Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
There has been an increase in the incidence of oesophageal and oesophagogastric junctional (OGJ) cancers in many Western countries, especially amongst White males. In contrast, stomach cancer incidence has declined except for proximal cancers in the gastric cardia. Both oesophageal and gastric cancers (GCs) are associated with < 20% survival at 5 years, mainly as a result of late diagnosis at advanced non-curable stages of disease. , Consequently, there is now a focus on early detection and secondary prevention, with the aim to move towards more personalised screening and surveillance strategies. These efforts have been supported by an increased understanding of the natural history of these cancers including risk factors, cellular and genetic pathology, and the identification of premalignant lesions and cancer precursors. In this chapter, we discuss the early detection of oesophago-GCs and provide evidence-based, practical suggestions for screening, surveillance, diagnosis, and management. Finally, we discuss genetic aberrations and their applications in biomarker discovery.
Oesophageal cancer continues to be classified on the basis of anatomical location and histopathology, although molecular features are starting to inform clinical management. It comprises of two distinct types—oesophageal adenocarcinoma (OAC), including OGJ adenocarcinomas, and oesophageal squamous cell carcinoma (OSCC).
![]() The risk of OAC associated with gastro-oesophageal reflux disease (GORD) is related to chronic injury of the distal oesophagus and the development of Barrett’s oesophagus (BO), now widely accepted as a key precursor to oesophageal and OGJ adenocarcinoma, with BO patients having a relative risk of around 11 compared to the general population. ,
The risk of OAC associated with gastro-oesophageal reflux disease (GORD) is related to chronic injury of the distal oesophagus and the development of Barrett’s oesophagus (BO), now widely accepted as a key precursor to oesophageal and OGJ adenocarcinoma, with BO patients having a relative risk of around 11 compared to the general population. ,
Most adenocarcinomas are diagnosed de novo and BO is not always evident histologically, leading to questions about whether all adenocarcinomas do in fact arise from Barrett’s metaplasia, or whether Barrett’s is a pre-requisite but in some cases the cancer overgrows the precursor. In a meta-analysis from 2018, only 11.8% of patients with OAC had a previous diagnosis of BO.
The latest British Society of Gastroenterology (BSG) guidelines define BO as an abnormal acquired state in which a segment of squamous epithelium in the distal oesophagus is transformed into columnar epithelium via a process known as metaplasia. While all pathological subtypes are included in the BSG definition, surveillance is limited to cases of intestinal metaplasia ([IM]; and longer segments where sampling bias may have missed focal IM). The American College of Gastroenterology (ACG) guidelines and the more recent position of the European Society of Gastrointestinal Endoscopy (ESGE) necessitate the histological presence of IM in order to make a diagnosis. ,
Assessing the prevalence of BO is challenging as it often asymptomatic. Therefore, rates are generally extrapolated from endoscopy performed for individuals with reflux symptoms. Overall, it appears that the population prevalence of BO, proved histologically, is around 1–2%. This rises to 5–15% for patients with GORD symptoms. According to a Dutch study of 500,000 medical records, the incidence of BO has been increasing, even when accounting for the number of upper GI endoscopies performed. This is likely to be related to the increase in GORD prevalence, a reduction in Helicobacter pylori and rise in obesity, and also increased recognition amongst endoscopists.
According to the Barrett’s Esophagus Study, a US-based multicentre consortium of 3643 patients with BO, at index endoscopy 70.1% of the cohort had non-dysplastic BO (ND-BO), 11.5% had low-grade dysplasia (LGD), 5.4% had high-grade dysplasia (HGD), and 5.1% had OAC. The study showed that there has been a significant increase in the prevalence of HGD and OAC, respectively, at index endoscopy over the last 25 years, whereas LGD levels remained stable.
A recent meta-analysis evaluating 102 studies and a total of 460 984 adults reported a pooled prevalence of 14.8% for GORD in the general population. Although the prevalence of BO is higher, ranging between 5% and 15%, in those that have a background of reflux symptoms compared to the general population, not all patients with GORD harbour BO or even oesophagitis. Similarly, around half of patients with BO do not report heartburn symptoms.
Other established risk factors include increasing age, male sex, White race, smoking, and obesity, but more specifically central adiposity. , A UK-based study looking at 21 899 index endoscopies reported that each additional year from age 20 to 59 led to a 7.4% increase in BO prevalence for male subjects, with similar findings but at 20 years later in life for females. Males between 60 and 70 years of age have a twofold increased likelihood of having BO compared to age-adjusted females. The incidence of BO was approximately five times higher in White compared to Black patients. A meta-analysis of five case-control studies showed that BO cases were significantly more likely to have been in smokers compared to controls, with additional evidence showing 1) a positive correlation between smoking duration and; 2) a synergistic effect between smoking and GORD. A recent pooled analysis from eight case-control studies revealed that waist circumference was associated with risk for BO risk. The latest meta-analysis, studying the impact of obesity, metabolic syndrome, and insulin resistance, included 119 273 subjects from 46 studies and showed that all three were significantly associated with an increased risk for BO.
In addition to environmental factors, there is also a genetic component that influences the development of BO. Familial clustering has been reported in 6–7% of BO/OAC cases in US and Dutch studies, , a finding consolidated by case-control genome-wide association studies (GWASs). Previous GWASs analysing Western populations have identified multiple genetic variants associated with the development of BO and OAC. A meta-analysis comprising 6 167 BO and 4 112 OAC cases, compared to 17 159 controls, identified 16 independent risk loci for BO and/or OAC, including locations related to the CFTR , Foxf1 , Foxp1 , ALDH1A2 , CTRC1 , TBX5 , and GDF7 genes and the major histocompatibility complex (MHC) region. Studies have also shown that the prevalence of GORD, a known risk factor for BO/OAC, has been shown to be higher in the relatives of patients with BO. , Twin studies have reported an estimated GORD heritability of 30–40%. , Similar findings have been reported with a high prevalence of BO, ranging between 18% and 28% amongst relatives of patients with BO/OAC. ,
It remains to be determined whether the genetic risk loci can be used as part of a clinical algorithm to identify individuals at risk.
Systemic mass endoscopic screening on a population level is not supported by any guidelines, given that upper GI endoscopy does not fulfil the criteria for a good screening test. Enriching the population for those at highest risk is therefore a serious consideration, and GORD has been generally advocated as the most useful risk factor. The BSG endorses screening in patients with chronic GORD symptoms plus a strong family history of BO or OAC (i.e. first-degree relatives), or chronic GORD symptoms and three other risk factors including male sex, White race, age more than 50 years, and obesity. Similarly, the ACG proposes selective screening in patients who have had GORD for at least 5 years with symptoms at least weekly, plus two or more of the following risks factors: White race, age over 50 years, central obesity, smoking history, or a first-degree relative with BO/OAC. Given that up to 40% of patients with OAC are asymptomatic, even with these criteria a large proportion of patients that might harbour BO/OAC are not screened. , Furthermore, a recent prospective study of 1 241 patients suggested that available clinical factor-based risk assessment tools, such as HUNT, M-BERET, and Kunzmann, are in fact more accurate than frequency and duration of GORD symptoms in identifying patients at risk of neoplastic BO.
In order to improve precision screening, a three-step approach has been proposed. The first stage enriches at-risk patients within the general population using risk-prediction algorithms. This is followed by triaging using an affordable, minimally invasive test in a primary care setting to find high-risk patients, and finally endoscopy to confirm diagnosis. Various options are currently being investigated for the second step in this screening process.
Non-sedated transnasal endoscopy (TNE) has been shown to have improved patient acceptability and similar clinical effectiveness to traditional upper GI endoscopy in assessing the oesophagus for endoscopic BO and histological IM, respectively. , TNE has been evaluated successfully for use in travelling vans for community BO screening. The use of TNE is supported in recent BO screening guidelines.
With advances in imaging, oesophageal capsule endoscopy (OCE) is another less invasive, clinic-based alternative that uses a wireless, pill-sized swallowed camera to visualise the oesophagus. However, a meta-analysis from 2009 assessing the original capsule showed poor sensitivity and specificity for BO diagnosis. Newer versions are currently under investigation.
An alternative to endoscopic imaging is the use of non-endoscopic cell sampling devices combined with genetic or cytological biomarker evaluation. The Cytosponge is a mesh-containing capsule attached on a string that can be swallowed by patients in an office-based setting. The capsule dissolves in the stomach about 5–7 minutes post ingestion, which allows the compressed mesh to expand and, while withdrawn by pulling the string, collect up to 1 million cells from the entire length of the oesophagus. These cells are evaluated by immunocytology for Trefoil Factor-3 (TFF3), a protein marker of IM overexpressed in BO to determine whether the test is positive or negative. The BEST1 trial established that this approach is feasible in primary care, with high levels of patient acceptability and encouraging accuracy data. The BEST-2 study recruited 1 100 subjects (647 BO cases vs 463 controls with heartburn symptoms) and reported a sensitivity of 79.9% and a specificity of 92.4% in a per-protocol analysis. When patients with an inadequate sample were offered a repeat Cytosponge, and for cases with circumferential BO segments ≥ 3 cm, sensitivity increased to 89.7% and 87.2%, respectively.
![]() The BEST-3 trial was a randomised trial to determine whether the offer of Cytosponge to patients on medication for heartburn increased the detection of BO compared to usual care, meaning endoscopy referral if the family doctor felt it was indicated. This showed that an offer of Cytosponge-TFF3 led to a tenfold increase in the number of BO diagnoses.
The BEST-3 trial was a randomised trial to determine whether the offer of Cytosponge to patients on medication for heartburn increased the detection of BO compared to usual care, meaning endoscopy referral if the family doctor felt it was indicated. This showed that an offer of Cytosponge-TFF3 led to a tenfold increase in the number of BO diagnoses.
A cost-effectiveness analysis showed that initial Cytosponge-based screening of GORD patients followed by upper GI endoscopy for positive tests was 27–29% less costly compared to endoscopy screening alone. Overall, these data are encouraging and implementation research is underway in the UK (NIHR ID: ISRCTN91655550).
Other investigators have since developed balloon and sponge devices coupled with methylation panels. A multicentre US-based case-control study of 295 patients showed that the EsophaCap, a sponge-on-a-string device, had a sensitivity of 92% and a specificity of 94% for diagnosing BO and was preferred to upper GI endoscopy by 94% of subjects. The test used a total of five methylated DNA biomarkers. The five-biomarker panel did not distinguish between dysplastic and ND-BO, and similar to Cytosponge the test was susceptible to BO length with some short length segments being missed. It is worth noting that, in contrast to the Cytosponge study, this was not an intention-to-treat analysis. A large case-control study using EsophaCap is currently underway (Clinical Trials ID: NCT04214119). EsoCheck is a balloon device deployed via a slim catheter that can be inflated and deflated such that you sample 5–6 cm above the oesophago-gastric junction (OGJ). The cells are analysed using a DNA methylation biomarker panel comprising of VIM and CCNA1 that showed a sensitivity of 90.3% and a specificity of 91.7% for detecting BO in a small pilot trial, despite poor DNA yields. Again, this was not an intention-to-treat analysis. Following device improvements, the use of EsoCheck in combination with the aforementioned two-marker panel for diagnosing BO in an at-risk screening cohort is being evaluated in a 1000-participant US-based study (Clinical Trials ID NCT04293458).
Finally, promising and even less invasive options currently under investigation for use in BO screening include liquid biopsy, looking for cancer-related biomarkers in the blood – particularly microRNAs , or circulating tumour DNA – as well as breath-sampling devices measuring exhaled volatile organic compounds. ,
Endoscopy is currently the gold-standard diagnostic test for BO.
![]() All international societal guidelines diagnostic criteria require that the BO columnar epithelium segment is visible endoscopically, at least 1 cm above the OGJ, and for any columnar metaplasia (BSG) and in particular IM (BSG, American Gastroenterological Association [AGA], ESGE) to be confirmed histologically on oesophageal biopsies.
All international societal guidelines diagnostic criteria require that the BO columnar epithelium segment is visible endoscopically, at least 1 cm above the OGJ, and for any columnar metaplasia (BSG) and in particular IM (BSG, American Gastroenterological Association [AGA], ESGE) to be confirmed histologically on oesophageal biopsies.
In the context of a healthy oesophagus, the top of the gastric fold coincides with the squamo-columnar junction (SCJ), but in the presence of BO the SCJ shifts at least 1 cm proximally to the OGJ, generating a characteristic salmon pink colour and velvety texture easily distinguishable from the pale, glossy appearance of squamous mucosa ( Fig. 2.1 ). An irregular Z-line describes the situation where the SCJ might lie above the OGJ but without any confluence, forming tongues shorter than 1 cm, thus not fulfilling the diagnostic criteria for BO ( Fig. 2.2 ). An irregular Z-line is found in 10–15% of patients undergoing upper GI endoscopy, and despite the fact that it can harbour histological IM in 39–44% of cases, , its neoplastic potential is low, with no patients progressing beyond ND-BO in one study and only 4% of patients progressing to BO with LGD and none progressing to HGD/OAC in another study. Therefore, current societal guidelines do not recommend sampling or further evaluation of irregular Z-lines.
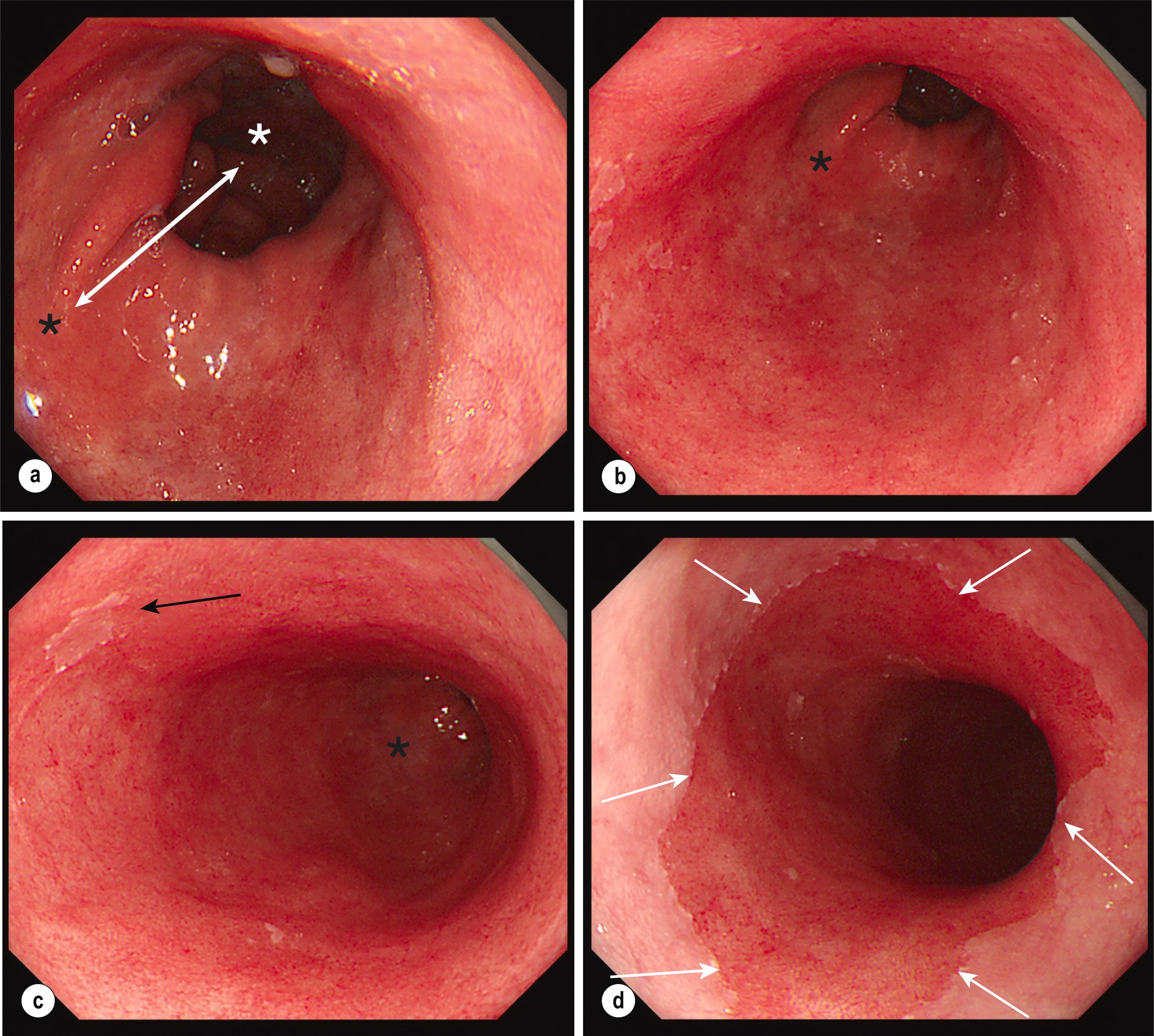
BO can be crudely categorised into short- (< 3 cm) and long-segment (≥ 3 cm) disease, with the latter being 3.5-fold more prevalent than the former (6.0% vs 1.6%, respectively). This distinction is relevant, as long-segment BO has a higher probability of harbouring dysplasia (prevalence 31% vs 10%) and a higher risk of neoplastic progression.
Equally important as making an accurate endoscopic diagnosis is the report of endoscopic findings to ensure precise clinical communication and tailored follow-up according to the risk of cancer progression.
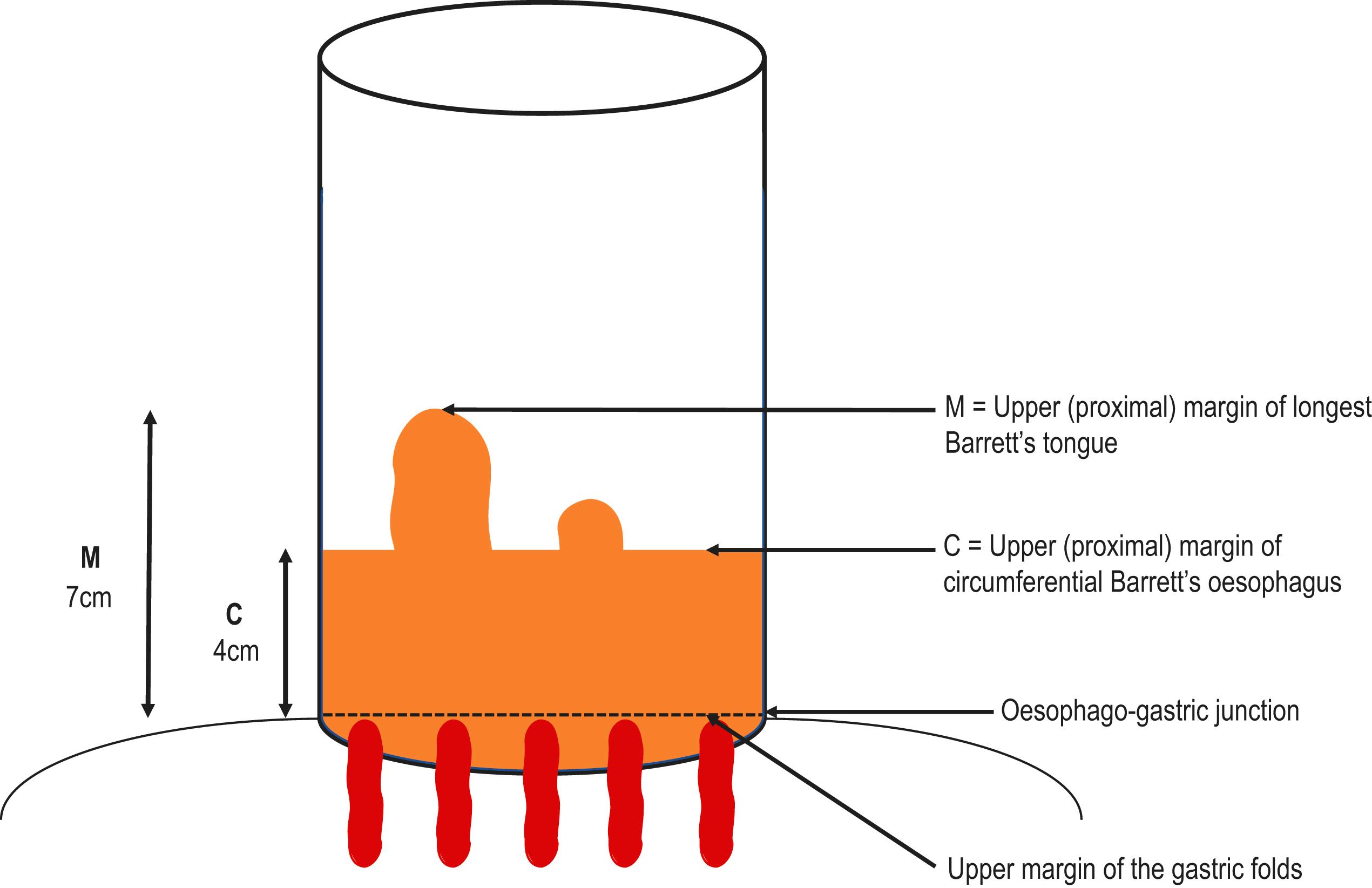
![]() The Prague C&M endoscopic classification scores the BO extent in terms of its circumference (C) and maximal tongue length (M), both measured in cm from the OGJ ( Fig. 2.3 ). Other islands of columnar-type mucosa above the main Barrett’s segment should be reported separately. The Prague classification has good inter-user reliability for BO segments larger than or equal to 1 cm.
The Prague C&M endoscopic classification scores the BO extent in terms of its circumference (C) and maximal tongue length (M), both measured in cm from the OGJ ( Fig. 2.3 ). Other islands of columnar-type mucosa above the main Barrett’s segment should be reported separately. The Prague classification has good inter-user reliability for BO segments larger than or equal to 1 cm.
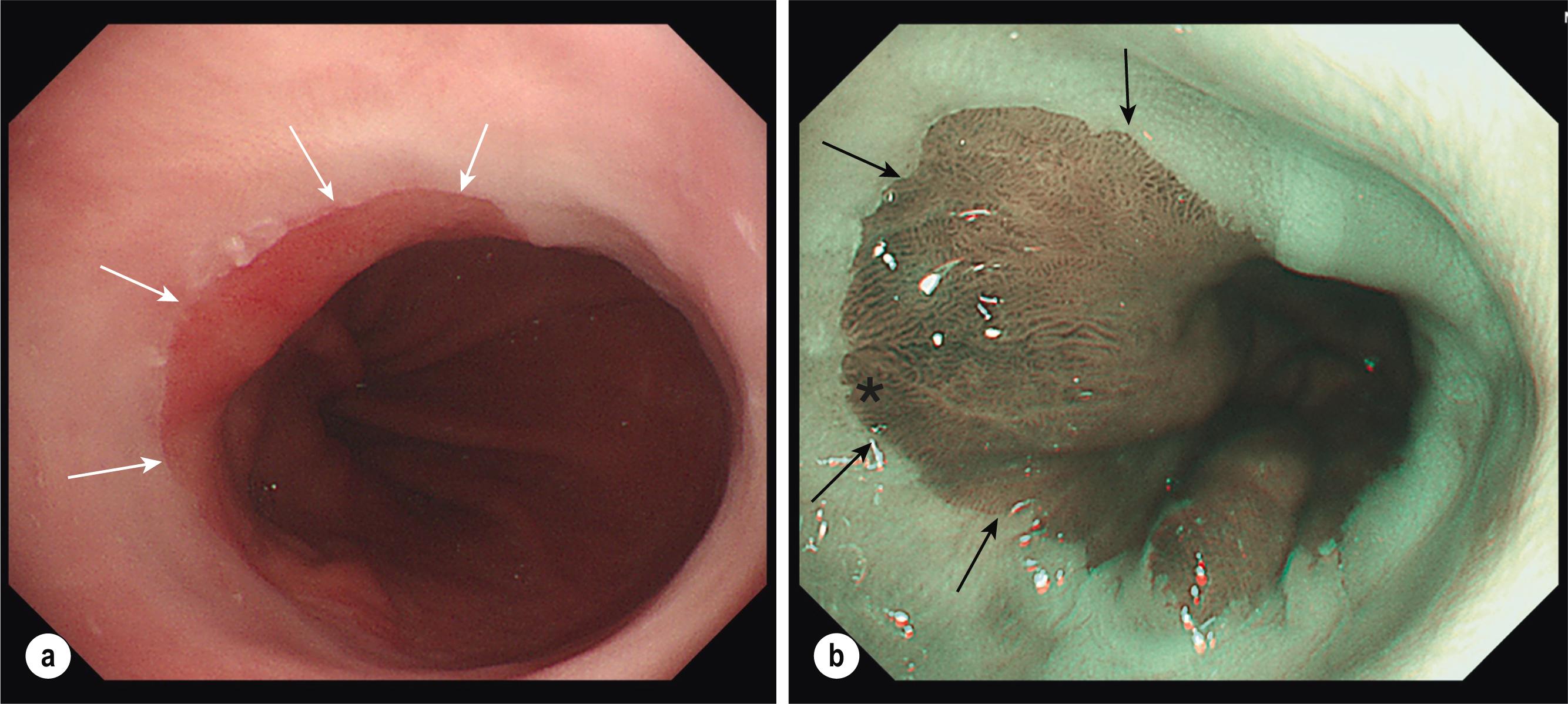
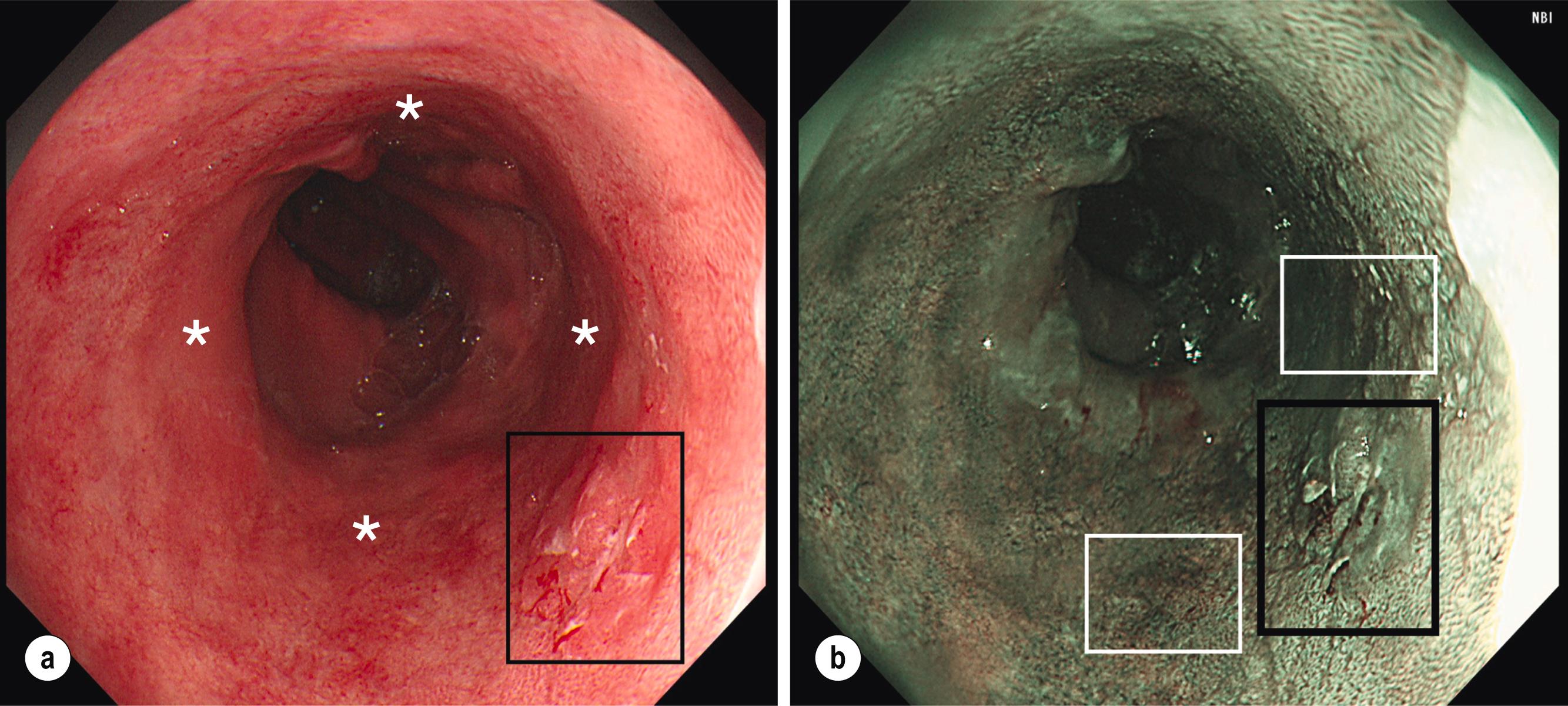
Any other visible lesions associated with the BO segment should be described independently according to the Paris classification and have their position mapped based on clock-face circumferential markings and distance from the incisors ( Figs. 2.4 and 2.5 ). Such lesions are often subtle, include ulcers and nodules, and are frequently enriched with dysplastic changes, , with sessile/depressed lesions more likely to already contain cancer. Additionally, in various studies the presence of endoscopically visible nodules was associated with a higher risk, up to fourfold, of neoplastic progression to HGD/OAC compared to no nodular appearances, , whereas HGD in the context of an ulcer conferred an enhanced risk of progressing to cancer compared to non-ulcer HGD. With improved magnification and narrow-band imaging (NBI), high-resolution images enable evaluation of the mucosal pit pattern and vessels within the Barrett’s segment, and any associated lesions (see Figs. 2.4 and 2.5 ). Compared with non-metaplastic columnar mucosa, which displays round pits, in BO with IM pits acquire a villous shape, whereas in the presence of dysplasia they have a totally irregular pattern. The microvasculature remains well structured in IM but tends to become disorganised with dilated, irregular, and diffusely distributed vessels not following the normal architecture of the mucosa (i.e. not along or between mucosal ridges) in areas of dysplasia. Complex NBI classification systems, such as Amsterdam and Kansas, , exist to predict the histologic presence and grade of dysplasia based on vascular and mucosal patterns but are mainly used in specialised, tertiary centres. The steps in evaluating BO endoscopically are summarised in Box 2.1 .
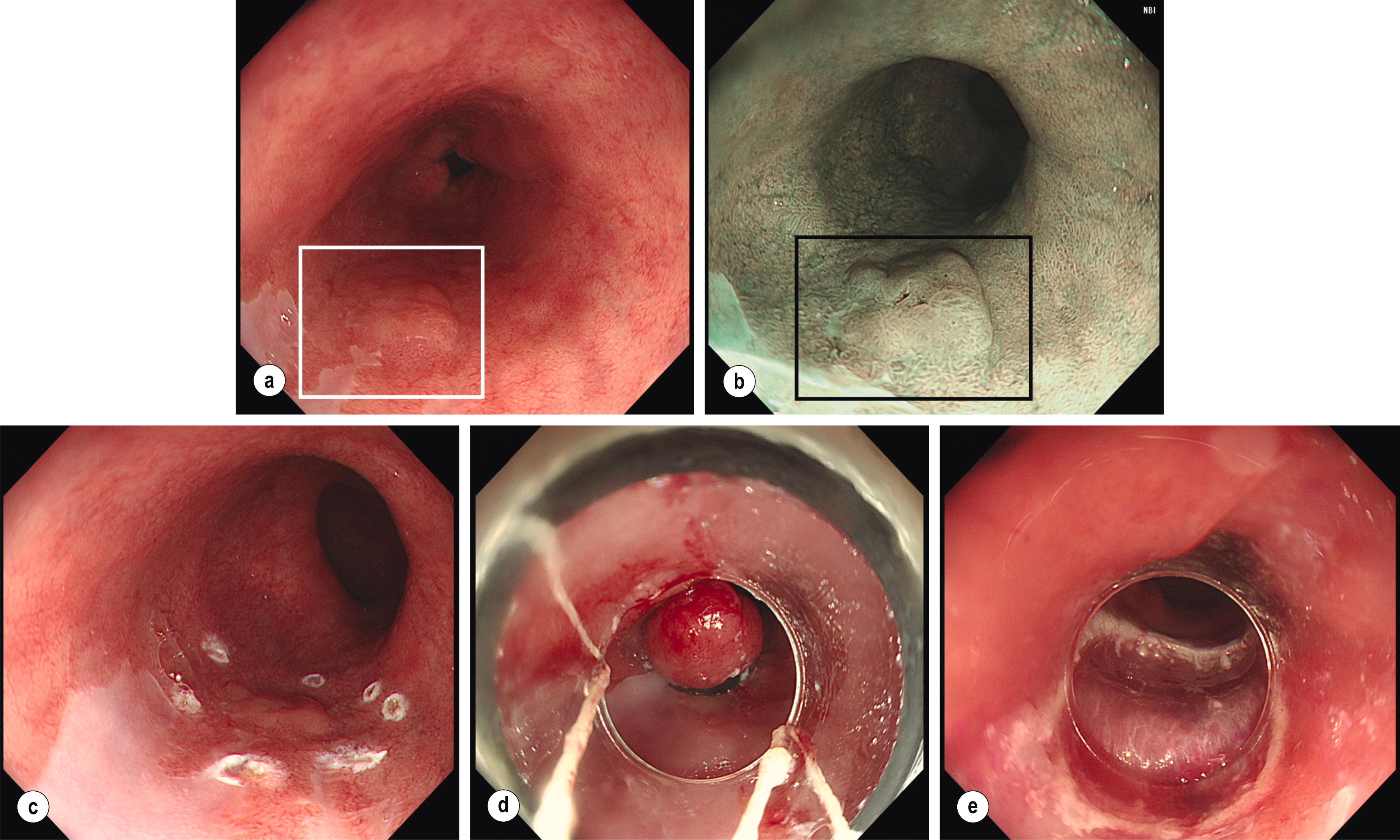
Identify key landmarks – oesophagogastric junction, squamous-columnar junction, and diaphragmatic pinch
Evaluate Barrett’s segment with white light endoscopy and report according to the Prague C+M classification
Look carefully for any lesions and characterise them according to the Paris classification
Evaluate Barrett’s segment with narrow-band imaging, looking at pit (round vs villous), mucosal (regular vs irregular), and microvasculature (regular vs irregular) patterns
Acquire random quadrantic biopsies every 2 cm and targeted biopsies of any lesions or suspected areas
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here