Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
In the minds of many students, the discipline of physiology is linked inextricably to images from its past. This prejudice is not surprising because many experiments from physiology's proud history, such as those of Pavlov on his dogs, have transcended mere scientific renown and entered the realm of popular culture. Some might believe that the science of physiology devotes itself exclusively to the study of whole animals and is therefore an antique relic in this era of molecular reductionism. Nothing could be further from the truth. Physiology is and always has been the study of the homeostatic mechanisms that allow an organism to persist despite the ever-changing pressures imposed by a hostile environment. These mechanisms can be appreciated at many different levels of resolution.
Certainly it would be difficult to understand how the body operates unless one appreciates the functions of its organs and the communication between these organs that allows them to influence one another's behaviors. It would also be difficult to understand how an organ performs its particular tasks unless one is familiar with the properties of its constituent cells and molecules.
The modern treatment of physiology that is presented in this textbook is as much about the interactions of molecules in cells as it is about the interactions of organs in organisms. It is necessary, therefore, at the outset to discuss the structure and characteristics of the cell. Our discussion focuses first on the architectural and dynamic features of a generic cell. We then examine how this generic cell can be adapted to serve in diverse physiological capacities. Through adaptations at the cellular level, organs acquire the machinery necessary to perform their individual metabolic tasks.
The chemical composition of the cell interior is very different from that of its surroundings. This observation applies equally to unicellular paramecia that swim freely in a freshwater pond and to neurons that are densely packed in the cerebral cortex of the human brain. The biochemical processes involved in cell function require the maintenance of a precisely regulated intracellular environment. The cytoplasm is an extraordinarily complex solution, the constituents of which include myriad proteins, nucleic acids, nucleotides, and sugars that the cell synthesizes or accumulates at great metabolic cost. The cell also expends tremendous energy to regulate the intracellular concentrations of numerous ions. If there were no barrier surrounding the cell to prevent exchange between the intracellular and extracellular spaces, all of the cytoplasm's hard-won compositional uniqueness would be lost by diffusion in a few seconds.
The requisite barrier is provided by the plasma membrane, which forms the cell's outer skin. The plasma membrane is impermeable to large molecules such as proteins and nucleic acids, thus ensuring their retention within the cytosol. It is selectively permeable to small molecules such as ions and metabolites. However, the metabolic requirements of the cell demand a plasma membrane that is much more sophisticated than a simple passive barrier that allows various substances to leak through at different rates. Frequently, the concentration of a nutrient in the extracellular fluid (ECF) is several orders of magnitude lower than that required inside the cell. If the cell wishes to use such a substance, therefore, it must be able to accumulate it against a concentration gradient. A simple pore in the membrane cannot concentrate anything; it can only modulate the rate at which a gradient dissipates. To accomplish the more sophisticated feat of creating a concentration gradient, the membrane must be endowed with special machinery that uses metabolic energy to drive the uphill movements of substances— active transport —into or out of the cell. In addition, it would be useful to rapidly modulate the permeability properties of the plasma membrane in response to various metabolic stimuli. Active transport and the ability to control passive permeabilities underlie a wide range of physiological processes, from the electrical excitability of neurons to the resorptive and secretory functions of the kidney. In Chapter 5 , we will explore how cells actively transport solutes across the plasma membrane. The mechanisms through which the plasma membrane's dynamic selectivity is achieved, modified, and regulated are discussed briefly below in this chapter and in greater detail in Chapter 7 .
Our understanding of biological membrane structure is based on studies of red blood cells, or erythrocytes, that were conducted in the early part of the 20th century. The erythrocyte lacks the nucleus and other complicated intracellular structures that are characteristic of most animal cells. It consists of a plasma membrane surrounding a cytoplasm that is rich in hemoglobin. It is possible to break open erythrocytes and release their cytoplasmic contents. The plasma membranes can then be recovered by centrifugation to provide a remarkably pure preparation of cell surface membrane. Biochemical analysis reveals that this membrane is composed of two principal constituents: lipid and protein.
Most of the lipid associated with erythrocyte plasma membranes belongs to the molecular family of phospholipids. In general, phospholipids share a glycerol backbone, two hydroxyl groups of which are esterified to various fatty-acid or acyl groups ( Fig. 2-1 A ). These acyl groups may have different numbers of carbon atoms and also may have double bonds between carbons. For glycerol-based phospholipids, the third glycerolic hydroxyl group is esterified to a phosphate group, which is in turn esterified to a small molecule referred to as a head group. The identity of the head group determines the name as well as many of the properties of the individual phospholipids. For instance, glycerol-based phospholipids that bear an ethanolamine molecule in the head group position are categorized as phosphatidylethanolamines (see Fig. 2-1 A ).
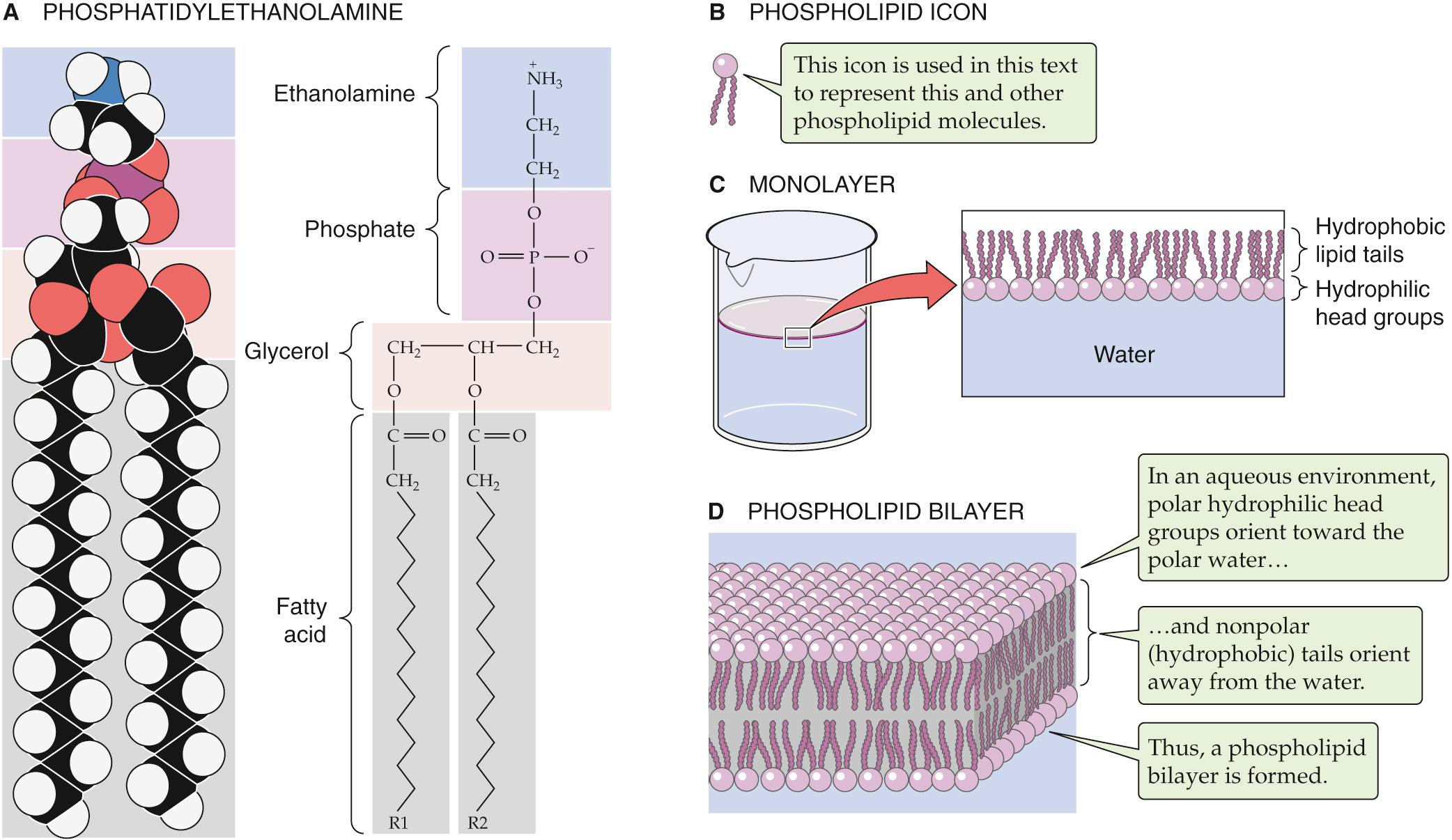
The unique structure and physical chemistry of each phospholipid (see Fig. 2-1 B ) underlie the formation of biological membranes and explain many of their most important properties. Fatty acids are nonpolar molecules. Their long carbon chains lack the charged groups that would facilitate interactions with water, which is polar. Consequently, fatty acids dissolve poorly in water but readily in organic solvents; thus, fatty acids are hydrophobic. On the other hand, the head groups of most phospholipids are charged or polar. These head groups interact well with water and consequently are very water soluble. Thus, the head groups are hydrophilic. Because phospholipids combine hydrophilic heads with hydrophobic tails, their interaction with water is referred to as amphipathic.
When mixed with water, phospholipids organize themselves into structures that prevent their hydrophobic tails from making contact with water while simultaneously permitting their hydrophilic head groups to be fully dissolved. When added to water at fairly low concentrations, phospholipids form a monolayer (see Fig. 2-1 C ) on the water's surface at the air-water interface. It is energetically less costly to the system for the hydrophobic tails to stick up in the air than to interact with the solvent.
At higher concentrations, phospholipids assemble into micelles. The hydrophilic head groups form the surfaces of these small spheres, whereas the hydrophobic tails point toward their centers. In this geometry, the tails are protected from any contact with water and instead are able to participate in energetically favorable interactions among themselves. At still higher concentrations, phospholipids spontaneously form bilayers (see Fig. 2-1 D ). In these structures, the phospholipid molecules arrange themselves into two parallel sheets or leaflets that face each other tail to tail. The hydrophilic head groups form the surfaces of the bilayer; the hydrophobic tails form the center of the sandwich. The hydrophilic surfaces insulate the hydrophobic tails from contact with the solvent, leaving the tails free to associate exclusively with one another.
The physical characteristics of a lipid bilayer largely depend on the chemical composition of its constituent phospholipid molecules. For example, the width of the bilayer is determined by the length of the fatty-acid side chains. Dihexadecanoic phospholipids (whose two fatty-acid chains are each 16 carbons long) produce bilayers that are 2.47 nm wide; ditetradecanoic phospholipids (bearing 14-carbon fatty acids) generate 2.3-nm bilayers. Similarly, the nature of the head groups determines how densely packed adjacent phospholipid molecules are in each leaflet of the membrane.
Detergents can dissolve phospholipid membranes because, like the phospholipids themselves, they are amphipathic. They possess very hydrophilic head groups and hydrophobic tails and are water soluble at much higher concentrations than are the phospholipids. When mixed together in aqueous solutions, detergent and phospholipid molecules interact through their hydrophobic tails, and the resulting complexes are water soluble, either as individual dimers or in mixed micelles. Therefore, adding sufficient concentrations of detergent to phospholipid bilayer membranes disrupts the membranes and dissolves the lipids. Detergents are extremely useful tools in research into the structure and composition of lipid membranes.
Despite its highly organized appearance, a phospholipid bilayer is a fluid structure. An individual phospholipid molecule is free to diffuse within the entire leaflet in which it resides. The rate at which this two-dimensional diffusion occurs is extremely temperature dependent. At high temperatures, the thermal energy of any given lipid molecule is greater than the interaction energy that would tend to hold adjacent lipid molecules together. Under these conditions, lateral diffusion can proceed rapidly, and the lipid is said to be in the sol state. At lower temperatures, interaction energies exceed the thermal energies of most individual molecules. Thus, phospholipids diffuse slowly because they lack the energy to free themselves from the embraces of their neighbors. This behavior is characteristic of the gel state.
The temperature at which the bilayer membrane converts from the gel to the sol phase (and vice versa) is referred to as the transition temperature. The transition temperature is another characteristic that depends on the chemical makeup of the phospholipids in the bilayer. Phospholipids with long, saturated fatty-acid chains can extensively interact with one another. Consequently, a fair amount of thermal energy is required to overcome these interactions and permit diffusion. Not surprisingly, such bilayers have relatively high transition temperatures. For example, the transition temperature for dioctadecanoic phosphatidylcholine (which has two 18-carbon fatty-acid chains, fully saturated) is 55.5°C. In contrast, phospholipids that have shorter fatty-acid chains or double bonds (which introduce kinks) cannot line up next to each other as well and hence do not interact as well. Considerably less energy is required to induce them to participate in diffusion. For example, if we reduce the length of the carbon chain from 18 to 14, the transition temperature falls to 23°C. If we retain 18 carbons but introduce one double bond (making the fatty-acid chains monounsaturated), the transition temperature also falls dramatically.
By mixing other types of lipid molecules into phospholipid bilayers, we can markedly alter the membrane's fluidity properties. The glycerol-based phospholipids, the most common membrane lipids, include the phosphatidylethanolamines described above (see Fig. 2-1 A ), as well as the phosphatidylinositols ( Fig. 2-2 A ), phosphatidylserines (see Fig. 2-2 B ), and phosphatidylcholines (see Fig. 2-2 C ). The second major class of membrane lipids, the sphingolipids (derivatives of sphingosine ), is made up of three subgroups: sphingomyelins (see Fig. 2-2 D ), ![]() N2-1 glycosphingolipids such as the galactocerebrosides (see Fig. 2-2 E ), and gangliosides (not shown in figure). Cholesterol (see Fig. 2-2 F ) is another important membrane lipid. Because these other molecules are not shaped exactly like the glycerol-based phospholipids, they participate to different degrees in intermolecular interactions with phospholipid side chains.
N2-1 glycosphingolipids such as the galactocerebrosides (see Fig. 2-2 E ), and gangliosides (not shown in figure). Cholesterol (see Fig. 2-2 F ) is another important membrane lipid. Because these other molecules are not shaped exactly like the glycerol-based phospholipids, they participate to different degrees in intermolecular interactions with phospholipid side chains. ![]() N2-2 The presence of these alternative lipids changes the strength of the interactions that prevents lipid molecules from diffusing. Consequently, the membrane has a different fluidity and a different transition temperature. This behavior is especially characteristic of the cholesterol molecule, whose rigid steroid ring binds to and partially immobilizes fatty-acid side chains. Therefore, at modest concentrations, cholesterol decreases fluidity. However, when it is present in high concentrations, cholesterol can substantially disrupt the ability of the phospholipids to interact among themselves, which increases fluidity and lowers the gel-sol transition temperature. This issue is significant because animal cell plasma membranes can contain substantial quantities of cholesterol.
N2-2 The presence of these alternative lipids changes the strength of the interactions that prevents lipid molecules from diffusing. Consequently, the membrane has a different fluidity and a different transition temperature. This behavior is especially characteristic of the cholesterol molecule, whose rigid steroid ring binds to and partially immobilizes fatty-acid side chains. Therefore, at modest concentrations, cholesterol decreases fluidity. However, when it is present in high concentrations, cholesterol can substantially disrupt the ability of the phospholipids to interact among themselves, which increases fluidity and lowers the gel-sol transition temperature. This issue is significant because animal cell plasma membranes can contain substantial quantities of cholesterol.
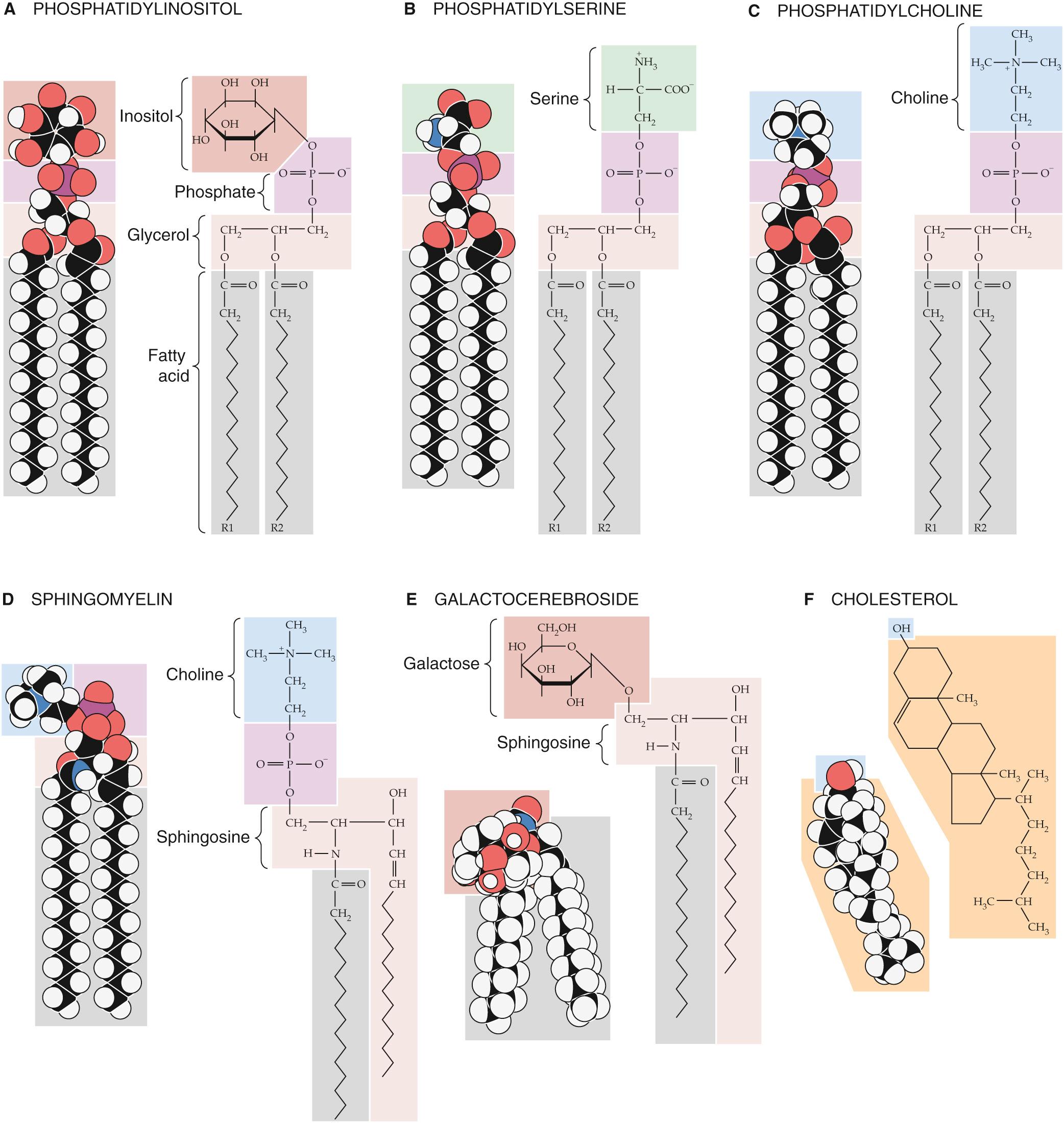
The polar head group of sphingomyelins can be either phosphocholine, as shown in Figure 2-2 D , or phosphoethanolamine (analogous to the phosphoethanolamine moiety in Fig. 2-1 A ). Note that sphingomyelins are both (1) sphingolipids because they contain sphingosine, and (2) phospholipids because they contain a phosphate group as do the glycerol-based phospholipids shown in Figures 2-1 A and 2-2 A – C .
Bilayers composed of several different lipids do not undergo the transition from gel to sol at a single, well-defined temperature. Instead, they interconvert more gradually over a temperature range that is defined by the composition of the mixture. Within this transition range in such multicomponent bilayers, the membrane can become divided into compositionally distinct zones. The phospholipids with long-chain, saturated fatty acids will adhere to one another relatively tightly, which results in the formation of regions with gel-like properties. Phospholipids bearing short-chain, unsaturated fatty acids will be excluded from these regions and migrate to sol-like regions. Hence, “lakes” of lipids with markedly different physical properties can exist side by side in the plane of a phospholipid membrane. Thus, the same thermodynamic forces that form the elegant bilayer structure can partition distinct lipid domains within the bilayer. As discussed below, the segregation of lipid lakes in the plane of the membrane may be important for sorting membrane proteins to different parts of the cell.
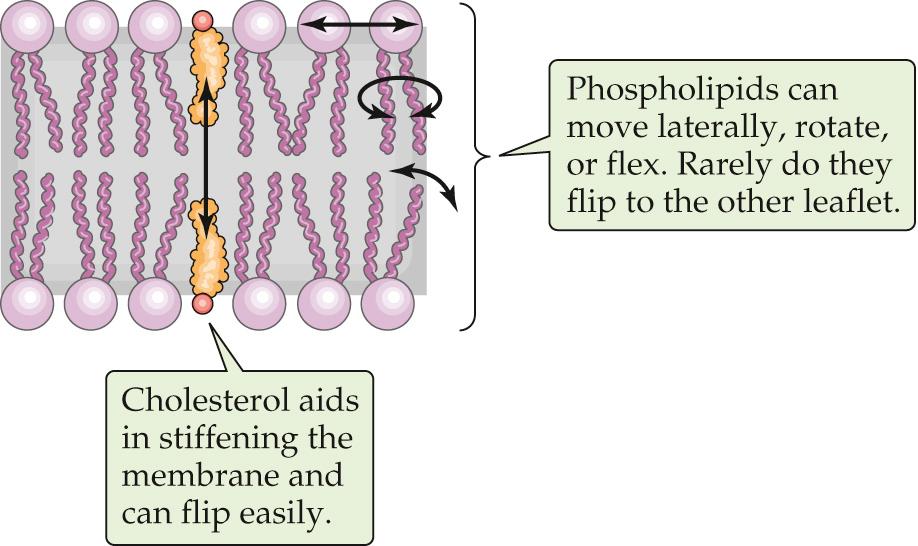
Although phospholipids can diffuse in the plane of a lipid bilayer membrane, they do not diffuse between adjacent leaflets ( Fig. 2-3 ). The rate at which phospholipids spontaneously “flip-flop” from one leaflet of a bilayer to the other is extremely low. As mentioned above, the center of a bilayer membrane consists of the fatty-acid tails of the phospholipid molecules and is an extremely hydrophobic environment. For a phospholipid molecule to jump from one leaflet to the other, its highly hydrophilic head group would have to transit this central hydrophobic core, which would have an extremely high energy cost. This caveat does not apply to cholesterol (see Fig. 2-3 ), whose polar head is a single hydroxyl group. The energy cost of dragging this small polar hydroxyl group through the bilayer is relatively low, which permits relatively rapid cholesterol flip-flop.
The lipid bilayer is ideally suited to separate two aqueous compartments. Its hydrophilic head groups interact well with water at both membrane surfaces, whereas the hydrophobic center ensures that the energetic cost of crossing the membrane is prohibitive for charged atoms or molecules. Pure phospholipid bilayer membranes are extremely impermeable to almost any charged water-soluble substance. Ions such as Na + , K + , Cl – , and Ca 2+ are insoluble in the hydrophobic membrane core and consequently cannot travel from the aqueous environment on one side of the membrane to the aqueous environment on the opposite side. The same is true of large water-soluble molecules, such as proteins, nucleic acids, sugars, and nucleotides.
Whereas phospholipid membranes are impermeable to water-soluble molecules, small uncharged polar molecules can cross fairly freely. This is often true for O 2 , CO 2 , NH 3, and, remarkably, water itself. Water molecules may, at least in part, traverse the membrane through transient cracks between the hydrophobic tails of the phospholipids without having to surmount an enormous energetic barrier. The degree of permeability of water (and perhaps that of CO 2 and NH 3 as well) varies extensively with lipid composition; some phospholipids (especially those with short or kinked fatty-acid chains) permit a much greater rate of transbilayer water diffusion than others do.
As may be inferred from the preceding discussion, the membrane at the cell surface is, in fact, a phospholipid bilayer. The truth of this statement was established by a remarkably straightforward experiment. In 1925, Gorter and Grendel measured the surface area of the lipids they extracted from erythrocyte plasma membranes. They used a device called a Langmuir trough in which the lipids are allowed to line up at an air-water interface (see Fig. 2-1 C ) and are then packed together into a continuous monolayer by a sliding bar that decreases the surface available to them. The area of the monolayer that was created by the erythrocyte lipids was exactly twice the surface area of the erythrocytes from which they were derived. Therefore, the plasma membrane must be a bilayer.
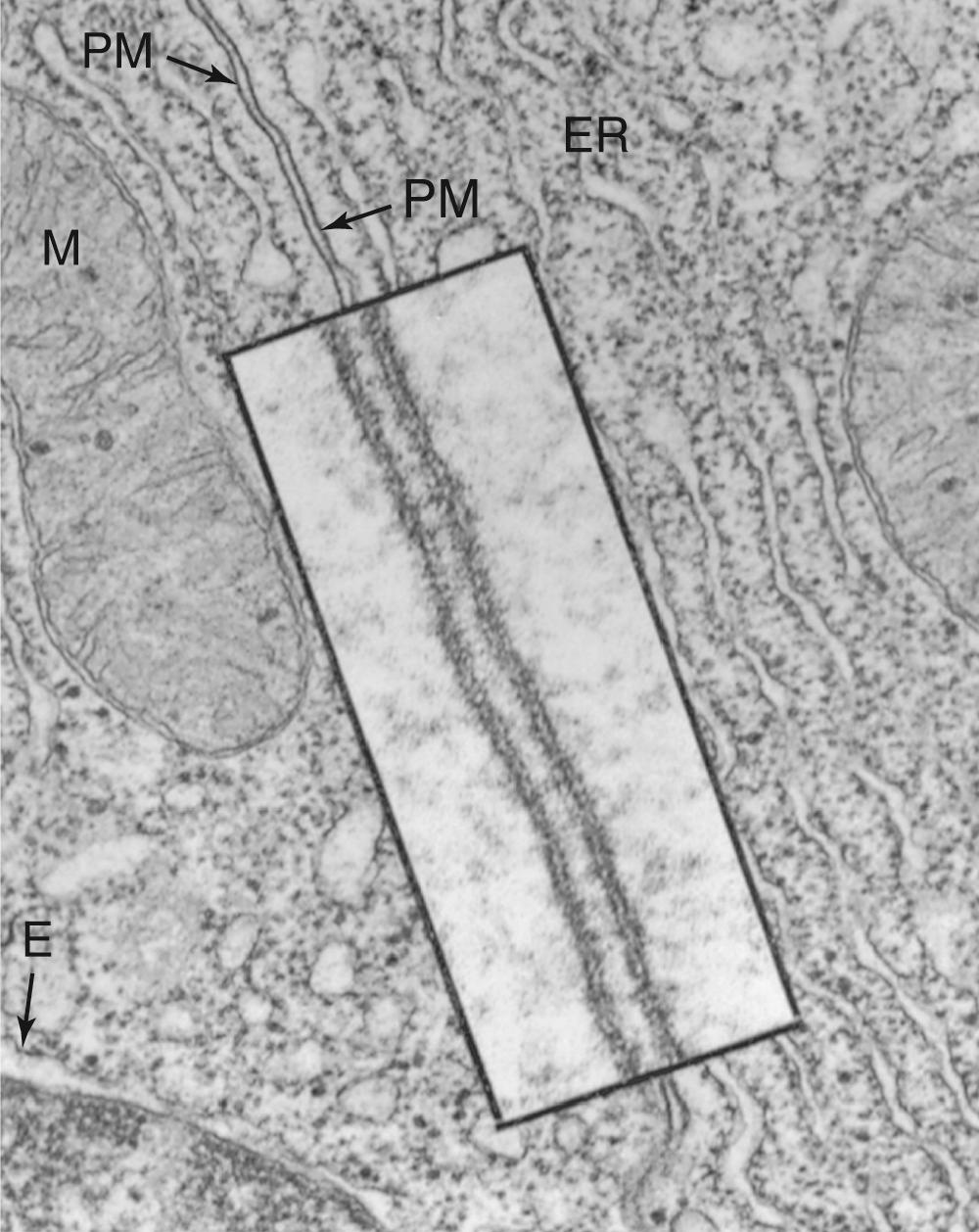
Confirmation of the bilayer structure of biological membranes has come from x-ray diffraction studies performed on the repetitive whorls of membrane that form the myelin sheaths surrounding neuronal axons (see pp. 292–293 ).
The membrane's bilayer structure can be visualized directly in the high-magnification electron micrograph depicted in Figure 2-4 . The osmium tetraoxide molecule (OsO 4 ) with which the membrane is stained binds to the head groups of phospholipids. Thus, both surfaces of a phospholipid bilayer appear black in electron micrographs, whereas the membrane's unstained central core appears white.
The phospholipid compositions of the two leaflets of the plasma membrane are not identical. Labeling studies performed on erythrocyte plasma membranes reveal that the surface that faces the cytoplasm contains phosphatidylethanolamine and phosphatidylserine, whereas the outward-facing leaflet is composed almost exclusively of phosphatidylcholine. As is discussed below in this chapter, this asymmetry is created during the biosynthesis of the phospholipid molecules. It is not entirely clear what advantage this distribution provides to the cell. The interactions between certain proteins and the plasma membrane may require this segregation. The lipid asymmetry may be especially important for those phospholipids that are involved in second-messenger cascades. Phosphatidylinositols, for example, give rise to phosphoinositides, which play critical roles in signaling pathways (see pp. 58–61 ). In addition, the phosphatidylinositol composition of the cytoplasmic face of an organelle helps to define the identity of the organelle and to govern its trafficking and targeting properties. Finally, the phospholipids that are characteristic of animal cell plasma membranes generally have one saturated and one unsaturated fatty-acid residue. Consequently, they are less likely to partition into sol-like or gel-like lipid domains than are phospholipids that bear identical fatty-acid chains. ![]() N2-3
N2-3
According to current models (see ; ), lipids and proteins are not uniformly distributed in the plane of the membranes that surround cells and organelles. Instead, certain lipids and associated proteins cluster to form microdomains that differ in composition, structure, and function from the rest of the membrane that surrounds them. These microdomains can be thought of as small islands bordered by the “lake” of lipids and proteins that constitute the bulk of the membrane. These two-dimensional structures are composed of lipids that tend to form close interactions with one another, resulting in the self-assembly of organized domains that include specific types of lipids and exclude others. The lipids that tend to be found in microdomains include sphingomyelin, cholesterol, and glycolipids. Proteins that are able to interact closely with microdomain-forming lipids can also become selectively incorporated into these microdomains. A number of different names are used to refer to these microdomains, the most common of which are caveolae and rafts.
Caveolae (see pp. 42–43 ) were originally identified in the electron microscope as flask-shaped invaginations of the plasma membrane. They carry a coat composed of proteins called caveolins, and they tend to be at least 50 to 80 nm in diameter. Caveolae have been shown to participate in endocytosis of specific subsets of proteins and are also richly endowed with signaling molecules, such as receptor tyrosine kinases.
Rafts are less well understood structures, which are defined by the biochemical behaviors of their constituents when the surrounding membrane is dissolved in nonionic detergents. Lipid microdomains rich in sphingomyelin, cholesterol, and glycolipids tend to resist solubilization in these detergents under certain conditions and can be recovered intact by density centrifugation. Once again, a number of interesting proteins involved in cell signaling and communication, including kinases, ion channels, and G proteins, tend to be concentrated in rafts, or to become associated with rafts upon the activation of specific signal-transduction pathways. Rafts are thought to collect signaling proteins into small, highly concentrated zones, thereby facilitating their interactions and hence their ability to activate particular pathways. Rafts are also involved in membrane trafficking processes. In polarized epithelial cells, the sorting of a number of proteins to the apical plasma membrane is dependent upon their ability to partition into lipid rafts that form in the plane of the membrane of the trans -Golgi network. Little is known about what lipid rafts actually look like in cell membranes in situ. It is currently thought that they are fairly small (<100 nm), although it is possible that they can be induced to coalesce into larger structures under certain circumstances. Much remains to be learned about the structures and functions of rafts and caveolae, but it is clear that they are dynamic and important entities that subdivide membrane into specialized regions that cells exploit for a wide variety of tasks.
The demonstration that the plasma membrane's lipid components form a bilayer leaves open the question of how the membrane's protein constituents are organized. Membrane proteins can belong to either of two broad classes, peripheral or integral. Peripherally associated membrane proteins are neither embedded within the membrane nor attached to it by covalent bonds; instead, they adhere tightly to the cytoplasmic or extracellular surfaces of the plasma membrane ( Fig. 2-5 A ). They can be removed from the membrane, however, by mild treatments that disrupt ionic bonds (very high salt concentrations) or hydrogen bonds (very low salt concentrations).
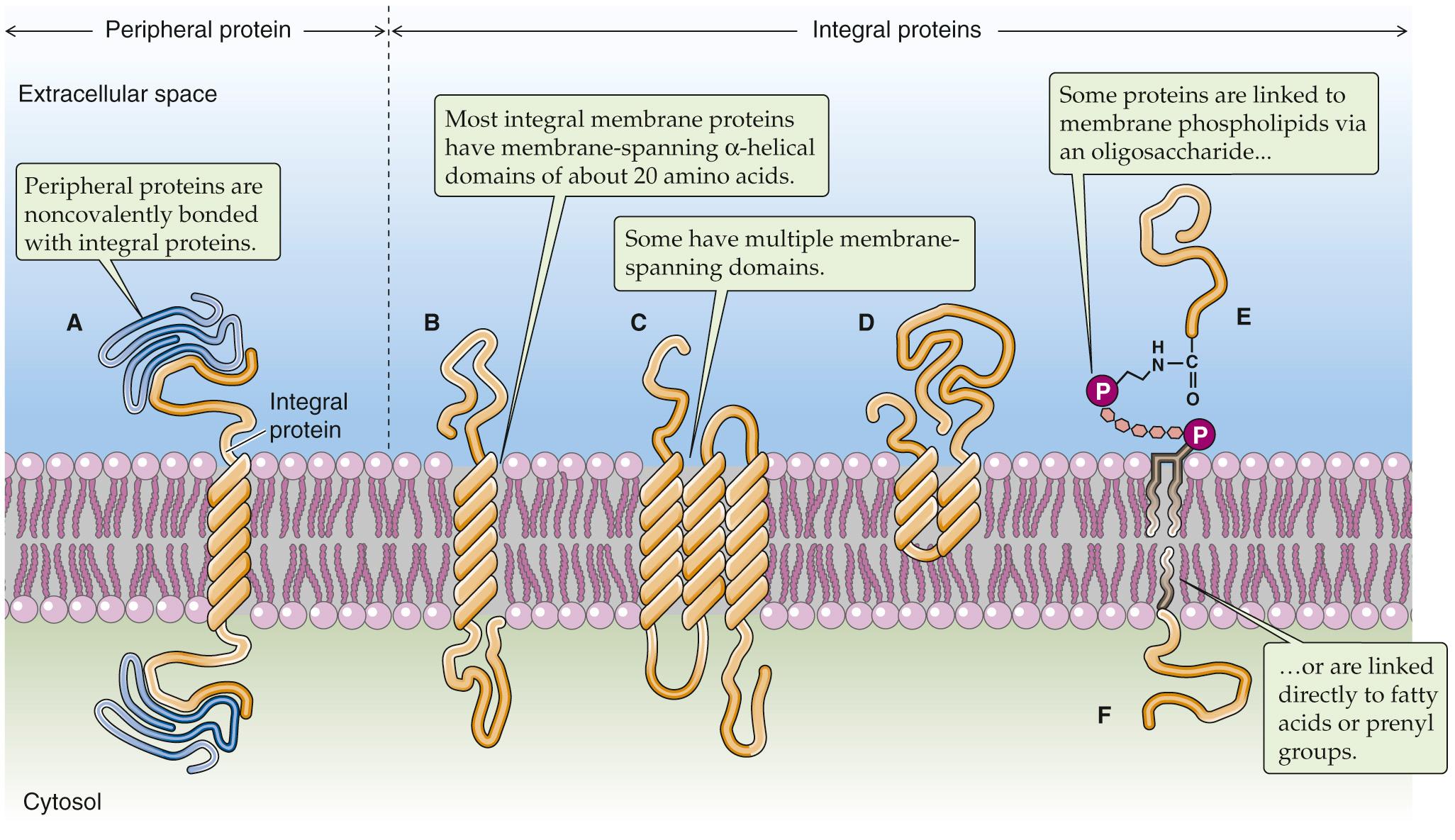
In contrast, integral membrane proteins are intimately associated with the lipid bilayer. They cannot be eluted from the membrane by these high- or low-salt washes. For integral membrane proteins to be dislodged, the membrane itself must be dissolved by adding detergents. Integral membrane proteins can be associated with the lipid bilayer in any of three ways. First, some proteins actually span the lipid bilayer once or several times (see Fig. 2-5 B, C ) and hence are referred to as transmembrane proteins. Experiments performed on erythrocyte membranes reveal that these proteins can be labeled with protein-tagging reagents applied to either side of the bilayer.
The second group of integral membrane proteins is embedded in the bilayer without actually crossing it (see Fig. 2-5 D ). A third group of membrane-associated proteins is not actually embedded in the bilayer at all. Instead, these lipid-anchored proteins are attached to the membrane by a covalent bond that links them either to a lipid component of the membrane or to a fatty-acid derivative that intercalates into the membrane. For example, proteins can be linked to a special type of glycosylated phospholipid molecule (see Fig. 2-5 E ), which is most often glycosylphosphatidylinositol (GPI), on the outer leaflet of the membrane. This family is referred to collectively as the glycophospholipid-linked proteins. Another example is a direct linkage to a fatty acid (e.g., a myristyl group) or a prenyl (e.g., farnesyl) group that intercalates into the inner leaflet of the membrane (see Fig. 2-5 F ).
How can membrane-spanning proteins remain stably associated with the bilayer in a conformation that requires at least some portion of their amino-acid sequence to be in continuous contact with the membrane's hydrophobic central core? The answer to this question can be found in the special structures of those protein domains that actually span the membrane.
The side chains of the eight amino acids listed in the upper portion of Table 2-1 are hydrophobic. These aromatic or uncharged aliphatic groups are almost as difficult to solvate in water as are the fatty-acid side chains of the membrane phospholipids themselves. Not surprisingly, therefore, these hydrophobic side chains are quite comfortable in the hydrophobic environment of the bilayer core. Most membrane-spanning segments —that is, the short stretches of amino acids that pass through the membrane once—are composed mainly of these nonpolar amino acids, in concert with polar, uncharged amino acids.
| NAME | THREE-LETTER CODE | SINGLE-LETTER CODE | STRUCTURE OF THE SIDE CHAIN | HYDROPATHY INDEX * | |
|---|---|---|---|---|---|
| Nonpolar | Alanine | Ala | A |
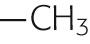
|
+1.8 |
| Valine | Val | V |
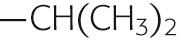
|
+4.2 | |
| Leucine | Leu | L |

|
+3.8 | |
| Isoleucine | Ile | I |
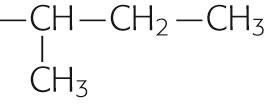
|
+4.5 | |
| Proline | Pro | P |
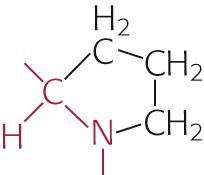
|
−1.6 | |
| Phenylalanine | Phe | F |
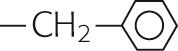
|
+2.8 | |
| Tryptophan | Trp | W |
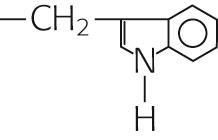
|
−0.9 | |
| Methionine | Met | M |
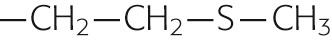
|
+1.9 | |
| Polar uncharged | Glycine | Gly | G |
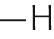
|
−0.4 |
| Serine | Ser | S |
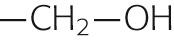
|
−0.8 | |
| Threonine | Thr | T |
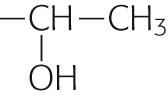
|
−0.7 | |
| Cysteine | Cys | C |
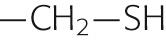
|
+2.5 | |
| Tyrosine | Tyr | Y |
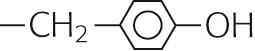
|
−1.3 | |
| Asparagine | Asn | N |
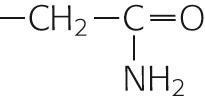
|
−3.5 | |
| Glutamine | Gln | Q |
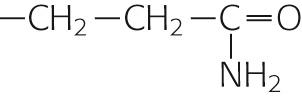
|
−3.5 | |
| Polar, charged, acidic | Aspartate | Asp | D |
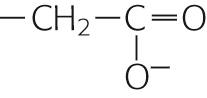
|
−3.5 |
| Glutamate | Glu | E |
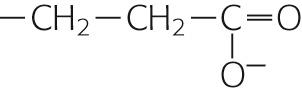
|
−3.5 | |
| Polar, charged, basic | Lysine | Lys | K |
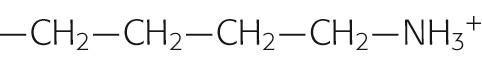
|
−3.9 |
| Arginine | Arg | R |
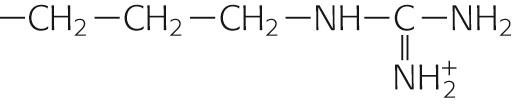
|
−4.5 | |
| Histidine | His | H |
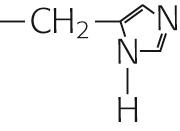
|
−3.2 |
* Kyte and Doolittle generated these values (arbitrary scale from −4.5 to +4.5) by averaging two kinds of data. The first is an index of the energy that is required to transfer the side chain from the vapor phase into water. The second indicates how likely it is to find the side chain buried in (as opposed to being on the surface of) 12 globular proteins, whose structures were solved by x-ray crystallography. A positive value indicates that the side chain is hydrophobic.
The hydrophobic, membrane-spanning segments of transmembrane proteins are specially adapted to the hydrophobic milieu in which they reside. The phospholipid molecules of the membrane bilayer actually protect these portions of transmembrane proteins from energetically unfavorable interactions with the aqueous environment. Transmembrane proteins tend to be extremely insoluble in water. If we separate the membrane-spanning segments of these proteins from the amphipathic phospholipids that surround them, these hydrophobic sequences tend to interact tightly with one another rather than with water. The resulting large protein aggregates are generally insoluble and precipitate out of solution. If, however, we disrupt the phospholipid membrane by adding detergent, the amphipathic detergent molecules can substitute for the phospholipids. The hydrophobic membrane-spanning sequences remain insulated from interactions with the aqueous solvent, and the proteins remain soluble as components of detergent micelles. This ability of detergents to remove transmembrane proteins from the lipid bilayer—while maintaining the solubility and native architectures of these proteins—has proved important for purifying individual membrane proteins.
Transmembrane proteins can have a single membrane-spanning segment (see Fig. 2-5 B ) or several (see Fig. 2-5 C ). Those with a single transmembrane segment can be oriented with either their amino (N) or their carboxyl (C) terminus facing the extracellular space. Multispanning membrane proteins weave through the membrane like a thread through cloth. Again, the N and C termini can be exposed to either the cytoplasmic or extracellular compartments. The pattern with which the transmembrane protein weaves across the lipid bilayer defines its membrane topology.
The amino-acid sequences of membrane-spanning segments tend to form α helices, with ~3.6 amino acids per turn of the helix (see Fig. 2-5 B ). In this conformation, the polar atoms of the peptide backbone are maximally hydrogen bonded to one another—from one turn of the helix to the next—so they do not require the solvent to contribute hydrogen-bond partners. Hence, this structure ensures the solubility of the membrane-spanning sequence in the hydrophobic environment of the membrane. Whereas most transmembrane proteins appear to traverse the membrane with α-helical spans, it is clear that an intriguing subset of membrane polypeptides makes use of a very different structure. For example, the porin protein (see p. 109 ), which serves as a channel in bacterial membranes, has membrane-spanning portions arranged as a β barrel.
In the case of multispanning membrane proteins, their transmembrane helices probably pack together tightly (see Fig. 2-5 C ). Molecular analysis of a number of known membrane-spanning sequences has helped in the development of algorithms predicting the likelihood that a given amino-acid sequence can span the membrane. These algorithms are widely used to assess the likelihood that newly identified genes encode transmembrane proteins and to predict the number and location of membrane-spanning segments.
Many membrane proteins form tight, noncovalent associations with other membrane proteins in the plane of the bilayer. These multimeric proteins can be composed of a single type of polypeptide or of mixtures of two or more different proteins. The side-to-side interactions that hold these complexes together can involve the membrane-spanning segments or regions of the proteins that protrude at either surface of the bilayer. By assembling into multimeric complexes, membrane proteins can increase their stability. They can also increase the variety and complexity of the functions that they are capable of performing.
As is true for phospholipid molecules (see Fig. 2-3 ), some transmembrane proteins can diffuse within the surface of the membrane. In the absence of any protein-protein attachments, transmembrane proteins are free to diffuse over the entire surface of a membrane. This fact was demonstrated by ( Fig. 2-6 ). They labeled the surface proteins of a population of mouse lymphocytes with a lectin (a plant protein that binds strongly to certain sugar groups attached to proteins) that was linked to the fluorescent dye fluorescein. They also tagged the surface proteins of a second population of human lymphocytes with a lectin that was conjugated to a different fluorescent dye, rhodamine. Because fluorescein glows green and rhodamine glows red when excited by the light of the appropriate wavelengths, these labeling molecules can be easily distinguished from one another in a fluorescence microscope. Frye and Edidin mixed the two lymphocyte populations and treated them with a reagent that caused the cells to fuse to each other. Immediately after fusion, the labeled surface proteins of the newly joined cells remained separate; half of the fused cell surface appeared red, whereas the other half appeared green. During a period of ~30 minutes, however, the green and red protein labels intermixed until the entire surface of the fused cell was covered with both labeling molecules. The rate at which this intermingling occurred increased with temperature, which is not surprising given the temperature dependence of membrane fluidity.
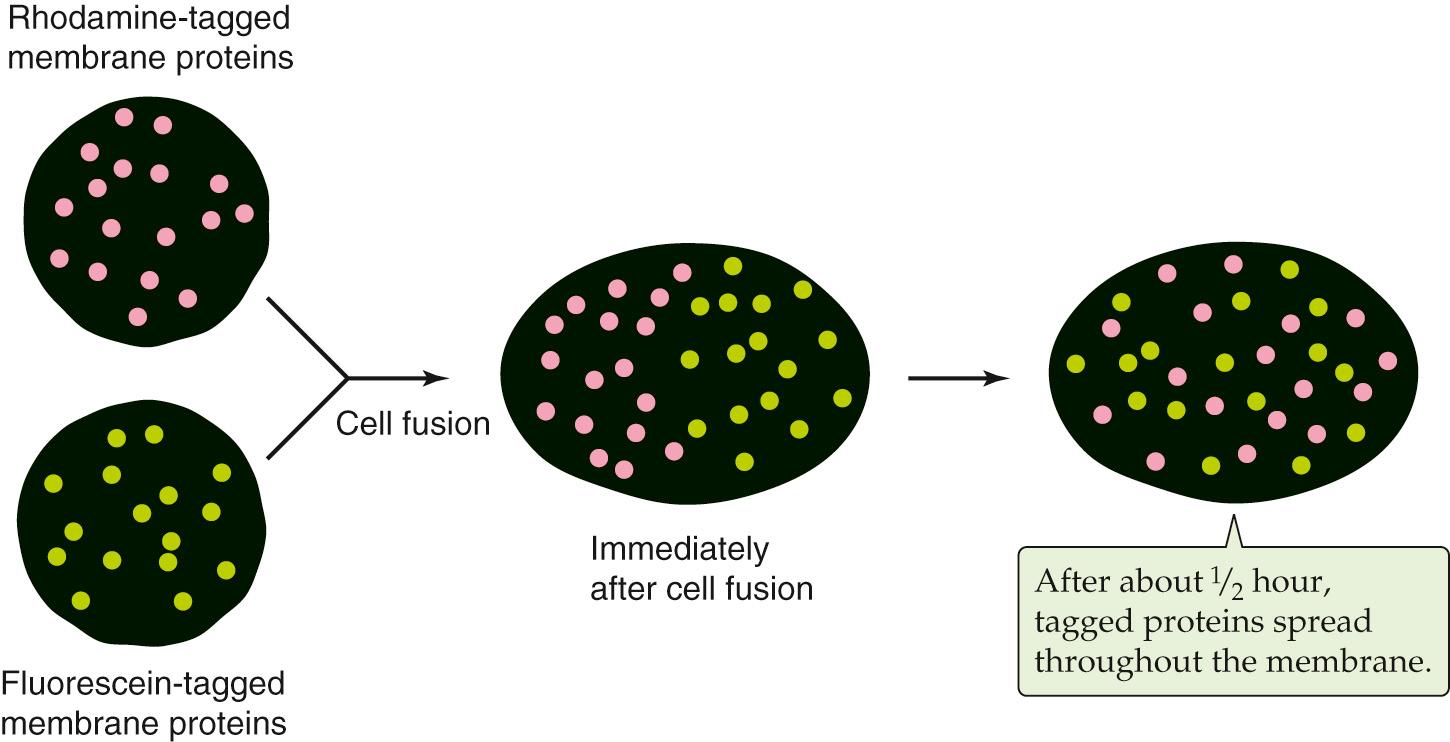
Because transmembrane proteins are large molecules, their diffusion in the plane of the membrane is much slower than that of lipids. Even the fastest proteins diffuse ~1000 times more slowly than the average phospholipid. The diffusion of many transmembrane proteins appears to be further impeded by their attachments to the cytoskeleton, just below the surface of the membrane. Tight binding to this meshwork can render proteins essentially immobile. Other transmembrane proteins appear to travel in the plane of the membrane via directed processes that are much faster and less directionally random than diffusion is. Motor proteins that are associated with the cytoplasmic cytoskeleton (discussed below) appear to grab onto certain transmembrane proteins, dragging them in the plane of the membrane like toy boats on strings. Finally, like phospholipids, proteins can diffuse only in the plane of the bilayer. They cannot flip-flop across it. The energetic barrier to dragging a transmembrane protein's hydrophilic cytoplasmic and extracellular domains across the bilayer's hydrophobic core is very difficult to surmount. Thus, a membrane protein's topology does not change over its life span.
All communication between a cell and its environment must involve or at least pass through the plasma membrane. For the purposes of this discussion, we define communication rather broadly as the exchange of any signal between the cell and its surroundings. Except for lipid-soluble signaling molecules such as steroid hormones, essentially all communication functions served by the plasma membrane occur via membrane proteins. From an engineering perspective, membrane proteins are perfectly situated to transmit signals because they form a single, continuous link between the two compartments that are separated by the membrane.
Ligand-binding receptors comprise the group of transmembrane proteins that perhaps most clearly illustrate the concept of transmembrane signaling ( Fig. 2-7 A ). For water-soluble hormones such as epinephrine to influence cellular behavior, their presence in the ECF compartment must be made known to the various intracellular mechanisms whose behaviors they modulate. The interaction of a hormone with the extracellular portion of the hormone receptor, which forms a high-affinity binding site, produces conformational changes within the receptor protein that extend through the membrane-spanning domain to the intracellular domain of the receptor. As a consequence, the intracellular domain either becomes enzymatically active or can interact with cytoplasmic proteins that are involved in the generation of so-called second messengers. Either mechanism completes the transmission of the hormone signal across the membrane. The transmembrane disposition of a hormone receptor thus creates a single, continuous communication medium that is capable of conveying, through its own structural modifications, information from the environment to the cellular interior. The process of transmembrane signal transduction is discussed in Chapter 3 .
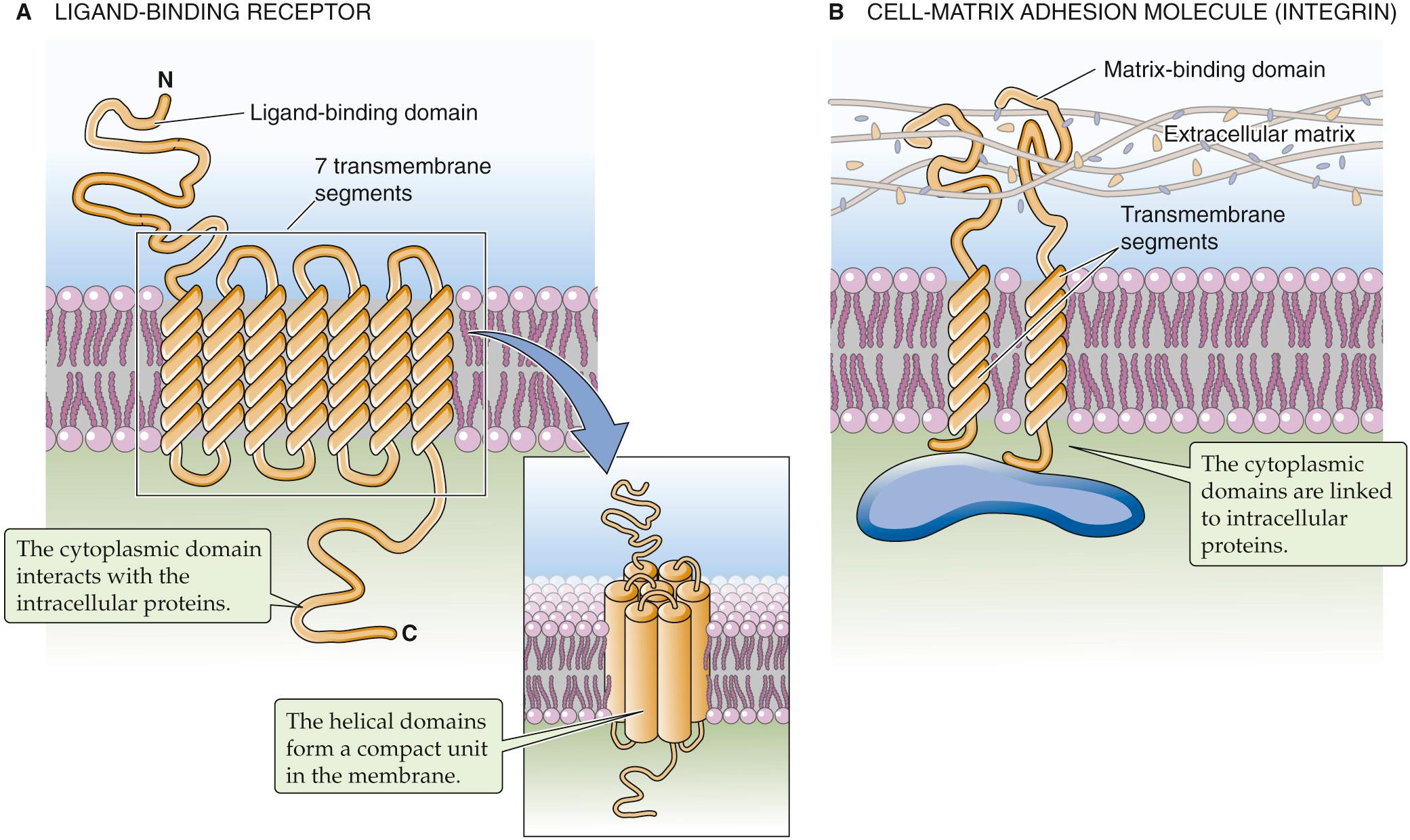
Cells can also exploit integral membrane proteins as adhesion molecules that form physical contacts with the surrounding extracellular matrix (i.e., cell-matrix adhesion molecules) or with their cellular neighbors (i.e., cell-cell adhesion molecules). These attachments can be extremely important in regulating the shape, growth, and differentiation of cells. The nature and extent of these attachments must be communicated to the cell interior so that the cell can adapt appropriately to the physical constraints and cues that are provided by its immediate surroundings. Numerous classes of transmembrane proteins are involved in these communication processes. The integrins are examples of matrix receptors or cell-matrix adhesion molecules. They comprise a large family of transmembrane proteins that link cells to components of the extracellular matrix (e.g., fibronectin, laminin) at adhesion plaques (see Fig. 2-7 B ). These linkages produce conformational changes in the integrin molecules that are transmitted to their cytoplasmic tails. These tails, in turn, communicate the linkage events to various structural and signaling molecules that participate in formulating a cell's response to its physical environment.
In contrast to matrix receptors, which attach cells to the extracellular matrix, several enormous superfamilies of cell-cell adhesion molecules attach cells to each other. These cell-cell adhesion molecules include the Ca 2+ -dependent cell adhesion molecules (cadherins) and Ca 2+ -independent neural cell adhesion molecules (N-CAMs). The cadherins are glycoproteins (i.e., proteins with sugars attached) with one membrane-spanning segment and a large extracellular domain that binds Ca 2+ . The N-CAMs, on the other hand, generally are members of the immunoglobulin superfamily. The two classes of cell-cell adhesion molecules mediate similar sorts of transmembrane signals that help organize the cytoplasm and control gene expression in response to intercellular contacts. Some cell-cell adhesion molecules belong to the GPI-linked class of membrane proteins (see p. 13 ). These polypeptides lack a transmembrane and cytoplasmic tail. Interactions mediated by this unique class of adhesion molecules may be communicated to the cell interior via lateral associations with other membrane proteins.
Adhesion molecules orchestrate processes that are as diverse as the directed migration of immune cells and the guidance of axons in the developing nervous system. Loss of cell-cell and cell-matrix adhesion is a hallmark of metastatic tumor cells.
As noted above, a pure phospholipid bilayer does not have the permeability properties that are normally associated with animal cell plasma membranes. Pure phospholipid bilayers also lack the ability to transport substances uphill (i.e., against electrochemical gradients; p. 105 ). Transmembrane proteins endow biological membranes with these capabilities. Ions and other membrane-impermeable substances can cross the bilayer with the assistance of transmembrane proteins that serve as pores, channels, carriers, and pumps. Pores and channels serve as conduits that allow water, specific ions, or even very large proteins to flow passively through the bilayer. Carriers can either facilitate the transport of a specific molecule across the membrane or couple the transport of a molecule to that of other solutes. Pumps use the energy that is released through the hydrolysis of ATP to drive the transport of substances into or out of cells against energy gradients. Each of these important classes of proteins is discussed in Chapter 5 .
Channels, carriers, and pumps succeed in allowing hydrophilic substances to cross the membrane by creating a hydrophilic pathway in the bilayer. Previously, we asserted that membrane-spanning segments are as hydrophobic as the fatty acids that surround them. How is it possible for these hydrophobic membrane-spanning domains to produce the hydrophilic pathways that permit the passage of ions through the membrane? The solution to this puzzle appears to be that the α helices that make up these membrane-spanning segments are amphipathic. That is, they possess both hydrophobic and hydrophilic domains.
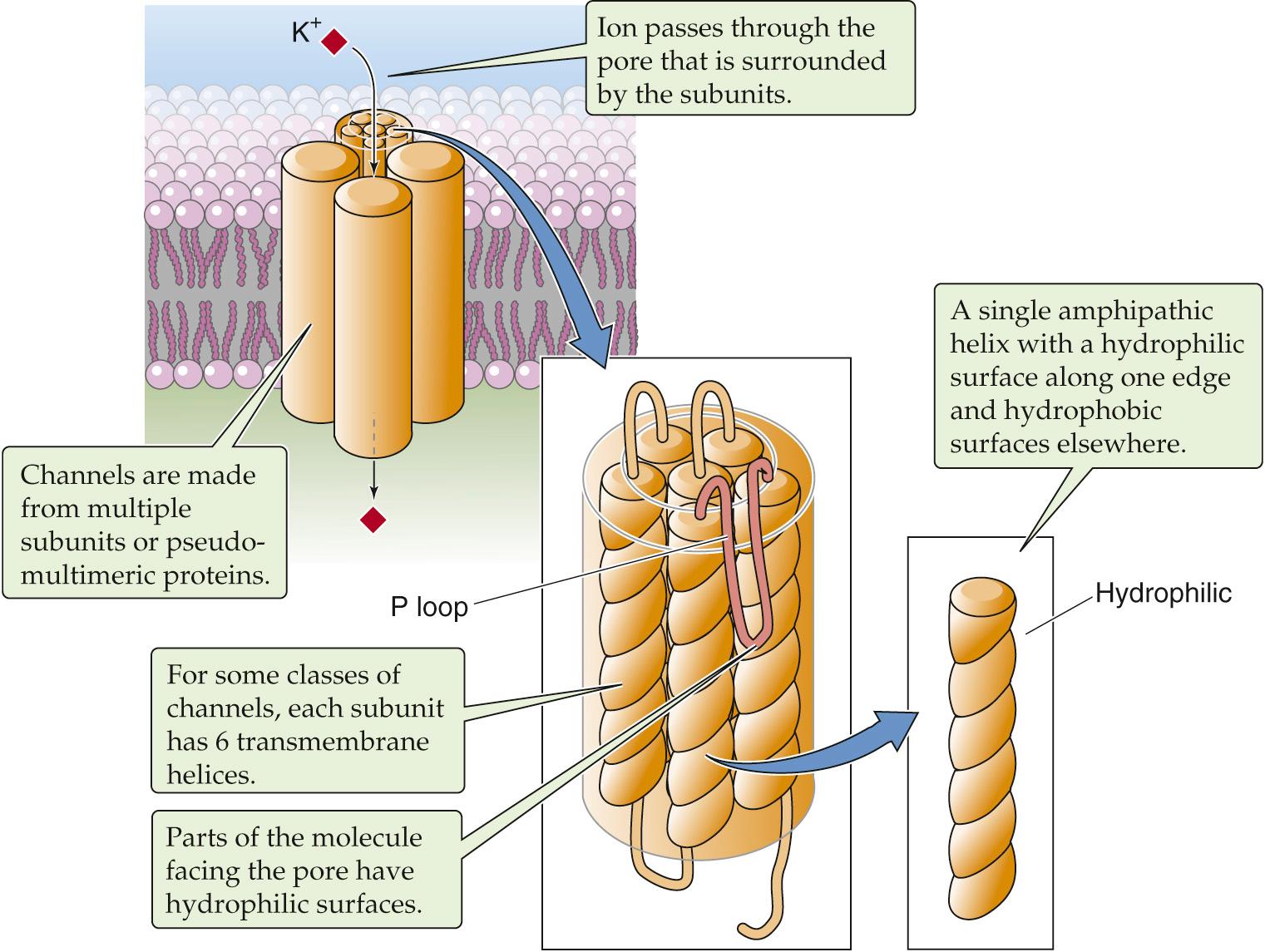
For each α helix, the helical turns produce alignments of amino acids that are spaced at regular intervals in the sequence. Thus, it is possible to align all the hydrophilic or hydrophobic amino acids along a single edge of the helix. In amphipathic helices, hydrophobic amino acids alternate with hydrophilic residues at regular intervals of approximately three or four amino acids (recall that there are ~3.6 amino acids per turn of the helix). Thus, as the helices pack together, side by side, the resultant membrane protein has distinct hydrophilic and hydrophobic surfaces. The hydrophobic surfaces of each helix will face either the membrane lipid or the hydrophobic surfaces of neighboring helices. Similarly, the hydrophilic surfaces of each helix will face a common central pore through which water-soluble substances can move. Depending on how the protein regulates access to this pore, the protein could be a channel, a carrier, or a pump. The mix of hydrophilic amino acids that line the pore presumably determines, at least in part, the nature of the substances that the pore can accommodate. In some instances, the amphipathic helices that line the pore are contributed by several distinct proteins—or subunits—that assemble into a single multimeric complex. Figure 2-8 shows an example of a type of K + channel discussed on page 184 . This channel is formed by the apposition of four identical subunits, each of which has six membrane-spanning segments. The pore of this channel is created by the amphipathic helices as well as by short, hydrophilic loops (P loops) contributed by each of the four subunits.
Ion pumps are actually enzymes. They catalyze the hydrolysis of ATP and use the energy released by that reaction to drive ion transport. Many other classes of proteins that are embedded in cell membranes function as enzymes as well. Membrane-bound enzymes are especially prevalent in the cells of the intestine, which participate in the final stages of nutrient digestion and absorption (see pp. 916–918 ). These enzymes—located on the side of the intestinal cells that faces the lumen of the intestine—break down small polysaccharides into single sugars, or break down polypeptides into shorter polypeptides or amino acids, so that they can be imported into the cells. By embedding these enzymes in the plasma membrane, the cell can generate the final products of digestion close to the transport proteins that mediate the uptake of these nutrient molecules. This theme is repeated in numerous other cell types. Thus, the membrane can serve as an extremely efficient two-dimensional reaction center for multistep processes that involve enzymatic reactions or transport.
Many of the GPI-linked proteins are enzymes. Several of the enzymatic activities that are classically thought of as extracellular markers of the plasma membrane, such as alkaline phosphatase, 5′-nucleotidase, and carbonic anhydrase IV (see p. 828 ), are anchored to the external leaflet of the bilayer by covalent attachment to a GPI.
Some integral proteins associate with the cytoplasmic surface of the plasma membrane by covalently attaching to fatty acids or prenyl groups that in turn intercalate into the lipid bilayer (see Fig. 2-5 F ). The fatty acids or prenyl groups act as hydrophobic tails that anchor an otherwise soluble protein to the bilayer. These proteins are all located at the intra cellular leaflet of the membrane bilayer and often participate in intracellular signaling pathways. The family of lipid-linked proteins includes the small and heterotrimeric GTP-binding proteins, kinases, and oncogene products (see Chapter 3 ). Many of these proteins are involved in relaying the signals that are received at the cell surface to the effector machinery within the cell interior. Their association with the membrane, therefore, brings these proteins close to the cytoplasmic sides of receptors that transmit signals from the cell exterior across the bilayer. The medical relevance of this type of membrane association is beginning to be appreciated. For example, denying certain oncogene products their lipid modifications—and hence their membrane attachment—eliminates their ability to induce tumorigenic transformation.
Peripheral membrane proteins attach loosely to the lipid bilayer but are not embedded within it (see p. 13 ). Their association with the membrane can take one of two forms. First, some proteins interact via ionic interactions with phospholipid head groups. Many of these head groups are positively or negatively charged and thus can participate in salt bridges with adherent proteins.
For a second group of peripheral membrane proteins, attachment is based on the direct binding of peripheral membrane proteins to the extracellular or cytoplasmic surfaces of integral membrane proteins (see Fig. 2-5 A ). This form of attachment is epitomized by the cytoskeleton. For instance, the cytoplasmic surface of the erythrocyte plasma membrane is in close apposition to a dense meshwork of interlocking protein strands known as the subcortical cytoskeleton. It consists of a long, fibrillar molecule called spectrin, short polymers of the cytoskeletal protein actin, and other proteins, including ankyrin and band 4.1 ( Fig. 2-9 ).
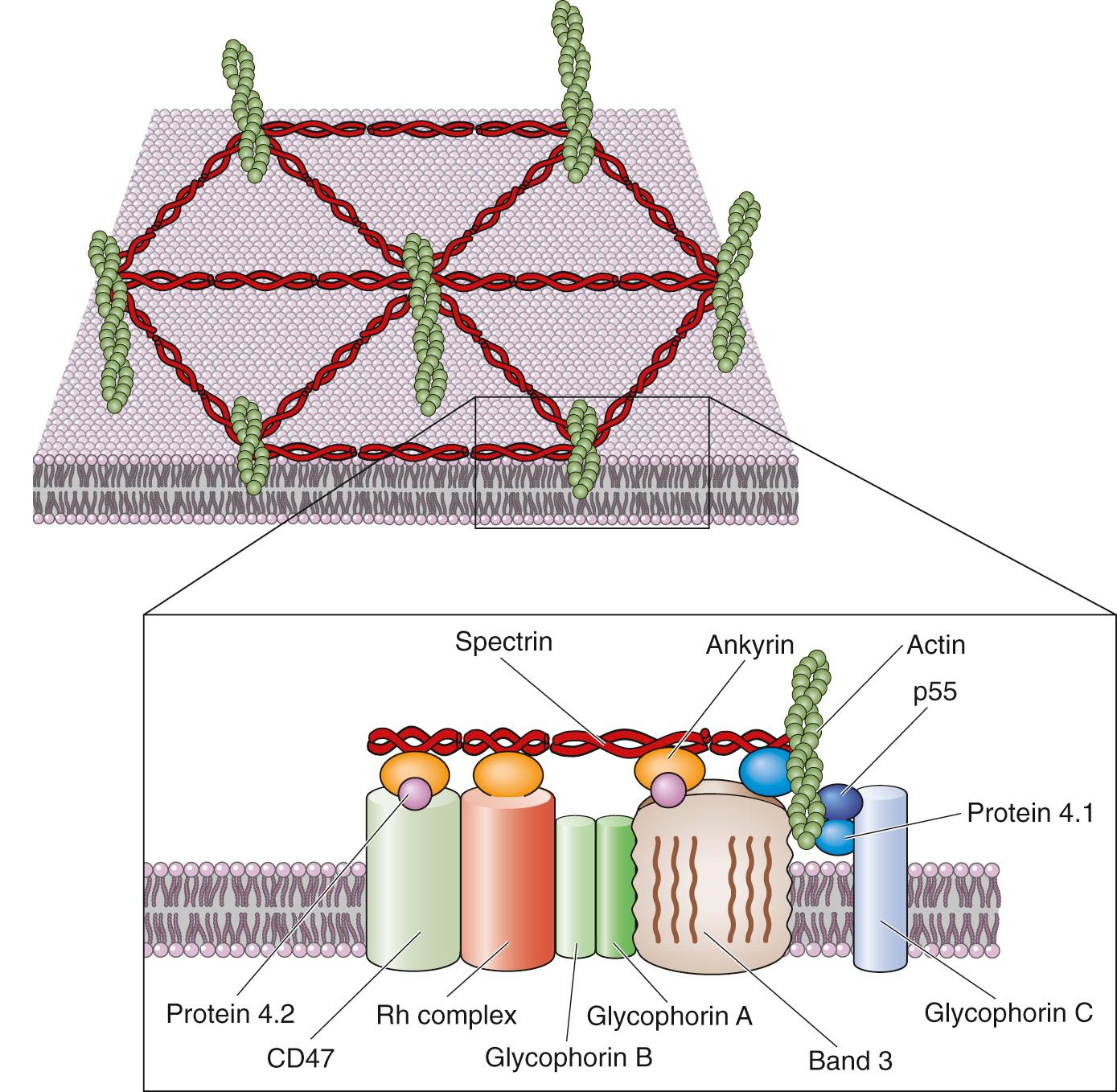
Two closely related isoforms of spectrin (α and β) form dimers, and two of these dimers assemble head to head with one another to form spectrin heterotetramers. The tail regions of spectrin bind the globular protein band 4.1, which in turn can bind to actin fibrils. Each actin fibril can associate with more than one molecule of band 4.1 so that, together, spectrin, actin, and band 4.1 assemble into an extensive interlocking matrix. The protein known as ankyrin binds to spectrin as well as to the cytoplasmic domain of band 3, the integral membrane protein responsible for transporting Cl – and  ions across the erythrocyte membrane. Thus, ankyrin is a peripheral membrane protein that anchors the spectrin-actin meshwork directly to an integral membrane protein of the erythrocyte.
ions across the erythrocyte membrane. Thus, ankyrin is a peripheral membrane protein that anchors the spectrin-actin meshwork directly to an integral membrane protein of the erythrocyte.
The subcortical cytoskeleton provides the erythrocyte plasma membrane with strength and resilience. People who carry mutations in genes encoding components of the cytoskeleton have erythrocytes lacking the characteristic biconcave disk shape. These erythrocytes are extremely fragile and are easily torn apart by the shear stresses (see p. 415 ) associated with circulation through capillaries. It would appear, therefore, that the subcortical cytoskeleton forms a scaffolding of peripheral membrane proteins whose direct attachment to transmembrane proteins enhances the bilayer's structural integrity.
The subcortical cytoskeleton is not unique to erythrocytes. Numerous cell types, including neurons and epithelial cells, have submembranous meshworks that consist of proteins very similar to those first described in the erythrocyte. In addition to band 3, transmembrane proteins found in a wide variety of cells (including ion pumps, ion channels, and cell adhesion molecules) bind ankyrin and can thus serve as focal points of cytoskeletal attachment. In polarized cells (e.g., neurons and epithelial cells), the subcortical cytoskeleton appears to play a critically important role in organizing the plasma membrane into morphologically and functionally distinct domains.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here