Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The incidence of intracranial aneurysms is variable throughout the world and is approximately 6% in the international population, with higher rates in Asian/Finnish populations and those with a high-risk profile. In patients without any risk factors, the incidence is approximately 2%. Once the aneurysm has ruptured, one-third will die, whereas 50% of survivors will lead independent lives.
The majority of intracranial aneurysms arise at branching points of large arteries. Hemodynamic stress likely contributes to both the initial development and subsequent growth. Aneurysm formation can have many other associated conditions, including autosomal dominant polycystic kidney disease, Ehlers-Danlos syndrome, Loeys-Dietz syndrome, Marfan syndrome, tuberous sclerosis, fibromuscular dysplasia, and other genetic and structural predispositions.
Approximately 25% to 50% of all patients have warning symptoms that herald the onset of a major subarachnoid hemorrhage (SAH). The patient's description of the headache is very important, as most involve a “thunderclap” onset or the “worst headache of my life.” Computed tomography (CT) scans detect SAH with high sensitivity. If the scan is negative but suspicion remains high, a lumbar puncture may be performed. Although CT angiography is good for imaging aneurysms greater than 2 mm, cerebral angiography remains the gold standard for intracranial vascular imaging.
The risk of rebleeding after the SAH is greatest on the first day (4.1%). By day 14, the cumulative rebleed incidence is 19%. Once an aneurysm is secure, the most significant cause of morbidity and death is cerebral vasospasm. The onset of cerebral vasospasm begins on day 3 and peaks between 7 and 14 days after the SAH. Seventy percent of patients develop radiographic signs of vasospasm, and approximately 30% develop clinical vasospasm requiring adjunctive therapy.
The treatment goal of intracranial aneurysms is to exclude them from the parent circulation. In ruptured aneurysms, this must be done early and safely so that maximum treatment of cerebral vasospasm can be administered if necessary. Symptomatic vasospasm can be treated with triple H therapy (involving hypertension, hypervolemia, and hemodilution) and endovascular means, namely intraarterial injection of vasodilators or angioplasty.
To achieve the goal of multidisciplinary cerebrovascular care, aneurysm patients are ideally treated at centers of excellence that employ expertise in all areas of neurovascular care, including endovascular, microvascular, and neurocritical care, as well as neuroanesthesia.
Intracranial aneurysms (IAs) are vascular lesions, and this poses a challenge for decision making and management. Morphologically and pathologically, they encompass a variety of entities including saccular, fusiform, and dissecting aneurysms with a wide variety of etiologic origins, including hemodynamic, traumatic, and infectious. Although intervention in most cases of ruptured intracranial aneurysms is indicated, controversy exists regarding the management of unruptured intracranial aneurysms (UIAs). Namely, the question for every UIA is whether the benefit of aneurysm occlusion via clipping or coiling outweighs the risk of a possible subarachnoid hemorrhage. Our understanding of the pathobiology and pathophysiology of this entity has significantly improved, leading to improvements in epidemiology, biology, genetics, therapeutic intervention, and clinical outcomes. However, our understanding of the natural history of this disease remains limited, rendering decision making regarding intervention complex. Moreover, as technology advances and new techniques become available for aneurysm obliteration, further questions arise regarding the optimal treatment choice. This chapter provides an overview of intracranial aneurysms, clinical presentations, current therapeutic options, and technical aspects of surgical treatment. Although intracranial aneurysms encompass a diverse disease entity, this chapter focuses on the most common type—that is, saccular aneurysms.
Aneurysm prevalence is difficult to assess, due to clustering in various high-risk groups and the presence of asymptomatic lesions. Unruptured aneurysms are common and are diagnosed in 2% to 3% of the population. They have been identified in as many as 1% to 9% of general population autopsies. However, extreme variability has been observed among different autopsy studies and reviews. Ethnicity-dependent clustering of aneurysms is observed, with a higher prevalence in the Asian and Finnish populations (4%–9%), and as a result, a 6% prevalence has been reported in the international population. This disease process affects females three times more frequently than males.
Although the most prevalent risk factors are modifiable, the strongest risk factors are thought to occur through genetic and familial predisposition. In fact, genetic predisposition through various systemic and syndromic conditions is thought to represent approximately 10% to 12% of all aneurysms. Aneurysms occur sporadically, but they may also occur as part as of familial syndromes such as adult polycystic kidney disease (ADPKD), Marfan syndrome, Ehlers-Danlos (EDS) type IV, Loeys-Dietz syndrome, fibromuscular dysplasia (FMD), Moyamoya, and sickle cell disease. Patients with one affected family member have a 4% risk, whereas patients with two affected family members have an 8% to 10% risk of harboring an aneurysm. A large meta-analysis of 19 studies identified 19 single nucleotide polymorphisms (SNPs) associated with sporadic UIAs, with strongest associations including the chromosome 9 CDKN2B antisense inhibitor gene, chromosome 8 near the SOX17 transcription regulator gene, and chromosome 4 near the EDNRA gene. Ruigrok and colleagues reviewed the genetics of intracranial aneurysms and reported that probable loci included chromosomes 5q and 17cen, from which the strongest associations code for versican, an extracellular matrix protein, and TNFRSF13B, a transmembrane activator and a calcium modulator ligand interactor, respectively. Other suggestive loci are 1p34.3–p36.13, 7q11, 19q13.3, and Xp22.
Smoking and hypertension are modifiable risk factors for the development of aneurysms due to an induction of vascular wall changes and a decrease in the size of the tunica media. Of note, female smokers are at risk of both aneurysm growth and de novo aneurysm formation. However, the prevalence of lesions is still thought to be approximately 2% in those without any known risk factors.
The incidence of aneurysmal subarachnoid hemorrhage varies from 6 to 26 per 100,000 per year. Women outnumber men by 1.6 to 1, and the peak age is 40 to 60 years. Modifiable risk factors for aneurysm rupture include alcohol use, smoking, and hypertension. Untreated ruptured aneurysms carry a mortality of 20% to 30% in the first 2 weeks, with a risk of rebleeding of 20% at 2 weeks, 33% at 1 month, and 50% at 6 months, and then 3% per year thereafter. Additionally, up to 60% of survivors remain functionally dependent.
Intracranial arteries are unique to the systemic vascular system in that they lack the external elastic lamina present in extracranial vessels. The pathogenesis of intracranial aneurysms has been thought to be due to a number of congenital factors, such as vascular wall defects (ie, tunica media and elastic lamina), hemodynamic load on tortuous segments and bends, and discontinuity of the tunica media at origins of small vessels from larger parent vessels. Moreover, systemic factors are thought to contribute to aneurysm pathogenesis, such as degenerative changes, hypertension, atherosclerosis, connective tissue disease, and hemodynamic stress.
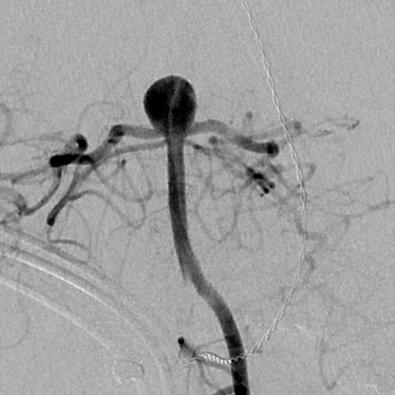
The majority of lesions are located at branching points along the proximal arterial tree, suggesting that hemodynamic factors play a significant role in aneurysm formation ( Fig. 16.1 ). In addition, formation of de novo aneurysms has been shown to occur at communicating arteries, and fatal rupture of aneurysms has occurred after recruitment of collateral blood flow through communicating arteries after internal carotid artery (ICA) occlusion. Furthermore, the incidence of aneurysms associated with high-flow states (eg, arteriovenous malformations) is higher than in the general population, and some of these lesions spontaneously regress after treatment of the vascular malformation. Finally, recurrent aneurysms can develop after incomplete treatment, whether by endovascular or microsurgical means.
Other factors related to aneurysm formation are trauma, infection, and tumor emboli. Traumatic aneurysms are more often fusiform and located near the skull base. Infectious aneurysms have a multitude of etiologic factors but most commonly occur secondary to bacterial endocarditis. Finally, aneurysms can result from tumor seeding of the arterial wall through hematogenous spread and are most often related to cardiac myxomas.
Hemodynamics has been shown to play an important role in aneurysm rupture. Using computational fluid dynamics, it has been demonstrated that high wall shear stress is responsible for growth and rupture in high-flow aneurysms, whereas the predominant factors causing rupture in low-flow aneurysms are high intraaneurysmal pressure and flow stasis. Indeed, ruptured aneurysms have unstable flow patterns and a higher average wall shear stress in the aneurysm sac than unruptured ones.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here