Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The extreme lateral infrajugular transcondylar (ELITE) approach was born out of the anatomical constraints and operative challenges presented by the cranial base. The transcondylar approach was devised as an avenue for access to the foramen magnum and ventral medulla. , In 1986, Heros described the extreme lateral inferior suboccipital approach for access to vertebral and vertebrobasilar artery lesions. The ELITE approach is a unique transcondylar and transjugular tubercle skull base approach developed and elaborated in Germany and Japan in 1987. , After years of refinement, this has become a very useful and well described approach for this region of the skull base.
The standard ELITE approach involves drill resection of the medial and superior medial third of the occipital condyle and jugular tubercle. With the exposure afforded by this dissection of the superior central spinal column and adjacent skull base, this is an excellent approach for vertebral artery and posterior inferior cerebellar artery aneurysms; vertebrobasilar junction aneurysms; and more complex foramen magnum, infrajugular, and anterior brainstem tumors. An anterolateral approach modification includes the addition of a high cervical exposure, which is ideal for large glomus jugulare tumors. Other modifications include a limited transcondylar approach for some smaller vertebral artery and posterior inferior cerebellar artery lesions and an extensive ELITE approach involving partial resection of the occipital condyle and resection of the C1 arch for access to chordomas, chondrosarcomas, and high cervical spine lesions. These modifications are beyond the scope of this chapter.
Paragangliomas of the temporal bone were first identified as a unique pathology in 1945. Attempts at characterization and ultimate surgical resection were initially fraught with high morbidity and mortality because of their location amid complex anatomical structures. Primary treatment with radiation therapy soon became the mainstay for therapy with the hopes of limiting complications. Radiation therapy has been shown to be superior to observation of these lesions; however, the long-term risks of malignancy and radiation morbidity limit its success. Definitive surgical resection remains the mainstay for attempted cure.
Attempts at surgical resection and cure led to the development of tumor classification strategies to help guide surgical planning and to improve communication of results. Two main classification schemes were devised and remain in use today, primarily in the otology and neurotology literature—the Fisch and Mattox and Glasscock-Jackson classifications. The Fisch scheme is as follows:
Type A: tumors limited to the middle ear cleft
Type B: tumors limited to the tympanomastoid area
Type C: tumors involving the infralabyrinthine region
Type D1: tumors with an intracranial extension less than 2 cm in diameter
Type D2: tumors with an intracranial extension greater than 2 cm in diameter
The Glasscock-Jackson scheme is divided for tympanicum and jugulare tumors. The following glomus jugulare scheme includes a superscript for the degree of intracranial extent (e.g., a paraganglioma type IV 2.0 is a lesion with 2 cm of intracranial extension):
Type I: small tumor involving the jugular bulb, middle ear, and mastoid process
Type II: tumor extending under the internal auditory canal; may have intracranial extension
Type III: tumor extending into the petrous apex; may have intracranial extension
Type IV: tumor extending beyond the petrous apex into the clivus or infratemporal fossa; may have intracranial extension
Glomus jugulare tumors with advanced stage (Fisch class C and D, Glasscock-Jackson class II-IV) are typically treated surgically with a classic Fisch infratemporal fossa approach (types A or B). Key to the surgical treatment of glomus jugulare tumors is gaining safe access to surgical margins while preserving vital nerves and vessels. The classic approaches described are associated with surgical morbidity, including facial nerve dysfunction owing to translocation, sacrifice of the external auditory canal, hearing loss, anterior dislocation of the mandible to gain access to the petrous portion of the carotid artery, and cerebrospinal fluid (CSF) leak. In addition, some tumors with intracranial extent can require a two-stage procedure to minimize complications.
We have used a simpler classification of tumors in this location, and modifications and additions to the ELITE approach—including transsigmoid, transjugular, infrajugular, and high cervical approaches—are tailored to provide needed exposure for tumor resection ( Fig. 54.1 ):
Type A: main tumor mass is intradural—primarily an ELITE approach is used
Type B: main mass is combined intradural and intrajugular—transjugular and ELITE approaches are used
Type C: main mass is intradural, intrajugular, and high cervical, typical of extensive glomus jugulare tumor—ELITE, transsigmoid, transjugular, infrajugular, and high cervical exposure approaches are used
The ELITE approach provides a one-stage transjugular posterior infratemporal fossa approach to resect complex glomus jugulare tumors and other lesions of the jugular foramen. This method can avoid rerouting of the facial nerve and may preserve the external auditory canal and hearing function. The petrous portion of the carotid is accessible without anterior dislocation of the mandible. Two modifications of this approach are described. The dorsolateral ELITE approach is used for intradural, type A tumors involving the vertebral artery; posterior inferior cerebellar artery aneurysms; foramen magnum meningiomas; and glomus jugulare tumors with a large intradural component. The anterolateral ELITE approach is used for type B and C tumors, including extensive glomus jugulare tumors. The approach can be broken down into a series of maneuvers, as follows:
Postauricular infratemporal incision
Retrolabyrinthine mastoidectomy
High cervical exposure
Skeletonization and (if needed) anterior translocation of the facial nerve
Lateral suboccipital and transcondylar transtubercular exposure
Removal of the internal jugular vein (IJV), jugular bulb, and sigmoid sinus
Intradural exposure
A preoperative arteriogram and embolization are performed 48 hours before the surgical procedure. Facial nerve monitoring is carried out in all cases. The surgeon may consider additional monitoring, including auditory brainstem evoked potentials, somatosensory evoked potentials, motor evoked potentials, and monitoring of CN X, XI, and XII. An electromyographic endotracheal tube can be used for CN X monitoring, and electrodes placed directly into the sternocleidomastoid muscle (SCM) and the tongue can be used for CN XI and XII monitoring. Before the incision, perioperative antibiotics and corticosteroids are administered.
Patient positioning and incision differ for the dorsolateral and anterolateral modifications. For the dorsolateral ELITE procedure, the patient is placed in a lateral decubitus position ( Fig. 54.2A and B ). For the anterolateral approach, the patient is positioned in a supine fashion with the head displaced away from the side of the lesion. A shoulder roll is placed to aid with the high cervical portion of the procedure and to raise the shoulder on the side of the lesion. Obese patients and those with very short necks may be placed in a lateral position if necessary. The patient should be padded and secured for ease of rotation of the table throughout the case.
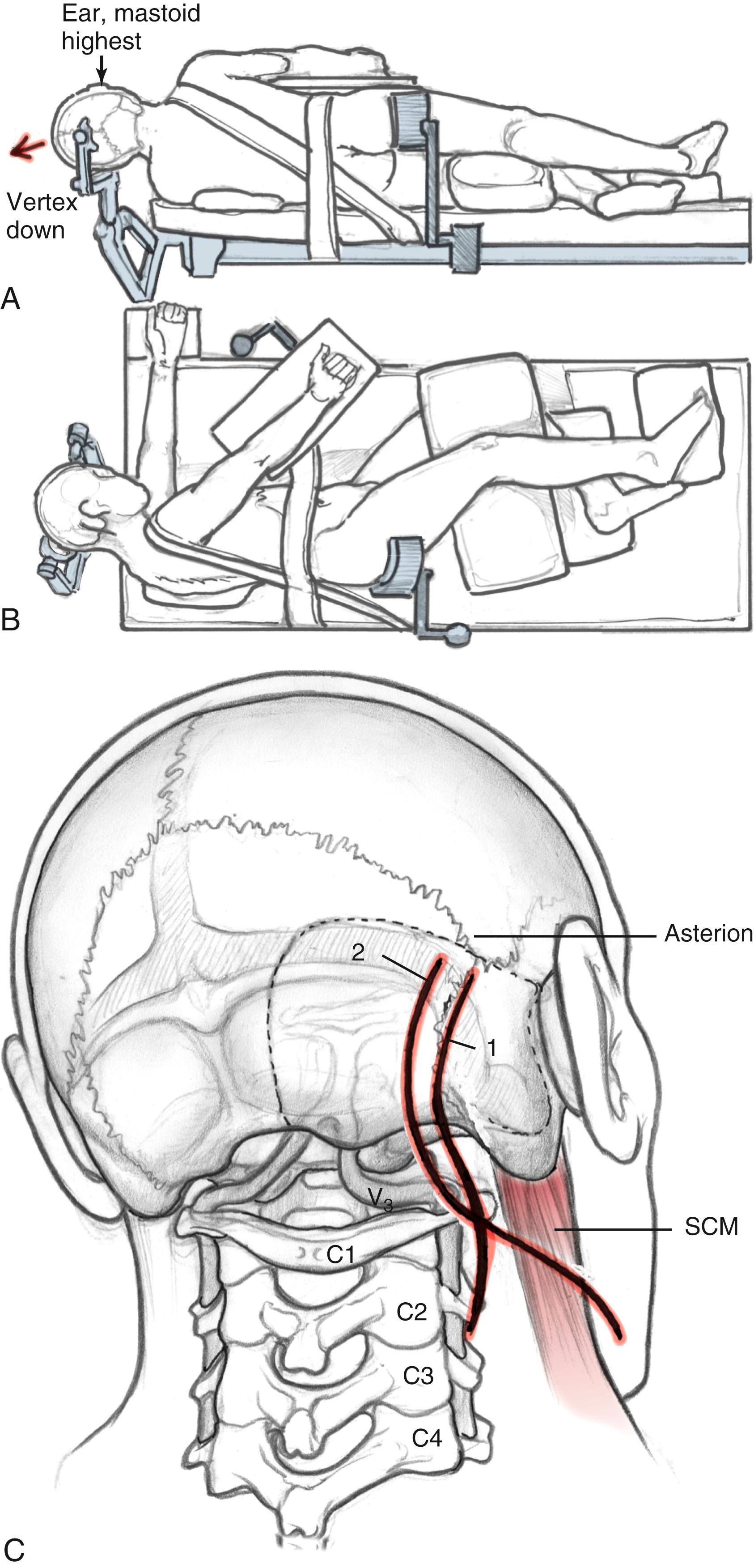
The incisions are shown in Fig. 54.2C . For the dorsolateral approach, a lazy S incision is used, 1 to 2 cm posterior to the mastoid bone, and extending inferiorly along the hairline. For the anterolateral ELITE procedure, a retroauricular curvilinear C -shaped, or question mark–shaped, skin incision is begun approximately 2 to 3 cm posterior to the upper border of the ear. Inferiorly, this incision is carried down into the neck, traversing the border of the SCM and running parallel to the body of the mandible, approximately two fingerbreadths below. The skin flaps are raised. For the dorsolateral approach, the SCM is retracted anteriorly; for the anterolateral approach, the SCM is retracted posteriorly. The inferior edge is raised in the subplatysmal plane. Superiorly, the temporoparietal fascia is elevated as a separate flap under the skin flap, after it is dissected off the bone with the periosteum.
The anterior flap is now reflected, and the posterior auricular muscle should be visible behind the external auditory canal. Inferiorly, the greater auricular nerve should be seen crossing the SCM approximately 3 cm below the mastoid tip. This nerve can be preserved for use as an interposition graft, if needed. The posterior flap is displaced, and the posterolateral neck muscles are reflected to expose the mastoid more fully. This group of muscles is composed of three layers ( Fig. 54.3 ). The superficial layer consists of the SCM and splenius capitis muscles and the middle layer of the longissimus capitis and semispinalis capitis. These muscles are reflected to expose the deep layer, which consists of the rectus capitis posterior major, the obliquus capitis superior, and the obliquus capitis inferior muscles. These muscles form the suboccipital triangle ( Fig. 54.4A ). The styloid diaphragm also is identified in this region. Beneath that diaphragm runs the occipital artery as it courses beneath the posterior belly of the digastric muscle ( Fig. 54.4B ).
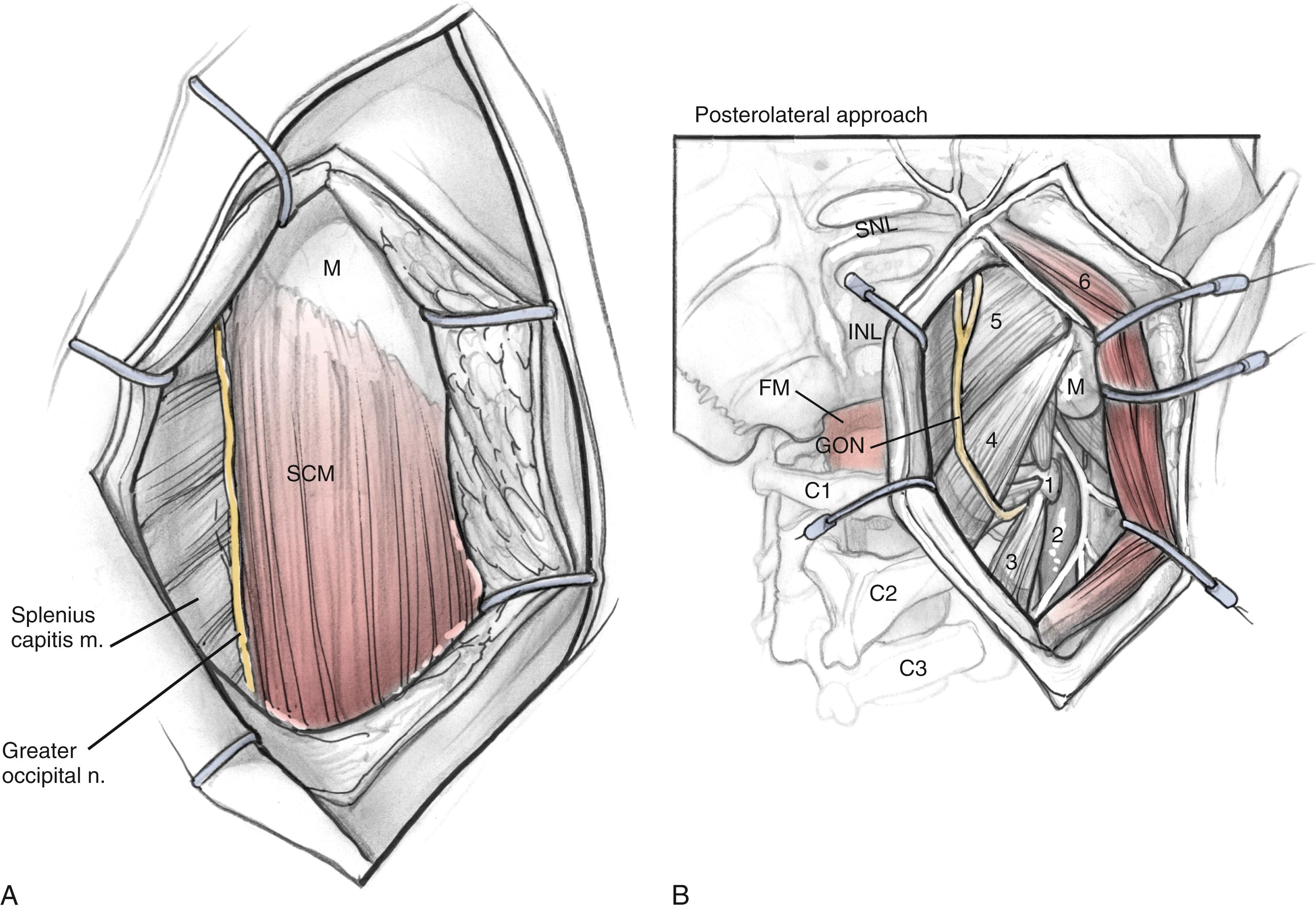
A mastoidectomy is performed. The sigmoid sinus and jugular bulb are thoroughly skeletonized. The mastoid cells overlying the middle fossa dura, presigmoid dura, superior petrosal sinus, and retrosigmoid dura are removed.
The digastric ridge is widely exposed and identified. The bony labyrinth should be skeletonized for ease of retrolabyrinthine dissection, as should the facial nerve. The retrofacial air tract must be drilled to expose the jugular bulb completely. The bone between the sigmoid sinus and jugular bulb can be quite adherent to these structures, and the dissection is prone to bleeding.
For the resection of glomus jugulare tumors, a high cervical approach is necessary to identify the lower cranial nerves (CN IX–XII) and to gain lower control of the internal carotid artery and IJV. When the subplatysmal flaps are raised, the posterior angle of the mandible is identified as the anterior limit of dissection. The mastoid tip is defined as the posterior limit. Dissection is carried anterior to the SCM to identify the posterior belly of the digastric muscle. The stylomastoid diaphragm is removed and the occipital artery, running just below, is controlled. The digastric muscle can be displaced superoanteriorly to cover the facial nerve and gain access to the transverse process of C1. The accessory nerve can be seen coursing posteroinferiorly from the lateral point (defined as 3 to 15 mm inferolateral to the transverse process of C1). The accessory nerve is usually seen running lateral to the IJV, medial to the digastric. The hypoglossal nerve is identified as it courses over the IJV. The carotid sheath is opened to identify the vagus nerve. The stylohyoid, stylopharyngeus, and styloglossus muscles lie anterior to the carotid artery. Detaching the styloid process from the base of the cranium improves the exposure of the carotid canal at the skull base. The glossopharyngeal nerve is identified 1 to 2 cm from this point, crossing the carotid inferomedially. Gently retracting the IJV can help expose the carotid branch of CN IX.
The facial nerve is skeletonized in its vertical portion and the mastoid tip removed. The styloid process (already detached) is reflected anteriorly to expose the infratemporal carotid artery. Facial nerve rerouting is rarely performed. Instead, a limited anterior translocation (approximately 5 mm) may be performed to gain anterior exposure in cases of tumor extension to the internal carotid artery (C7 portion). Preserving the periosteum of the facial nerve can minimize the risk of postoperative paresis with this procedure. The carotid canal is drilled out to expose the petrous carotid. CN IX exits dorsal to the carotid in this location, and it can be injured without proper care. The Jacobson nerve can be seen branching from CN IX before reaching the jugular bulb.
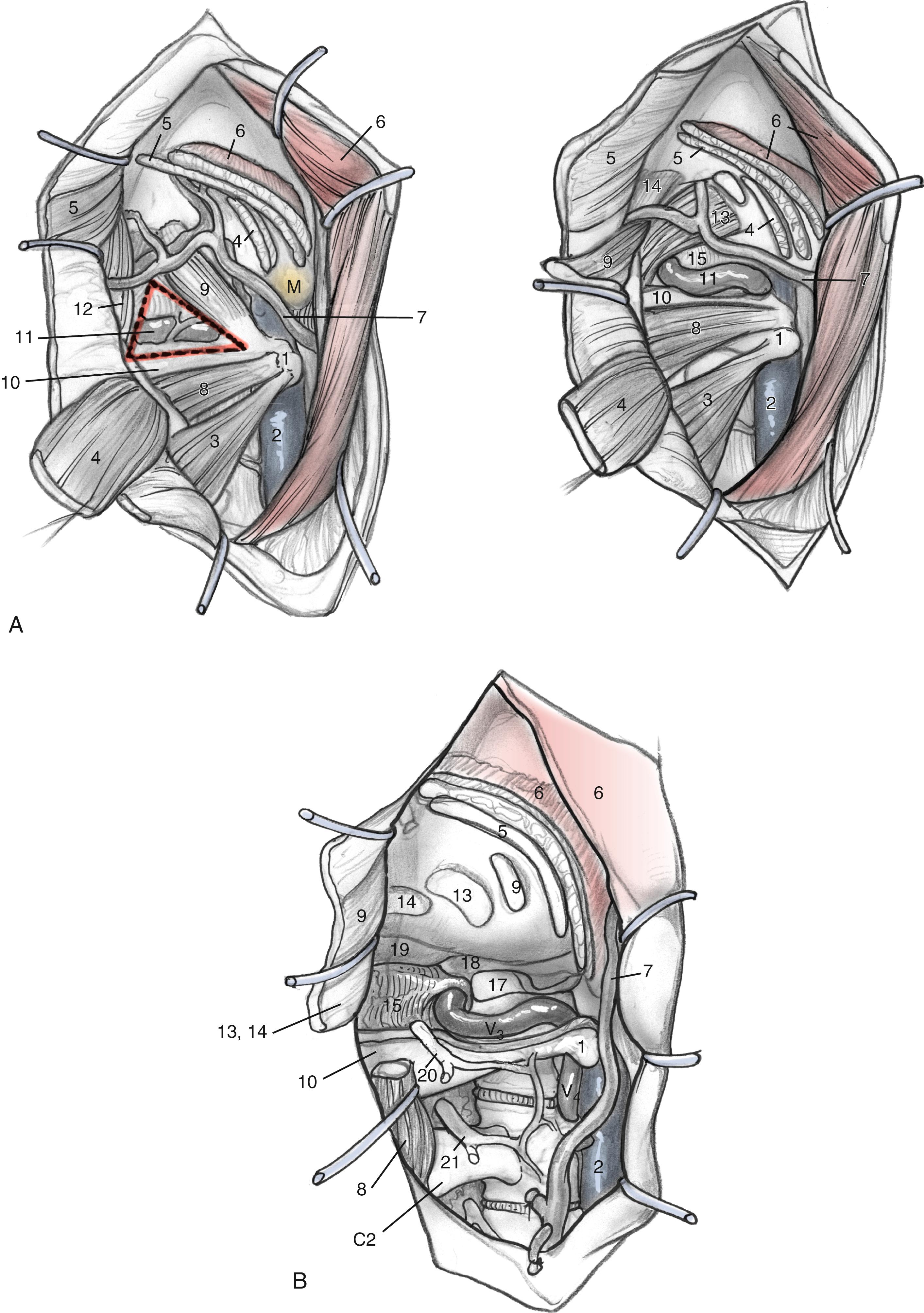
It is essential to identify the V 3 segment of the vertebral artery to avoid injury during this exposure. V 3 can be identified in one of three ways: by dissection of the suboccipital triangle, by identification of the vertebral sulcus or J groove of the C1 lamina, or by the use of a Doppler probe ( Fig. 54.5 ). For identification of the suboccipital triangle, the deep layer of muscles mentioned previously serves as the guide to this anatomical region. The rectus capitis posterior major inserts superiorly on the nuchal line and attaches inferiorly on the spinous process of C2. The obliquus capitis inferior muscle inserts superiorly on the transverse processes of C1 and inferiorly on the spinous process of C2. The obliquus capitis superior attaches superiorly at the temporooccipital suture and inferiorly on the transverse process of C1. The suboccipital triangle is opened by detaching the superior and inferior oblique muscles off the transverse process of C1 and reflecting them posteriorly. The rectus capitis posterior major is reflected posteriorly as well, after freeing it from the inferior nuchal line; this helps identify the C1 lamina and the vertebral artery.
Alternatively, the vertebral artery can also be identified by following the C1 lamina from the posterior point along its superior edge until the vertebral sulcus or J groove is identified. The vertebral artery is encased by a venous plexus as it exits the foramen transversarium of C1, and it then travels in the vertebral sulcus until its medial turn into the atlanto-occipital membrane. The venous plexus can be avoided by dissecting in the subperiosteal plane as the vertebral artery is freed from the sulcus. Venous plexus bleeding can be controlled by using bipolar cautery or absorbable knitted fabric (Surgicel). Care should be taken to avoid injuring an extradural posterior spinal artery or the posterior inferior cerebellar artery. The atlanto-occipital membrane is incised to expose the craniocervical dura.
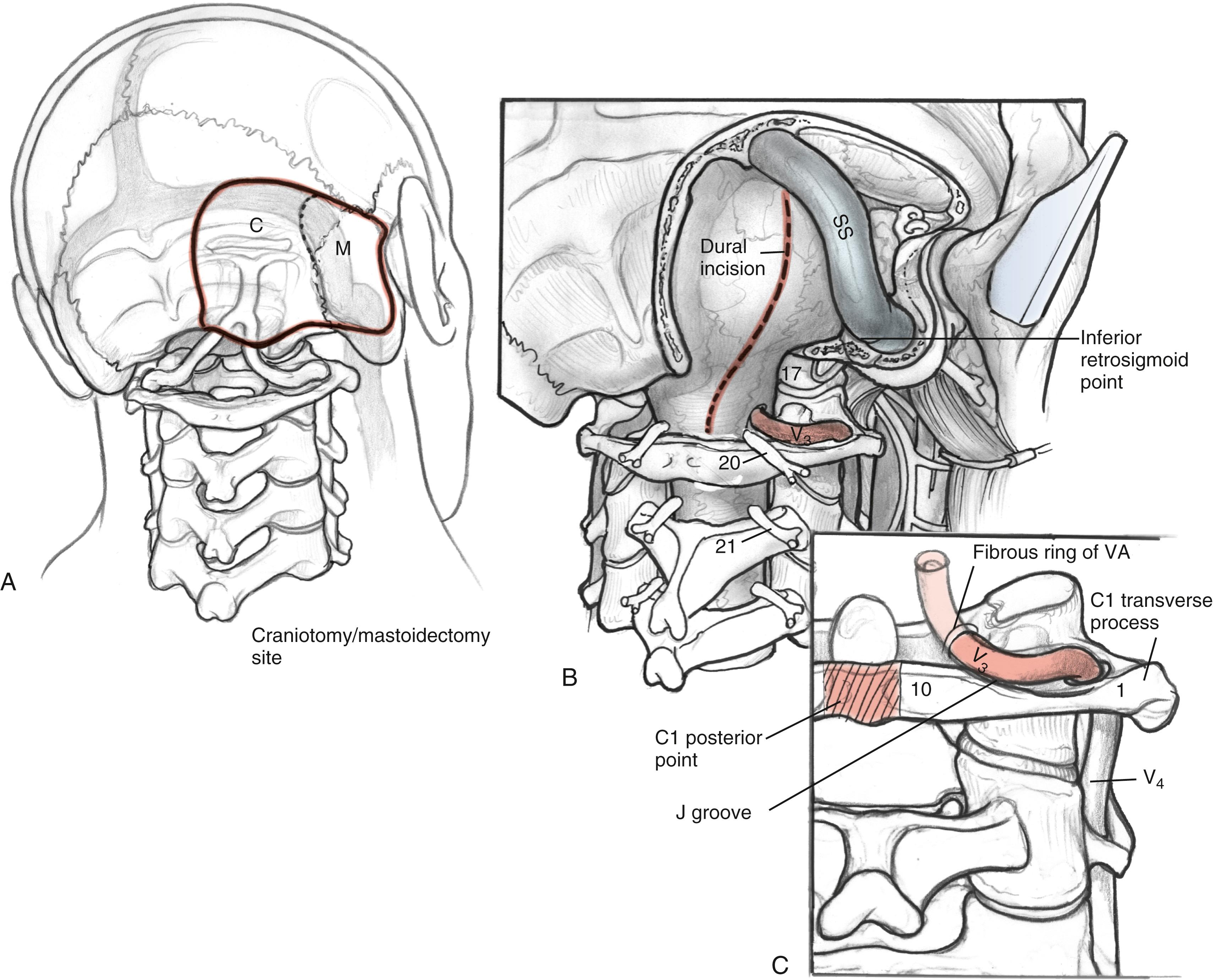
The lateral suboccipital craniotomy is performed with a craniotome. The boundaries of the craniotomy are about 3 cm posterior to the retrosigmoid line, superior to the nuchal line (transverse sinus), inferior to the posterior rim of the foramen magnum, and lateral to the occipital condyle. Exposure of the jugular bulb and sigmoid sinus is crucial. The posterior condylar emissary vein is encountered as it exits the jugular bulb, and it can be controlled by bipolar cautery and Surgicel packing. In most cases, the lateral edge of the foramen magnum must be removed entirely.
The posterior and medial one-third of the occipital condyle is removed in front of the C1 dura, using a high-speed diamond drill. Resecting more than half of the condyle can cause instability of the craniovertebral junction, and stabilization should be considered on resection. As the condyle is removed, the surgeon proceeds through a cortical layer leading to a cancellous bony layer. Venous bleeding may be present and should be controlled with bone wax and Surgicel before proceeding. The next cortical layer overlies the hypoglossal canal. The hypoglossal nerve, branch of the ascending pharyngeal artery, and venous plexus of the canal are located within the canal. Because the canal sits superior to the condylar facet and inferior to the jugular tubercle and is directed slightly cephalad at a 60-degree anterolateral plane, skeletonization of the canal to its lateral extent indicates a one-third resection of the superomedial portion of the occipital condyle. The removal of this portion of the condyle provides easier access to the ventral foramen magnum ( Fig. 54.6A and B ).
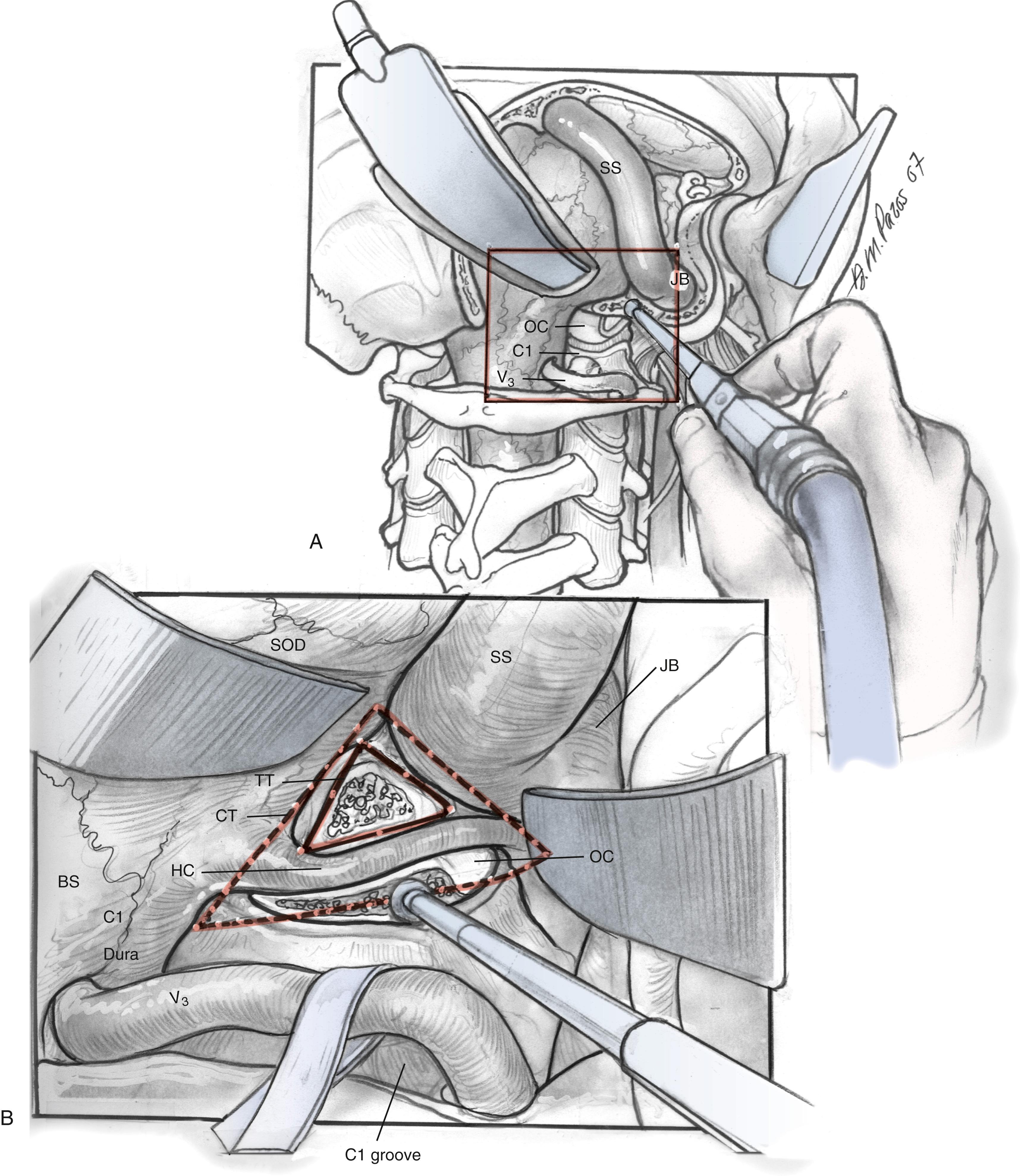
The dissection continues superiorly toward the jugular tubercle. The jugular tubercle is a smooth osseous protrusion on either side of the occipital bone. The base of the tubercle is defined by the hypoglossal canal and extends anteriorly 20 mm toward the clivus. Reduction of the tubercle permits access to the anterior clivus. Because the tubercle lies medial to the jugular foramen, CN IX, X, and XI are at risk as the tubercle is drilled out. The risk of injury can be minimized by drilling within a core of the tubercle and working anteriorly, deeper toward the clivus, minimizing heat and potential stretch injury to the nerves. Drilling should be carried 20 to 25 mm deep toward the clival junction. A 3-mm smooth diamond burr is ideal here, and it can be sized down to a 2-mm burr. Remaining thin bone can be dissected; inner cortical bone can be preserved to avoid damage to CN IX and X. It is important to have flat access from the lateral edge of the foramen magnum under CN IX, X, and XI to the inferior clivus and ventral medullary area ( Fig. 54.6C–E ). Between the jugular bulb and the C1 transverse process, the small rectus lateralis muscle must be removed. Occasionally, the C1 transverse process is shaved to expose the V 3 –V 4 genu for wider exposure. In this process, care should be taken not to injure CN XI.
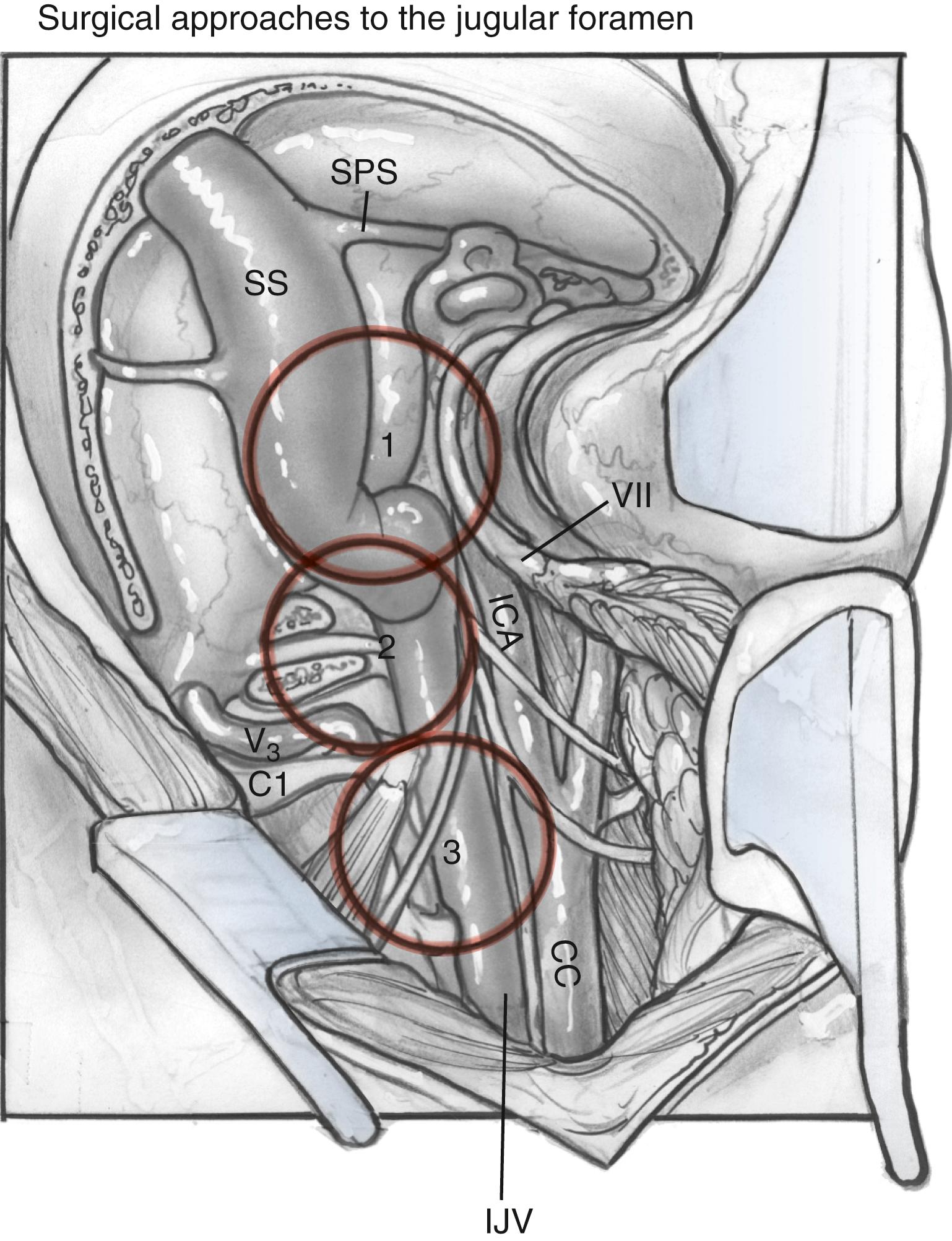
Adequate exposure of the sigmoid sinus, jugular bulb, and IJV, and all the lower cranial nerves should allow for the preservation of vital functions and easy identification of the tumor within these structures. Fig. 54.7 shows the exposure of these structures provided by this approach. The approach used for the resection of large glomus jugulare tumors includes (1) the suprajugular, retrofacial, infralabyrinthine approach; (2) the infrajugular transcondylar approach; and (3) the high cervical approach.
Before tumor resection, it is important to control the arterial feeders to the tumor. After obvious arterial feeding vessels have been ligated, the IJV is then ligated inferiorly. The sigmoid is occluded above the tumor mass. The IJV lateral wall can be incised, and the tumor specimen can be resected up to the jugular bulb. The medial wall of the jugular bulb is preserved, and the tumor is excised from the pars nervosa. The dural sheath over the lower cranial nerves should be left intact. Bleeding should be controlled with gentle packing because bipolar cautery can damage the cranial nerves.
The intradural portion of the tumor is addressed next. The dural incision is curvilinear and is placed several millimeters posterior to the sigmoid sinus. It is carried inferiorly to the point where the vertebral artery pierces the dura. The dura is reflected back with multiple tacking sutures. With sufficient drilling of the occipital condyle and jugular tubercle, the accessory nerve is readily identifiable. Dissection can expose the medullary branch of the accessory nerve, vagal vein and vagus nerve, and glossopharyngeal nerve. The tumor is dissected free from the cranial nerves after the jugular foramen is inspected for further extension. The blood supply from the posterior inferior cerebellar artery can be cauterized ( Fig. 54.8 ).
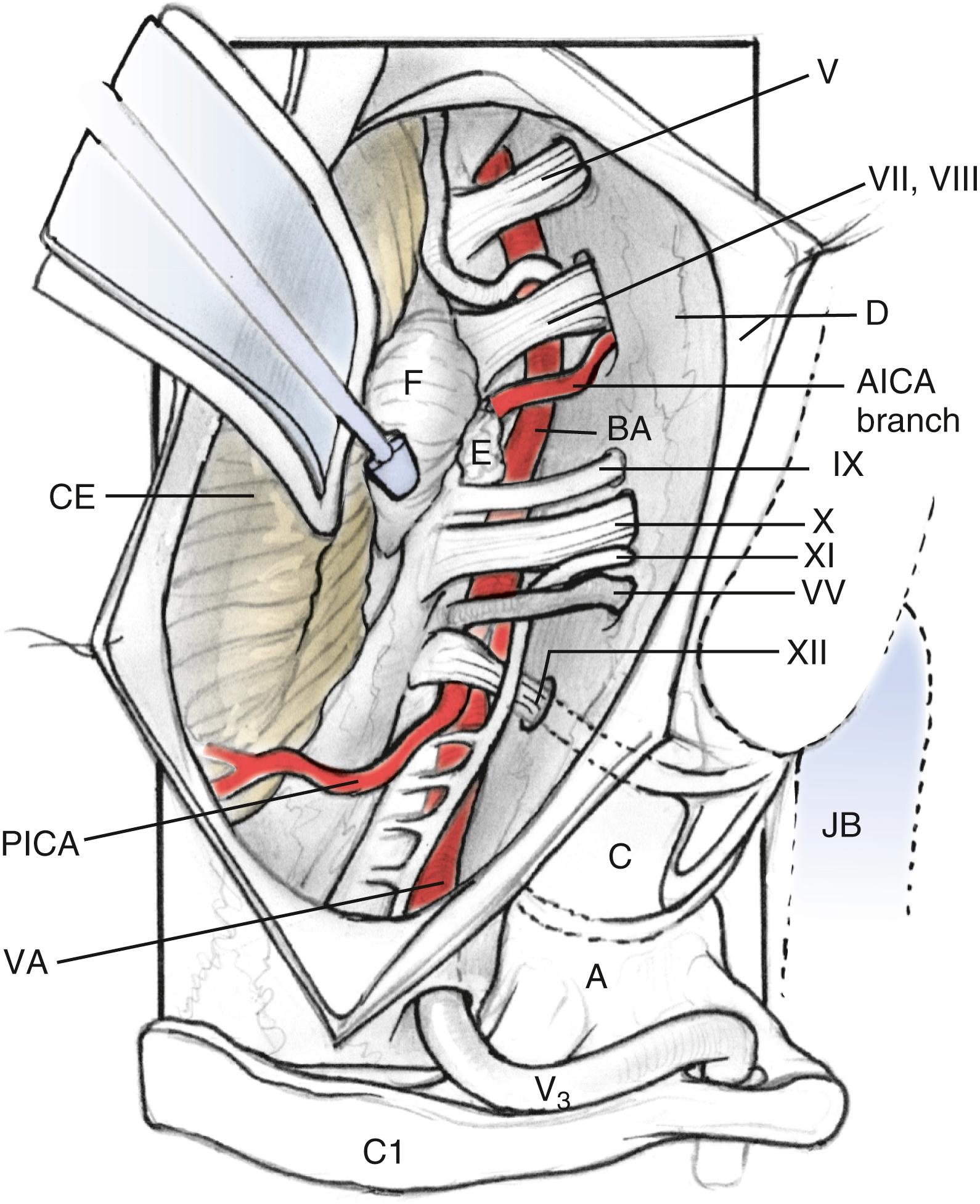
The lower clivus can be accessed, if necessary, by inferior translocation of the lower cranial nerves and anterior translocation of the vertical petrous segment of the internal carotid artery. The dural sleeve over the lower cranial nerves can protect them from trauma. The resection of the clivus can continue toward the petrous apex.
The wound is irrigated and reconstruction of the cranial base performed to obtain a watertight dural closure. The options include fascia for smaller defects, a pericranial flap, or a free flap reconstruction for larger defects. The use of vascularized myofascial flaps for reconstruction is thought to reduce the incidence of postoperative CSF leak. Abdominal fat should be harvested to fill the mastoid defect. Lumbar drainage is used in most patients.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here