Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Most benign and malignant neoplasms of the esophagus are epithelial in origin ( Box 24.1 ). Overall, an estimated 17,650 new diagnoses of esophageal carcinoma, and 16,000 deaths, occurred in the United States in 2019. The incidence of esophageal carcinoma has increased in the United States and Europe in the past 30 years, principally because of a marked rise in Barrett’s esophagus (BE)–associated adenocarcinoma.
Squamous papilloma
Adenoma (gland/duct)
Barrett’s esophagus–associated polypoid dysplasia
Squamous dysplasia
Squamous cell carcinoma NOS
Squamous cell carcinoma variants
Basaloid carcinoma
Spindle cell carcinoma
Verrucous carcinoma
Esophageal carcinoma cuniculatum
Adenocarcinoma NOS
Barrett’s esophagus–associated
Non–Barrett’s-associated
Heterotopia-associated
Esophageal gland duct associated
Adenoid cystic carcinoma
Mixed squamous and glandular tumors
Adenosquamous carcinoma
Mucoepidermoid carcinoma
Neuroendocrine neoplasms
Neuroendocrine tumor (grades 1, 2, 3)
Neuroendocrine carcinoma (grade 3)
Large cell neuroendocrine carcinoma
Small cell neuroendocrine carcinoma
Mixed neuroendocrine–nonneuroendocrine carcinoma
Mixed adenocarcinoma–neuroendocrine carcinoma
Mixed squamous cell carcinoma–neuroendocrine carcinoma
Undifferentiated carcinoma
Lymphoepithelioma-like carcinoma
Choriocarcinoma
Pleomorphic giant cell carcinoma
Metastases
Esophageal squamous papilloma is the most common benign type of epithelial tumor of the esophagus. , The prevalence rate is estimated to be 0.1% to 0.5%, with some suggesting a recent increase in incidence in some patient populations. These lesions may affect patients of all ages and both sexes. Most are asymptomatic, but large lesions may cause epigastric pain, dysphagia, or symptoms related to luminal obstruction. Key features are summarized in Box 24.2 .
Clinical/endoscopic
M = F, wide age range
Asymptomatic, usually incidental finding
Solitary whitish polypoid lesion (up to 5 mm), rarely multiple
Benign
Histological
Mature squamous epithelium and fibrovascular cores
Subtypes: exophytic (most common), spiked/verrucous, endophytic
Special/immunohistochemical stains: none
Molecular
HPV (low risk or high risk) in 10% to 50% of patients
Differential diagnosis
Pseudoepitheliomatous hyperplasia (associated with esophagitis)
Verrucous carcinoma (large plaquelike lesion)
Prognostic factors
(Rare: Multiple papillomas or papillomatosis may require resection)
The pathogenesis of esophageal squamous papilloma is controversial. Many studies have shown an association (10% to 50%) with human papillomavirus (HPV) infection. When detected, both low-risk and high-risk subtypes (e.g., types 6/11 and 16, respectively) have been reported. However, the prevalence of HPV is variable among studies, likely resulting from differences in the techniques used to detect viral antigens or DNA and the possibility that as yet unidentified subtypes of HPV may be involved. Others have proposed that at least a subset of these lesions develop as a result of chronic mucosal irritation, perhaps secondary to gastroesophageal reflux disease (GERD).
Grossly, esophageal squamous papillomas are usually small, discrete, sessile, soft, tan lesions that are most commonly 0.3 to 0.5 cm in size but may be larger. They occur most commonly in the distal or middle esophagus but may develop anywhere in the esophagus ( Fig. 24.1 ). As many as 20% of cases are multiple, and rare cases of diffuse papillomatosis have also been reported. , Microscopically, three distinct histopathological types have been recognized: exophytic, endophytic, and spiked (verrucoid) ( Fig. 24.2 ). The exophytic type, which is the most common, is composed of finger-like papillary fronds. The endophytic type shows a smooth, round surface contour and an inverted papillomatous proliferation. Least common is the spiked or verrucoid type, which has a spiked surface contour, a prominent granular cell layer, and marked hyperkeratosis.
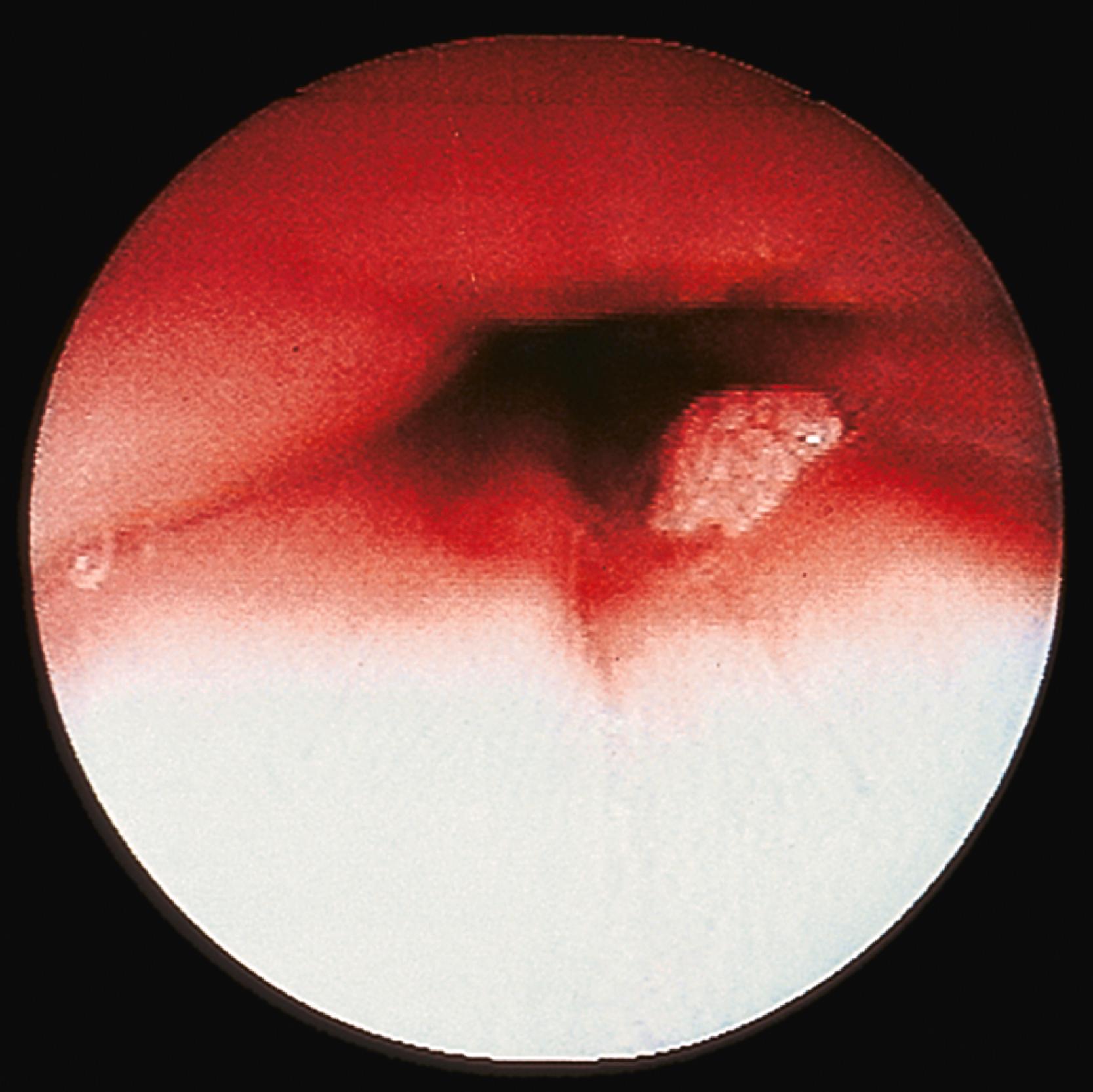
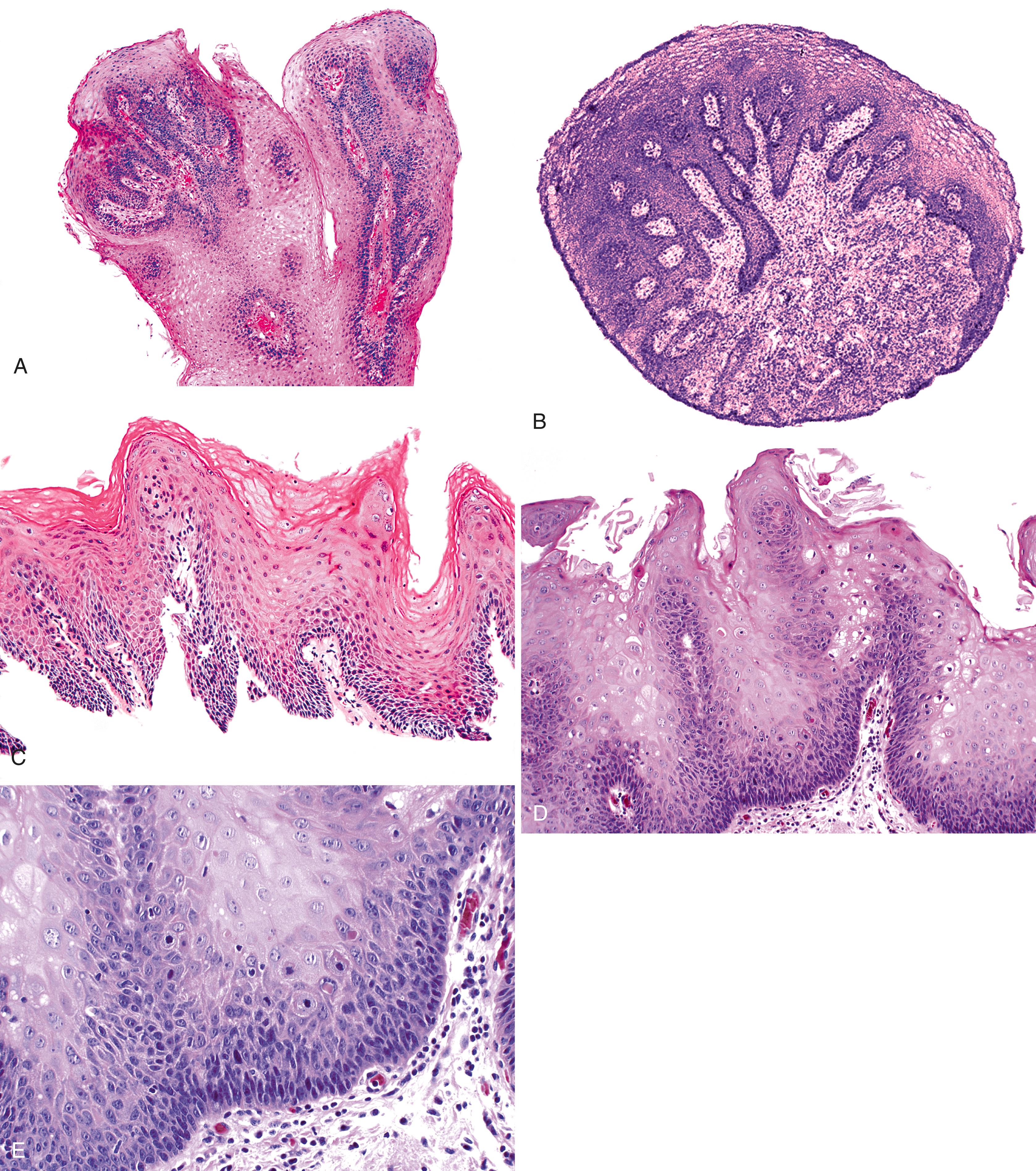
All types are characterized by the presence of a branched fibrovascular core of lamina propria with a variable degree of acute and chronic inflammation and vascular congestion. The overlying squamous epithelium in all types is acanthotic, often reactive, showing a prominent basal cell zone and complete surface maturation, with occasional parakeratosis or dyskeratosis (see Fig. 24.2E ). Up to 50% of lesions show histological features of HPV infection, such as koilocytosis or bi- or multinucleation. Mitoses may be present in the basal and suprabasal epithelial layers, particularly in cases with active inflammation.
A papilloma that is large, is oriented poorly, or shows marked reactive changes may, on occasion, be difficult to distinguish from a well-differentiated squamous cell carcinoma or a verrucous carcinoma. However, esophageal squamous papillomas lack cytological atypia, mitoses in the middle and upper levels of the squamous epithelium, atypical mitoses, and an infiltrative growth pattern characteristic of carcinoma. Furthermore, their gross (endoscopic) appearance is distinctive. In contrast to carcinoma, papillomas are well localized and well demarcated, smooth, discrete polyps without necrosis, stricture formation, or heaped-up borders. They lack the central keratinous crater characteristic of verrucous carcinoma ( Table 24.1 ).
| Feature | Reactive Hyperplasia | Papilloma | Verrucous Carcinoma |
|---|---|---|---|
| Clinical | |||
| Frequency | Common | Common | Rare |
| Age | All ages | All ages | 50-70 |
| Sex | M = F | M = F | M > F (80%) |
| Location | Distal > mid | Mid = distal | Mid |
| Pathological | |||
| Size | Grossly inapparent | <3 cm | >3 cm |
| Polyp | − | ++ | − |
| Ulceration | + | − | +/− |
| Mucosal inflammation | + | − | − |
| Stricture | − | − | +/− |
| Circumferential involvement | − | − | +/− |
| Growth pattern | Endophytic | Exophytic > endophytic > spiked | Exophytic |
| Fibrovascular core | − | ++ | − |
| Pushing margin | − | − | + |
| Extension into submucosa | − | − | + |
| Lymph node metastases | − | − | +/− |
| Cytological atypia | − | − | + |
| Koilocytosis | − | +/− | − |
| Outcome/treatment | |||
| Clinical behavior | Benign | Benign | Aggressive |
| Therapy | None | None/excision | Surgical resection |
Papillomas may also be confused with pseudoepitheliomatous hyperplasia occurring adjacent to a healing ulcer or overlying a granular cell tumor. However, the latter type of lesion lacks a fibrovascular core and other features of HPV infection such as koilocytosis. Unlike esophageal squamous papillomas, pseudoepitheliomatous hyperplasia often shows abundant inflammation and surface erosion.
Squamous cell papillomas are benign lesions with little or no malignant potential. , However, rare cases associated with squamous dysplasia, or even squamous cell carcinoma, have been reported, often in association with extensive esophageal papillomatosis, and may warrant ablation or resection.
Adenomas of the esophagus are defined as benign epithelial proliferations located within the esophagus. True adenomas develop from neoplastic transformation of the submucosal gland or duct system. Polypoid dysplastic lesions arising in BE, which have been referred to in the past as adenomas, are not true adenomas and are better regarded as a neoplastic complication of the underlying metaplastic disorder.
Adenomas that develop from the submucosal glands or gland ducts are rare. They are most often histologically similar to those that arise from the minor salivary glands. This is not surprising because, embryologically, the esophageal submucosal glands are considered a continuation of the minor salivary glands of the oropharynx. They are usually submucosal and develop as well-circumscribed lesions that resemble pleomorphic adenomas, Warthin tumors ( Fig. 24.3B ), or, more rarely, pancreatic or ovary-like serous cystadenomas.
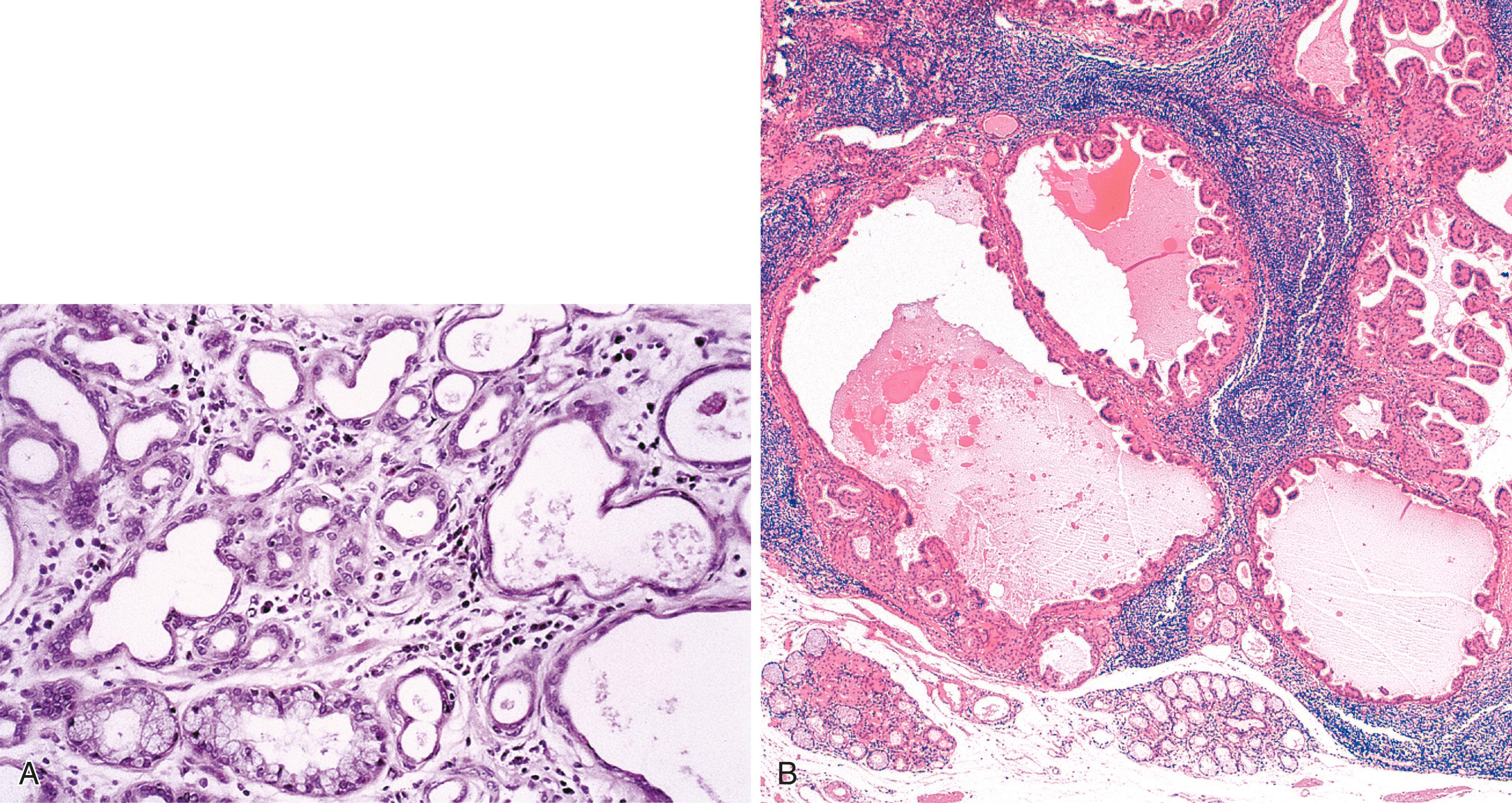
Other examples exhibit tubal, cystic, and papillary growth patterns similar to intraductal papillomas of the breast or sialadenoma papilliferum of the salivary glands. The epithelium typically contains two cell layers (similar to the normal submucosal gland ducts) with only mild to moderate cytological atypia and infrequent mitoses (see Fig. 24.3A ). The immunophenotype of the tumor cells is also similar to that of the normal esophageal gland ducts. A mixed inflammatory infiltrate may be present as well. In all instances, adenomas should be distinguished from nonneoplastic cystic dilatation of esophageal glands or ducts, which can occur secondary to duct obstruction. Most of these tumors behave in a benign fashion, although rare carcinomas have been reported.
Polypoid dysplastic lesions resembling adenomas may develop in BE (see Chapter 14 ). Endoscopically, these lesions usually appear as well-defined sessile or pedunculated polyps that range in size from 0.5 to 1.5 cm and usually occur in the middle or distal esophagus within areas of BE ( Fig. 24.4 ). Histologically, these polyps are composed of a tubular or tubulovillous proliferation of low- or high-grade dysplastic epithelium. Similar to the surrounding flat nonpolypoid BE, the epithelium is most commonly of the intestinal type, but it may also show a gastric (foveolar) phenotype or mixed features of both.
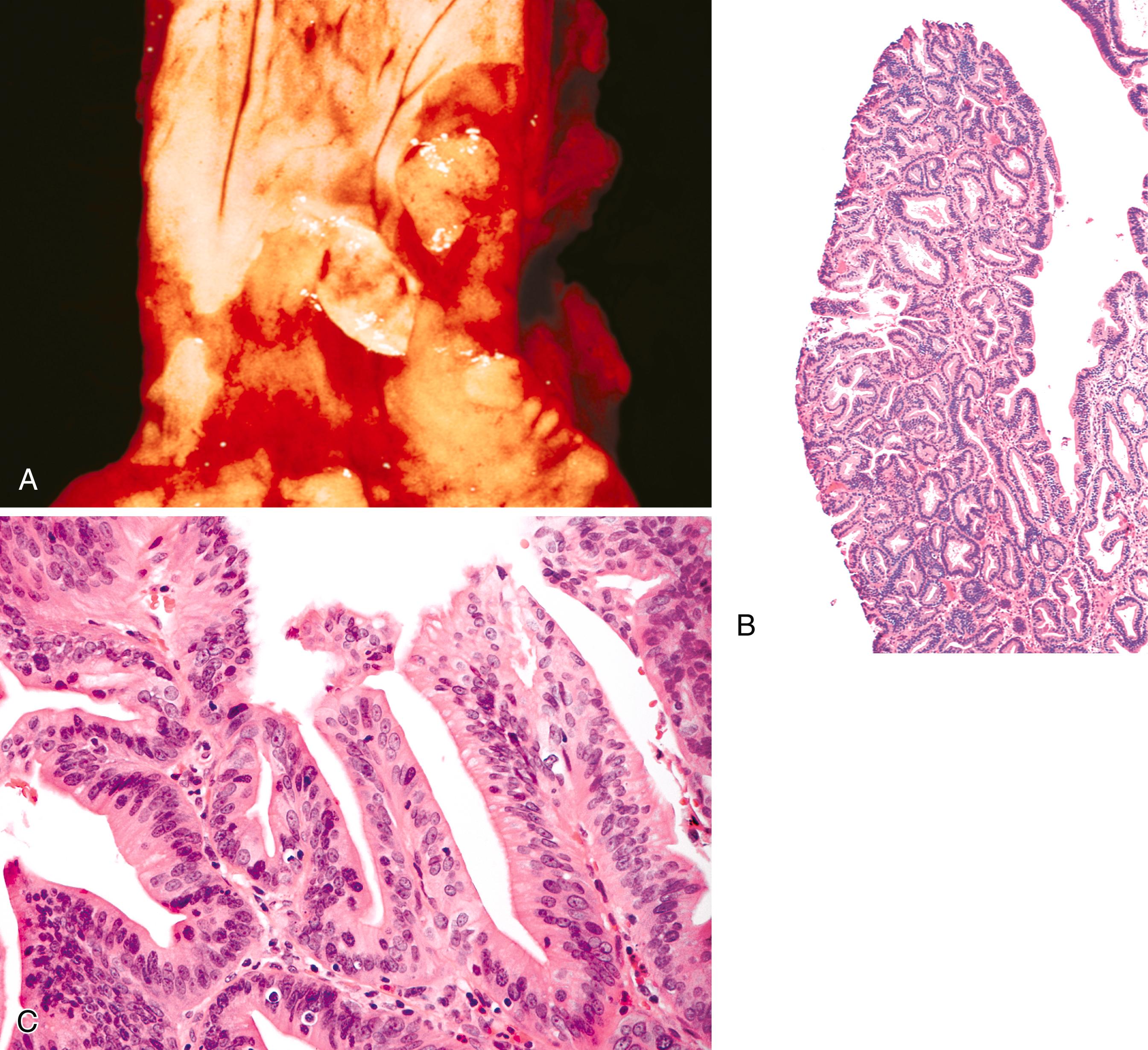
In one study, these lesions showed proliferative and molecular abnormalities (loss of heterozygosity of APC and TP53 ) similar to those of flat BE-associated dysplasia. Polypoid or nodular mucosal abnormalities in BE are associated with an increased risk of underlying adenocarcinoma or subsequent progression. In fact, in one study of 10 cases, 9 were associated with adenocarcinoma. Patients with nodular or polypoid dysplastic foci in the BE segment should be treated according to accepted protocols for Barrett’s dysplasia, with a preference for endoscopic mucosal resection to adequately stage the lesion and guide further therapy.
Congenital cystic lesions and duplications may occur in the esophagus and mimic a malignant tumor because of their mass effect. These lesions, which are discussed more thoroughly in Chapter 8 , are categorized into two principal groups: duplications and neurenteric (dorsal enteric) remnants. They may occur anywhere in the esophagus but are most common in the upper third. Clinically, they may be asymptomatic, or they may cause dysphagia or pain secondary to compression of nearby respiratory or alimentary tract structures.
Duplications most likely develop as a result of abnormal recanalization of the esophageal lumen during embryogenesis, and often present during childhood but may also be noted in adults. , They may be extramural or intramural, and may become cystic if there is no communication with the esophageal lumen. They are usually isolated but may also be associated with other congenital foregut lesions such as pulmonary cystic malformations. Histologically, these unilocular cysts may be lined by respiratory, gastric, oxyntic, squamous, or simple cuboidal epithelium, and usually contain all layers of the gastrointestinal (GI) tract (muscularis propria, enteric nerve plexuses) in the cyst wall. A lymphoepithelial cyst, lined by squamous or cuboidal epithelium and surrounded by a dense mononuclear inflammatory infiltrate, has also been reported in the cervical esophagus.
Tracheobronchial duplications (also known as bronchogenic cysts) are a duplication subtype resulting from anomalous budding of bronchial structures derived from the embryonic foregut. They are typically unilocular and located in the mediastinum or in the wall of the esophagus. Microscopically, they are lined by ciliated columnar epithelium and frequently contain cartilage, smooth muscle, and mucous glands in the surrounding cyst-lining tissue ( Fig. 24.5 ).
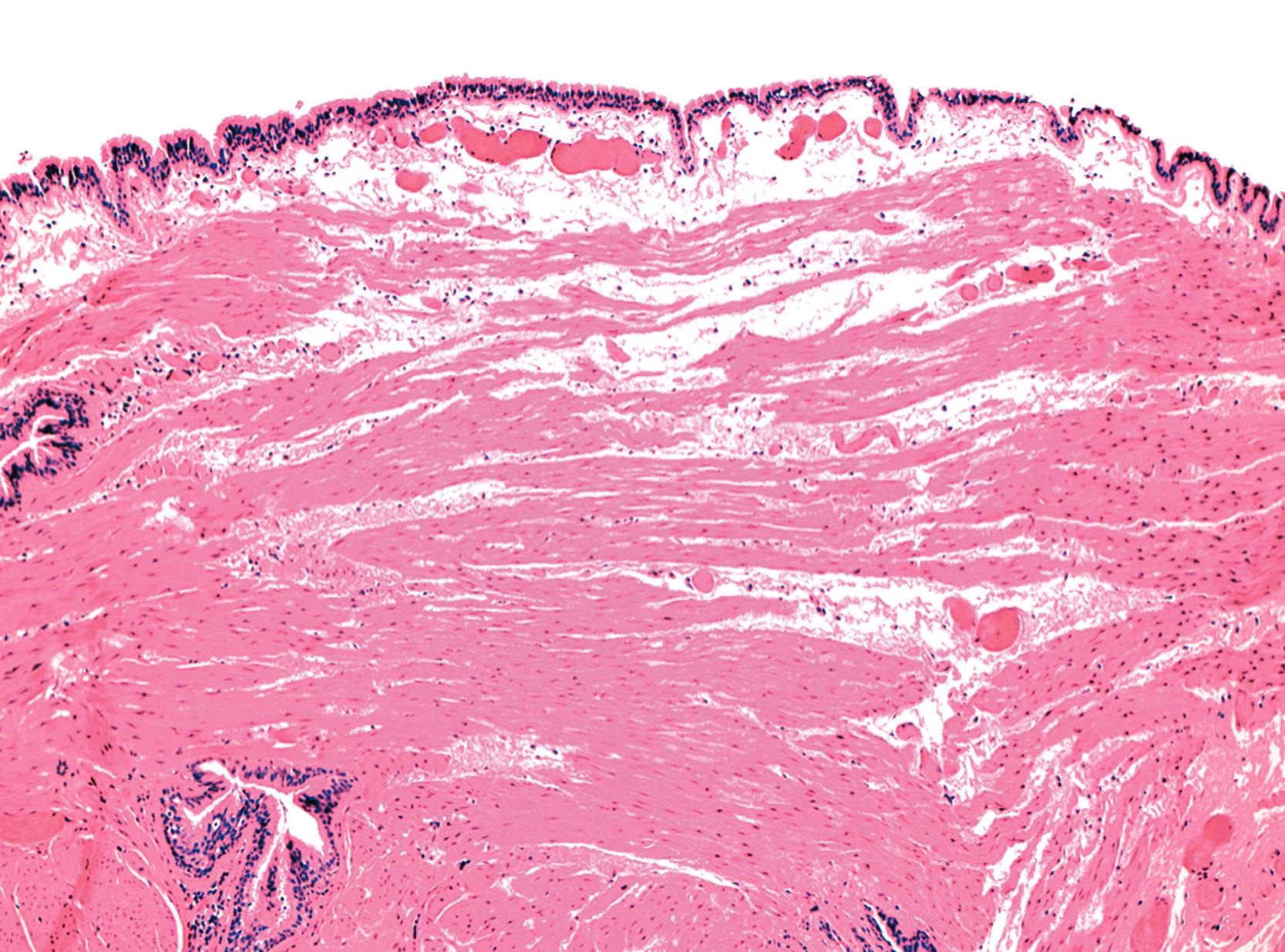
Neurenteric remnants (also termed dorsal enteric cysts ) occur primarily in the posterior mediastinum of infants and are thought to arise from incomplete closure of the notochordal remnant. These cysts may be associated with additional congenital defects, including spina bifida and vertebral anomalies. Microscopically, they are lined by gastric, intestinal, squamous, or respiratory epithelium and are usually surrounded by all the normal tissue layers of the bowel wall.
Developmental cysts and duplications are benign. However, they may become infected as a result of rupture, or may produce symptoms due to mass effect. Extremely rarely, adenocarcinoma or squamous cell carcinoma may develop from these lesions. ,
Among acquired cystic lesions, mucoceles are not uncommon. They may develop in excluded segments of esophagus created after surgery for esophageal atresia or perforation. In large or chronic cases, the cystic lining may not be apparent microscopically, revealing only inflammation and fibrous tissue.
Gastric, thyroid, parathyroid, pancreatic, or even sebaceous tissue may be present as heterotopias in the esophagus. These deposits are presumed to be congenital in origin, although, as discussed later, some possible relationships with clinical conditions such as GERD and BE have been proposed.
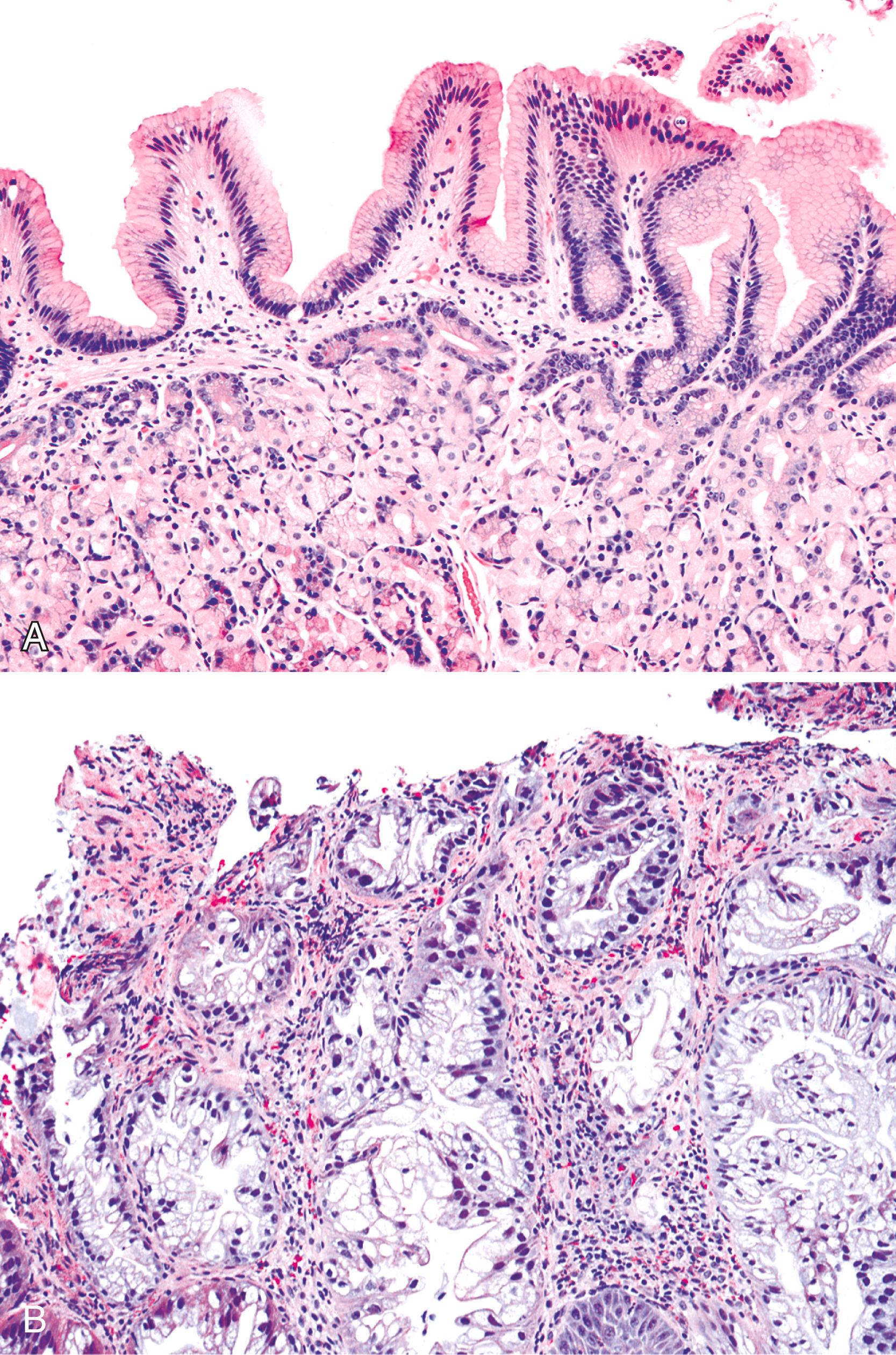
Gastric heterotopia (“inlet patch”) is by far the most common type, being present in an estimated 2% to 14% of the general population. These lesions are usually located in the upper third of the esophagus. Although they are often asymptomatic, they may give rise to symptoms that result from complications of acid secretion, such as heartburn, dysphagia, active esophagitis, ulceration, bleeding, stricture, and perforation. Patients with Helicobacter pylori gastritis frequently show concurrent colonization of the heterotopic mucosa. Although heterotopias are often identified endoscopically, the diagnosis is confirmed by finding gastric glandular (usually oxyntic type) and surface epithelium in the esophagus in patients without intervening BE ( Fig. 24.6A ). Some investigators have found a positive correlation with the presence of BE, although the basis for this association, if any, is unknown. , No therapy is required in most patients, although large inlet patches, or those causing symptoms, may be ablated. Rarely, adenocarcinoma may develop in gastric heterotopia (see Fig. 24.6B ), , and an intraductal papillary mucinous neoplasm arising from a pancreatic heterotopia has been reported.
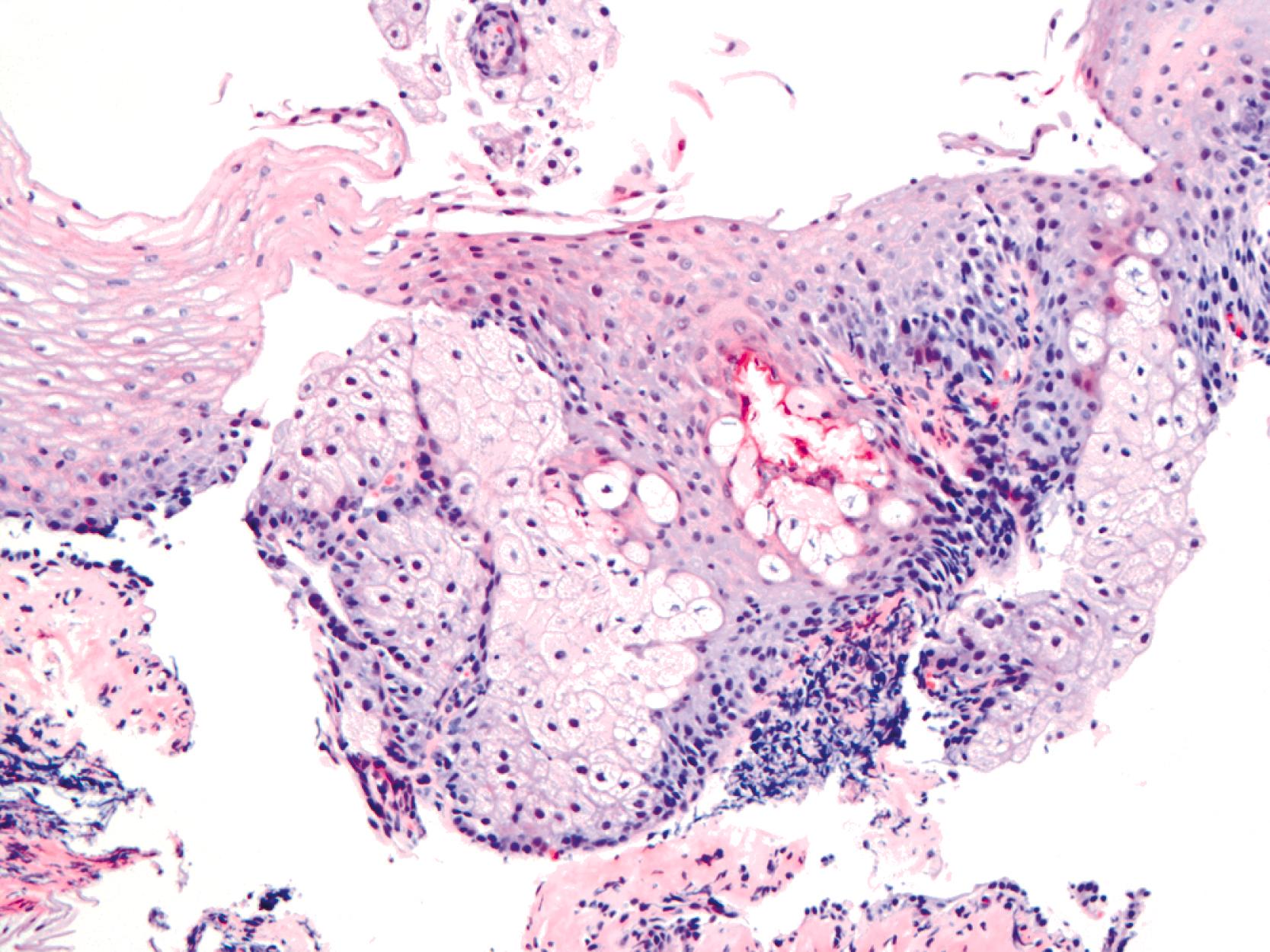
Heterotopic sebaceous glands are the second most common form of heterotopia in the esophagus. They may occur at any level of the esophagus, are frequently multiple, and usually appear as slightly elevated, yellowish lesions, 1 to 2 mm in diameter. Microscopically, these lesions show sebaceous cells within the epithelium or in the lamina propria ( Fig. 24.7 ). Sebaceous cells are microvesicular and contain vacuolated cytoplasm filled with lipid substances. An excretory duct, with or without a connection to the surface epithelium, may be present as well. Although it has been suggested that sebaceous glands may represent a metaplastic process in the setting of GERD, no clear relationship to symptoms or other pathological findings has been established in large population-based studies of asymptomatic individuals.
Pseudoepitheliomatous hyperplasia is a morphological pattern of reactive squamous epithelium that most commonly occurs adjacent to healing ulcers. It is characterized by parallel, elongated, and typically evenly spaced and uniform columns (pegs) of highly reactive squamous cells with prominent nucleoli and mitoses, which may extend deep into the lamina propria ( Fig. 24.8 ). Inflammation, both acute and chronic, is usually present, and this assists in recognizing the reactive nature of the cell proliferation. Lack of surface maturation (nucleated cells at surface) and parakeratosis may be present as well. Its appearance may simulate invasive squamous cell carcinoma, although the lack of either cytological atypia or deeply infiltrative growth supports its benign nature. Furthermore, the squamous cells in reactive hyperplasia usually retain their polarity with respect to each other (absence of overlapping nuclei) and with respect to the basement membrane. Nevertheless, if the columns (pegs) of squamous epithelium are irregular and nonuniform, particularly in areas immediately adjacent to ulcers, and this feature is present in abundance, suspicion for a malignant process should be higher.

On occasion, this pseudoepitheliomatous reaction may be endoscopically visible and polypoid; some authors refer to these lesions as “hyperplastic” or “inflammatory” polyps. In addition, polypoid foci of pseudoepitheliomatous hyperplasia may be confused with the endophytic type of squamous papilloma. However, the latter lesions usually lack the significant inflammation and epithelial damage associated with hyperplasia. Finally, pseudoepitheliomatous hyperplasia may be encountered in squamous mucosa overlying subepithelial granular cell tumors.
Esophageal squamous cell carcinoma, similar to its counterparts in the skin or cervix, is believed to develop through a progression of premalignant or dysplastic precursor lesions. Dysplasia is defined as the presence of unequivocal neoplastic cells confined to the epithelium. The terms dysplasia and intraepithelial neoplasia (IEN) are synonymous, the former being used in the United States and the latter being preferred in Europe and Asia. Squamous dysplasia is more common in patients at high risk for squamous cell carcinoma and is adjacent to squamous cell carcinomas in 60% to 90% of cases. In addition, dysplasia is frequently multifocal, and carcinomas associated with dysplasia are more likely to be multifocal in origin. Dysplasia is currently classified as low-grade or high-grade, based on the degree of cytological atypia and the proportion of the epithelial thickness involved by dysplasia. In this two-tiered system, low-grade squamous dysplasia roughly corresponds to the previously used terms mild and moderate dysplasia, and high-grade squamous dysplasia includes severe dysplasia and carcinoma in situ (see later discussion).
Dysplastic epithelium appears erythematous, friable, and irregular in more than 80% of cases. , Erosions, plaques, and nodules may also be present. However, dysplasia may appear completely normal endoscopically. When available, enhanced endoscopic visualization techniques such as narrow-band imaging are also helpful in highlighting dysplastic mucosa that is otherwise grossly nondescript. Mucosal staining with Lugol iodine may also be helpful to highlight dysplastic mucosa, but this technique has proven inferior to narrow band imaging in comparative studies. Dysplastic cells may also be harvested by exfoliative balloon cytology, a technique that is often used for screening in high-risk areas such as China (see Chapter 3 ).
Dysplastic squamous epithelium is characterized by a combination of architectural and cytological abnormalities that vary in extent and severity, and this is reflected in the grade ( Fig. 24.9 ). The epithelium is usually hypertrophic but may be atrophic in rare circumstances. In 20% of cases, dysplastic epithelium spreads into esophageal mucosal gland ducts and simulates stromal invasion. Low-grade dysplasia reveals involvement of the basal (lower) half of the squamous epithelium with neoplastic cells with mild cytological atypia, whereas high-grade lesions involve more than half of the epithelial thickness. High-grade dysplasia is diagnosed when severe cytological atypia (markedly increased nucleus-to-cytoplasm [N:C] ratio, significant nuclear pleomorphism and hyperchromasia) is present regardless of the extent of epithelial involvement (see later). In addition, dysplastic cells may occasionally grow as isolated cells in a horizontal pagetoid fashion, although this pattern must be distinguished from the more common pagetoid involvement of the squamous epithelium by adenocarcinoma. ,

Cytological changes of low-grade lesions include nuclear enlargement, varying degrees of nuclear hyperchromasia, mildly increased N:C ratio, and increased mitotic rate. High grade lesions show markedly increased N:C ratio, coarsened chromatin, greater degrees of nuclear hyperchromasia, pleomorphism, and irregularity. Nucleoli may be present but are not a consistent feature and are not specific because they are also frequently present in reactive squamous epithelium (discussed later). When present, nucleoli may be unusually large in size and irregular in shape. Architecturally, dysplastic cells display disorganization, loss of polarity, overlapping nuclei, and lack of surface maturation, which are key features in helping to distinguish true dysplasia from nonneoplastic (reactive) processes (see Fig. 24.9C ). Mitotic figures are usually increased in number and may be found at any level of the epithelium (base, midepithelium, or surface). Abnormal (tripolar or disorganized) mitotic figures may be present as well, particularly in high-grade lesions.
Rarely, precursor dysplastic lesions may reveal a proliferation of disorganized large cells (with a normal or even low N:C ratio) with open irregular nuclei, prominent enlarged and irregular single or multiple nucleoli, peripheral condensation of chromatin, and multinucleation (see Fig. 24.9D,E ). In these cases, the border between the epithelium and the lamina propria is often highly irregular, showing sharp, budding, or bulbous expansions of epithelium protruding deep into the lamina propria. This type of dysplastic epithelium is often associated with inflammation and surface maturation, although the latter to a much lesser degree than in either normal or reactive squamous epithelium. In these cases, distinguishing dysplastic epithelium from either markedly reactive squamous epithelium or very well-differentiated squamous cell carcinoma may be extremely difficult. Often, repeat biopsies from deeper portions of the lesion and close correlation with the gross endoscopic appearance are necessary to help establish a final diagnosis. In these cases, invasion should not be diagnosed unless there is unequivocal evidence of irregular clusters of neoplastic cells within the deep lamina propria that are clearly disconnected from the overlying surface epithelium (on deeper levels of tissue sectioning) and associated with a peritumoral stromal desmoplastic response.
Squamous dysplasia must be distinguished from reactive epithelial changes associated with esophagitis. Although regenerating squamous cells may show mild nuclear enlargement, hyperchromasia, and expansion of the basal cell layers, they lack significant nuclear pleomorphism, overlapping nuclei, and nuclear crowding and do not display abnormal mitoses (see Fig. 24.9 ). Unlike in dysplasia, the chromatin is typically fine and homogeneous, and nucleoli, when present, are small and regular in shape. Architecturally, reactive squamous epithelium often displays some degree of surface maturation. Essential to the benign, nonneoplastic diagnosis is the fact that the basal and suprabasal layers maintain their polarity and orderly spacing in reactive lesions. Mucosal inflammation frequently accompanies reactive squamous epithelium, and in the presence of inflammation, a diagnosis of dysplasia should be rendered with caution. In biopsy specimens in which the epithelial changes appear sufficiently marked to suggest dysplasia but a reactive process cannot be excluded because of inflammation, a diagnosis of “indefinite for dysplasia” is appropriate. In such cases, follow-up biopsies after treatment of the underlying esophagitis frequently help resolve the diagnostic uncertainty. The results of ancillary stains, such as strong nuclear staining for TP53 (indicative of inactivating mutations) and extensive, suprabasal cell proliferation highlighted by the proliferation marker Ki-67, may also support a diagnosis of dysplasia in problematic cases. Histological features useful in the differential diagnosis of squamous dysplasia are summarized in Table 24.2 .
| Feature | Reactive | Low-Grade Dysplasia | High-Grade Dysplasia |
|---|---|---|---|
| Clinical/Endoscopic | |||
| Esophageal SCC risk factors | +/− | + | + |
| Normal appearance | +/− | +/− | +/− |
| Erythema | +/− | +/− | +/− |
| Irregular/nodular | − | +/− | +/− |
| Abnormal Lugol/chromoendoscopy | − | + | + |
| Pathological | |||
| Nuclear pleomorphism | − | +/− | ++ |
| Increased N:C ratio | +/− | + | ++ |
| Nuclear hyperchromasia | +/− | + | ++ |
| Increased mitotic rate | +/− | + | ++ |
| Abnormal mitoses | − | + | ++ |
| Nuclear crowding, disarray | − | + | ++ |
| Surface maturation | + | + | − ∗ |
| Inflammation | + | +/− | +/- |
| Immunohistochemical/Molecular | |||
| TP53 mutation/abnormal p53 staining | − | + | + |
| Full-thickness Ki-67 staining | − | − | + |
| DNA aneuploidy | − | + | + |
∗ Some examples of high-grade dysplasia show surface maturation, with high-grade dysplastic epithelial cells limited to the lower half of the epithelium.
In biopsy specimens from patients who have received chemotherapy or radiotherapy, the squamous epithelium may contain markedly atypical cells with enlarged hyperchromatic nuclei that raise the possibility of dysplasia. However, in contrast to dysplasia, these cells do not have an increased N:C ratio and often contain distinctive vacuolization in their cytoplasm. Furthermore, unlike in dysplasia, mesenchymal cells showing similar changes may also be present in the lamina propria, as well as other characteristic stromal changes of chemotherapy or radiotherapy ( Fig. 24.10A ). Therefore awareness of the patient’s clinical history is critical.

Biopsy specimens from patients with esophagitis caused by GERD or other causes such as drug effects occasionally contain multinucleated epithelial giant cells, raising the possibility of dysplasia (see Fig. 24.10B,C ). In these cases, no other cytological or architectural features of dysplasia are present, and results of special studies for viral inclusions are negative. In inflammation-induced multinucleation, the nucleated squamous cells are typically basal or suprabasal in location and may be increased in number adjacent to areas of ulceration. In contrast, multinucleated virally infected cells (e.g., herpes simplex) are common in the surface epithelium and adjacent to sloughing epithelium.
Squamous dysplasia frequently occurs adjacent to invasive carcinoma. For example, 30% of patients with a biopsy diagnosis of high-grade dysplasia show invasive squamous cell carcinoma in subsequent endoscopic resections. In addition, patients with squamous dysplasia are at increased risk for squamous cell carcinoma on follow-up. In a 13-year follow-up study of patients from China, 106 with low-grade dysplasia and 23 with high-grade dysplasia, those with low-grade dysplasia had a 3- to 8-fold increased risk, and patients with high-grade dysplasia had a 28- to 34-fold increased risk for invasive carcinoma. In another study, 15% of patients with low-grade dysplasia progressed to high-grade dysplasia, whereas invasive carcinoma developed in 30% of patients with high-grade dysplasia during an 8-year follow-up period. However, dysplasia may also regress or disappear, although sampling error certainly may account for a normal finding on follow-up biopsy in a patient with dysplasia in a prior biopsy specimen. Therefore patients with a diagnosis of squamous dysplasia require thorough endoscopic examination with biopsies, first to exclude synchronous invasive squamous cell carcinoma (particularly if associated with a visible mass lesion or ulceration) and second to detect the development of early invasive tumor on follow-up. Low-grade dysplasia without a mass or associated carcinoma may be managed with repeated biopsies and continued surveillance in most patients. Mucosal ablation is an option in patients with flat dysplasia, and this technique provided durable remission in 86% of patients over a period of 5 years in one study. A patient with any grade of squamous dysplasia associated with a mass lesion should be considered as having carcinoma until proven otherwise, and managed accordingly. Some endoscopically subtle invasive carcinomas have been reported that apparently arose from low-grade dysplasia, further emphasizing the need for careful endoscopic scrutiny and adequate sampling in patients with any grade of squamous dysplasia.
Endoscopic mucosal resection or endoscopic submucosal dissection, which permits removal of dysplastic lesions or even early superficial cancers, aids in the diagnosis and staging of early invasive tumors and is performed increasingly in specialized centers. Use of both methods have been associated with complete resection rates of greater than 90%, though endoscopic submucosal dissection may be preferable for larger (>15 mm) lesions.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here