Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
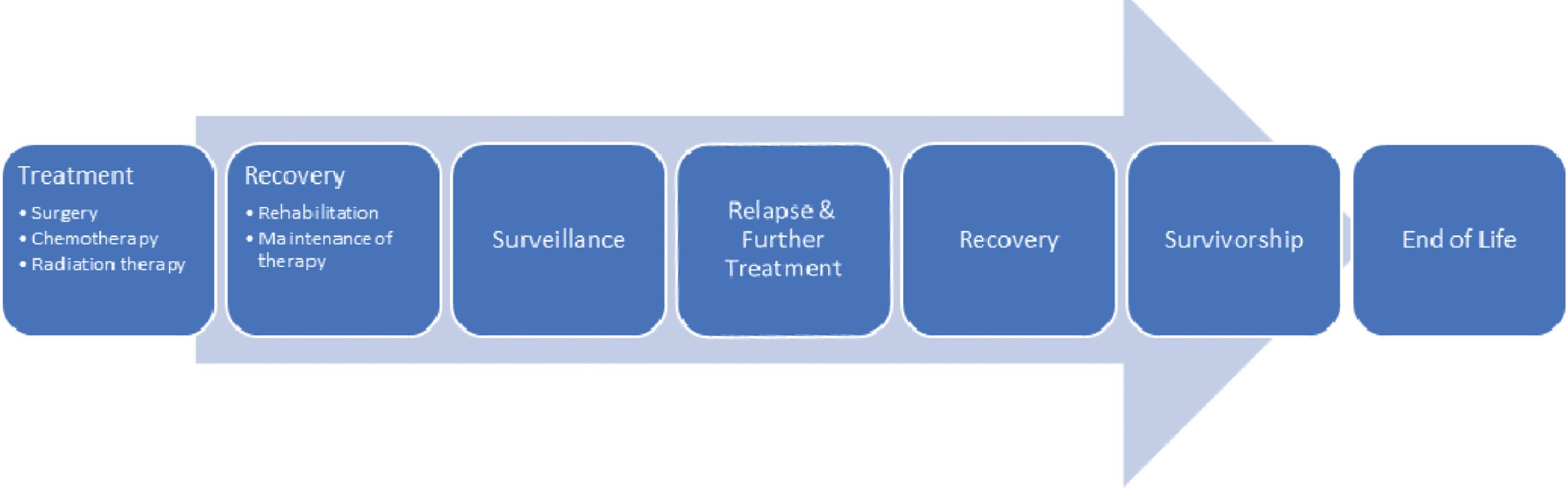
Cancer is a disease process that results when cellular changes cause uncontrolled growth and division of cells. It continues to be a major health problem and is the second leading cause of death in the United States. The three most prevalent cancers in 2019 were prostate, colon, and rectum, and melanoma of the skin among males, and breast, uterine corpus, and colon and rectum among females. Common side effects of cancer and its treatment are pain, fatigue, and emotional distress, , all of which can affect functional status. Pain can be problematic at different phases of cancer treatment. Van den Beuken‐van Everdingen et al. reported pain prevalence rates of 39% after curative treatment; 55% during anticancer treatment; 66% in advanced, metastatic, or terminal disease; and 51% in all cancer stages. There was a prevalence rate of 38% of moderate to severe pain in studies reporting pain intensity. Hematologic patients also have high prevalence rates of pain at diagnosis, during therapy, and in the last month of life. Despite the availability of opioids and other treatment strategies, undertreatment of cancer pain remains problematic, , with some authors estimating that one in three patients does not receive an analgesic prescription to match the reported level of pain. Despite increased attention to cancer pain, pain prevalence in cancer patients has not significantly changed over the last decade compared to the prior four decades. This continues into survivorship, where the prevalence of pain also is high. At initial diagnosis, oncology patients face a continuum of care that may progress from disease-oriented, curative, life-prolonging treatment through symptom-oriented, supportive, and palliative care extending to terminal-phase hospice care ( Fig. 33.1 ).
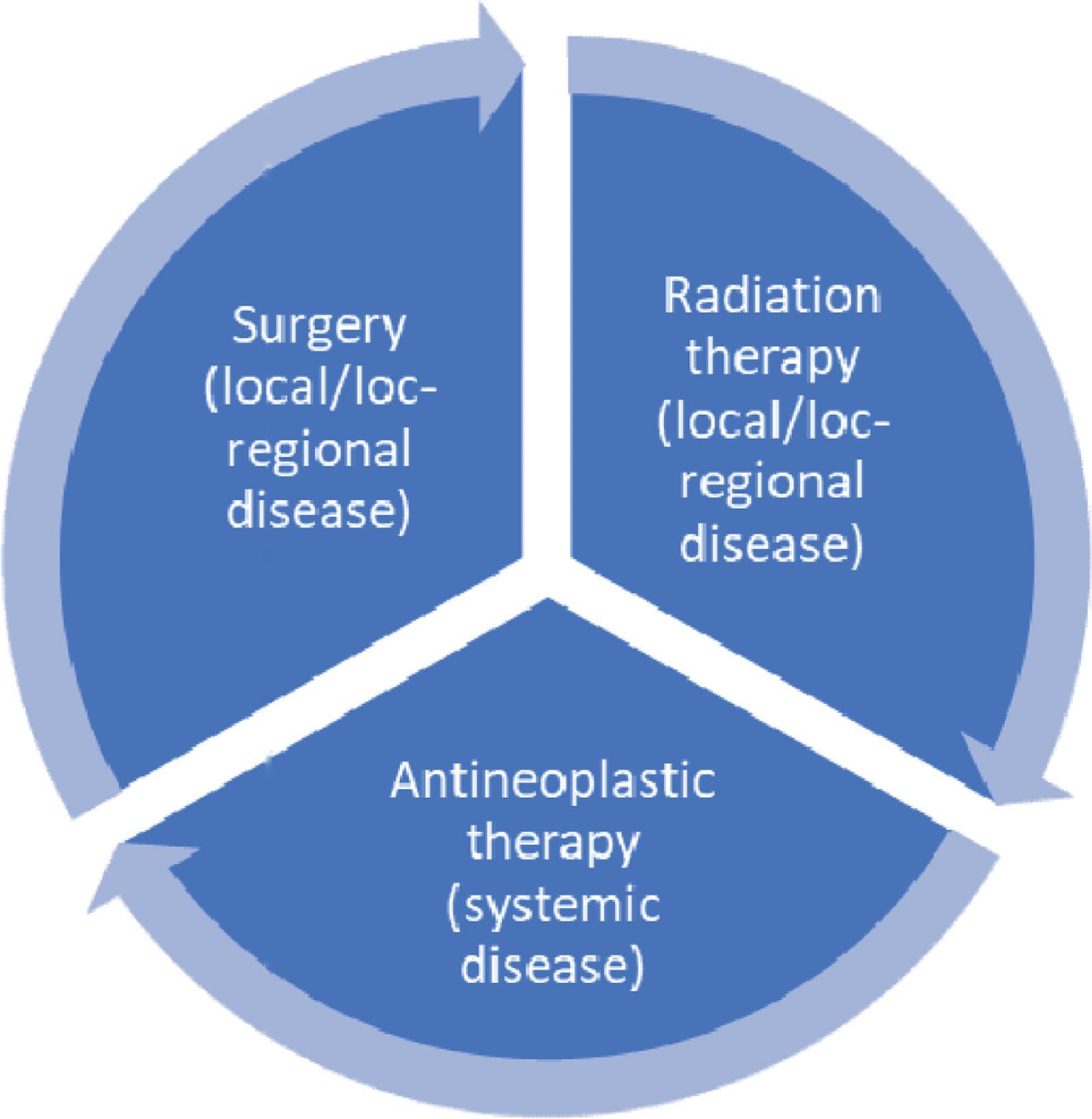
Treatments are usually multifaceted and often include surgery, radiation therapy, and systemic treatment, which includes the use of chemotherapeutic cytostatic drugs, targeted therapy with small molecule inhibitors, monoclonal antibodies that involved targeted immunotherapy, and inhibition of tumor-associated angiogenesis or immune regulation by checkpoint inhibitors ( Fig. 33.2 , Table 33.1 ).
| Surgery | Open versus minimally invasive Primary tumor resection Resection of metastases Cytoreduction Palliative Reconstructive |
| Radiation Therapy | External beam (photon, proton, neutron) * Internal radiation therapy (brachytherapy) Systemic radiation therapy (radioactive iodine, radium, radiostrontium) |
| Chemotherapy | Cytotoxic (oral, IV, injection, intrathecal, intraperitoneal, intra-arterial) |
| Immunotherapy | Immune checkpoint inhibitors Cellular immunotherapy (CAR T-cell, TIL) Monoclonal antibodies (rituximab) Cancer treatment vaccines Immune system modulators (cytokines – interferons, interleukins; BCG; immunomodulatory drugs – thalidomide, lenalidomide, pomalidomide, which are also angiogenesis inhibitors) |
| Targeted Therapy | Tyrosine kinase inhibitors (e.g. erlotinib for EGFR, venetoclax for BCL2, imatinib for BCR-ABL mutations in CML, ibrutinib in CLL) Monoclonal antibody-drug conjugate (e.g. brentuximab-vedotin for lymphoma, obinutuzumab-ozogamicin for ALL) Angiogenesis inhibitors primarily target VEGF (e.g. bevacizumab, everolimus) Hormone therapy Gene therapy |
| Hormone Therapy | Oral Injection Surgery (oophorectomy, orchiectomy) |
| Stem Cell Transplant | Autologous Allogenic Syngeneic |
| Precision Medicine | Certain tumors, including melanoma, some leukemias, and breast, lung, colon, and rectal cancers, may be tested for certain genetic changes |
Once treatment is completed, patients frequently require an extended interval of surveillance, resulting in additional treatments for recurrences or the appearance of new diseases or problems. Advances in cancer prevention and treatment with an improved understanding of the biology of cancer have resulted in improved patient survival and quality of life. Cancer survival has improved since the mid‐1970s for all of the most common cancers except uterine cervix and uterine corpus, and there has been an overall drop of 29% in the mortality rate since 1991. Even after completion of care, pain can be a persistent problem, and the ability to address this is an increasing issue because of advances in cancer treatment with a record number of survivors in the United States. Pain management should be an integral component of the cancer care continuum. For oncology patients, the ultimate goal is to cure disease and pain, often an undesirable consequence of disease or treatment. Although pain management should be integrated into oncology care, it should not be the focus of care in contrast to many chronic pain models. Interdisciplinary collaboration is essential for the comprehensive care of cancer patients with pain. Aggressive therapy of both cancer and pain is mutually beneficial and is best done by skilled, interdisciplinary teams that understand and respond to the changing demands of oncology care that include pain management.
The most successful strategy for managing tumor-related pain is identifying the source and directing appropriate anti-tumor measures (which may include all care options in oncology) toward the source. Symptomatic management of the pain may be appropriate while implementing these measures. However, such measures may not be appropriate for patients with pre-existing chronic non-cancer-related pain who present for oncology care. These issues can usually be differentiated by a comprehensive pain assessment with a clear understanding of the current tumor location(s) and correlation with the patient’s pain complaint. The goal of any pain management strategy is to manage pain that allows for the optimization of function and enable an acceptable quality of life. Although a reduction in pain intensity is ideal, the guiding principle for pain management should be an improvement in function. Adequate management of cancer pain depends on making an accurate diagnosis of the underlying cause(s) of pain and contributing factors. In every situation, this requires a comprehensive assessment of these issues. The diagnosis will include a reason for the pain complaint, a summary of the current disease status, the appropriateness and efficacy of the current pain medication regimen, and an assessment of functional and social status. It is important to recognize that the source of pain in the cancer patient is usually not a single entity but often has multiple sources that may result from different issues such as tumor staging, treatment phase, preexisting conditions causing chronic pain, and posttreatment complications. Tumor-related pain may be classified according to the tissue source as nociceptive (somatic or visceral) or neuropathic. Once a treatment plan is implemented, assessment and re-assessment at regular intervals are key to ensuring that treatment is appropriate and safe, as well as minimizing and addressing side effects related to treatment throughout care. The concurrent use of adjunctive, specialized, or complementary therapies should be considered, and referral for specialized surgical, anesthetic, or psychological intervention benefits selective patients. However, most patients can attain adequate management of pain (and improvement in function) using appropriate oral pharmacotherapy. Cancer (tumor-related) pain management guidelines typically incorporate pharmacologic, anesthetic, neurosurgical, and behavioral approaches. The mainstay of cancer pain therapy is pharmacologic, but radiotherapeutic, anesthetic, neurosurgical, psychological, physiotherapeutic, spiritual, and social interventions all play roles in adequate cancer pain management ( Table 33.2 ). The basic principles of tumor-directed pain control include modifying the source of pain by treating cancer, altering the central perception of pain (for example, by the use of analgesics, anti-depressants, anxiolytics, and psychotherapy), and, when appropriate, interfering with nociceptive transmission outside of and within the central nervous system, for example, with anesthetic techniques (e.g. neurolytic celiac plexus block, neuraxial analgesia, and spinal neurolysis), or neurosurgery procedures (e.g. cordotomy, dorsal root entry zone lesioning). Options for disease modification should always be foremost.
In 1986, the World Health Organization (WHO) developed a simple model for the introduction and upward titration of analgesics, which became known as the WHO analgesic ladder. In 2018, WHO updated its guidelines for pharmacologic and radiotherapeutic management of cancer pain (WHO 2018). Pharmacologic options should be used by following the WHO analgesic ladder ( Fig. 33.3 ).
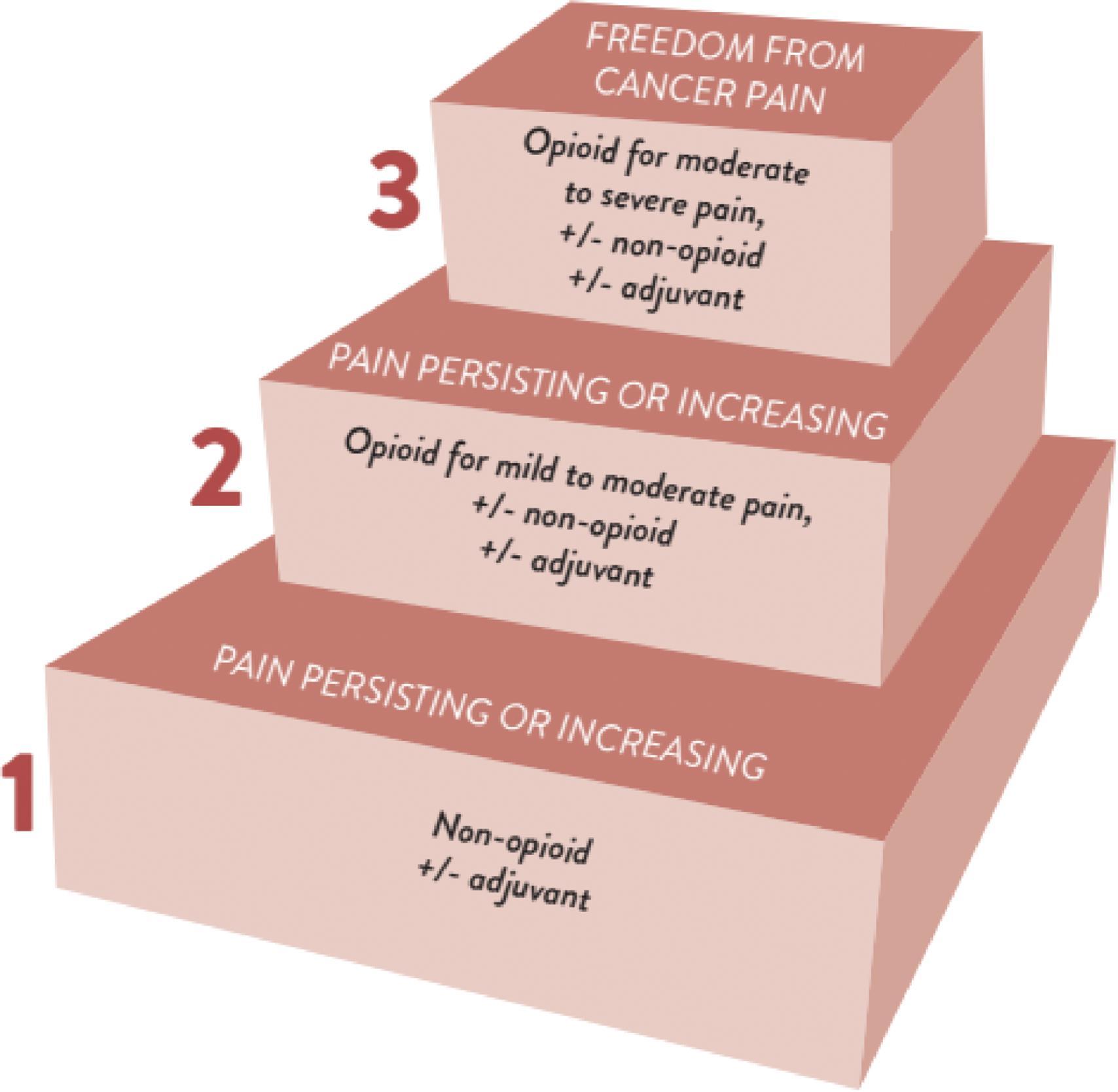
Implementation of the three-step ladder, in combination with appropriate dosage guidelines, should result in adequate pain relief for 70%–90% of patients. Successful implementation of the ladder requires adherence to basic principles of medication use ( Table 33.3 ).
The ladder advocates for the use of analgesics (non-opioids including automatic positive airway pressure [APAP] and nonsteroidal anti-inflammatory drugs [NSAIDs], adjuvant analgesics such as anti-depressants and anticonvulsants, and opioids) should be used incrementally according to pain intensity with the focus on the use of opioids for moderate to severe intensity pain. Opioids are commonly used to treat moderate or severe cancer pain, and the most commonly used opioid drugs are buprenorphine, codeine, fentanyl, hydrocodone, hydromorphone, methadone, morphine, oxycodone, tramadol, and tapentadol. For initiation and maintenance of pain relief, NSAIDs, APAP, and opioids either alone or in combination may be used with a strength appropriate for patient-reported pain severity. The use of steroids for the shortest time possible is recommended if deemed appropriate for care, but evidence for the efficacy of steroids in cancer pain is weak, and the side effect profile of prolonged use needs to be defined. Optimal dosing of steroids likely depends on location and type of pain, presence of or risk of infection, stage of illness, presence of diabetes mellitus, and goals of care. In patients with pain related to bone metastases, bisphosphonates or monoclonal antibodies (denosumab) should be considered to prevent and treat bone pain. Regarding the use of anti-depressants and anti-convulsants for cancer pain management, the WHO report did not make a recommendation for these classes of medications for the treatment of cancer-related neuropathic pain. Adequate data from clinical trials regarding the use of anti-depressants and anti-convulsants in oncology pain was not available. In the absence of clear evidence in favor of use, practitioners may consider an individual trial of therapy and prescribe for those patients without adequate pain relief or have severe/unmanageable side effects to standard analgesic regimens. Drugs or drug classes recommended for the management of neuropathic pain are listed in Table 33.4 .
| Serotonin noradrenaline reuptake inhibitors duloxetine or venlafaxine a | 60–120 mg, once a day (duloxetine); 150–225 mg, once a day (venlafaxine extended release) | First line |
| Tricyclic anti-depressants | ||
| 25–150 mg, once a day or in two divided doses | First line b | Weak Recommendations for Use |
| Capsaicin 8% patches | One to four patches to the painful area for 30–60 min every three months | Second line (peripheral neuropathic pain) c |
| Lidocaine patches | One to three patches to the region of pain once a day for up to 12 h | Second line (peripheral neuropathic pain) |
a Duloxetine is the most studied, and therefore recommended, of the serotonin noradrenaline reuptake inhibitors.
b Tricyclic anti-depressants generally have similar efficacy; tertiary amine tricyclic anti-depressants (amitriptyline, imipramine, and clomipramine) are not recommended at doses greater than 75 mg/day in adults aged 65 years and older because of major anticholinergic and sedative side effects and potential risk of falls; an increased risk of sudden cardiac death has been reported with tricyclic anti-depressants at doses greater than 100 mg daily.
c The long-term safety of repeated applications of high-concentration capsaicin patches in patients has not been clearly established, particularly with respect to degeneration of epidermal nerve fibers, which might be a cause for concern in progressive neuropathy.
N-acetyl-p-aminophenol (APAP) is an antipyretic/analgesic drug that is discussed extensively in Chapter 49. Hepatotoxicity is the most well-known severe adverse effect of APAP. Particularly in cases of overdose, APAP characteristically shows direct hepatocellular injury. Hepatocellular injury may result in elevated aspartate aminotransferase (AST) and alanine aminotransferase (ALT) blood levels, usually 24–48 h after ingestion. Elevated ALT level greater than three times the upper limit of normal was seen in 33%–44% of normal volunteers who were administered 4 g of APAP daily for 14 days. The maximum daily therapeutic dose of 3900–4000 mg was established in separate actions in 1977 and 1988, respectively, via the Food and Drug Administration (FDA) monograph process for nonprescription medications. The 3900 mg maximum daily dose, as recommended originally, was deemed to be safe and is five to seven times lower than the estimated median lethal dose (LD50) of 400 mg/kg. Some studies have indicated that daily APAP doses of 4000 mg for 6 to 12 months demonstrated that no patient exceeded liver function tests beyond two times the upper limit of normal. The FDA has conducted multiple advisory committee meetings to evaluate acetaminophen and its safety profile and has suggested (but not mandated) a reduction in the maximum daily dosage from 3900–4000 mg to 3000–3250 mg, which appears based on the potential of an overdose occurring if a patient was not using acetaminophen properly or if, unknowingly, a patient was using multiple acetaminophen-containing products. The concern was not with therapeutic dosing (≤4000 mg/24 h) but with excessive dosing when two or more products containing acetaminophen are taken inadvertently and the potential for hepatotoxicity with chronic use at excessive doses. In a Cochrane review, there was no high quality evidence to support or refute the use of APAP alone or in combination with opioids for the first two steps of the three-step WHO cancer pain ladder. It was not clear whether any additional analgesic benefit of acetaminophen could be detected in the available studies. In an era of concern for opioid prescribing, there is a tendency to prescribe regular and higher doses of APAP (up to 4 G/day) in cancer patients. Toxicity from high daily doses of APAP is of concern. Unintentional fever suppression in patients receiving chemotherapy is also an issue. APAP should be used cautiously and not for extended periods in oncology patients receiving active treatment.
NSAIDs exhibit their action by inhibiting the cyclooxygenase (COX) enzyme, which consists of two isoforms named as COX-1 and COX-2. Most NSAIDs are nonselective and inhibit both COX-1 and COX-2. Celecoxib is the only marketed drug belonging to the class of selective COX-2 inhibitors approved by the FDA. COX-2 overexpression is demonstrated throughout various steps of tumorigenesis to metastasis, and COX-2 expression directly co-related with increased prostaglandin levels in neoplastic tissues, particularly in the gastrointestinal tract, and possibly breast. Long-term use of (low dose) aspirin and non-selective NSAIDs are potential candidates for chemoprevention of gastrointestinal cancer. The anti-inflammatory activity of NSAIDs in descending order is indomethacin > diclofenac > piroxicam > ketoprofen > lornoxicam > ibuprofen > ketorolac > acetylsalicylic acid. The greater the degree of COX-1 block, the greater the tendency to cause gastrointestinal (GI) ulceration and the promotion of bleeding. From the first day of use, all NSAIDs increase the risk of GI bleeding, myocardial infarction, and stroke. The risk of bleeding is low for patients using NSAIDs intermittently. NSAIDs increase the risk of GI bleeding by inhibiting platelet aggregation, causing gastric mucosa lesions, ulceration, overt bleeding, enteropathy, and occult bleeding associated with a decrease in hemoglobin. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occur in approximately 1% of patients treated for three to six months and in about 2%–4% of patients treated for one year. Even short-term (zero to three days) treatment with NSAIDs was associated with increased risk of bleeding compared with no NSAID use in patients receiving antithrombotic therapy. Non-selective NSAIDs increase the risk of a GI bleed four fold, whereas COX-2 inhibitors increase this risk three fold. Co-prescription of NSAIDs with corticosteroids increases bleeding risk 12-fold, spironolactone 11-fold, and SSRIs 7-fold. Monotherapy with non-selective NSAIDs increased the risk of upper GI bleeding by an incidence rate ratio (IRR) of 4.3 to a greater extent than monotherapy with COX-2 inhibitors (IRR, 2.9) or low-dose aspirin (IRR, 3.1). Combination therapy generally increased the risk of GI bleeding; concomitant NSAID and corticosteroid therapies increased the IRR to the greatest extent (12.8) and produced the greatest excess risk relative excess risk because of an interaction (RERI of 5.5. Concomitant use of NSAIDs and aldosterone antagonists produced an IRR for upper GI bleeding of 11.0 (RERI 4.5). The excess risk from the concomitant use of NSAIDs with selective serotonin reuptake inhibitors (SSRIs) was 1.6, whereas that from the use of COX-2 inhibitors with SSRIs was 1.9, and that for the use of low-dose aspirin with SSRIs was 0.5. Excess risk of concomitant use of NSAIDs with anticoagulants was 2.4, of COX-2 inhibitors with anticoagulants was 0.1, and of low-dose aspirin with anticoagulants was 1.9. Concomitant use of non-selective NSAIDs or low dose aspirin, but not COX-2 inhibitors, with corticosteroids, aldosterone antagonists, or anticoagulants produces a significant excess risk of upper GI bleeding. Castellsague et al. pooled relative risks (RRs) of upper GI complications associated with individual NSAID use. Agents such as celecoxib and ibuprofen have a low RR (1.5 and 1.8, respectively), while piroxicam and ketorolac have a higher RR (7.4 and 11.5, respectively).
Approximately 10% of the total drug-induced hepatotoxicity is NSAID-related. Sulindac and diclofenac are the NSAIDs most associated with hepatotoxicity, but virtually all NSAIDs have been linked to at least rare cases of clinically apparent drug-induced liver injury. The most common adverse effects affect the gastric mucosa, renal system, cardiovascular system, hepatic system, and hematologic system. The mechanism of injury to the liver is usually hepatocellular, but cholestatic injury can also occur. COX-2 selective inhibitors and non-selective NSAIDs are associated with an increased risk of acute cardiovascular events. Even one week of NSAID use is associated with an elevated risk of myocardial infarction (MI), and this phenomenon may apply to those with no prior history of MI. While the risk increases with increasing dosage, the risk appears to plateau after a one-month duration of use. In older patients with arthritis and established coronary artery disease or risk factors who used chronic NSAID therapy (celecoxib 100–200 mg twice daily, ibuprofen 600–800 mg thrice daily, or naproxen 375–500 mg twice daily), naproxen and ibuprofen at moderate doses were associated with more cardiovascular events that with celecoxib. The vascular risks of high-dose diclofenac (150 mg/day) and possibly ibuprofen (2400 mg/day) are comparable to coxibs. In contrast, high-dose naproxen (1000 mg/day) is associated with less vascular risk than other NSAIDs. NSAIDs can cause serious renal adverse effects, which include sodium and water retention with edema, worsening of heart failure, hypertension, hyponatremia, hyperkalemia, acute kidney injury, renal papillary necrosis, and acute interstitial nephritis. Acute forms of kidney injuries are transient and often reversible upon drug withdrawal. Approximately 1%–8% of patients taking NSAIDs develop renal adverse effects such as a reduction in glomerular filtration rate, acute renal failure, renal papillary necrosis, nephrotic syndrome, acute interstitial nephritis, and chronic renal failure. NSAIDs, unlike aspirin, bind reversibly at the active site of COX-1, usually depressing platelet thromboxane formation to the degree that platelet function is impaired for only a portion of the dosing interval. Platelet function normalizes within 24 h of the last dose of ibuprofen in healthy volunteers who took 600 mg q 8 h for one week.
Studies on the use of NSAIDs in cancer (including both aspirin and non-aspirin NSAIDs) show inconsistent and contradictory findings in terms of the role of these drugs in cancer, with some reporting an increased risk in certain types of cancer and others showing a reduction in cancer risk. Although many NSAIDs are available to treat various painful conditions, it is unclear which agent is most clinically efficacious for relieving cancer-related pain and if there are clinical differences between these agents that justify their cost differences. In a Cochrane review of the use of NSAIDs either alone or in combination with opioids, the quality of evidence supporting the use of NSAIDs was poor and suggested that moderate to severe cancer pain was reduced to no worse than mild pain after one to two weeks in approximately 1:3 patients. One in four patients stopped taking NSAIDs because the drug did not work, and 1:20 stopped because of side effects. In effect, there is no evidence to support or refute the use of NSAIDs alone or in combination with opioids for cancer pain. , The long-term use of NSAIDs for pain management is not advisable in patients with cancer, particularly because of the additional bleeding risks in cancer patients undergoing active treatment with myelosuppressive regimens and the probability that a patient with cancer may be on a broad regimen of medications, some of which may increase NSAID-related toxicity (myocardial infarction, gastrointestinal bleeding, and renal failure).
The pharmacologic mechanism of action responsible for therapeutic effects in pain management is unclear ( Table 33.5 ) and may involve enhancement of GABAergic inhibition, decreased glutamatergic excitation, modulation of voltage-gated sodium and calcium channels, and effects on intracellular signaling pathways.
| Blocks voltage-gated sodium channels. | Epilepsy (especially partial seizures), bipolar disorder, and trigeminal neuralgia. | Yes | Carbatrol or Tegretol-XR | Side effects include hyponatremia, leukocytosis, thrombocytopenia, dizziness, drowsiness, ataxia, nausea/vomiting, and blurred vision. Dosing starts at 100–200 mg twice a day and is titrated up by 200 mg/day every three to five days until pain relief is achieved. The maximum dose is 1200 mg/day. | Phenytoin |
| Partial and generalized seizures; monotherapy for trigeminal neuralgia. | Yes | ER | Non-linear pharmacokinetics. | Oxcarbazepine | |
| Blocks voltage-gated sodium channels. | Focal seizures with or without secondary generalization. | Yes | ER | ER peak concentrations are 19% lower than with OXC IR. It can be started at 300 mg twice a day and titrated up by 300 mg/day every three days for therapeutic effect. The maximum dose is 2400 mg/day. | Lamotrigine |
| Blocks Na+ channels, neuronal α-4-β2-nicotinic acetylcholine receptors may be a target. | Partial seizures, the generalized seizures of Lennox-Gastaut syndrome, primary generalized tonic-clonic seizures. Mood stabilizer in bipolar. | Yes | XR | Risk of Stevens-Johnson. Start at low doses (25 mg/day) with weekly titration to target dose. Syndrome, blood dyscrasia. | Lacosamide |
| Selectively enhances the slow inactivation of voltage-gated sodium channels and interacts with the collapsin-response mediator protein-2. | Primary generalized tonic-clonic seizures, partial onset seizures. | Dosing starts at 50 mg twice daily and can be increased to 400 mg/day. Abrupt discontinuation can precipitate seizures. No convincing evidence of efficacy in neuropathic pain and fibromyalgia at doses of 200–400 mg daily. | Gabapentin | ||
| Binds to calcium channels and modulating calcium influx. Exert analgesic effect through high affinity binding and modulation of the calcium channel α2-δ proteins in the dorsal root ganglion. | Partial seizures, postherpetic neuralgia. | Yes | ER | Compared with other anti-convulsant drugs, the α2-δ ligands do not have significant drug interactions, predominantly because of their lack of hepatic metabolism or their modulation of cytochrome P450 activity. Nonlinear pharmacokinetics (absorbed in the proximal small bowel by the L-amino acid transporter, which is a saturable mechanism). |
Pregabalin |
| Binds to calcium channels and modulating calcium influx. Exert analgesic effect through high affinity binding and modulation of the calcium channel α2-δ proteins in the dorsal root ganglion. | Painful diabetic peripheral neuropathy, postherpetic neuralgia, partial onset seizures, fibromyalgia, neuropathic pain associated with spinal cord injury. | Yes | ER | Compared with other anti-convulsant drugs, the α2-δ ligands do not have significant drug interactions, predominantly because of their lack of hepatic metabolism or their modulation of cytochrome P450 activity. Unlike gabapentin, it has linear pharmacokinetics over its recommended dose ranges. Start at low doses (50 mg daily), adding 50 mg each week on a twice a day basis to a max of 600 mg per day. | Topiramate |
| Blocks activity-dependent, voltage-gated sodium channels; enhance the action of GABA receptors; inhibit L-type voltage-gated calcium channels; presynaptically reduce glutamate release, and postsynaptically block kainate/α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors. | Generalized tonic-clonic seizures, partial seizures, migraine. | Yes | ER | Dosing starts at 50 mg/day and can be titrated up to 400 mg/day. No convincing evidence of efficacy in neuropathic pain. Serious adverse events thought to be related to topiramate included convulsion and bradycardia plus syncope. Additional adverse effects include sedation, nausea, diarrhea, and metabolic acidosis. | IR = Immediate release.ER/XR = Extended release.DR = Delayed release. |
Antiepileptic drugs (AEDs) are often administered as polytherapy or combined with other treatments. The pharmacokinetics of carbamazepine is nonlinear because of autoinduction that completes within three weeks and can result in a threefold increase in elimination. It is extensively metabolized in the liver, primarily by CYP3A4. Because of this, it is readily inducible and potently inhibited. Therefore it is subject to many pharmacokinetic drug-drug interactions. As well as being an enzyme-inducing drug, carbamazepine is affected by the autoinduction of CYP3A4-mediated metabolism. This mainly occurs at the beginning of treatment when clearance can increase up to three times during the first two to three weeks. Carbamazepine, oxcarbazepine, phenobarbital, and phenytoin may reduce the levels of cyclosporin, tacrolimus, and corticosteroids, delaying the immunosuppressant effect by up to ten days. Gabapentin, lacosamide, levetiracetam, pregabalin, and vigabatrin are essentially not associated with clinically significant pharmacokinetic interactions.
Gabapentinoids include gabapentin and pregabalin. The α2δ-1, commonly known as a voltage-activated Ca2+ channel subunit, is a binding site of gabapentinoids. Gabapentinoids commonly cause adverse effects such as sedation, confusion, dizziness, ataxia, visual disturbances, and altered cognition. Combined with opioids, this side effect profile can be potentiated. For first-time pregabalin users, dizziness and somnolence were reported by 25% and 14% of patients, respectively. Gabapentinoids have the potential to be abused with an estimated prevalence of 1.6% in the general population with ranges from 3% to 68% among opioid abusers, and are associated with euphoria, sedation, and dissociation. Gabapentin was misused primarily for recreational purposes, self-medication, or intentional self-harm and was misused alone or combined with other substances, especially opioids, benzodiazepines, and/or alcohol. Risk factors for abuse include a history of substance abuse, particularly opioids, and psychiatric comorbidities. Sedation and dizziness are relatively common, and some patients experience cognitive difficulties while taking these drugs. In 2008, the FDA issued a warning on all AEDs that these drugs have an increased risk of suicidal thoughts and behaviors. The relative risk was higher in the epilepsy patient group when compared to the groups of patients receiving AEDs for psychiatric and other conditions. However, patients and clinicians should not avoid the use of AEDs because of the risk of suicidality. However, the high prevalence of comorbid behavioral health issues in patients treated with AEDs suggests that the association is confounded by indication, preexisting mental health conditions, and medications received. In a cohort analysis, Patorno et al. found an increased risk for these events in new users of gabapentin, lamotrigine, oxcarbazepine, and tiagabine compared with topiramate. A secondary analysis confirmed the increased risk and identified an excess of 5.6 cases of attempted or completed suicide per 1000 person-years among new users of gabapentin, 10.0 cases per 1000 person-years among new users of oxcarbazepine, and 14.1 cases per 1000 person-years among new users of tiagabine compared with topiramate. The risk remained increased for gabapentin in subgroups of younger and older patients, patients with a mood disorder, and patients with epilepsy or seizure disorders.
Gabapentinoids are indicated for the treatment of epilepsy, peripheral and neuropathic pain, fibromyalgia, and generalized anxiety disorder in adults. Recommendations for use and doses in neuropathic pain are listed in Table 33.4 . For gabapentin, the only pain related indication approved by the FDA is postherpetic neuralgia. For pregabalin, FDA-approved indications related to pain are limited to postherpetic neuralgia, neuropathic pain associated with diabetic neuropathy or spinal cord injury, and fibromyalgia. Despite these limited indications, gabapentin and pregabalin are widely prescribed off-label for multiple different pain complaints. A common mistaken perception is that an effective drug for one type of neuropathic pain is beneficial for all neuropathic pain, regardless of underlying etiology or mechanism. Recommendations across guidelines consistently endorse gabapentinoids as first line agents for general neuropathic pain complaints but that the overall quality of these guidelines is poor. Similarly, the evidence does not support gabapentinoid therapy for low back pain or radiculopathy, suggesting that pregabalin was ineffective. A Cochrane review showed that some patients with neuropathic pain would benefit substantially from pregabalin, while more are having moderate benefit, and many are without any benefit. Gabapentin can provide good levels of pain relief to some people with postherpetic neuralgia and peripheral diabetic neuropathy, but evidence for other types of neuropathic pain is very limited. , Similarly, there is little evidence to support the effectiveness of oxcarbazepine in painful diabetic neuropathy, neuropathic pain from radiculopathy, and a mixture of neuropathies. Overall, there is low quality evidence that gabapentinoids are effective in reducing pain intensity in patients with cancer pain.
Anti-depressants can be classified into different classes based on their mechanisms of action ( Table 33.6 ).
| Desvenlafaxine | Major active metabolite of venlafaxine. X10 times more potent inhibiting serotonin than norepinephrine uptake. | Selective serotonin reuptake inhibitors (SSRIs) |
| Proven efficacy in panic disorder, generalized anxiety disorders, obsessive-compulsive disorder, and bulimia, with encouraging findings in social phobia, post-traumatic stress disorder. Side effects of SSRIs may include nausea, sedation, decreased libido, sexual dysfunction, headache, and weight gain. | Citalopram | |
| QT prolongation. | Escitalopram | (S)-enantiomer of citalopram. |
| Fluoxetine | Can cause activation (take during daytime). Because of a long half-life and active metabolites, rare symptoms of discontinuation or withdrawal symptoms if abruptly stopped. |
|
| Paroxetine | Can be sedating and thus taken at bedtime. Its anticholinergic side effects can affect cognition, especially in the elderly. Discontinuation/ withdrawal symptoms could develop if abruptly discontinued. | |
| Sertraline | ||
| Serotonin receptors antagonist with serotonin reuptake inhibition (SARI) | Trazodone | Moderate-to-strong serotonin receptor(s) antagonism with a weak serotonin reuptake transporter inhibition. Side effects include sedation and drowsiness, tachycardia, and priapism (rarely). |
| Tricyclic anti-depressants (TCAs) | ||
| “Gold standard” anti-depressants. Among the best studied and most commonly used are the first-generation TCAs, which are mixed reuptake inhibitors with both NE and serotonergic 5-HT effects such as amitriptyline, imipramine, and doxepin. | Amitriptyline | |
| Most potent anticholinergic; cautious use in elderly. | Clomipramine | |
| Desipramine | ||
| As secondary amine, fewer anticholinergic effects. | Doxepin | |
| Most potent antihistamine effects. | Imipramine | |
| Nortriptyline | Acts by inhibiting the reuptake of NE and serotonin 5-HT and possesses central anticholinergic activities. As secondary amine, fewer anticholinergic effects. | |
| Desipramine | ||
| Noradrenergic α2-receptor antagonist with specific serotonergic receptors-2 and -3 antagonism (NASSA) | Mirtazapine | |
| Enhances release of NE and 5-HT1A-mediated serotonergic transmission. Side effects include drowsiness, dizziness, anxiety, confusion, increased appetite, and weight gain. | ||
The two classic mechanisms are demonstrated by tricyclic antidepressants and monoamine oxidase inhibitors. The nonclassical anti-depressants, the most widely prescribed agents, are SSRIs and dual serotonin norepinephrine reuptake inhibition (SNRI). Anti-depressants have virtually no antinociceptive effects but are prescribed for neuropathic pain. Tricyclic anti-depressants and serotonin noradrenaline reuptake inhibitors are used to treat chronic pain, such as neuropathic pain and fibromyalgia. SSRIs are generally the first line anti-depressants for the treatment of depression in cancer patients because of their tolerability profile. SSRIs, which are frequently used to treat depression, are not effective against chronic pain. The inhibitory effects of anti-depressants for neuropathic pain manifest more quickly than their anti-depressant effects, suggesting different modes of action. The analgesic effect on chronic pain may manifest in as little as a few days to one week, whereas the anti-depressant effects may take two to four weeks. Anti-depressants that inhibit the reuptake of both noradrenaline and 5-HT have stronger analgesic effects than a drug that selectively inhibits the reuptake of only one of these neurotransmitters, and norepinephrine plays a greater role than 5-HT in the analgesic action. Anti-depressants have several other actions in addition to increasing monoamines that may contribute to the inhibition of neuropathic pain, such as sodium channel blockers, N -methyl-d-aspartate (NMDA) receptor antagonists, effects on α-1 receptors, calcium channel blockers, potassium channel activators, modulation of the adenosine system, and activation of GABA-B receptors. Serotonin and dopamine may reinforce the noradrenergic effects to inhibit neuropathic pain. Increasing norepinephrine in the spinal cord by reuptake inhibition directly inhibits neuropathic pain through α2-adrenergic receptors. Also, increasing norepinephrine acts on the locus coeruleus and is an essential element of both the ascending and descending pain modulator systems regulated by these anti-depressants. TCAs (amitriptyline, nortriptyline, and desipramine) have been the first line treatment for neuropathic pain for many years, but only a minority of people will achieve satisfactory pain relief. Finnerup et al. reviewed the pharmacotherapy for neuropathic pain in adults. Outcomes were generally modest even for effective drugs: in particular, NNTs were 3.6 (95% confidence interval [CI] 3.0-4.4) for TCAs and 6.4 (95% CI 5.2-8.4) for the SNRI anti-depressants duloxetine and venlafaxine. There was no evidence for a dose-response effect for amitriptyline. Combined NNH was 13.4 (9.3-24.4) for TCAs and 11.8 (9.5-15.2) for SNRIs. Low-dose amitriptyline (25 mg/day) for chronic low back pain did not improve outcomes at six months with no improvement in pain intensity, but there was a reduction in disability at three months. When anti-depressants are used for chronic pain, the most prevalent adverse effects from anti-depressants were dry mouth, dizziness, nausea, headache, and constipation. Risk for withdrawal because of adverse effects was highest for desipramine followed by venlafaxine and duloxetine. TCAs are associated with increased mortality in cases of intentional or accidental overdose, and their side effects profile and tolerability require careful dose titration and frequent monitoring, which substantially limits their wide use. Minor side effects are common and more common with duloxetine 60 mg and particularly with 120 mg daily than 20 mg daily, but serious side effects are rare.
Chemotherapy-induced peripheral neuropathy (CIPN) is common among cancer patients who undergo chemotherapy with platinum analogs, taxanes, vinca alkaloids, epothilone, bortezomib, and thalidomide. Chemotherapeutic agents can damage nervous system structures and, depending on the individual compound, can cause various neuropathies that affect different structures, including large and small fibers and autonomic function. The effects of chemotherapy on the nervous system vary among the different classes of drugs, depending on the specific physical and chemical properties of the drug used and its single or cumulative doses. The mechanism by which chemotherapeutics damages nervous system structures and causes CIPN is multifactorial and involves microtubule disruption, oxidative stress and mitochondrial damage, altered ion channel activity, myelin sheath damage, DNA damage, immunologic processes, and neuroinflammation. The mechanisms of injury are complex and interested readers are referred to the review by Carozzi et al. When patients experience drug-induced chronic neurotoxicity that necessitates intervention beyond dose reduction or discontinuation, the strongest evidence supports the use of duloxetine. However, other anti-depressants have not shown a similar benefit. Furthermore, the best available data support a moderate recommendation for treatment with duloxetine. CIPN trials were also inconclusive regarding tricyclic anti-depressants (such as nortriptyline) and a compounded topical gel containing baclofen, amitriptyline HCL, and ketamine. Anti-convulsants, such as gabapentinoids, have not been proven effective for patients with CIPN. Duloxetine, when used for diabetic peripheral neuropathy and fibromyalgia, has efficacy for pain associated with diabetic neuropathy at doses >60 mg/day (not at lower doses). The effect in fibromyalgia was likely achieved through a greater improvement in mental symptoms than in somatic physical pain.
Opioids are extensively discussed in Chapter 48. Although some suggest that opioids are relatively ineffective for the management of neuropathic in chronic pain, , this has not been our experience and of others in cancer pain. Clinical pathways have emerged as one of the key approaches to minimize unwarranted variation in care and improve both the quality and efficiency of care. When appropriately designed and implemented, oncology pathways are detailed with evidence-based treatment protocols for delivering quality cancer care for particular patient presentations that include the type and stage of disease. Extrapolation from opioid prescribing recommendations, including the guideline for prescribing opioids for chronic pain from the Centers for Disease Control and Prevention, and applying these to the cancer population is problematic as most of the recommendations have not included opioid outcomes in this particular population. Consequently, some professional oncology organizations and societies have issued policy and position statements on opioid therapy emphasizing continued access for cancer-related pain. Continued use of opioids in oncology requires risk evaluation and strategies for continued monitoring in all patients, including those considered at high risk for opioid misuse or abuse or illicit substance use ( Table 33.7 ).
| No initiation of controlled substance prescriptions with ongoing illicit substance use, including alcohol Referral for addiction assessment and management Frequent (at least four times per year or more frequently as clinically indicated) urine drug screening (to include alcohol, illicit drug, comprehensive panel for opioids) Elimination of other psychoactive medications when possible Consequences for unsanctioned substance/non-prescribed medication use, including alcohol |
|
Opioids have been classified as weak and strong opioids, based upon their affinity for the µ-receptor. , Distinct opioids may elicit very different intracellular responses and may ultimately have very different outcomes on behavior, including analgesic ability, addiction liability, and the risk for acute negative side effects such as respiratory depression. The wide-ranging drug-specific differences among opioids in their pharmacokinetics and pharmacodynamics include opioid receptor biases and ligand biases at the opioid receptors, as well as differential interactions with the non-opioid system. These result in differential coupling and activation of intracellular signaling and likely accounts for diverse interactions with various biologic systems, such as the immune system and the dopaminergic reward system. Three distinct opioid receptors have been cloned, mu (MOR), kappa (KOR), and delta (DOR), with different selectivity for individual endogenous peptides and the various opioids. The three opioid signaling systems have unique and counterbalancing roles as they relate to their regulation of pain, stress, and affect. Though the MOR is the main target for opioid analgesics, the DOR and KOR also regulate pain, and analgesia and the relative affinities of opioid analgesics for these receptors confer their unique properties. MOR agonists produce euphoria and promote stress coping. KOR agonists produce dysphoria and are associated with stress and negative affect. DOR is on the opposite end of the continuum describing mood, and DOR agonists have anxiolytic and anti-depressant activity. Opioids mimic endogenous opioids and act by binding to the opioid receptors, thereby activating them with individual differences in receptor binding and signal transduction. Additionally, G protein signaling can be selectively targeted.
Opioids are the mainstay of pharmacotherapy for patients with moderate or more intense pain resulting from virtually any cancer-related etiology. The specific pathogenic mechanism that underlies a patient’s cancer pain should not be a factor in deciding which opioid to use, as the mechanism of pain does not reliably predict the response to opioid therapy. This particularly applies to situations in which tumor-associated neuropathic mechanisms dominate the pain complaint. Opioids should be used as first line therapy in such situations, particularly if the pain is considered moderate to severe in intensity. In oncology, opioids have demonstrable benefits for neuropathic pain. Factors influencing the selection of a particular opioid are listed in Table 33.8 .
All clinicians who prescribe opioids for the treatment of cancer pain should be familiar with at least three different agents appropriate for the management of moderate to severe pain. The regimen for opioid medications should generally provide around the clock analgesia with provision for rescue doses for the management of exacerbations of the pain not covered by the regular dosage. Predicting the effectiveness of extended release opioids requires knowledge of associated pharmacokinetics. The onset time and time to maximal concentration (Tmax) are particularly helpful. For the extended release opioids, the elimination half-life is not particularly useful because of altered absorption characteristics into the central compartment.
Opioids are effective after peripheral (topical, intra-articular), neuraxial (intrathecal, epidural, intracerebroventricular), or systemic (intravenous, oral, subcutaneous, sublingual, intranasal, transdermal) administration, although the final effects are highly dependent on the particular pharmacokinetic and pharmacodynamic features of each drug.
All clinicians involved in the treatment of cancer patients should be familiar with the most common types and dosages of opioids, as well as the management of the most common side effects ( Table 33.9 ).
| Opioid-induced constipation | Enteral activation of opioid receptors resulting in gastric emptying, sphincter tone, and peristalsis Consider peripherally acting MOR antagonist (PAMORA) agents |
|---|---|
| Endocrine effects | ? direct effect on opioid receptors within hypothalamus, pituitary, and testes Hypothalamic effect, gonadotropin-releasing hormone |
| CTZ = Chemoreceptor trigger zone.MOR = Mu opioid receptor. | |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here