Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Fungal infections in the neonatal intensive care unit (NICU) remain an important health problem associated with substantial morbidity and mortality. Invasive fungal infections encompass infections largely caused by Candida species and with a small portion caused by Aspergillus , Zygomycetes , Malassezia , and Trichosporin . Invasive Candida infections occur in two main patient groups in the NICU: (1) the extremely premature infant and (2) NICU patients with complex gastrointestinal disease such as necrotizing enterocolitis (NEC), gastroschisis, and spontaneous bowel perforation. Strategies for prevention and management of invasive Candida infections are paramount for improving outcomes for neonates ( Fig. 49.1 ).
The incidence of candidiasis correlates with advances in medical therapy and increased survival of extremely low birth weight and gestational age preterm neonates. In developed countries, increases occurred in the 1980s and 1990s, then began to fall after 2001, primarily in infants less than 1000 g, owing to various factors including antifungal prophylaxis, antibiotic and medication stewardship, and central line–associated bloodstream infection (CLABSI) bundles.
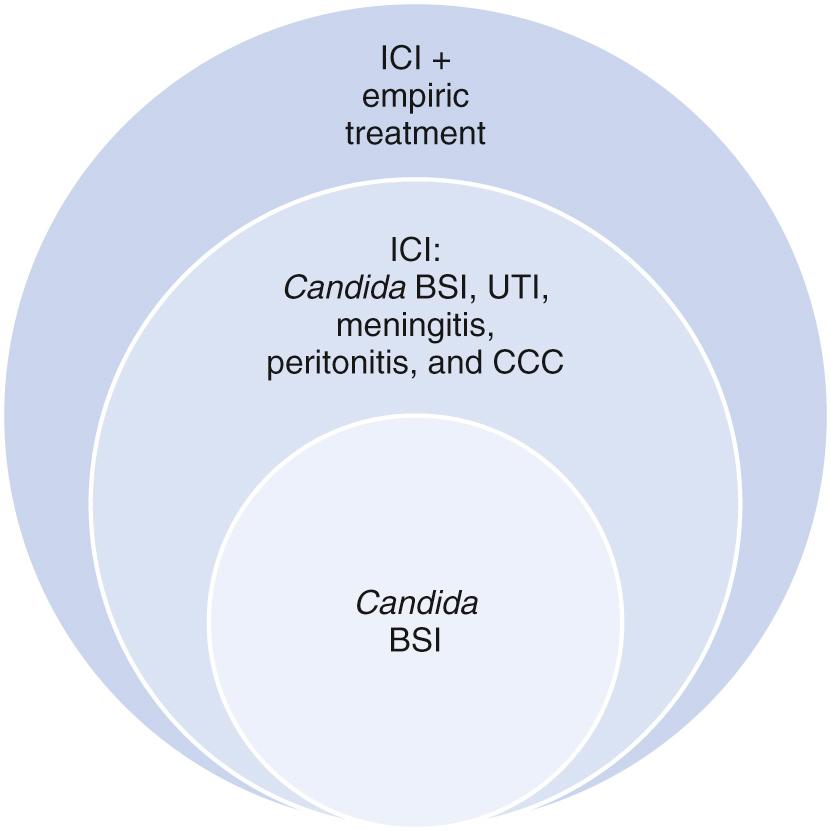
Invasive Candida infections (ICI) in neonates include: congenital cutaneous candidiasis (CCC) and late-onset cutaneous candidiasis, bloodstream infections (BSIs), urinary tract infections (UTIs), meningitis, peritonitis, and infection of other sterile sites (bone and joint infections). These infections are often classified similar to other neonatal infections into early-onset (<72 hours after birth) and likely vertically transmitted and late onset (≥72 hours after birth) and potentially horizontally transmitted infections. This is a worthy system to standardize definitions, recognizing that Candida species are slow-growing organisms and some infections in the first weeks of life have been shown by molecular studies to be vertically transmitted.
Candida albicans and C. parapsilosis account for 80%-90% of invasive Candida infections in centers not using antifungal prophylaxis. Less-common Candida species include C. glabrata and C. tropicalis , and a smaller percentage of infections are caused by C. lusitaniae , C. guilliermondii, and C. dubliniensis. C. parapsilosis isolates have been found to be genetically heterogeneous, with 10% being identified as two different species: C. metapsilosis and C. orthopsilosis . These three species are phenotypically indistinguishable, and molecular methods are needed for their detection. Centers using antifungal prophylaxis have a very low incidence of infections, and the few that occur are primarily by non- albicans species in NICUs using fluconazole prophylaxis. The change in epidemiology of Candida species with antifungal prophylaxis is important because fluconazole prophylaxis has virtually eliminated the more virulent C. albicans infections.
Most Candida species are commensal organisms and part of the normal human microbiome in the skin and gastrointestinal tract. They may exist primarily as budding yeast cells, filamentous hyphal structures, or both. Adherence and colonization are normal, and fungi become pathogenic owing to fungal and host factors ( Fig. 49.2 ).

The slow-growing nature of Candida facilitates its ability to progress from colonization to dissemination into the bloodstream and body tissues before clinical signs and symptoms of infection become apparent. In preterm neonates, similar to other hosts, four major steps can be identified in infected infants after exposure to Candida spp.: (1) adhesion, (2) colonization, (3) dissemination, and (4) abscess formation (see Fig. 49.2 ). These steps are related to the size of the inoculum (e.g., the number of fungal organisms), their virulence, and the characteristics of the host response.
Adhesion is the key step in colonization. Candidal species can adhere to epithelial and endothelial cells as well as catheters. Biofilm formation limits penetration of antifungals and shielding Candida from the host response. Surface glycoproteins such as INT1p, which binds to beta-integrins present on the endothelium and white blood cells, facilitate fungal adherence. The absence of a functional INT1 gene diminishes adherence in yeast cells.
The preterm infant has an immature immune system and is frequently exposed to broad-spectrum antibacterial and other medications that can suppress their immune response. Studies examining the effect of steroids and antibiotics have been performed in mice orally inoculated with C. albicans to mimic conditions in the preterm infant. Antibiotic treatment alone led to increased Candida colonization but did not affect dissemination. When dexamethasone was added to the antibiotic regimen, both colonization and dissemination increased in these animal models. Additionally, dexamethasone plus antibiotics led to an increase in the percentage of filamentous forms in the gastrointestinal tract compared with antibiotics alone. These studies also showed that C. albicans strains with two functional copies of the INT1 gene increased the number of fungi colonizing the cecum and disseminating to extraintestinal sites.
Studies consistently demonstrate higher associated mortality in preterm infants with C. albicans compared with non- albicans BSIs. Its virulence encompasses a combination of factors involving adhesion, cytotoxicity, hydrolytic enzymes (aspartyl proteinases, phospholipases, lipases, and hemolysins), morphologic switching, and genes. In addition to the adhesion factors discussed, C. albicans produces higher concentration of hydrolytic enzymes compared to non- albicans species.
C. albicans can alternate its morphology in response to specific environmental cues, has an obligate diploid genome, lacking a complete sexual cycle, and has several genes important for virulence. Studies have found a strong correlation between virulence and the ability to alternate among three morphological forms—yeasts, hyphae, and pseudohyphae. The morphologic switch is a complex process involving changes in cellular shape, mechanical properties, interactions with other cells, and differential expression of multiple genes. Because of their size and shape, filamentous forms increase virulence of C. albicans in part by escaping phagocytosis.
To further examine the role of yeast and filamentous forms, three strains of C. albicans were studied : (1) a wild-type strain that had both yeast cell and filamentous forms, (2) a strain only producing yeast cells, and (3) a strain that was constitutively filamentous. The mortality rate was significantly greater in both the wild-type (92%) and yeast-cell (56%) strains compared with the filamentous strain alone (0%). The filamentous strain had no dissemination, and cecal colonization was significantly less than that of the other two strains. The wild-type strain had diffuse hyphal invasion with increased tissue necrosis compared with the yeast-cell strain. The researchers speculated that the yeast forms are critically important for adherence and tissue dissemination and that hyphal formation in the tissues contributes to parenchymal destruction.
The immature lymphocyte and antibody system predisposes preterm infants to skin and mucosal fungal adherence and colonization, whereas deficient innate host defense mechanisms predispose them to dissemination and overwhelming infection. With compromise of the barrier defense system, invasive Candida infections may occur, owing to deficient neutrophil function and when neutropenia is present. Neutrophils provide the major role in antifungal defense, ingesting and killing Candida in a process requiring production of oxygen metabolites, antibodies, cytokines, and activation of the C3 complement component, all of which are decreased in preterm infants compared with term infants and adults. Also deficient are neutrophil granules and cytokines that play a critical role in the lysis of Candida hyphae and pseudohyphae, which are too large to be engulfed by phagocytosis. Macrophages have impaired adherence, phagocytosis, and oxidative killing in preterm infants, affecting their ability to contain fungal colonization and infection. In addition, dysregulation of the cytokine response of the innate immunity against fungal infection impairs cytokine enhancement of cell-mediated fungicidal activity. Pulmonary host defense is also diminished by immature or deficient alveolar macrophages, cilia and mucous, and surfactant in preterm infants.
Additionally, studies in animal models showed that the hypoxic insult effect on cellular function and tissues that can be common in preterm infants around delivery or with apnea events is associated with increased gut fungal colonization and dissemination to mesenteric lymph nodes.
Candida species produce proteases that may be lytic for the thin keratin layer produced by the immature stratum corneum in the first weeks after birth and phospholipases against lipid membranes that both may facilitate epithelial invasion. Recently the first cytolytic peptide toxin of C. albicans that can directly damage epithelial membranes was identified. Increased transepidermal water loss from preterm skin creates a moist environment that facilitates fungal colonization and growth. Because of the increased permeability of the preterm skin, substrates such as glucose may diffuse to the epithelial surface, facilitating Candida growth. Candida may alter its surface structure in the presence of high glucose, increasing its adherence and proliferative properties. Skin maturation occurs by 2 weeks of life in extremely preterm infants, after which new fungal skin colonization occurs less frequently.
There is variation between rates in different NICUs and the incidence reported in the literature. These differences reflect a wide range of factors and are not a mere indicator of the overall quality of care. Based on patient demographics; use of antifungal prophylaxis; surgical population; resuscitation of extremely preterm infants; and practices related to feeding, medication, and antibiotic usage of individual NICUs, considerable variation exists between rates and risk for invasive Candida infections. Fridkin et al. reported the rate of Candida BSIs ranged between 3% and more than 23% among NICUs in infants less than 1000 g, and other studies have reported similar findings ( Fig. 49.3 ). Centers that care for infants with gastroschisis, NEC, and other complex gastrointestinal diseases have an increased risk for invasive Candida infections. Resuscitation practices may be the most significant area of practice variation affecting invasive Candida infection rates; NICUs that do not resuscitate infants less than 25 weeks, for example, would have a lower rate of invasive Candida infections in infants less than 1000 grams compared with centers caring for 23- and 24-week-gestation infants.
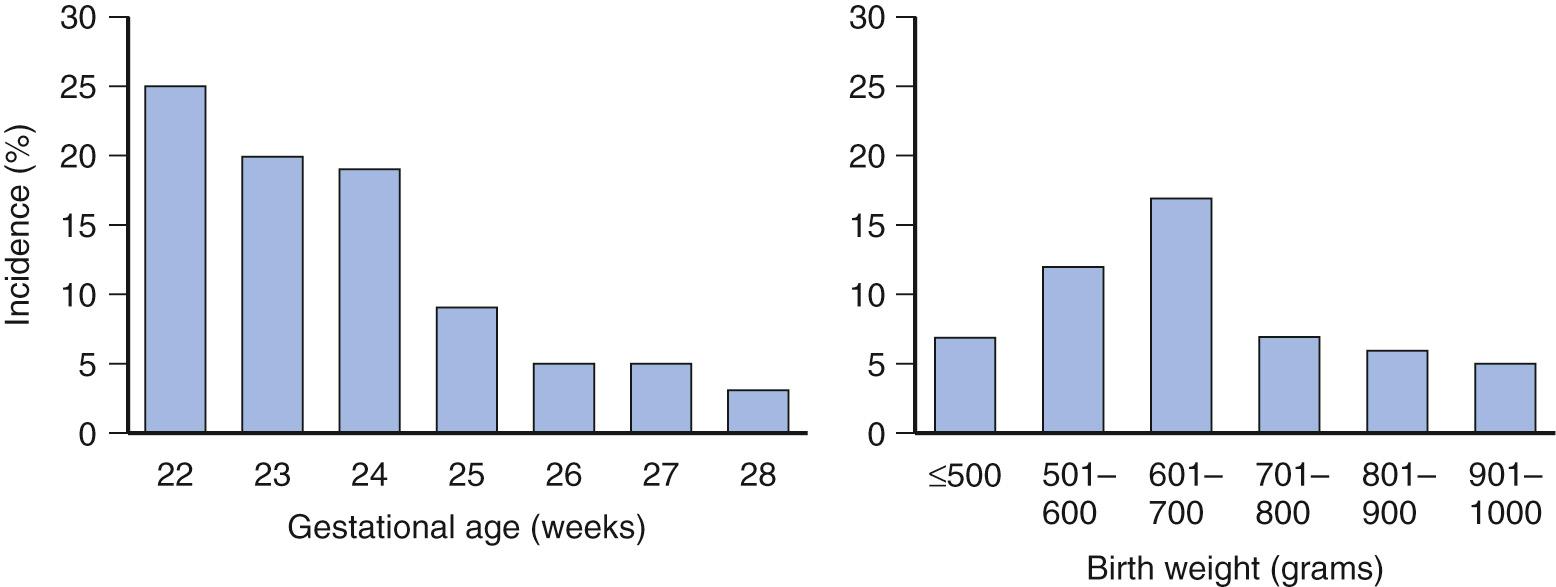
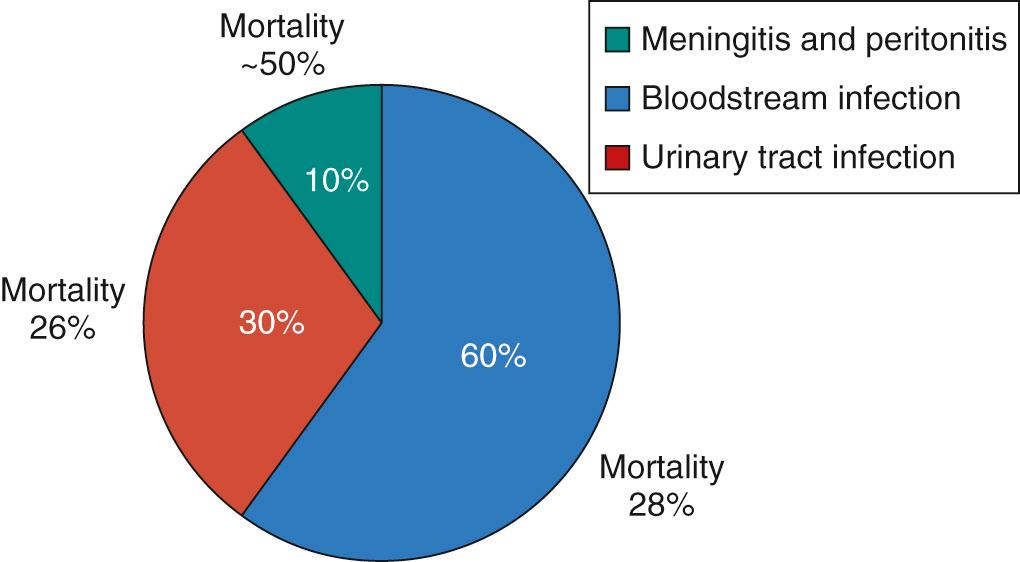
The literature has limitations owing to definitions of incidence. Studies including all invasive Candida infections in both preterm and full-term infants are limited, and most studies of incidence or risk factors focus on only Candida BSIs. The burden of all invasive Candida infections is nearly twice as high as BSI rates ( Figs. 49.4 and 49.5 ).
The incidence of invasive Candida infections in extremely low birth weight (ELBW, <1000 grams at birth) infants, not including congenital cutaneous candidiasis, is around 10% in the absence of antifungal prophylaxis. The highest incidence of invasive Candida infections occurs in the most extremely preterm infants and is greater than 20% for infants less than 25 weeks’ gestation (see Fig. 49.3 ). Many studies only report BSI incidence, but Candida UTIs account for an additional 3%-4% of invasive Candida infections in ELBW infants. Multicenter studies have reported that Candida UTIs have a high associated mortality similar to Candida BSIs in ELBW infants, confirming their invasive nature in neonates. Finally, meningitis and peritonitis contribute an additional 1%-2% to the incidence of invasive Candida infections in ELBW infants.
Examining candidemia alone, the largest is a report from 128 US NICUs using US National Nosocomial Infections Surveillance (NNIS) system data from 1995-2004 ( N = 130,523 neonates). For infants less than 1000 g, the median infection rate was 7.5%, whereas 25% of NICUs had rates 13.5% or higher. The incidence decreases significantly for infants greater than 1000 g. Candidemia rates for infants of birth weights 1001-1500 g is 1.32%, for 1501-2500 g is 0.36%, and for those greater than 2501 g is 0.29%. The incidence increases in an inverse linear pattern when examining rates by gestational age. The rate of invasive Candida infections increases from around 3% at 28 weeks’ gestation to 24% at 23 weeks’ gestation (see Fig. 49.3 ).
Since 2001, several epidemiologic studies have tracked the incidence of Candida blood and cerebrospinal infections and have correlated a decrease in these infections with an increased use of fluconazole prophylaxis, decrease in broad spectrum antibiotics, and initiation of CLASBI prevention interventions.
Complex gastrointestinal diseases can be complicated by invasive Candida infections. Feja et al. found that candidemia was increased in patients with gastrointestinal pathology (OR = 4.57; 95% CI = 1.62-12.92). Gastrointestinal pathology was defined in their study as tracheoesophageal fistula, gastroschisis, omphalocele, Hirschsprung disease, intestinal atresia, or episodes of NEC. The incidence of candidemia has been reported between 10 and 16.5% in patients with NEC. Peritoneal cultures isolating Candida species have been found in ~50% of focal bowel perforations and 15% of surgical NEC cases. In a study of the pathology from 66 surgical NEC cases, 7.5% had intestinal candidiasis with C. albicans .
Congenital heart disease is an emerging patient population that may develop invasive Candida infections. Candidemia accounts for 9% of BSIs in patients with congenital heart disease cared for in NICUs in the first 4 months of life. Candida BSI occurred at a rate of 6.3 per 1000 admissions and had an overall mortality rate of 21%.
Candida species are the second most common cause of infection in neonates on extracorporeal membrane oxygenation (ECMO), responsible for 10% of infections. Compared with the first 7 days on ECMO, Candida infections are twofold and fourfold more common between 8 and 14 days and more than 14 days on ECMO, respectively.
For preterm infants, invasive Candida infections are associated with neurodevelopmental impairment and high mortality.
Invasive Candida infections may lead to significant neurodevelopmental impairment even in the absence of documented fungal meningitis. Neurodevelopmental impairment in most studies is the presence of one or more of the following: low mental and motor development using Bayley Scales of Infant Development II, cerebral palsy, deafness, and/or blindness. Using these criteria, the incidence of neurodevelopmental impairment is 57% with candidemia versus 29% in noninfected ELBW infants. The incidence is similar for Candida meningitis as well as with candiduria. In another study demonstrating similar neurodevelopment impairment of 59% of Candida -infected patients, earlier treatment within 2.1 ± 1.3 days of the blood culture was associated with normal or mildly impaired cases compared with later antifungal therapy (5.1 ± 3.0 days after blood culture was drawn), which was associated with severe disability or death ( p < .0001).
Identifying Candida -related mortality is difficult to determine and in the literature may include attributable mortality, death within a specified number of days from onset of infection (3, 7, 14, or 30 days) or review of each death using predetermined definitions. Many studies simply report overall mortality. In infants less than 1000 g with invasive Candida infections, these rates range from an attributable mortality of 13% to 20% to overall mortality rates of 23% to 66% when examining overall mortality rates of patients in the placebo or control groups in the prophylaxis studies. Mortality increases at lower gestational ages, and variation of mortality rates by NICU, similar to infection rates, is influenced by admission rates of these extremely preterm infants. Additionally, a standardized approach with prompt treatment with appropriate drug dosing and line removal with BSIs decreases Candida -related mortality rates.
Mortality is high with all types of invasive Candida infections in ELBW infants. In a multicenter epidemiologic study of patients less than 1000 grams, all-cause mortality rates were 28% for Candida BSI, 26% for Candida UTI, 50% for other sterile sites (meningitis and peritonitis), and if two or more culture sites were involved (BSI + UTI or UTI + meningitis), the mortality rate was 57% (see Fig. 49.5 ). In this study, the overall mortality was 34% for infants with invasive Candida infections compared with 14% without infections.
There is a marked difference in overall and attributable mortality between infants less than 1000 grams and larger and more mature infants with invasive Candida infections. In infants less than 1000 g with infections, all-cause mortality rate was 26% compared with 13% in infants without candidiasis. For infants greater than 1000 g with invasive Candida infections versus those without, mortality was 2% compared with 0.4%.
The Candida species causing the infection also affects survival. C. albicans is more virulent than nonalbicans species. In infants less than 1000 grams, Candida -associated mortality was 43% in infants with C. albicans candidemia compared with 19% with C. parapsilosis sepsis.
The financial costs of invasive fungal infections are high. Two case control studies have examined the effect of these infections on cost of hospitalization and length of hospital stay. They are limited, because they were based on ICD-9 codes, which may not have captured all invasive Candida infections. The mean increase in hospital costs was $39,045 for infants less than 1000 g with no difference in length of stay, and for infants ≥1000 g, there was an increase of $122,302 with an additional length of stay of 16 days.
As neonatal candidiasis can be fatal among premature infants, an understanding of the risk factors that lead to infection is critical ( Table 49.1 ). Conditions that facilitate fungal colonization and proliferation increase the risk for invasive infection and include gastrointestinal dysmotility, ileus, antibiotics, acid inhibition, steroids, and the immaturity of the patient's immune system. Several risk factors have been identified, with gestational age having the strongest effect. This correlates with the degree of underdevelopment of the immune system and immaturity of the skin and gastrointestinal and respiratory tracts in these infants.
| Immunity | Medications That Facilitate Fungal Growth | Medications That Suppress Immune Defense | Diseases | Conditions |
|---|---|---|---|---|
| Undeveloped immune system Immunosuppression Neutropenia |
Cephalosporins Carbapenems Postnatal steroids |
Acid suppression (H 2 antagonists and proton pump inhibitors) Postnatal steroids |
Necrotizing enterocolitis Focal bowel perforation Complicated gastrointestinal diseases (e.g., gastroschisis) Prior blood stream infection |
Extreme prematurity Maternal vaginal colonization Vaginal delivery Male Central venous catheter Intubated Lack of enteral feedings Hyperglycemia |
The extremely preterm infant has the unique combination of being immune compromised, requiring prolonged intensive care (parenteral nutrition, mechanical ventilation/endotracheal tube, central venous access), being exposed to medications that promote fungal growth (H 2 blocking agents, proton pump inhibiting agents, postnatal corticosteroids, and broad-spectrum antibiotics), and being predisposed to gastrointestinal dysmicrobism, dysmotility, and disease (NEC and focal bowel perforation). Infants, both term and preterm, with complicated gastrointestinal diseases such as gastroschisis or NEC also have multiple risk factors of prolonged ileus, central venous access, parenteral nutrition, surgery, and exposure to prolonged and/or broad-spectrum antibiotics.
Medications such as broad-spectrum antibiotics and third- and fourth-generation cephalosporin and carbapenem antibiotics and the lack of feeding are associated with increased risk for candidemia. It is hypothesized that these medications accumulate in high concentrations in bile and alter the balanced gastrointestinal microflora, eradicating competitive gram-negative and anaerobic bacteria, thus facilitating proliferation and dissemination of fungal organisms. Other classes of drugs found to be risk factors for neonatal fungal infection are H 2 antagonists and postnatal corticosteroids. In examining risk factors present at 3 days after birth in infants less than 1000 g, lack of enteral feedings and third-generation cephalosporin use increased risk for infecion. For infants not feeding by day of life 3, 8.7% developed Candida BSI or meningitis, compared with 3.4% of those receiving enteral feedings by day of life 3.
A few multivariate analyses have examined risk factors, finding that central venous catheter use, previous bacterial BSIs, broad spectrum antibiotics, presence of an endotracheal tube, and gastrointestinal pathology were significantly associated with candidemia. Gastrointestinal pathology was defined in the study as tracheoesophageal fistula, gastroschisis, omphalocele, Hirschsprung disease, intestinal atresia, or episodes of NEC.
The microflora of the oral mucosa and gastrointestinal tract play an important role in fungal colonization and infection. A normal bacterial flora inhibits Candida growth by competing for both adhesion sites and nutrients as well as various anticandidal actions. The commensal gut microflora can also enhance immunomodulatory activities against the most common pathogens, including Candida spp. This may explain, in part, the protective role of enteral feeding in the absence of antibiotics. In athymic compared to euthymic adult neonatal mice exposed to Candida spp. , probiotics ( Lactobacillus acidophilus , L. reuteri , L.casei GG , or Bifidobacterium animalis ) decreased the incidence of systemic candidiasis and were associated with prolonged survival. Probiotic bacteria also affected antibody- and cell-mediated immune responses to C. albicans . This highlights the fact that the intestinal microflora may be as important as a mature immune system in preventing fungal colonization and infection.
There are some differences in risk factors for infection by species. Risk factors for C. albicans and C. parapsilosis include exposure to third-generation cephalosporins, central vascular catheters, parental nutrition and lipid emulsions, and high acuity while, additionally, vaginal delivery increases risk for C. albicans and H 2 antagonists for C. parapsilosis infection. Risk for C. glabrata infection is increased with gastrointestinal disease, exposure to fluconazole or antibiotics, prolonged hospitalization, and prior infection with other fungi. C. tropicalis infections are associated with gastrointestinal mucosal injury, antibiotic suppression of bacterial flora, neutropenia, and parenteral nutrition .
Colonization is the key step in the pathway that leads from exposure to disseminated infection. Colonization may occur via vertical (maternal) or horizontal (nosocomial) transmission. Most fungal colonization occurs by 2-3 weeks of life. Fungal colonization of the skin and respiratory or gastrointestinal tract occurs in 10% of full-term infants compared with 26.7%-62.5% of very low birth weight (VLBW, <1500 grams at birth) infants in the first weeks of life. Fungal colonization and subsequent infection depend on exposure, size of inoculum, host susceptibility, and properties of the pathogen (see Fig. 49.2 ).
Vertical transmission leading to colonization is common and has been confirmed in studies using genotyping techniques. Candida yeast cells adhere preferentially to intermediate layers of the vaginal tract that are increased during pregnancy, increasing maternal fungal colonization and exposure of vaginally delivered infants. Maternal risk factors include gestational diabetes and need for steroid treatment outside of antenatal steroids. The incidence of vaginal fungal colonization during pregnancy has been reported to be between 25% and 50%, with up to 90% due to C. albicans and the remainder predominantly C. glabrata and C. tropicalis . Despite the frequent fungal colonization, chorioamnionitis caused by Candida occurs less frequently, with almost all vertical transmission occurring when the infant passes through the birth canal via mucocutaneous contact, swallowing, or aspiration of fungi. Treatment of maternal Candida vaginosis and UTIs during pregnancy may decrease the inoculum the infant is exposed to and potentially prevent vertical transmission. However, maternal exposure during pregnancy to repeated courses of antifungal products (mainly topical azoles) may alter yeast susceptibility and if maternal symptoms persist, follow-up cultures to confirm clearance and/or identify resistance should be performed.
Candida colonization may also be acquired horizontally, primarily from the hands of health care workers. In a multicenter trial examining fungal colonization in six NICUs, Candida species were isolated on the hands of 29% of health care workers. Whereas C. albicans was the more common fungal isolate in all NICU patients (14% versus 7%), C. parapsilosis was the most common species isolated from the hands of NICU staff. C. parapsilosis was isolated from 19% and C. albicans from 5% of the cultures from health care personnel ( p < .001). C. lusitaniae (2%), C. guilliermondii (1%), C. tropicalis (<1%), and C. glabrata (<1%) were also recovered from hand cultures. Increased handling required by sicker preterm infants increases the risk of acquiring fungal colonization, because there is an average of 32 direct infant touches during a 12-hour shift.
Candida species causing invasive infection rarely colonize environmental sources. Despite the tendency of fungus to grow in moist environments, studies have not isolated fungi from incubators, wash basins, water faucets, chest tubes, intravenous drug pumps, ventilators, inanimate objects, or radiant warmers.
Risk factors for Candida colonization and sepsis are similar, as adhesion and colonization of the skin, mucosal membranes, and/or vascular catheters occur prior to most infections. Colonization is inversely proportional to gestational age and birth weight. Both C. albicans and C. parapsilosis colonize multiple sites in the majority of infants, whereas colonization at three or more sites occurs more frequently with C. albicans .
Biofilm formation on catheters inhibits the host's defense mechanisms and the penetration of antifungal agents. Elements of catheter care related to sterile placement; hub and dressing care; sterile preparation of parenteral nutrition, intravenous fluids, and medications; and line changes at the bedside are critical infection control practices. Infusates may also become contaminated and directly seed the bloodstream. In vitro growth curves demonstrate that Candida species have a selective growth advantage compared with bacteria in parenteral nutrition fluid.
Candida colonization characteristics aid in identifying which colonized infants have the highest odds to progress toward invasive Candida infections ( Table 49.2 ). Although skin and gastrointestinal colonization are more common and precede respiratory tract colonization, endotracheal colonization has a higher risk for infection. The highest risks for progression are from colonization of indwelling devices (endotracheal tubes and central catheters) and when more than one site is colonized. Rowen et al. demonstrated that with endotracheal fungal colonization, candidemia was 15.4 times more likely to occur compared with infants without any fungal colonization. Controlling for fungal colonization at other sites, endotracheal colonization alone increased the risk for fungal sepsis nearly six-fold.
| Odds Ratio | Reference | |
|---|---|---|
| Endotracheal tube | 15.4 | Rowen et al., 1994 |
| Respiratory tract colonization | 35.5 | Kaufman et al., 2001 |
| Colonization in multiple sites | 6.2 (3 or more) 3.0 (for each additional site) |
Manzoni et al., 2006 Kaufman et al., 2001 |
| Urine | 11.6 | Manzoni et al., 2006 |
| Colonization of central venous catheter | 10.8 | Manzoni et al., 2006 |
Cultures of blood, urine, cerebrospinal, and peritoneal fluid or other sterile body fluids remain the best method for diagnosing invasive Candida infections ( Fig. 49.6 ). Focus on obtaining sufficient blood culture volumes (≥1 mL) and performing urine and cerebrospinal fluid cultures at the time of evaluation for sepsis remains critical to making prompt diagnoses. One study of neonates demonstrated that isolation of fungus in blood cultures occurred at 37 ± 14 hours, and 97% of blood cultures were positive by 72 hours. Laboratory capabilities to culture fungi, identify Candida species, and perform susceptibility testing are critical to diagnosis and management.
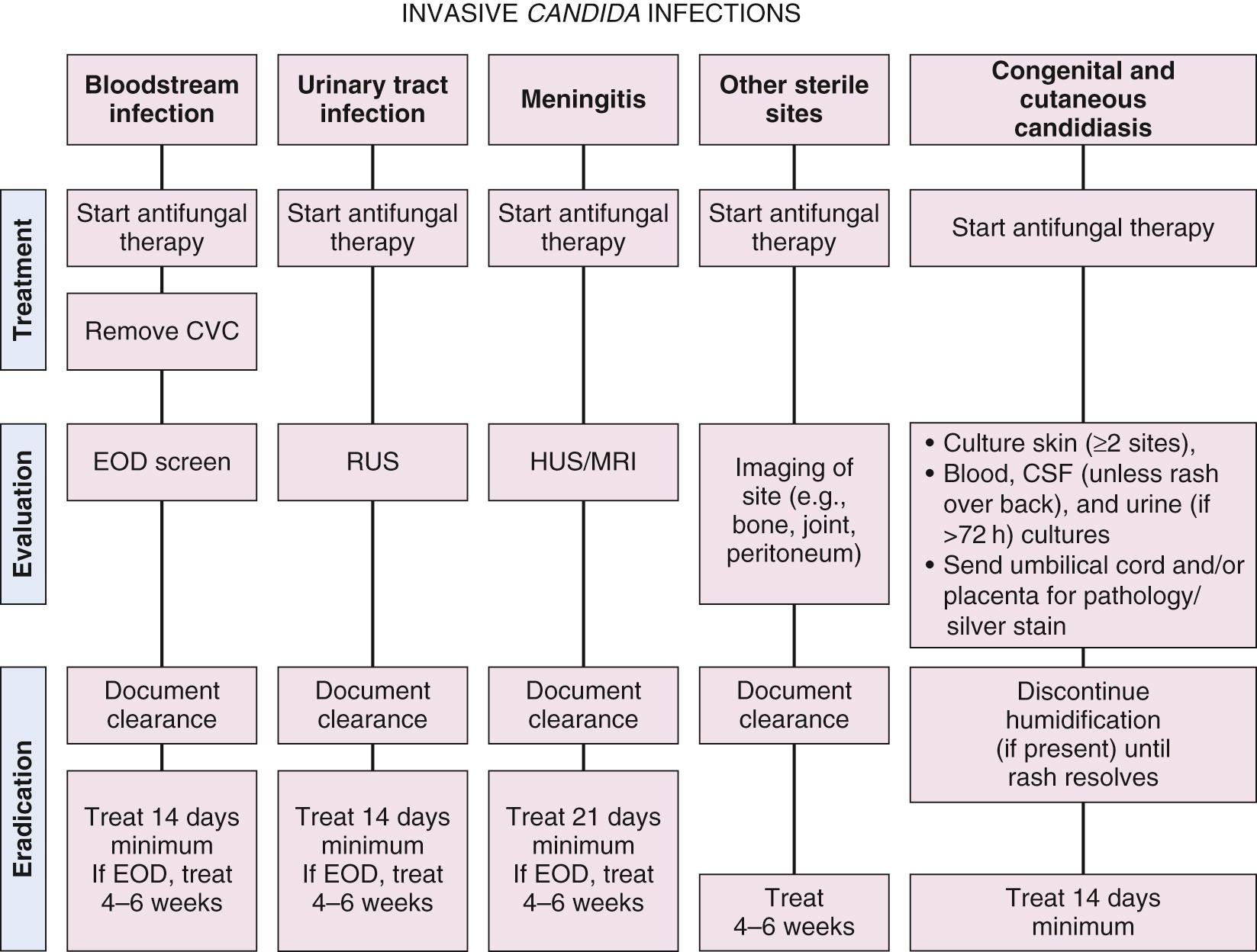
Most commonly, with bloodstream infections, an initial end-organ dissemination (EOD) screen including an echocardiogram, liver and renal ultrasound, cranial ultrasound, and ophthalmologic examination should be performed. This could be performed at presentation or after 5-7 days of antifungal therapy. If there is significant bowel pathology such as NEC or focal bowel perforation, a complete abdominal ultrasound should be performed to rule out peritoneal, liver, or splenic involvement. If signs and symptoms of septic arthritis (joint swelling) or osteomyelitis (swelling or immobility) are present, a clinical diagnosis can be made, and evaluation with joint aspiration, bone scan, or magnetic resonance image (MRI) may help define the extent of involvement but should not be used to rule out joint or bone involvement in neonates. If candidemia persists, end-organ dissemination is even more likely, and surveillance after 5-7 days should also include: (1) ultrasound at location of the tip of any previous or current central catheters for infected thrombus, (2) complete abdominal ultrasound for abscesses (laparotomy is sometimes considered if high clinical suspicion), and (3) cranial ultrasound/MRI to detect brain dissemination. Abscesses that are amenable to drainage should be drained.
Laboratory adjunctive tests have not been able to replace cultures but can be useful in identifying high-risk patients who would benefit from early empiric antifungal therapy (pending culture results) and to follow response to antifungal therapy. Polymerase chain reaction (PCR) and fungal cell wall polysaccharides such as (1-3)-beta-D-glucan (BDG) and mannan can be extremely helpful in high-risk patients in determining the need for early empiric antifungal therapy while cultures are pending, detecting infections when there is a high suspicion for infection that is not in the bloodstream (e.g., urine, peritoneal cavity, abscesses), and determining if length of treatment needs to be extended.
Molecular techniques to identify fungi and their antifungal susceptibilities more rapidly and with higher sensitivity than with blood cultures are being studied. Examples include polymerase chain reaction (PCR) and DNA microarray technology. One challenge is that these tests are mainly compared only with blood culture results, but often detect infection or only colonization at other sites. Fungal PCR to detect the gene for 18S ribosomal RNA in VLBW infants has yielded promising results but requires additional study. Results of PCR tests showed a broader number of infections, because they not only detect patients with candidemia but are also positive in those with Candida peritonitis, candiduria, previous candidal infections, and endotracheal colonization.
BDG levels are helpful if there is uncertainty in deciding need for empiric antifungal therapy and in following response to therapy as levels decrease over time with antifungal therapy. Various cut-off points have been recommended for interpreting BDG levels in neonates. A recent neonatal study of BDG found higher levels in infants with ICI (364 pg/mL vs. 89 pg/mL in non-infected neonates), and levels decreased significantly with antifungal therapy to 58 pg/mL (28-81). They suggested the cut-off for BDG be higher (>125 pg/mL) for neonates than adults (>80 pg/mL) due to the effect colonization and other infections (Gram-negative and coagulase-negative Staphylococcus [CoNS]) can have on BDG levels. The BDG levels in infants infected with CoNS were 116 pg/mL (46-128) and 118 pg/mL (52-304) in patients without bacteremia. One challenge is that BDG can be elevated to the same degree with fungal colonization as with ICI, and studies have not critically examined this effect. As several studies have demonstrated there are high-risk sites that when colonized (e.g., the respiratory tract) may benefit from empiric treatment. One study used endotracheal lavage aspirates with mannan levels ≥0.5 ng/mL to decide on preemptive treatment and significantly decreased ICI. Further study of preemptive treatment at certain BDG levels may be beneficial.
BDG may also give false-positive results following transfusion of blood products in adults and neonates. A study of 133 VLBWs found BDG to be higher in transfused (red blood cells or fresh frozen plasma) neonates (170 pg/mL, 65-317) compared to nontransfused infants (57 pg/mL, 34-108; p < 0.001).
Another method that may help with the decision to start early empiric therapy is direct fluorescent assay in buffy coat. This test is a fluorescent stain that binds to structures containing cellulose and chitin. This diagnostic test has been successfully used for identifying hyphae and spores, and results are obtained after only 1-2 hours. Other markers of fungal disease being studied include anti- Candida antibodies, D-arabinitol (candidal metabolite), and fungal chitin synthase. These markers have some of the same challenges, because they may be present with bloodstream infections, nonbloodstream infections, previous infections, or with colonization alone.
Congenital cutaneous candidiasis (CCC) ( Table 49.3 , Fig. 49.7 ) presents most commonly at birth but can occur within the first week. Dermatologic findings include desquamating maculopapular, papulopustular, and/or erythematous rashes. The most common finding from a recent study included desquamation alone (scaling, peeling, flaking, or exfoliation) or with other rash presentations. CCC usually occurs only as a rash but dissemination such as pneumonia or bloodstream infection may also be present. Without prompt identification and systemic treatment, dissemination to the blood, urine, or CSF can occur in infants with CCC ranging from 11% in term infants to 33% in infants 1000-2500 grams and highest at 66% in infants <1000 grams. A study examining the pathology and pathogenesis of CCC demonstrate a high burden of yeast with invasion into the epidermis and dermis with inflammation and injury including granulomas, focal necrosis, and hemorrhage. These data from biopsies give insight into the invasive nature of the cutaneous involvement. For these reasons, preterm and term infants should be treated promptly at the time of rash presentation with systemic antifungal therapy and for a minimum of 14 days similarly to other invasive fungal infections. Delaying systemic treatment, solitary use of topical therapy (nystatin), and treating for <10 days is associated with Candida dissemination to the bloodstream.
| Presentation | Extensive Candida Skin Rash |
|
|
| Affected Areas | Skin |
|
|
| Umbilical Cord and Placenta | |
|
|
| Skin Rash |
|
| Timing | Congenital Cutaneous Candidiasis |
|
|
| Cutaneous Candidiasis | |
|
|
| Evaluation | Congenital Cutaneous Candidiasis |
|
|
| Cutaneous Candidiasis | |
|
|
| Diagnosis | Skin Findings and 1 or More of the Following: |
|
|
| Treatment |
|
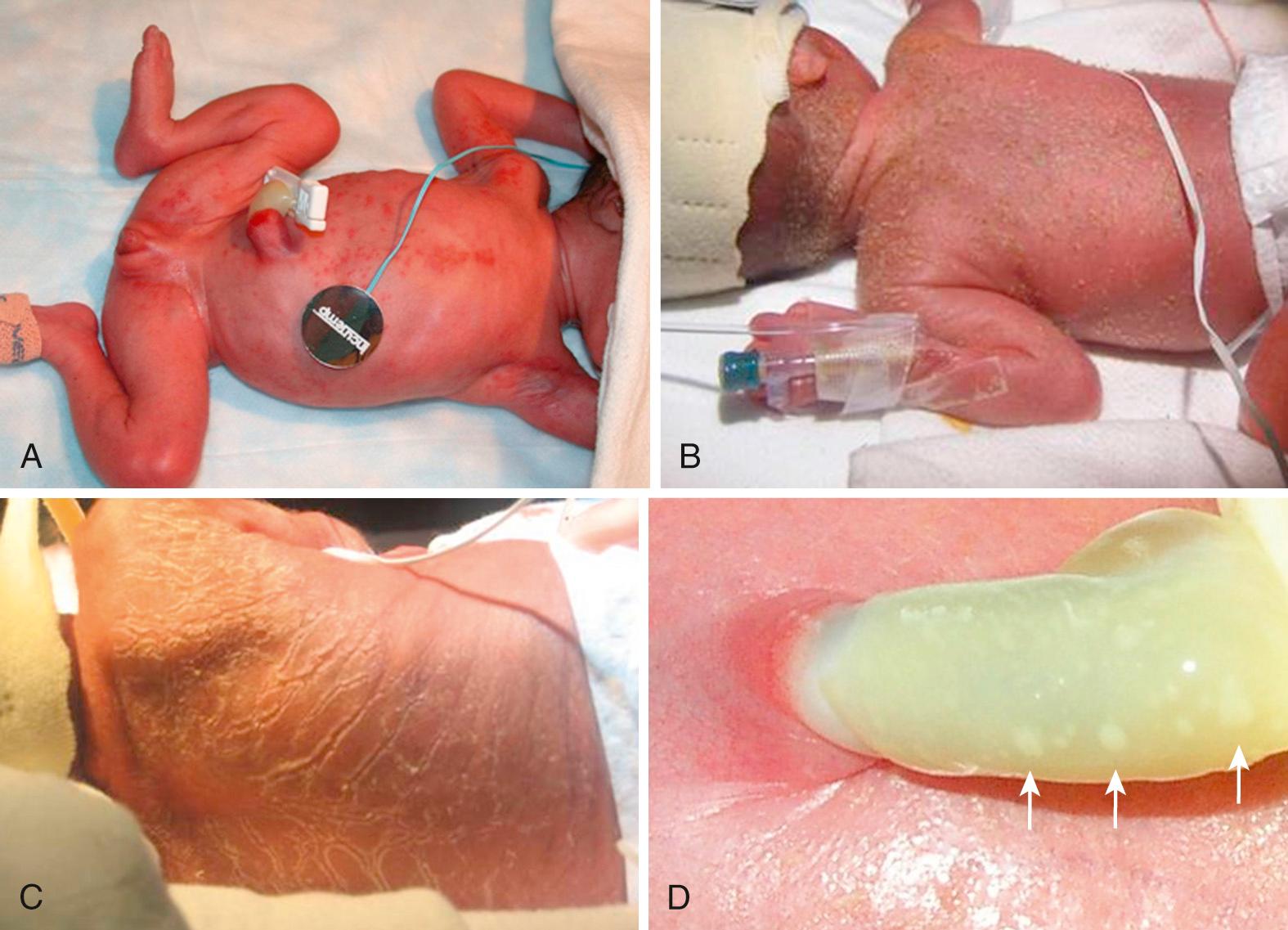
By culturing for both fungal and bacterial organisms by performing aerobic skin cultures of the rash, the source of infection can be confirmed in a timely fashion, but empiric therapy should be administered at time of rash presentation. Differential diagnosis includes staphylococcal as well as other bacterial and fungal skin infections. Pathology with fungal staining of the umbilical cord and placenta can also aid in diagnosis.
Cutaneous candidiasis, referred to in past literature as mucocutaneous infection, presents with similar skin manifestations to CCC but occurs later than CCC. In the era prior to antifungal prophylaxis, the incidence was reported to be as high as 8% in VLBW infants. Risk factors include extreme prematurity, vaginal birth, postnatal steroids, and hyperglycemia. The importance of candida-like dermatitis often goes unrecognized prior to dissemination to the blood. Cutaneous candidiasis, similar to CCC, is an invasive infection of the skin; empiric therapy should be started at the time of rash presentation and needs treatment for a minimum of 14 days in preterm infants.
Clinical signs and symptoms of candidemia are similar to bacteremia ( Table 49.4 ). Most importantly, candidemia can be associated with disseminated disease (see End-Organ Dissemination ). Evaluation of cardiac, liver, renal, ophthalmologic, and central nervous systems is warranted and discussed in the following.
Common (>50%)
|
Frequent (~33%)
|
Less Common (10–15%)
|
Can Occur (3%)
|
Candida infection of the kidney may occur because of an ascending urinary tract infection or via hematogenous spread. Studies demonstrate that Candida UTIs without bloodstream involvement are more common than with a BSI, suggesting that the ascending route via the urinary tract is more common than hematogenous spread. Candida can attach to human uroepithelial cells. Colonization and proliferation is augmented by proteinuria, nitrogenous compounds, acidic pH, hydrophobic Candida cells, and the presence of Enterobacteriaceae such as Escherichia coli . Signal-activated phospholipases (SAPs) and phospholipases play a role in the renal system as well, attacking structural and immunologic defenses leading to tissue injury and invasion. Risk factors for candiduria include vaginal birth, lower birth weight and gestational age, male gender, and prolonged antibiotic therapy.
Evaluation of late-onset sepsis should include a urine culture obtained via sterile catheterization or suprapubic bladder aspiration. Infection is defined as growth of 10,000 or more colony-forming units (CFU)/mL from a sterile catheterization or 1000 or more CFU/mL for bladder aspiration. Many neonatal studies have used lower CFUs as well. Lower CFUs, while likely not infection, indicate colonization at a high-risk site in which a follow-up urine culture should be obtained and empiric antifungal therapy started pending culture results. Preemptive treatment for 14 days would be appropriate as well for lower CFUs. Additionally, empiric antifungal therapy should be used with subsequent sepsis evaluations.
Signs and symptoms of urinary tract infections can be similar to sepsis and near-term and term infants may also present with fever. Additionally, an elevated creatinine value in the absence of other pathology should prompt evaluation for a urinary tract infection. If the urine culture is positive for fungus, renal ultrasonography is needed to evaluate for abscess formation. In the absence of antifungal prophylaxis, candiduria occurs in approximately 2% of VLBW infants and up to 6% of ELBW infants. Mortality is similar in infants with Candida UTI alone (26%) compared with Candida BSIs (28%) in ELBWs and older infants and studies showing increased neurodevelopmental impairment as well. These findings emphasize the need for prompt treatment for a minimum duration of 14 days.
Renal abscess formation may occur by dissemination of candidemia or as an ascending infection with candiduria. Two studies of candiduria from 1982-1993 and 2001-2003 found that renal abscesses developed in 42% (36 screened) and 12% (26 screened) of infants with candiduria (±candidemia), while a more recent study during 2004-2007 found the incidence to be 0% (0 of 23 evaluated) with candiduria alone and 22% (2 of 9) with concordant Candida infection. One study found ultrasound examinations were normal in one-third of patients who later developed renal abscesses 8-39 days later, which may have been related to antifungal dosing used or delay in treatment. Between these two study periods, antifungal prophylaxis, pharmacokinetic and safety studies of appropriate initial antifungal dosing (e.g., amphotericin B deoxycholate at 1 mg/kg compared with 0.25 mg/kg in the previous era), and increased empiric antifungal therapy may be attenuating the formation of renal abscesses. These studies have small numbers but suggest that in infants with candiduria, prompt initial antifungal therapy has improved outcomes, and renal imaging should be performed at presentation and repeated in cases with persistent candiduria.
Central nervous system fungal infection may involve meningitis, ventriculitis, or abscess formation in infants. Studies have found around 50% of meningitis cases occur in the absence of candidemia, pointing to the importance of CSF evaluation when performing sepsis evaluations. Culture of the CSF is important in diagnosing fungal meningitis prior to the initiation of antifungal therapy, because CSF cell counts and chemistries often are not abnormal. This may be because of the number of organisms in the CSF or difficulty of interpretation caused by blood contaminating the lumbar puncture, the location of the central nervous system infection (brain tissue versus spinal fluid), and host response of the preterm or term infant.
Fungal abscesses of the central nervous system have been reported to be microscopic and not readily detectable by ultrasonography. In a study of 46 ELBW patients with fungal sepsis and/or meningoencephalitis, only 6 of 13 cases with fungal central nervous system abscesses (detected by neuroimaging or on autopsy) had abnormal results on lumbar puncture. Studies have found an association between invasive fungal infection and periventricular leukomalacia in preterm infants, possibly related to release of cytokines, which may damage the periventricular white matter. These findings and animal studies demonstrate the need for central nervous system imaging (ultrasonography or MRI) in all patients with fungal sepsis regardless of the results of CSF studies in addition to meningitis cases.
In patients presenting with NEC or focal bowel perforation, invasive Candida infections can complicate these gastrointestinal diseases. Peritonitis with bowel perforation owing to NEC or focal bowel perforation often presents with abdominal distention with or without erythema. Candida species are the predominant organism causing peritonitis in 15% of the perforated NEC cases and 44% of focal bowel perforation patients. Radiographs help confirm perforation, but at times ultrasound or an exploratory laparotomy may be needed if clinically indicated. Persistent leukopenia, severe thrombocytopenia, hypotension, or low urine output (<1 mL/kg/hr) after 48 hours of treatment even if radiographs and ultrasound are negative should also prompt consideration for an exploratory laparotomy to evaluation for perforation(s) and/or abscesses. When exploratory laparotomy is performed or drains are placed, cultures should be obtained to determine organisms associated with bowel perforation, peritonitis, and potential abscesses.
Necrotizing enterocolitis can be complicated by fungal infections, and associated bloodstream infections occur in up to 16.5% cases of NEC. Screening the gastrointestinal tract for the presence of Candida species (by culture of rectum, stool, or oral flora) should prompt the addition of a systemic antifungal to the antimicrobials selected as part of the treatment of NEC. If Candida is not detected on screening, antifungal prophylaxis should be considered while treating NEC until full enteral feedings are reached.
The diagnosis of pneumonia remains difficult in ventilated preterm and term infants with chronic lung disease, as atelectasis, fluid, scar tissue, and infection have similar radiologic findings. Candida colonization of the respiratory tract occurs after colonization of the skin and gastrointestinal tract. Respiratory colonization is a high-risk site for invasive Candida infections (see Table 49.2 ). Pre-emptive treatment when Candida species are isolated in the lungs by culture, polymerase chain reaction ( PCR), or Candida mannan antigen have been shown to prevent dissemination.
At the time fungal infection is clinically apparent, the organisms have often disseminated from the blood, urine, or CSF to adhere and proliferate in body fluids, tissues, and organs. Candida species can cause endocarditis, endophthalmitis, dermatitis, peritonitis, osteomyelitis, and septic arthritis, and fungal abscesses may form in the central nervous system, kidneys, liver, spleen, skin, bowel, and peritoneum (see Fig. 49.2 ). Fungal end-organ dissemination has been noted since the earliest reports of Candida sepsis in neonates, being as high as 66% in the 1970s, reflecting prolonged periods of fungemia, uncertainty of significance of positive fungal cultures, underdosing of antifungals, and poor diagnostics, as many infections were diagnosed late or at the time of autopsy. A meta-analysis of studies reporting fungal end-organ dissemination in neonates spanning various practices from 1979-2002 found median prevalence of cardiac vegetations or thrombi was 5%, endophthalmitis 3%, renal involvement 5%, and central nervous system abscesses 4%. Dissemination may be higher in the more extremely preterm infant.
Persistent candidemia of 5-7 days was associated with increased end-organ dissemination, as each day of candidemia increases the risk by 10%. When amphotericin B was administered and central vascular catheters removed within 2 days of the first positive blood culture, outcomes such as end-organ dissemination and mortality were decreased.
Candida endocarditis or infected vascular thrombi have been reported in 5.5%-15.2% of cases of fungal sepsis, with equal prevalence for C. albicans and C. parapsilosis . Fungal endocarditis may be associated with higher mortality than fungemia alone. Central vascular catheters place neonates at increased risk. Catheters can cause local trauma to valvular, endocardial, or endothelial tissue, creating a nidus for thrombus and infection at insertion and while in situ. When antifungal therapy alone is unsuccessful in resolution of the endocarditis or thrombus, thrombolytic or anticoagulation therapy may be indicated in some cases, depending on infant's gestational age and accompanying conditions.
Endophthalmitis represents intraocular dissemination from the bloodstream. Exogenous infection can also occur secondary to retinopathy of prematurity (ROP) surgery or trauma. Endophthalmitis begins as a chorioretinal lesion that gradually elevates and breaks free in the vitreous, appearing as a white fluffy ball. The infection appears as solitary or multiple yellow-white elevated lesions with indistinct borders that are most often seen in the posterior retina and vitreous. The clear cell-free vitreous becomes hazy, owing to an influx of inflammatory cells. This vitreous reaction is more difficult to recognize in preterm infants because of the vitreous haze that is present in the first weeks of life. The most immature infants appear to be at highest risk for fungal endophthalmitis. The incidence of retinal endophthalmitis with candidemia has decreased over the years, likely because of more rapid diagnosis, treatment, and prevention of invasive Candida infections. Incidence may be affected by timing of screening, as antifungal treatment effectively treats this infection. The incidence has decreased from 6% to 0.8% of preterm infants with candidemia who had indirect ophthalmoscopic examination.
Even in the absence of visible retinal abscesses or chorioretinitis, there is some epidemiologic evidence that Candida sepsis may predispose preterm infants to severe ROP. Noyola and colleagues, in a case control study of VLBW infants less than 28 weeks’ gestation, found that 52% (24 of 46) of infants with candidemia developed threshold ROP compared with 24% (11 of 46) of controls without candidemia ( p = .008). Although this has been shown only as an association between fungal sepsis and ROP, early and frequent screening for retinal pathology is recommended in preterm infants with candidemia.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here