Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Infections caused by fungi and yeasts are common in neonates and infants. Among the most frequent are Candida infections such as thrush and diaper dermatitis. More extensive manifestations, such as congenital and systemic candidiasis, are much less common. Improvements in preterm infant survival due to advances in neonatal intensive care and an increase in bone marrow and organ transplantation have resulted in more frequent systemic fungal infections. Aspergillus is second only to Candida as a cause of opportunistic fungal infections in immunocompromised pediatric hosts. Zygomycosis and trichosporonosis are seen almost exclusively in premature infants. Malassezia species colonize the newborn skin and plays a role in a variety of clinical presentations such as neonatal cephalic pustulosis, tinea versicolor, or fungemia. Dermatophyte infections (mainly tinea capitis and tinea corporis) are the predominant cutaneous fungal infections during infancy in otherwise healthy infants and children.
Candida is a common fungal pathogen. In newborns, infection may be acquired vertically from the mother or horizontally by nosocomial transmission in the nursery. The increasing use of antifungal prophylaxis with azoles has changed the epidemiology of candidemia in immunocompromised pediatric patients. C. albicans is responsible for approximately 45% of neonatal fungal infections, while non-albicans candidal species are increasing in incidence. Other Candida species associated with neonatal disease include C. tropicalis , C. parapsilosis , C. lusitaniae , and C. glabrata . Normally, these yeasts are saprophytes, inhabiting the skin or gastrointestinal tract without invasion unless host defenses are altered. Candida spp. may also colonize endotracheal tubes and catheters without causing systemic illness, but associated clinical illness is common in very low-birthweight (VLBW) infants. In extremely low-birthweight (ELBW) infants (<1000 g), invasive candidiasis is more common. In one prospective study, 7% of 4379 infants had Candida isolated from blood or cerebrospinal fluid.
Virulence mechanisms associated with Candida infections include fungal proteinase, increased adherence of yeast to epithelial cells due to similarity to mammalian cell ligands, and resistance to neutrophil ingestion of hyphal forms. Secretory IgA, functional T lymphocytes, and phagocytic cells are important in defense against Candida infections. Hence, there is increased susceptibility to these infections in patients with secretory IgA deficiency, primary T-cell deficiency such as DiGeorge syndrome, severe combined immunodeficiency, chronic granulomatous disease, myeloperoxidase deficiency, and human immunodeficiency virus (HIV) infection. Recurrent or persistent yeast infections can be presenting symptoms of immunodeficiency. Host resistance to fungal infections also depends on activated macrophages, which in turn rely on T-lymphocyte release of interferon (IFN)-γ. Incomplete activation of macrophages by IFN in neonates contributes to increased susceptibility to invasive fungal disease.
Predisposing factors for Candida infections include excessive humidity, maceration, diabetes, and broad-spectrum antibiotics. Risk factors for systemic candidiasis in neonates include low birthweight, prematurity, broad-spectrum antibiotic therapy, indwelling catheters, prolonged endotracheal intubation, tracheostomy, prior fungal colonization, gastrointestinal pathology or abdominal surgery, enteral feeding, immunosuppression, defective neutrophil function or neutropenia, and steroid therapy. Similarly, pediatric patients with hematologic malignancies and neutropenia, short bowel syndrome with central vascular catheters or recent surgery or trauma (namely gastrointestinal/abdominal) are at increased risk of invasive candidiasis.
Various techniques in addition to culture are used to evaluate the epidemiology of fungal pathogens. They include polymerase chain reaction (PCR), restriction fragment endonuclease digestion of chromosomal DNA, electrophoretic karyotyping, and Southern blot hybridization analysis using DNA probes, β-glucan assay (β- d -glucan is a major component of the fungal cell wall) and gas chromatography mass spectrometry for d -arabinitol (a major metabolite of most Candida species).
Clinical presentations of candidal infections are discussed in this chapter, followed by diagnostic and treatment recommendations.
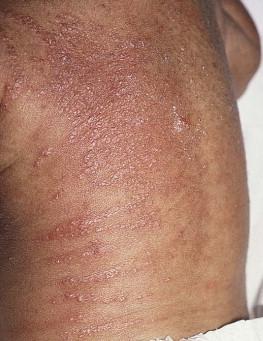
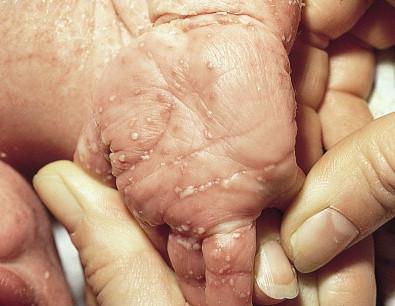
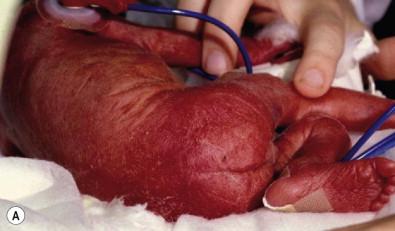
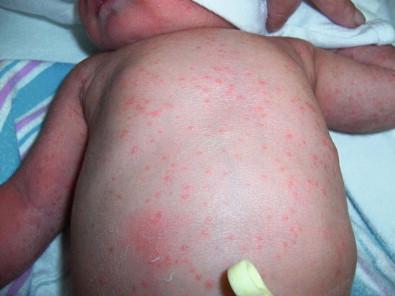
Congenital candidiasis (CC) refers to Candida infection acquired in utero and presenting with symptoms in the first days of life. Classic congenital candidiasis presents as a diffuse cutaneous infection, presumed to arise from an ascending intrauterine chorioamnionitis. The typical patient is an otherwise healthy term neonate who, within the first 12 h of life, develops a monomorphous papulovesicular eruption that is intensely erythematous ( Figs. 14.1, 14.2, 14.3 ![]() ) progressing to pustules, crusting and desquamation. Any area of the body surface, including the face, palms, and soles, can be involved, and widespread involvement is often evident ( Fig. 14.4 ). Candida paronychia and onychomycosis have been reported. Congenital candidiasis is surprisingly uncommon given the 33% rate of vaginal colonization with Candida in pregnant women. The presence of a foreign body in the maternal uterus or cervix is a risk factor for CC. In the majority of term infants, systemic dissemination of CC is rare. The prognosis of a term infant with CC is excellent, with rapid clearance of the eruption using topical treatment. Occasionally, CC can present as pneumonia and sepsis without cutaneous manifestations. CC with systemic involvement is more commonly seen in premature, low-birthweight infants (<1500 g). Skin findings in these infants may be variable, with a burn-like dermatitis (similar to staphylococcal scalded skin syndrome) in addition to the usual red scaly or vesiculopustular eruption ( Fig. 14.5 ). A widespread rash in a premature or ill-appearing infant, respiratory distress in the immediate postnatal period, leukocytosis, or hyperglycemia should alert the physician to the possibility of systemic candidiasis despite negative blood cultures.
) progressing to pustules, crusting and desquamation. Any area of the body surface, including the face, palms, and soles, can be involved, and widespread involvement is often evident ( Fig. 14.4 ). Candida paronychia and onychomycosis have been reported. Congenital candidiasis is surprisingly uncommon given the 33% rate of vaginal colonization with Candida in pregnant women. The presence of a foreign body in the maternal uterus or cervix is a risk factor for CC. In the majority of term infants, systemic dissemination of CC is rare. The prognosis of a term infant with CC is excellent, with rapid clearance of the eruption using topical treatment. Occasionally, CC can present as pneumonia and sepsis without cutaneous manifestations. CC with systemic involvement is more commonly seen in premature, low-birthweight infants (<1500 g). Skin findings in these infants may be variable, with a burn-like dermatitis (similar to staphylococcal scalded skin syndrome) in addition to the usual red scaly or vesiculopustular eruption ( Fig. 14.5 ). A widespread rash in a premature or ill-appearing infant, respiratory distress in the immediate postnatal period, leukocytosis, or hyperglycemia should alert the physician to the possibility of systemic candidiasis despite negative blood cultures.
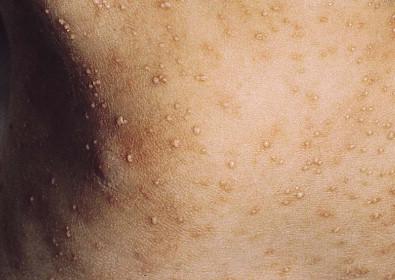
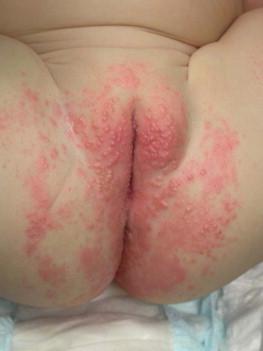
Systemic candidiasis (SC), defined as Candida infection in an otherwise sterile body fluid such as blood, urine, or cerebrospinal fluid, affects 2–7% of VLBW newborns. Skin manifestations occur in up to 60% of these infants. These infections can be acquired in utero (CC) or postnatally. In a multicenter prospective study, median gestational age and birthweight of neonatal invasive candidiasis was 30 weeks and 740 g, respectively. Only half of these patients had a central venous catheter at the time of diagnosis. Baley and Silverman described skin manifestations of VLBW infants with SC to include an extensive burn-like dermatitis followed by desquamation, progressive diaper dermatitis involving papules and pustules, and isolated diaper rash with or without thrush ( Fig. 14.6 ). Cutaneous abscesses at the site of intravascular catheters may also be seen. Systemic signs include apnea, bradycardia, abdominal distension, guaiac-positive stools, hyperglycemia, temperature instability, leukemoid reaction, and hypotension. During childhood, invasive candidiasis occurs in immunocompromised individuals, especially those having had a central vascular catheter placed within 7 days of diagnosis. After hematopoietic stem cell transplantation, the median time to invasive candidiasis diagnosis is 74.5 days. Systemic candidiasis in immunocompromised children presents as randomly distributed deep, firm and erythematous papules and nodules.
Invasive fungal dermatitis (IFD) is a clinicopathologic entity of erosive crusting lesions in VLBW infants. It is described by Rowen and colleagues as a primary skin condition that leads to secondary dissemination and systemic disease. It is primarily due to Candida albicans or other Candida species. Aspergillus , Trichosporon asahii , and Curvularia are also etiologic agents of invasive fungal dermatitis. Skin biopsy demonstrates fungal invasion beyond the stratum corneum, well into the epidermis, and at times extending into the dermis. Onset several days after birth, the presence of erosions and crusts, and typical histologic findings help to differentiate IFD from congenital candidiasis. Risk factors include extreme prematurity (<25 weeks' gestational age), vaginal birth, steroid administration, and hyperglycemia.
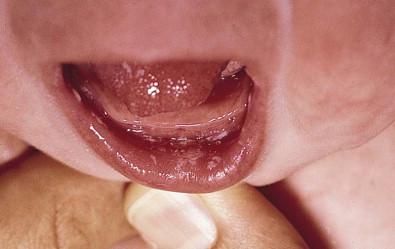
Acute oral candidiasis appears on the oropharyngeal mucosa as white adherent curd-like plaques resembling milk or formula ( Fig. 14.6 ). Plaques can be scraped off only with difficulty, leaving a bright erythematous base (pseudomembranous and erythematous forms, respectively). Extensive infection can lead to feeding difficulties, particularly if the esophagus is involved. Perlèche is a candidal infection involving the lateral angles of the mouth.
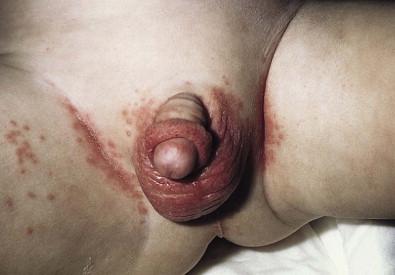
Candida infection of the diaper area may occur alone or in conjunction with thrush. Bright, erythematous plaques, papules, and pustules affect the moist intertriginous areas of the perineum, with a predilection for inguinal creases. White scale and satellite pustules are common along the periphery, often prominent at the border of involved and uninvolved skin ( Figs. 14.7 , 14.8 ![]() ). Perianal involvement is common. Pustules may be very superficial and rupture easily leaving collarettes of scale. Candidal dermatitis may be seen in 4–6% of term newborns, with the incidence peaking at 3–4 months of age. Similar bright erythema may be seen in inverse psoriasis. Further differential diagnoses including infectious and noninfectious entities are outlined in Chapter 17 .
). Perianal involvement is common. Pustules may be very superficial and rupture easily leaving collarettes of scale. Candidal dermatitis may be seen in 4–6% of term newborns, with the incidence peaking at 3–4 months of age. Similar bright erythema may be seen in inverse psoriasis. Further differential diagnoses including infectious and noninfectious entities are outlined in Chapter 17 .
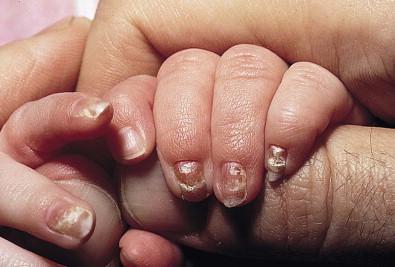
Candidal infection of the nail plate and related structures may occur alone or in conjunction with systemic and congenital candidiasis. Candidal paronychia is common in finger-sucking children and presents with erythematous, edematous and tender nail folds with loss of cuticle. If present for months (chronic paronychia), resulting nail plate dystrophy can be seen distinguishing it from acute bacterial paronychia.
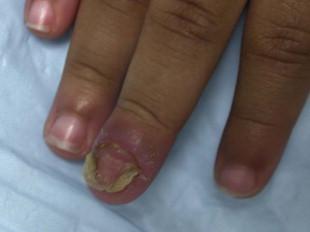
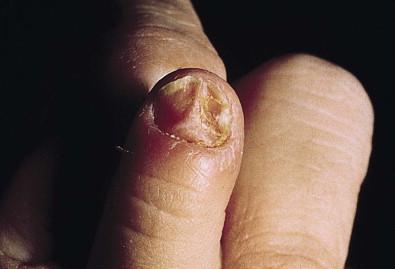
For onychomycosis secondary to Candida , finger sucking is a potential predisposing event ( Figs. 14.9, 14.10 ![]() , 14.11 ). The resultant onychodystrophy may lead to proximal-, distal-, and lateral-subungual, superficial white, or total dystrophic onychomycosis. The latter presents with a crumbling nail and an abnormally thickened nail bed. It is frequently seen in patients with chronic mucocutaneous candidiasis and other immunodeficiency states. Tinea unguium, hereditary onychodystrophy, ectodermal dysplasia, epidermolysis bullosa, psoriasis, and acrodermatitis may present with similar nail findings.
, 14.11 ). The resultant onychodystrophy may lead to proximal-, distal-, and lateral-subungual, superficial white, or total dystrophic onychomycosis. The latter presents with a crumbling nail and an abnormally thickened nail bed. It is frequently seen in patients with chronic mucocutaneous candidiasis and other immunodeficiency states. Tinea unguium, hereditary onychodystrophy, ectodermal dysplasia, epidermolysis bullosa, psoriasis, and acrodermatitis may present with similar nail findings.
Skin scrapings from pustules or peripheral scale should be examined using KOH solution, Giemsa, Gram, or calcofluor stains. Pseudohyphae and spores may be visualized with direct staining. Satellite pustules are most likely to yield positive results. Cultures from multiple sites, including skin, blood, cerebrospinal fluid, and urine, should be collected if systemic disease is suspected in premature infants or immunocompromised children. Culture yield is inconsistent, and negative cultures do not rule out systemic disease in the symptomatic infant. Buffy coat smear microscopy, a rapid bedside test with 100% specificity, can confirm candidemia within 1–2 h. Sensitivity is 62%, compared with 44% for peripheral blood smear examination. In some centers, buffy coat culture may yield results faster than whole blood cultures. A skin biopsy specimen may reveal a subcorneal pustule with neutrophils, and periodic acid–Schiff (PAS) staining will highlight the organisms. Invasive fungal dermatitis demonstrates invasion and inflammation of the epidermis, and possibly invasion of the dermis. In both neonates and children, Candida albicans is the etiologic organism in approximately 50% of candidal infections. As a group, the non-albicans species comprise the remaining causes.
Localized forms of candidiasis can be treated topically in most term infants and healthy children. For thrush, nystatin solution (100 000 units/mL) is applied to the oral mucosa four times per day for at least 1 week. Resistant thrush may respond to once-daily oral fluconazole (2–3 mg/kg per day) or itraconazole (2 mg/kg per day), particularly in immunocompromised children. Oral amphotericin B has been studied for treatment of recurrent thrush. Imidazole creams are useful for diaper dermatitis and nail infection. Nystatin, allylamines (including naftifine and terbinafine), or aqueous solutions of 1% gentian violet or 2% eosin are alternatives for localized disease. Inflamed or erosive monilial diaper dermatitis may require a combination of the above with a 1% hydrocortisone cream or ointment and barrier paste containing zinc oxide. Oral nystatin or fluconazole may also be a useful adjunct for the treatment of diaper dermatitis, especially for recurrent disease, concurrent oral candidiasis or persistent cutaneous candidiasis in the setting of prolonged oral antibiotics thereby reducing the load of gastrointestinal Candida . Congenital candidiasis in asymptomatic term infants can be treated with topical agents alone. However, for ill and VLBW preterm infants, systemic treatment is recommended.
In pediatric patients with invasive candidiasis, successful treatment outcomes are related to the candidal species causing the infection. C. parapsilosis and C. albicans have significantly better treatment outcomes than the more virulent C. glabrata infections. The drug of choice for systemic antifungal therapy evolves with candidal species, concurrent medical conditions and balance of potential medication side-effects. Early empiric systemic therapy reduces mortality in pediatric patients with candidemia. In ELBW infants, empiric treatment improves survival without associated neurodevelopmental impairment. Once the candidal species is indentified, adjustment of the antifungal agent may be necessary. In neutropenic patients, therapy should be continued for at least 14 days after last positive blood culture.
Amphotericin B, a polyene macrolide antibiotic, has been a major therapeutic agent in systemic candidasis in immunocompromised patients. It is typically utilized in immunocompromised neutropenic patients with prior failed fluconazole prophylaxis or those at high risk of C. glabrata or C. krusei infections. Less commonly seen in neonates, the potential for nephrotoxicity warrants monitoring of renal function. Hepatotoxicity and bone marrow suppression are also potential side-effects. Lipid-associated amphotericin B formulations have less direct renal tubular toxicity and allow delivery of greater dosages, limits of infusion volume, and less toxicity. In addition to use in children, successful treatment of systemic fungal disease with these products has been reported in neonates. Both the liposomal and colloidal dispersion forms of amphotericin B were shown to be safe and effective in ELBW infants with candidemia and renal dysfunction. Furthermore, high-dose (5–7 mg/kg per day) liposomal amphotericin B may add a therapeutic advantage with more rapid eradication of infection when used as first-line therapy.
Systemic fluconazole is commonly used in pediatric invasive candidal infections and has even been used successfully for treatment of systemic candidiasis in neonates. The recommended daily dose is 6 mg/kg per day for patients over 4 weeks of age, with less-frequent dosing for infants with compromised renal function and for those less than 4 weeks old. It is particularly useful in those with C. albicans infection and in whom amphotericin B is ineffective or contraindicated. When given in the first trimester of pregnancy, fluconazole has been reported to be teratogenic, leading to multiple malformations. Resistance to fluconazole can be seen especially in non-albicans Candida species ( C. krusei , C. tropicalis , C. glabrata , C. parapsilosis ), and also some C. albicans . Susceptibility testing can be performed to document the utility of these agents.
Oral itraconazole administered at 5 mg/kg per day has been safe and well tolerated in infants and children; however, there are limited reports documenting its use in newborns with candidiasis. Oral ketoconazole (3–6.5 mg/kg per day) has been largely supplanted by the newer triazoles for candidiasis due to its risk of fulminant hepatitis.
The echinocandins such as caspofungin and micafungin are a relatively new class of pediatric-approved antifungal agents, which irreversibly inhibit 1,3-β- d -glucan synthesis. These medications are fungicidal against Candida spp., including fluconazole-resistant strains, making them especially important for cases of non-albicans candidal infection. In general, the echinocandins have similar efficacy as amphotericin, with fewer adverse side-effects. In one study of caspofungin use in neonates with invasive candidiasis unresponsive to amphotericin B, all cultures cleared after 3–7 days of therapy without adverse events.
Especially in C. parapsilosis infections, adjunctive therapy for systemic Candida infection includes changing or removing indwelling catheters when feasible. Possibly due to the inability to sterilize the subcutaneous tract of existing lines, exchange of peripherally inserted catheters is associated with a 25-fold increased risk of bloodstream infections. Placement of new vascular access at a different site is recommended.
Because ELBW infants and immunocompromised children are susceptible to and have a higher mortality risk from systemic Candida infections, prevention of infection by means of fluconazole prophylaxis should receive strong consideration. Potential concerns include unknown short- or long-term risks of fluconazole; uncertainty regarding the group of patients that would benefit most and the optimum dose and duration of therapy; and the potential for increased fluconazole resistance. Fluconazole has been shown to reduce rectal colonization with Candida . Bertini and colleagues gave fluconazole 6 mg/kg every 72 h for week 1, then daily for weeks 2–4 in neonates <1500 g with central venous access, and reduced the rate of candidemia from 7.6% to zero. Healy and coworkers initiated fluconazole prophylaxis in all ELBW infants of <5 days of age and reduced invasive candidiasis-related mortality from 12% to zero. As for dosing, twice-weekly fluconazole (3 mg/kg per dose) in ELBW infants with a central venous catheter or endotracheal tube was as effective as daily prophylaxis in decreasing Candida colonization and bloodstream infections. Meta-analysis of prophylactic fluconazole in this population found both agents to be safe and highly effective in preventing invasive candidiasis. Although these studies appear promising, larger multicenter studies looking closely at the morbidity and mortality rates from all causes, the emergence of resistant Candida strains, and unexpected consequences are still required.
Malassezia (previously referred to as ‘ Pityrosporum ’ species) are saprophytic yeasts found on 90% of adults as normal skin flora. Skin colonization of newborns usually occurs in the first 1–3 months of life. By means of genetic analysis, the species formerly called Malassezia furfur has now been reclassified as M. furfur plus at least six other species. Although M. furfur is the most commonly associated species, M. pachydermatis , M. sympodialis , and M. globosa have also been associated with infections. Three clinical forms of Malassezia infection may present in children: tinea versicolor, cephalic pustulosis, and catheter-associated sepsis without cutaneous lesions.
Malassezia furfur is the main species responsible for human skin colonization and infection. Other Malassezia species, including M. sympodialis , M. globosa , and M. pachydermatis have also been implicated in human disease. Malassezia colonizes the skin of adults who are usually asymptomatic. Skin colonization begins in infancy, and the prevalence of colonization increases with age. The most common site of colonization in both infants (>78%) and neonates is the ear. Ashbee and collegues showed that 97.6% of infants (>28 days of age) and 31.8% of neonates were colonized; the mean age of colonization was 14 days. Risk factors for colonization include gestational age of <28 weeks and length of hospital stay >10 days. Fungal growth from skin and catheter cultures is not always associated with clinical sepsis.
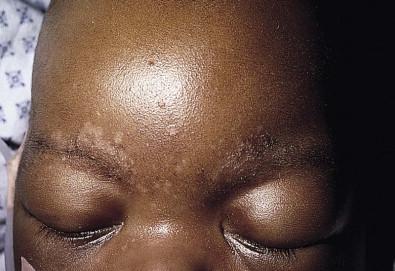
Due to the variation of sebum production during life, tinea versicolor (pityriasis versicolor) is less common in infants than in older children, adolescents, and young adults. However, young infants, particularly those less than 6 months of age, can have more sebum production under the influence of the fetal adrenal glands (so-called ‘mini-puberty of infancy’) and thus are more likely to develop tinea versicolor than older infants or pre-pubertal children. Facial involvement is very typical in affected infants and young children, although lesions may also be seen on the neck and upper trunk. Warm humid weather and immunosuppression induce greater growth of this yeast. A positive family history is variably present. Most commonly caused by M. globosa and M. furfur , tinea versicolor presents with multiple, 0.3–1 cm oval macules or plaques with fine scaling ( Figs. 14.12 ![]() , 14.13 ). Relative to normal skin, lesions may be hypopigmented, skin-colored, or hyperpigmented. Wood's light examination highlights the pigmentary changes and may produce a golden fluorescence. Differential diagnoses include pityriasis alba and postinflammatory hypopigmentation and hyperpigmentation.
, 14.13 ). Relative to normal skin, lesions may be hypopigmented, skin-colored, or hyperpigmented. Wood's light examination highlights the pigmentary changes and may produce a golden fluorescence. Differential diagnoses include pityriasis alba and postinflammatory hypopigmentation and hyperpigmentation.
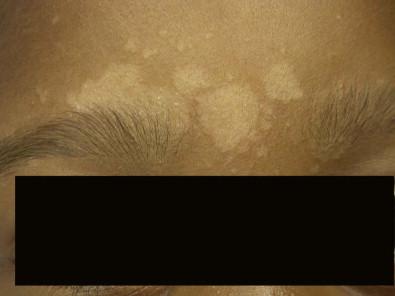
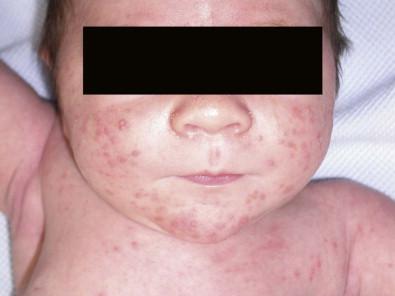
Neonatal cephalic pustulosis (see Chapter 7 ) is a condition that was previously considered to be neonatal acne. During the second or third week of life, multiple, tiny, monomorphous papulopustules on an erythematous base begin on the face, scalp, and neck ( Fig. 14.14 ). Contrary to classic acne, comedones are not a feature, and follicular accentuation is absent. Both M. globosa and M. sympodialis have been reported in association with neonatal cephalic pustulosis. Higher rates of colonization were associated with increased severity of pustulosis. Diagnosis is suggested by onset at 1 month of age, cephalic distribution, microscopic findings of yeast forms suggestive of Malassezia , exclusion of other pustular eruptions, and response to topical ketoconazole. The differential diagnosis of this pustular eruption is discussed more extensively in Chapters 7 and 10 .
Malassezia fungemia is seen primarily in premature infants receiving intralipids through intravenous catheters. Skin colonization rates are much higher in premature infants than in full-term newborns, and the pathogenesis of disease probably involves organisms on the skin gaining venous access through indwelling catheters. Although in one study of VLBW infants (<1250 g), skin colonization with M. furfur was not predictive of catheter infection or systemic illness, the skin colonization rate was 63%, and positive M. furfur blood cultures were common (9.6%) in infants with central venous catheters. Clinical presentation ranges from asymptomatic colonization of the indwelling catheter to sepsis and death. Fever, apnea, bradycardia, and thrombocytopenia in the presence of negative routine bacterial cultures suggest fungal disease. Clusters of cases of infantile bronchopneumonia in neonatal units have also been attributed to M. furfur .
Malassezia can be identified by KOH preparation or Giemsa stain examination of the fine scales or pus; this will reveal clusters of spherical yeast and associated filaments. Malassezia is differentiated from Candida and other yeasts by a broader budding base. Malassezia furfur is a lipophilic yeast that requires fatty acid supplementation for growth. Modified Dixon agar or an olive oil overlay on routine fungal media is used for isolation of these fungi from blood specimens.
Cutaneous infections can be treated with an imidazole cream. A high level of suspicion for Malassezia sepsis is appropriate in premature infants with clinical signs of sepsis and negative bacterial and viral cultures, especially if intravenous lipid emulsions are being infused through venous catheters. Removal of the catheter and cessation of intravenous lipids, without systemic antifungal therapy, may be sufficient therapy for catheter-associated sepsis. Systemic antifungals may be considered if the infant does not show rapid clinical improvement.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here