Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Connexins are the subunit proteins of gap junction (GJ) channels. Connexin43 (Cx43) is the most broadly expressed connexin in the tissues and organs of the human body and the main GJ protein of the heart. , Gap junctions provide a direct, gated pathway for cytoplasmic coupling between cells. This capacity is the basis of the long-standing consensus view on the main significance of connexins and GJs to the physiology of the heart. Namely, GJs enable direct electrotonic coupling between cardiomyocytes. GJ-mediated intercellular communication is thus widely held to be critical, if not obligate, in facilitating the propagation of action potential (AP) through cardiac tissues, starting within the sinoatrial (SA) nodal pacemaker and culminating in the synchronized activation of the ventricular chambers. The clinical importance of connexins to the electrical activation of myocardial tissues has been reinforced by a growing body of information correlating disruptions to the three-dimensional pattern of GJ coupling between myocytes with the generation of cardiac arrhythmias, including those of the pacemaking and conduction system and the working myocardial tissues of the atria and ventricle. ,
GJs consist of tightly packed aggregates of membrane channels, which, in the electron microscope, appear as areas of distinct and extended intercellular contact. The robust junctional ultrastructure of the GJ belies the fact that Cx43 is relatively short lived. The half-life of Cx43 has been estimated to be around 1.5 hours based on studies of cultured myocytes and isolated hearts. , This is more than 100 and 1000 times shorter, respectively, than the half-lives of actin and collagen. , To put the implications of the high rate of Cx43 turnover in more concrete terms, the total mass of Cx43 produced in a year by the ventricle of an adult human female can be estimated to be around 80 grams ( Table 8.1 ), which would be just shy of one-third of the weight of her heart. The cumulative annual totals for cardiac actin and collagen, using steps similar to those iterated in Table 8.1 for Cx43, can be calculated at approximately 2.5 grams and approximately 8.5 grams, respectively. These numbers may seem surprising and care should be taken not to overinterpret their implications, especially in light of findings that Cx43 is exported by cells in exosomes, as discussed later. Nonetheless, even if taken as a rough guide, the calculations suggest that myocardial tissues in mammals dedicate significant amounts of energy to the task of producing, maintaining, and turning over Cx43.
| The steady-state mass of 0.03 g of Cx43 in a human female ventricle is calculated from: | |
|---|---|
| 1. μm 2 GJ/μm 3 of ventricle | 0.0055 |
| 2. Total μm 2 of GJ in ventricle | 1.12E × 10 12 |
| 3. Cx43 molecules per μm 2 of GJ | 344800 |
| 4. Total Cx43 molecules in the ventricle | 3.87E × 10 17 |
| 5. Total Moles of Cx43 in the ventricle | 6.42E × 10 -07 |
This chapter surveys the literature to help us better understand why the mammalian heart may commit such a high level of resources to Cx43. Addressing this question is not just academic. We posit that insight into this phenomenon has profound implications for understanding the mechanisms of heart disease. It may also point the way to new approaches to clinical treatments, including for arrhythmias, where there continues to be a critical unmet need for safe and effective therapies. Aspects covered will include an overview of basics of the molecular biology, structure, and function of cardiac Cx43 and the gene that encodes it: GJA1. We will touch on, but not comprehensively survey, other cardiac connexins. This topic has been extensively reviewed by others over the years, , including by the late Felix Bukauskas, who dealt with this area in his trademark scholarly manner in the last edition of this textbook. Instead, our focus is on Cx43, with particular emphasis on its role in the myocardium and how understanding has expanded since 2000. Progress in the new millennium has provided a number of new and unexpected insights into how the function of connexins in the heart may be more complex and multifaceted than once imagined. Included in the advances discussed are the identification of a novel domain of the Cx43 GJ called the perinexus ; the growing understanding of the pathogenic role of Cx43 hemichannels; the posttranscriptional regulation of Cx43 expression and its implications for cardiac function; and the emergence of Cx43-based and/or selective pharmacologic agents. We will also cover the identification of Cx43 channels at unexpected locations, including at junctions between myocytes and immune cells and in mitochondria and exosomes. Last, but certainly not least, we review the growing body of evidence that gap junctions formed by Cx43 may not represent the sole obligate mechanism of cardiac conduction.
The connexins are a family of integral membrane proteins encoded by 21 different homologous genes in humans. The myocardial tissues of mammals express a number of these genes, including Cx40/ GJA5 , which occurs mainly in the atria and His-Purkinje tissues of the cardiac conduction system , ; Cx45/ GJA3, which is preferentially expressed in pacemaking and conduction tissues, albeit at low levels , ; and Cx43/ GJA1 , , which comprises most (estimated in the mouse to exceed 99%) of the connexin produced by ventricular myocytes. A fourth isoform Cx30.2 has been described at the mouse AV junction, but the cognate of this protein is not found in the human heart. There are also reports of expression of low levels of Cx46/ GJA6 in myocardial tissues. Cx46 is a GJ protein that was first identified in the avascular tissues of the eye lens. It should be noted that connexins are also found in abundance in the heart’s nonmyocardial tissues, including Cx43, Cx40, and Cx37 in endocardial cells, vascular cells, fibroblasts, and macrophages. , Multiple connexins can be expressed by the same cell. , For example, Purkinje fibers in the ventricle can coexpress Cx43 and Cx40. This has led some to point to the potential for connexin-formed channels to have properties defined by different combinations of connexins, including connexon channels that are coassembled from multiple connexins (heteromeric connexons) or connexons of differing connexin composition that dock to form heterotypic GJ channels. Reviews of this topic have been provided in Molica et al. and others. ,
The basic structure of the intercellular channel formed by connexins is highly conserved. , Six connexin subunits oligomerize to form a connexon (also known as a hemichannel or half-channel) in the membrane. Two connexons dock back-to-back across a narrow extracellular gap between the membranes of neighboring cells to form an intercellular channel; many of the cells are packed tightly into the channel aggregate or plaque that forms the GJ proper (the terms GJ and GJ plaque are used synonymously). Connexins share the same integral membrane protein structure comprising four membrane-spanning alpha-helical domains (M1 to M4), a cytoplasmic loop (CL), two extracellular loops (E1 and E2), and cytoplasmic amino (NT) and carboxyl terminal (CT) domains. Based on a 3.5 Å resolution crystal structure of human Cx26, M1 and E1 line the channel pore. The channel vestibule, in turn, is constricted by six NT domains, narrowing the pore. Cysteine scanning has provided further support for this high resolution topology with some caveats, suggesting that the NT, M1, and E1 domains are directly involved in defining the unitary conductance, gating properties, and perm-selectivity of GJ channels. Electron cryo-crystallography studies of the structure of channels formed by Cx43 with a truncated CT domain at 7.5 Å resolution has confirmed that each connexon possesses 24 helices, which is consistent with the long-established understanding of the structural configuration of the channel. , In addition to intercellular GJ channels, undocked connexon hemichannels within the cell membrane have emerged as having assignments in vivo—particularly in the cardiac injury response—a topic that we will return to later.
The electrical conductances of GJ channels composed of subunits representing a single connexin type range from 9 picoSiemens (pS) for Cx30.2 to around 20 pS for Cx45, 30 to 110 pS for Cx43, and approximately 200 pS for Cx40. , , It is worth noting that these measurements are derived from KCl-predominant solutions; channel conductances are somewhat lower when large ions (tetraethylammonium [TEA], glutamate, aspartate, etc.) are substituted for either K or Cl (Dr. Janis Burt, personal communication). Mixed heteromeric and/or heterotypic channels display conductances and other electrical and perm-selectivity properties that appear to be determined, at least in part, by the stoichiometry of their constituent connexins. , The channel is gated and activity is characterized by flickering between open and closed states over millisecond and submillisecond time scales. Conductance is determined by the amplitude of this opening and closing activity, and for some connexins, multiple conductance states can occur. For example, open state amplitudes for Cx43 GJ channels have a main peak at around 110 pS, but there is also a smaller substate peak centered at approximately 30 pS. Among other factors, it has long been known that channel opening and closure shows sensitivity to [Ca 2+ ] and pH. , , The CL and CT domains of Cx43 appear to be particularly important in the regulation of channel gating in response to Ca 2+ and H + ion concentrations, respectively. The amino-terminus of Cx43 has been reported to be a determinant of a modest level of charge selectivity by the channel. Studies of Cx43 suggest posttranslational modification is a further key factor that regulates channel and nonchannel functions of Cx43, of which the best studied so far is phosphorylation of the CT domain. The extent of the GJ plaque may also modify channel conductance for constituent channels because of access resistance, which is theoretically highest for channels near the center of aggregates. This being said, there is evidence that channels in small Cx43 GJ plaques (<0.1 μm, containing <500 channels) may not be able to mediate significant levels of intercellular coupling, and that even for larger aggregates of Cx43-formed GJ channels, only a small fraction of these are functional. Work by Falk et al. suggests that the small fraction of active Cx43 channels within GJs is located at the circumference of the aggregate, with those further toward the plaque center being in a closed state. Interestingly, the authors suggest that closure of inner channels within the aggregate results from a hierarchical series of Cx43 phosphorylation/dephosphorylation events that occur concentrically from the outer edge of the GJ plaque inward.
The naming of connexins and the genes encoding these proteins has been a subject of debate within the field and a product of the vagaries of history and language. In the 1970s and early 1980s, it was not anticipated that connexins would emerge as a large and diverse family of proteins. The first connexin gene was cloned by Paul in 1986; based on the predicted 32 KDa molecular weight encoded by its gene sequence, the protein was named connexin32 (Cx32). The convention of naming connexins by predicted molecular mass has stuck even though, in retrospect, it is now viewed as less than ideal. By contrast, the genes encoding connexins are named in numerical sequence divided into five classes defined by Greek notation based on sequence similarity—α, β, γ, δ, and ε—a number of examples of which we have already introduced. For example, the genes encoding human Cx32 and Cx43 are, respectively, GJB2 and GJA1. Mutations in connexin genes are associated with inherited disease in humans, including a growing array of mutations in GJA1 linked to a pleiotropic syndrome called oculo-dental-digital dysplasia (ODDD). Its appellation notwithstanding, the syndrome is associated with heart disease. Mutations in Cx40 and Cx45 have also been associated with the occurrence of atrial arrhythmia in humans. The genomic structure of connexin genes, including Cx43 /GJA1, is relatively simple with the protein sequence of most family members being encoded by a single exon (an organization that possibly reflects the emergence of these genes at the divergence of vertebrate and invertebrate lineages). Nevertheless, as will be discussed subsequently, the posttranscriptional regulation of Cx43/ GJA1 mRNAs is anything but straightforward and has been among the most significant new findings in the field of the last years.
Cx43, with its predicted molecular mass of 43 KDa, was the second connexin to be identified. The group responsible soon followed up with a second report demonstrating the iconic polarized distribution of immunolocalized Cx43 at intercalated disks in ventricular myocardium. A year or so after that, our group provided further details on the three-dimensional (3D) organization of GJs in the ventricle, using the then relatively new technique of laser scanning confocal microscopy. , This approach showed that large Cx43 GJs formed distinct ovoid rings circumscribing the edge of intercalated disks (the specialized zones of electrical and mechanical interaction characteristically found between myocytes). This highly organized distribution of Cx43 GJs at intercalated disks is disrupted in diseases of the heart, resulting in prominent localization of Cx43 in the lateral sarcolemma of myocytes—a process referred to as lateralization . , Interestingly, lateralized GJs are a feature of myocytes in the developing mammalian heart. Cx43 GJs in the ventricle differentiate into their characteristic polarized distributions slowly, over a time course that spans almost the entire period of postnatal growth in mammals, lagging behind the accumulation of other junctional proteins (e.g., N-cadherins, desmoplakin) and ion channels (e.g., sodium channels) that come to be concentrated in intercalated disks. , It should also be noted that in homeostasis, a significant proportion of cellular Cx43 is not localized in GJs. An estimate from cultures of neonatal rat ventricular myocytes based on quantification of polymerization ligation assay signals indicated that Cx43 molecules divide evenly between the cell membrane (49%) and cytoplasmic compartments (51%), with GJs accounting for most (85%) of the sarcolemmal pool of Cx43. Cell membrane-associated Cx43 appears to reside largely in connexon channels, which are assembled in the trans-Golgi (TGN) before cytoskeletal transport to the sarcolemma. Removal and degradation of Cx43 is thought to mainly occur via ubiquitination and endocytosis of part or all of the GJ. This is contributed to by clathrin-based mechanisms, , and possibly by ZO-1-actin-mediated processes, , and associated with the formation structures called annular junctions or connexosomes , which are then processed by the degradative machinery of the cell. This being said, evidence suggests that internalized Cx43 also may be recycled from the endosome via the TGN back to the cell membrane by an EHD1-based mechanism.
The original model of GJ structure proposed by Caspar, Makowski, and colleagues has largely stood the test of time. , Based on electron microscopy (EM) and x-ray diffraction, these workers proposed that the GJ was a semicrystalline array of hexameric channels, packed with around 20 nm spacing, which were symmetrically aligned one-to-one with partnering hexamers in a neighboring cell. GJs can vary in size from a cluster of a few channels to densely packed domains that in the largest cases extend across tens of square micrometers of cell membrane. , GJ plaques account for 1.7% of external sarcolemma of ventricular cardiomyocytes in adult mammals, with each usually being adjoined to neighboring cells by hundreds of such plaques of GJ, the majority of which are concentrated in intercalated disks.
The 2 nm separation between cell membranes at GJs is probably one of the closest stable interactions between any two cells anywhere on the tree of life, short of membrane fusion. The second closest zone of intercellular interaction in cardiomyocytes is the narrow 2 to 30 nm extracellular cleft that circumscribes each GJ channel aggregate, a domain that, in 2011, we dubbed the perinexus ( Fig. 8.1 ). The perinexus ranges variably up to 500 nm from the circumference of the Cx43 GJ plaque, with an average extent of 200 nm. Given the large number of GJs between ventricular myocytes, simple geometrical considerations suggest that perinexi are also an extended and frequent feature of cardiomyocyte ultrastructure. Accordingly, in recent years, evidence has mounted for the importance of the perinexus to cardiac function. The perinexus was first identified via the preferential localization of the PDZ-containing scaffolding protein ZO-1 at the edge of Cx43 GJs. , Subsequently, it was shown that ZO-1 participates in the regulation of the size, number, and localization of GJs by controlling the rate at which Cx43 hemichannels, concentrated within the perinexus, dock with connexons in the neighboring cell membrane to form GJ channels. In a related and potentially synonymous action, ZO-1 has been identified as having a role in Cx43 hemichannel exchange from lipid rafts. , In line with such assignments, loss of ZO-1, or Cx43-ZO-1 binding competence, is associated with unregulated recruitment of channels to the plaque and increases in the size of the GJ aggregate, , a process also governed by phosphorylation, particularly at the S373 residue of the protein, a consensus AKT phosphorylation site. Studies in cardiac-specific knockout mice indicate that ZO-2 may compensate for ZO-1 in ventricular myocardium, but there appears to be no similar ZO protein redundancy in SA and atrioventricular (AV) nodal tissues. , Of that fraction of Cx43 localized in neonatal myocyte cell membranes as connexons, it has been estimated that around 14% occur as undocked hemichannels in the perinexus. Thus the perinexus may be a niche in which undocked hemichannels are concentrated in the cell membrane, at least under homeostatic, nonpathologic conditions (see Fig. 8.1 ).
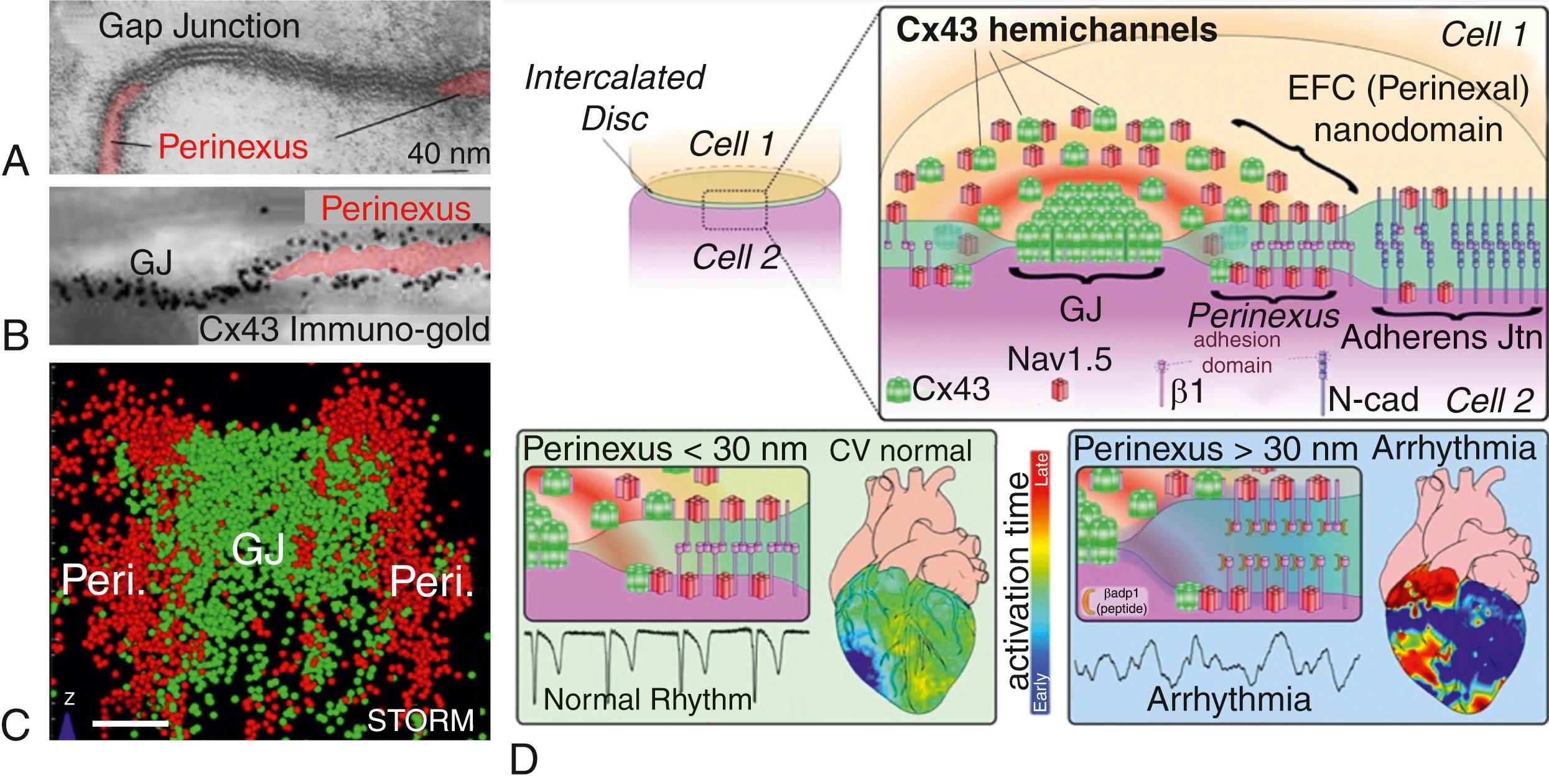
Other proteins reported in the perinexus include the water channel aquaporin4 (AQP4), the glutamate transporter EAAT2b, the desmosome-associated protein PKP2, and the ankyrin family member AnkG/ANK3. As with ZO-1 loss of function, when AnkG expression was silenced by small interfering RNA (siRNA), GJ-PKP2 interacting areas increased in size. AnkG interacts with the voltage-gated sodium channel α-subunit Na v 1.5/SCN5A, , as does Cx43, with the latter protein–protein association being localized in the perinexus. , Similarly, super-resolution microscopy has demonstrated that the potassium channel protein K ir 2.1 is preferentially concentrated at perinexal locations within intercalated disks. An addition of late to the growing list of perinexal proteins is the SCN1B/β1-subunit of the voltage-gated sodium channel (VGSC). Interestingly, quantitative analysis of stochastic optical reconstruction microscopy (STORM) images indicated that 97% of sarcolemmal SCN1B/β1 occurred at perinexal and interplicate regions of the intercalated disk, where the preponderance of GJs are located. A smaller relative proportion (48%) of Na v 1.5 was found proximal to Cx43 GJs, suggesting that this population of α-subunits was closely associated with SCN1B/β1, particularly in the perinexus. This difference in distribution of the two VGSC proteins suggests they may have independent roles in addition to their assignments within the sodium channel complex. SCN1B/β1 is a cell adhesion protein with an extracellular immunoglobulin (Ig) domain that shares homology with other Ig-containing adhesion molecules, including CAMs, JAMs, cadherins (e.g., desmoglein-2) and the myelin adhesion family of proteins that include P0. , , As summarized in Fig. 8.1 , we have provided evidence that VGSC β-subunits mediate intercellular adhesion and may approximate transapposed Na v 1.5 channels within the perinexus. This arrangement could be key to a novel channel-independent role for Cx43 in cardiac conduction, a concept that we will explore in the next section.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here