely the organisms are identified on Pap smears. Biopsy of the ulcer base, performed for smear-negative cases, should be stained with Giemsa or Warthin-Starry stain. A PCR test has been developed for granuloma inguinale. Ulcers caused by multiple infectious agents have been reported.
Tuberculosis of the vulva is the rarest form of genital tuberculosis and is usually associated with foci of infection elsewhere in the genital tract. Hypertrophic lesions with sinus formation may develop. Surgery may be necessary in conjunction with antituberculosis drugs. Necrotizing granulomas are found on biopsy. Nucleic acid techniques including real-time PCR are used to diagnose Mycobacterium tuberculosis in clinical samples.
Malakoplakia, a chronic histiocytic inflammatory response characterized by calcified Michaelis-Gutmann bodies, usually occurs in the urinary tract, more often in women than in men. Malakoplakia has been reported on rare occasions in the female genital tract, including the vulva and, more often, the vagina. Diagnosis of vaginal malakoplakia has been made by cytology, including fine-needle aspiration cytology.
Candida is found in the vaginal tract of up to 50% of asymptomatic women; before menopause, the majority of women will have at least one episode of Candida vulvovaginitis, which is not considered to be an STD. Candida albicans accounts for up to 90% of infections; Candida glabrata and Candida parapsilosis are less commonly the cause. Candida vulvovaginitis manifests as a pruritic, erythematous swelling of the vulva with a thick, white vaginal exudate. Diagnosis is usually achieved in the clinic by means of microscopic demonstration of yeast forms, with pseudohyphae in the case of C. albicans , after application of 10% potassium hydroxide to vaginal secretions (to disaggregate squamous cells) with a normal vaginal pH of less than 4.5. Biopsy occasionally is performed on long-standing, chronically pruritic, hyperkeratotic lesions; often, only a few organisms are found in the stratum corneum. Periodic acid–Schiff (PAS) with diastase or Grocott methenamine silver (GMS) stain can be used to highlight the yeast and pseudohyphae. Candida spp. can cause pustular lesions in immunocompromised patients ( Table 17.3 ).
| Organism (Disease) | Characteristics |
|---|---|
| Candida albicans | Erythematous swelling |
| Schistosoma haematobium | “Sandy patches” appear as tiny rice grains beneath skin; also ulcers and polyps |
| Sarcoptes scabiei | Pruritic, erythematous, scaly tracts; also blisters and pustules |
| Corynebacterium minutissimum (erythrasma) | Erythematous lesion similar to candidiasis |
| Malassezia furfur (pityriasis or tinea versicolor) | Oval or irregular, flat, hypopigmented or hyperpigmented, 0.5- to 2.5-cm lesions |
With the advent of the 21st century a nationwide increase in incidence of coccidioidomycosis has been reported in the United States, most particularly in endemic regions, including Arizona and California. Infection, which is four times more prevalent in men than in women, occurs after inhalation of spores. Most infections (60%) are asymptomatic, but flulike symptoms can occur; less than 1% of patients develop disseminated disease. Although vulvar lesions have not been reported, scrotal lesions characterized by necrotizing granulomatous inflammation containing Coccidioides immitis spherules have been described.
Histoplasma capsulatum is a dimorphic fungus that is found worldwide but is most common in North and Central America. In the United States the fungus is endemic to the Mississippi and Ohio River valleys. Soil and caves contaminated by bird or bat guano are especially rich in this fungus. Infection, usually asymptomatic, occurs by the respiratory route. Less than 1% of infected patients develop clinically recognized symptoms, mainly pulmonary. Disseminated histoplasmosis usually occurs in immunocompromised patients, such as patients with AIDS or transplant recipients. The liver, spleen, bone marrow, gastrointestinal tract, skin, and mucous membranes may be involved. Ulcers of the female genital tract due to H. capsulatum have only rarely been described; the organisms were recognized as small intracellular budding yeast on routinely stained smears and biopsy samples. Disseminated histoplasmosis can also occur in nonimmunocompromised older patients, in whom the slow disease progression is usually fatal. Disease in older adults is not always accompanied by constitutional symptoms. A case of venereal spread provoking an isolated vulvar ulcer has been described.
Cryptococcus neoformans , an encapsulated yeast, is found worldwide, especially in soil harboring pigeon guano, but infection usually causes symptoms only in immunocompromised patients. With symptomatic infection, a granulomatous response develops. The fungus occurs as a yeast form in tissue and can be recognized on routine staining by its more variable size compared with other fungi (2 to 20 µm in diameter). Special stains used to highlight Cryptococcus include PAS and methenamine silver. The capsule can be seen in negative relief on smears with India ink and is demonstrated in tissue sections with mucicarmine stain. Cryptococcus contains phenol oxidase, which is involved in the metabolism of phenolic compounds to melanin. Phenol oxidase is a virulence factor, and the melanin that is produced can be detected by the Fontana-Masson stain on histologic sections. Isolated lesions of the female genital tract have rarely been reported. A vaginal nodular lesion was found in a 20-year-old, nonimmunocompromised woman, and a painless ulcer of the labia major occurred in a 60-year-old renal transplant recipient. In both instances, biopsy was performed because the lesions were clinically suspected to be carcinoma, and in both cases, granulomatous inflammation with mucin-positive fungi was found.
Trichophyton mentagrophytes has been reported to cause a case of Majocchi granuloma of the vulva in a young woman. Majocchi granuloma, or nodular granulomatous perifolliculitis, is an uncommon intradermal infection by dermatophytes that usually cause follicular lesions.
Genital schistosomiasis, usually caused by the bladder trematode, Schistosoma haematobium , which is endemic to Africa, has been reported to involve the vulva of women living in endemic areas, such as Malawi, and in travelers. The eggs gain access to the stromal tissue of the vulva via the venous drainage from the bladder and engender granulomatous inflammation. A variety of lesions are associated with genital schistosomiasis, including polyps and ulcers, but the most common lesion is the “sandy patch,” which appears as an aggregate of tiny rice grains beneath smooth mucosa. Regional lymphadenopathy often accompanies the genital lesions. The diagnosis relies on demonstration of the characteristic egg with a terminal spine in tissue biopsy. It can be seen with routine hematoxylin and eosin (H&E) stain, but silver stains highlight the egg. Schistosoma mansoni , the cause of intestinal schistosomiasis, is found in many of the same regions of Africa as S. haematobium , as well as in South America and the Caribbean, and has also been associated with genital disease, although the incidence is less certain. The ova of S. mansoni are differentiated from those of S. haematobium by the presence of a lateral spine; both are approximately 60 µm wide by 160 µm long ( Fig. 17.6 ).
![Figure 17.6, Vulvar biopsy of 7-year-old with swelling of the vulva. No purulent exudate was obtained on incision and drainage, so a biopsy was performed. It showed an intact, albeit acanthotic squamous epidermis (hematoxylin and eosin [H&E] stain) (A) . Within the dermis were numerous, mostly embryonated, schistosome eggs, a few of which showed the terminal spine of Schistosoma haematobium (H&E stain) (B) . Within the subcutis, a distended vein contained a pair of Schistosoma worms (male and female) (H&E stain) (C) . Figure 17.6, Vulvar biopsy of 7-year-old with swelling of the vulva. No purulent exudate was obtained on incision and drainage, so a biopsy was performed. It showed an intact, albeit acanthotic squamous epidermis (hematoxylin and eosin [H&E] stain) (A) . Within the dermis were numerous, mostly embryonated, schistosome eggs, a few of which showed the terminal spine of Schistosoma haematobium (H&E stain) (B) . Within the subcutis, a distended vein contained a pair of Schistosoma worms (male and female) (H&E stain) (C) .](https://storage.googleapis.com/dl.dentistrykey.com/clinical/GynecologicInfections/5_3s20B9780323445856000175.jpg)
Vulvovaginitis secondary to Enterobiasis vermicularis has been reported in prepubertal girls and rarely in older women.
Scabies is caused by the mite Sarcoptes scabiei , which is spread by person-to-person contact and rarely by fomites. The mites burrow into the epidermis to lay eggs. Clinically the burrow is linear and intensely pruritic. On biopsy, diagnostic fragments of mites and eggs are found in the epidermis, and a polymorphous inflammatory infiltrate including eosinophils is present in the epidermis and dermis. Additional histologic findings include spongiosis and superficial fibrin thrombi.
Myiasis is the cutaneous infestation by larvae of several fly species. The larvae can infest devitalized tissue of open wounds or burrow into healthy skin of the vulva, resulting in furuncular myiasis. The lesion is dome-shaped, with a central pore from which a variable number of larvae exit. The larvae can be plucked from the lesion for identification by a parasitologist. The infestation occurs mainly in tropical climates and is associated with poor hygiene, diabetes, and travel.
Fixed drug eruptions are circumscribed erythematous plaques that are usually located on the hands, feet, and external genitalia; the lesions recur in the same site with repeated exposure to the drug. A central vesicle may develop. Biopsy of a vulvar lesion may be necessary, if the association with the offending drug is not recognized, to exclude similar-appearing infectious lesions such as HSV or secondary syphilis. The histologic appearance of a fixed drug eruption consists of basal cell hydropic change, interface lymphoid infiltrate occasionally with eosinophils, and individual keratinocyte necrosis.
Behçet disease, a rare cause of vulvar ulcers, occurs in Japan and Eastern Mediterranean countries. The diagnosis is made in the presence of recurrent oral ulcers and at least one of the following: genital ulcers, skin pustules or nodules, and uveitis/retinitis. Herpetiform or aphthous vulvar ulcers show necrotizing vasculitis of all calibers of vessels, with acute or chronic inflammation. The diagnosis is one of exclusion; ulcerative lesions of the vulva may be the presenting symptom.
The differential diagnosis of vulvar and perianal abscesses, sinus tracts, and fistula formation includes Crohn disease. Crohn disease can be particularly challenging to diagnose if the genital manifestations precede the gastrointestinal lesions, in which case a diagnosis of granulomatous vulvitis may be made. Sarcoidosis of the vulva has been reported.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here