Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
To understand the diseases of the peripheral nerve system, physicians require anatomic awareness about the course of the peripheral nerves, bony landmarks, and the relevant ligaments and key muscles/tendons. Furthermore, a working knowledge about how the muscles function is crucial in order to properly diagnose peripheral nerve system problems.
Entrapment neuropathy is a subgroup of acquired peripheral neuropathies. It occurs when a peripheral nerve injury results from compression, either by external forces or more commonly from adjacent anatomic structures.
Clinical history and specific physical signs are of utmost importance to establish a diagnosis, whereas electrophysiologic studies are required to confirm entrapment neuropathies. Magnetic resonance imaging and ultrasound are useful for entrapment neuropathies and are indispensable for the diagnosis of patients with presumed nerve tumors.
Conservative treatment can be a first step before surgical intervention for most entrapment neuropathies, and it involves postural and dynamic considerations to reduce pressure or nerve irritation; surgical treatment aims to decompress the nerve and is indicated when conservative treatment fails or in advanced clinical conditions.
Carpal tunnel syndrome is the most common entrapment neuropathy and the most frequent cause of neurologic symptoms in the hand. This is followed by ulnar nerve compression at the elbow; nonetheless, its incidence is extremely less than that for carpal tunnel syndrome.
Our understanding of peripheral nerve tumors has evolved considerably since the 1990s, particularly with advancements in neuroimaging and genetics.
Patients with malignant peripheral nerve sheath tumors should be evaluated and ideally managed by a team of specialists that include the neurosurgeon, oncologist, geneticist, and oncologic surgeon.
The peripheral nerve system (PNS) includes cranial nerves III through Xll; the spinal nerves; the cervical, brachial, and lumbosacral plexi; and nerves of the extremities. In other words, it is the system of fibers or axons that connect the central nervous system (CNS) with motor and sensory, somatic, and visceral end organs.
The term peripheral neuropathy (PNP) is used when diffuse lesions of peripheral nerves produce sensory disturbance, neuropathic pain, motor weakness, and reflex abnormalities. This can be inherited or acquired. Entrapment neuropathy (EN) is used when peripheral nerve injury or dysfunction results from compression, either by nearby anatomic structures or from external forces.
Some nerves are particularly vulnerable at specific locations. Entrapment of nerves usually occurs as they traverse a confined space or pass beside a joint, such as the elbow and wrist. Repetitive motion and dynamic factors may exacerbate entrapment in an anatomically constricted space. These two considerations are as follows:
The fibro-osseous tunnel, where the compression of a nerve is static ( Fig. 62.1A ), is one consideration. The space for the nerve within the tunnel may become constricted in two ways. The first is by enlargement of the tunnel contents, as in condition of tenosynovitis. The second is by the walls of the tunnel compressing the tunnel's content, as when a segment of enlarged or displaced carpal bone displaces into the carpal tunnel.
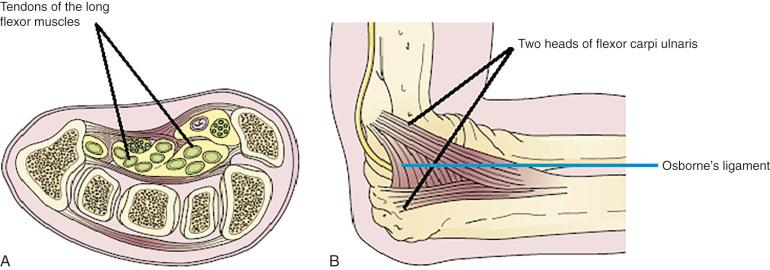
The fibrotendinous structure and compression of the nerve are dynamic ( Fig. 62.1B ). The nerve passes between two heads or bellies of a muscle that under static conditions do not compress the nerve. However, they constrict the space around the nerve when they contract, which results in nerve compression. This can occur, for instance, at the two heads of the flexor carpi ulnaris (FCU), at the distal part of the cubital tunnel for the ulnar nerve, and at the arcade of Frohse, in the supinator muscle for the radial nerve.
The most common symptom is pain (frequently at rest, more severe at night, with the possibility of retrograde radiation causing the more proximal lesion to be suspected), with tenderness at the point of entrapment the most common finding. EN might be associated with some systemic diseases ( Box 62.1 ).
Diabetes mellitus
Hypothyroidism
Acromegaly
Amyloidosis
Carcinomatosis
Polymyalgia rheumatica
Rheumatoid arthritis
Gout
Ischemia and mechanical compression are the two mechanisms believed to lead to this type of injury. Correspondingly and for the sake of simplicity, the pathologic changes will be divided in two main parts, and certain facts should be considered to embrace the mechanisms from all aspects ( Box 62.2 ).
The pathophysiologic changes are dependent on the degree, rate, and duration of compression. For instance, mild and brief compression causes a reversible nerve conduction block, whereas prolonged and sustained compression produces structural changes.
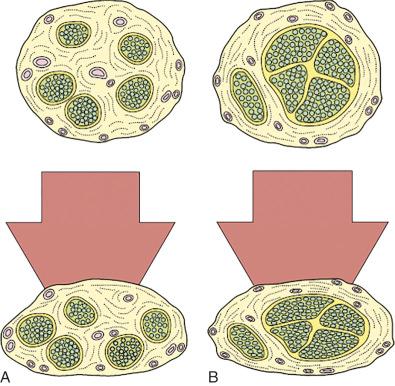
Susceptibility to a given degree of compression is different according to (a) the nerve component: a nerve with a greater amount of epineurium in relation to fascicles is less susceptible to compression than a nerve with scanty epineurium ( Fig. 62.2 ), (b) the location of fibers within the nerve: superficial fibers show more degenerative changes, and (c) myelinated versus unmyelinated fibers: the former (touch and motor function) are more sensitive to compression than the latter (cold and sharp pain sensation).
Ischemia has a couple of functions:
Impediment to microvascular flow is a major factor in the pathophysiology of nerve entrapment. Venous and capillary obstruction herald progressive compression. This leads to nerve ischemia, which in turn leads to endothelial impairment and progressive edema; the edema compounds the ischemia and swelling of the nerve in a vicious cycle.
Critical swelling of a nerve within the constraints of its surroundings may lead to further nerve compression, a phenomenon that can be termed minicompartmental syndrome.
This effect is reversible and little or no histologic change is seen unless ischemia persists for more than approximately 8 hours. If ischemia lasts more than this duration, it produces widespread axonal loss and wallerian degeneration.
Nerve compression blocks axonal transport (changes at neuronal cellular level):
The antegrade transport from the nerve cell to the axon toward the synapse can be divided into fast and slow components; the fast component carries the membrane-associated materials, and the slow components carry the cytoskeletal proteins. Nerve compression impedes both the fast and slow components of the antegrade flow, resulting in a swelling of the nerve proximal to the compression as a result of the damming up of the moving axoplasm within the fibers. Thus the distribution of cytoskeletal elements, axolemma constituents, and the transmitter substances required for synaptic conduction is impaired by a block of antegrade flow.
Retrograde axonal flow from the synaptic level to the cell body of the nerve is similarly blocked by compression of the nerve. This results in a loss of transfer of end organ–derived neurotrophic factors to the nerve cell body. The impairment of retrograde axonal transport results in changes in the nerve cell body comparable to those that occur after peripheral nerve section, which include an eccentrically placed nucleus, dispersion of Nissl substance (chromatolysis), and a decrease in nuclear and whole cell volumes. The overall result of the impediment to axoplasmic flow is impaired membrane permeability and conduction block.
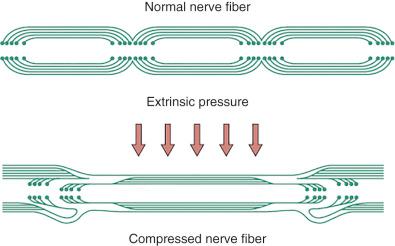
Pathologic changes at myelin sheath level (myelinated fibers) can be another consideration. With acute and severe compression, one observes a characteristic sequential invagination or telescoping of the myelin sheath ( Fig. 62.3 ). The polarity of invagination is reversed at the edges of the compression. With chronic compression, segmental demyelination occurs within the compressed segments, accounting for the slowing of the conduction velocity of the nerve.
Subperineurial and endoneurial edema
Impairment of blood-nerve barrier
Connective tissue changes causing perineurial and endoneurial thickening
Demyelination
Wallerian degeneration
In the early phases, the nerve fibers distal to the compression show normal morphology. With sustained compression, axolysis occurs within the compressed segment, leading to distal wallerian degeneration (see Box 62.3 ).
Several peripheral nerves are vulnerable for entrapment (listed in Table 62.1 ). The clinically pertinent entrapment neuropathy syndromes are discussed in this chapter.
| Nerve | Affected Level | Culprit | Comment |
|---|---|---|---|
| Suprascapular nerve | Suprascapular notch | Idiopathic; ganglia, callus formation from healing fracture | Weakness in abduction and external rotation of arm |
| Spinoglenoid notch | Ganglia, hypertrophied inferior transverse scapular ligament | Weakness of only external rotation of arm | |
| Axillary nerve | Lateral aspect of the humerus and quadrilateral space | Fibrous band, prosthetic devices | Deltoid atrophy |
| Musculocutaneous neuropathy | At the level of the coracobrachialis | Osteochondroma of the humerus | Elbow flexor and forearm supination weakness |
| Median nerve | Supracondylar | Ligament of Struthers or supracondylar spur | |
| Elbow | Bicipital aponeurosis (lacertus fibrosus), hypertrophic pronator teres muscle, sublimis bridge (tendinous arch of flexor digitorum superficialis) | Pronator syndrome | |
| Wrist | Transverse carpal ligament or element of carpal tunnel contents | Carpal tunnel syndrome | |
| Anterior interosseous nerve | Forearm | Fibrous bands from pronator teres or flexor digitorum superficialis muscles. Often no abnormalities may be found | Anterior interosseous syndrome |
| Ulnar nerve | Elbow | The fascial band bridging the two heads of the flexor carpi ulnaris | Cubital tunnel syndrome |
| Elbow | Old fracture, valgus deformity | Tardy ulnar palsy | |
| Wrist | Intraneural or wrist joint ganglia, repetitive trauma, fracture of hook of hamate | Guyon canal syndrome | |
| Radial nerve | Forearm | Supinator edge, fascial band, lipoma, | Radial tunnel or supinator syndrome |
| Posterior interosseous nerve | Forearm | Arcade of Frohse, lipoma, fibroma | Posterior interosseous syndrome |
| Superficial radial nerve | Forearm | Deep fascia of the forearm between the extensor carpi radialis longus and brevis muscles, trauma | Superficial radial nerve syndrome, Wartenberg syndrome |
| Brachial plexus elements | Neck | Cervical rib, fibrous band, scalenus muscle tendons | Thoracic outlet syndrome |
| Lateral femoral cutaneous nerve | Hip | Inguinal ligament and associated fascia | Meralgia paresthetica |
| Peroneal nerve | Knee | Local pressure, mass lesion | Peroneal neuropathy |
| Posterior tibial nerve | Ankle | Soft tissue irregularities, mass lesion | Tarsal tunnel syndrome |
| Saphenous nerve | Thigh | Deep fascia roofing Hunter canal | Saphenous nerve entrapment |
Carpal tunnel syndrome (CTS) is the most common entrapment neuropathy. It involves compression of the median nerve within the carpal tunnel located at wrist. The carpal tunnel is bounded dorsally by the carpal bones and ventrally by the transverse carpal ligament. The carpal bones form a shallow concavity that is converted into a tunnel by the transverse carpal ligament. The contents of the tunnel are the median nerve and tendons of the flexor digitorum profundus (FDP) and the superficialis muscles and tendon of flexor pollicus longus (see Fig. 62.1A ). Any lesion affecting the synovial sheath tends to compromise the cross-sectional diameter of the carpal canal and may induce compression neuropathy. Some studies that include magnetic resonance imaging (MRI) and computed tomography (CT) scans have revealed that patients with CTS tend to have a small cross-sectional diameter of the carpal tunnel that could be either on a congenital or on a developmental basis. Its small size in women may account for the higher incidence of CTS in females.
Most patients are middle aged at the onset of symptoms with a woman-to-man ratio of 3 : 1. The main symptom is an aching, burning, tingling, numb sensation in the hand, usually described in the entire hand, but it may be noted in the lateral half of the hand and the radial three and a half digits including the dorsal side of corresponding fingers to the proximal interphalangeal joint. The pain may extend to the proximal forearm or even to the arm up to the shoulder and could be confused with cervical radiculopathy. Nocturnal pain (a reliable diagnostic clue) is typical in CTS, and patients describe waking up at night due to pain and then shaking, manipulating, or even washing their hand with cold water to obtain relief. Bilateral presentation is frequent and worse in the dominant hand. In advanced cases, patients complain of weakness of grip and a tendency to drop objects, the latter due to reduced sensibility in the thumb and index finger. The local and systemic risk factors of CTS should be reviewed during history taking ( Table 62.2 ).
| Local Factors | Systemic Factors |
|---|---|
| Hypertrophic tenosynovitis | Alcoholic or diabetic polyneuropathy |
| Masses: neurofibroma, hemangioma, lipoma, ganglion cyst | Hereditary neuropathy |
| Anomalous muscles and tendons | Amyloidosis |
| Persistent median artery with or without thrombosis, aneurysm, | Proximal lesions of the median nerve (“double crush” syndrome) |
| Acute palmar space infections | Pregnancy |
| Hemorrhage | Menstrual cycles |
| Congenitally small carpal canal | Eclampsia |
| Idiopathic or familial thickening of the transverse carpal ligament | Menopause |
| Malunion or callus following Colles fracture or fracture of the carpal bones | Contraceptive pills |
| Unreduced dislocations of the wrist | Raynaud disease |
| Improper immobilization of the wrist | Obesity |
| Compression by cast | Long-term hemodialysis |
| Exostoses | |
| Burns at the wrist | Myxedema |
| Acromegaly | |
| Rheumatoid arthritis | |
| Dermatomyositis | |
| Scleroderma | |
| Polymyalgia rheumatica | |
| Mucopolysaccharidoses | |
| Mucolipidoses | |
| Amyloidosis | |
| Chondrocalcinosis | |
| Gout |
Physical finding are few in the early stages. There could be impairment of pinprick, light touch sensation, and two-point discrimination in the tips of the fingers in the median nerve distribution. Atrophy of the thenar muscles, particularly the abductor pollicis brevis, can be seen in advanced cases. The Tinel sign may be elicited by tapping over the median nerve at the wrist crease; it is positive (60% of cases) if this results in tingling in the distribution of the median nerve. The Phalen test is performed by asking the patient to flex the wrist to 90 degrees for about 60 seconds; it is positive (80% of cases) if this results in paresthesia in the distribution of the median nerve. Both of these tests are supportive but not conclusive.
CTS should be differentiated from cervical radiculopathy (which may coexist in many patients), thoracic outlet syndrome, pronator teres syndrome, de Quervain syndrome, and reflex sympathetic dystrophy.
Electromyography (EMG) and nerve conduction velocity (NCV) may help confirm the diagnosis and distinguish CTS from other conditions. NCV sensory latency is more sensitive than motor. The earliest finding is the prolongation of sensory latency > 3.7 m/sec and motor > 4.5 m/sec due to demyelination. The sensory evoked response will show diminution of amplitude and may even be absent. Motor latency abnormalities occur late in the course of the disease. Needle electromyography may show a loss of motor unit potentials and the presence of denervation potentials in the thenar eminence muscles due to axonal loss. Clinically it corresponds to the impairment of two-point discrimination.
Imaging tests are not routinely ordered and are only indicated when a mass lesion or structural deformity is suspected. Wrist MRI may reveal swelling or flattening of the nerve and ganglion cysts. Ultrasound can be used with limited indications. Similarly, blood tests are indicated when risk factors suggest diabetes mellitus or thyroid disease.
Widely acceptable management strategies based on the evidence-based medicine approach were summarized in the American Academy of Orthopaedic Surgeons practice guidelines last updated in 2011 ( http://www.aaos.org/cc_files/aaosorg/research/guidelines/cts_treatment_reissue.pdf ). Some of the pertinent studies are summarized in Table 62.3 for both median and ulnar nerves.
| Study | Conclusions |
|---|---|
Carpal Tunnel Syndrome
|
|
Cubital Tunnel Syndrome
|
|
In early cases with minimal symptoms or for patients in whom the syndrome is expected to be transient (as in peripartum), conservative treatment is the first option. The recurrence within 1 year is almost 90% in severe CTS compared to 60% in mild cases. Nonsurgical treatment consists of a wrist splint in neutral position at night (initial relief in approximately 50%), a course of vitamin B 6 treatment, and antiinflammatory drugs (NSAIDs). Injection of local anesthesia and steroids around the median nerve may be beneficial, yet accidental injection into the nerve may result in annoying paresthesias in the median nerve distribution, so the authors do not advocate injection therapy.
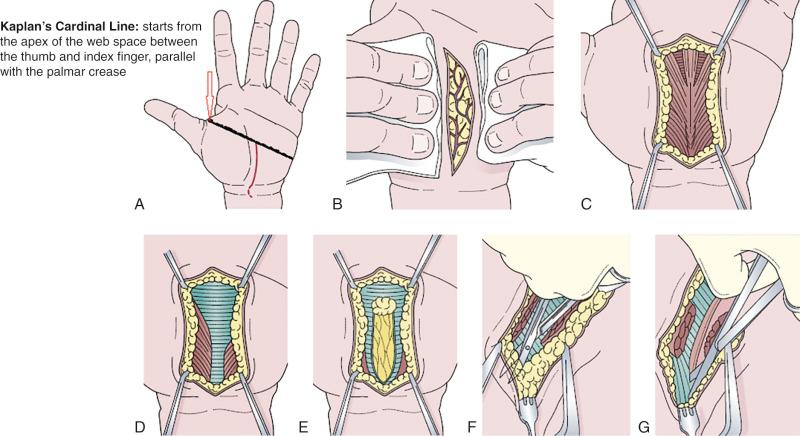
Surgical treatment is indicated when conservative measures fail, symptoms are present for > 1 year, or in cases of thenar muscle atrophy. The procedure can be either open surgery or an endoscopic technique for release. The steps in the surgical sectioning of the transverse carpal ligament are shown in Fig. 62.4 . Usually local or regional anesthesia is used; our preference is local with conscious sedation. General anesthesia may be used if the patient is extremely nervous.
Endoscopic carpal ligament release (ECTR) was first described in 1989, and since then it has been in practice. It may shorten the postoperative recovery period and minimally disturb the structural integrity of the carpal tunnel mechanism; however, there is a greater risk of injury to the ulnar artery and nerve. Low-quality evidence revealed that patients treated with ECTR return to work on average 10 days earlier than those treated with open carpal tunnel release (OCTR). The quality of evidence is low to very low regarding major complications; recurrence and reoperations and the advantage of ECTR with respect to minor complications are uncertain.
The patient should move the fingers postoperatively to minimize scar formation. A good outcome is to be expected in up to 90% of cases, but if there is significant weakness and wasting at the time of presentation the results are less satisfactory.
The ulnar nerve (UN) has components mainly from C8 and T1 spinal nerve roots. It can be entrapped at the elbow and the wrist levels. Around the elbow it can be compressed by the (1) arcade of Struthers, (2) medial intermuscular septum, (3) medial epicondyle, (4) cubital tunnel, and (5) deep flexor aponeurosis. Among these, the cubital tunnel location is the most common. It is the second most common EN after CTS.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here