Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Shock has been defined in a multitude of ways, but they all come down to inadequate tissue perfusion, and the diagnosis is made at the bedside using clinical judgment. Early identification of the type of shock, understanding of the cause, and institution of early treatment and supportive care including thoughtful patient-tailored fluid resuscitation using a combination of endpoints are recommended. The goal is to restore adequate end-organ perfusion with a matching metabolic supply and demand, while avoiding complications from overresuscitation. In the case of hemorrhagic shock, early hemostasis and balanced blood product resuscitation while minimizing crystalloid are indicated.
In 2001 when early goal-directed therapy for sepsis was first introduced, it was uniformly believed that early aggressive resuscitation of critically ill patients would limit tissue hypoxia and progression to organ failure and would improve outcomes. The surviving sepsis campaign and subsequent updates have recommended earlier intervention, progressing from interventions in the first 3 to 6 hours to a combined 1-hour bundle in the most recent update in 2018. However, the most recent guideline still recommends a 30 mL/kg fluid bolus for all hypotensive patients or those with a lactate >4 within the first hour and uses lactate and MAP as endpoints. There are no high-quality data supporting these recommendations and they remain controversial. We now know after the ProCESS, ARISE, and ProMISe studies evaluating early goal-directed resuscitation that there is no benefit to fixed protocols and that there is a higher likelihood to overresuscitate, resulting in increased morbidity and mortality.
Protocols that do not consider the patient’s clinical response to treatment and resuscitation have the potential to do harm especially when both the disease process and patient population are so diverse. In fact, some studies have reported rates of fluid overload in critically ill patients of up to 67% resulting in worse outcomes.
ARISE Fluids, a phase II feasibility observational study in a large number of hospitals, found that restricted fluid and early vasopressor regimens resulted in a significantly lower volume of fluid administered over the first 24 hours of care, with no harm. The results of the Phase III trial are pending.
Additionally, Maitland et al in the FEAST trial where over 3000 African children in septic shock were randomized to receive albumin or saline fluid boluses vs. no fluid demonstrated a statistically significant increase in mortality in the fluid bolus group.
Balancing fluid resuscitation and fluid overload in a patient in shock is complex. The judgment of the clinician attuned to the clinical manifestations, physiologic parameters, trends of laboratory markers, and other useful endpoints and resuscitation markers come into play to be successful in this balancing act (see Fig. 1 ) where the only goal is to restore perfusion and systemic oxygen delivery normalizing cellular metabolism.
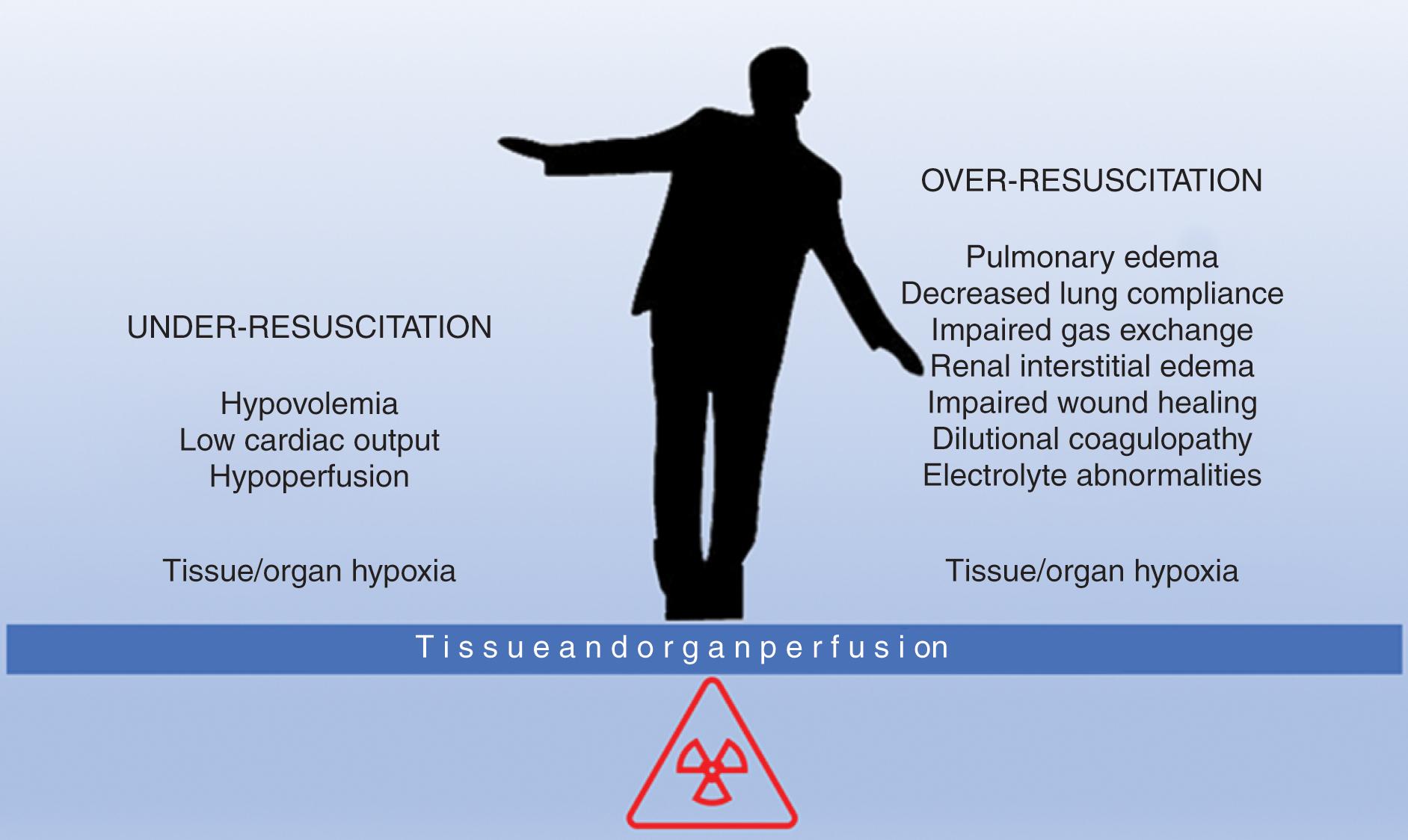
Significant research has been done in the last few decades looking at all the available endpoints for resuscitation and to this date we haven’t found an ideal single endpoint that would be easily accessible, reproducible, cheap, and noninvasive. Instead, using a few markers, trends, targets, and fluid response determinants in conjunction with the overall clinical picture may be a more valuable resuscitation strategy. The goal of this chapter is to review the most commonly used resuscitation endpoints, their roles, and their limitations.
The six basic Advanced Trauma Life Support physiologic parameters that have been used to identify shock are heart rate, respiratory rate, blood pressure, urine output, level of consciousness, and pulse pressure. They are unreliable in a large number of patients and must be placed in the context of each individual patient. For example, hypotension was traditionally believed to be the presenting sign of shock; however, what is considered a “normal” blood pressure may be dangerously low in a chronically hypertensive patient. Similarly, heart rate and respiratory rate can be misleading in cases of anxiety, pain, and stress. Renal blood flow correlates with arterial pressure but can be subject to significant autoregulation during periods of hypoperfusion. Additionally, the patient’s level of consciousness is less reliable when influenced by intoxication, central nervous system injury, and medication.
The presence of relatively normal vital signs does not guarantee that regional perfusion is adequate, and a high index of suspicion should be maintained while additional data are collected.
Oxygen delivery is a function of hemoglobin concentration, oxygen saturation, and cardiac output. Hemoglobin concentration and oxygen concentration are relatively easy to manipulate and monitor, but cardiac output evaluation and monitoring requires an advanced assessment. Based on the following equation, changes in the hemoglobin or SaO 2 will produce minimal changes in oxygen delivery contrary to CO.
Therefore heart rate is generally at maximum compensation. Adequate cardiac performance is largely a function of preload, and intravascular volume status assessment is of utmost importance and not always straightforward. The goal of fluid resuscitation in shock is to increase preload, stroke volume, and cardiac output, assuming that the patient is in the ascending part of the Frank-Starling curve ( Fig. 2 ) and identifying the flattening of the curve is critical to avoid overresuscitation, since it is at this point, where they become fluid nonresponders and more fluid begins to cause harm.
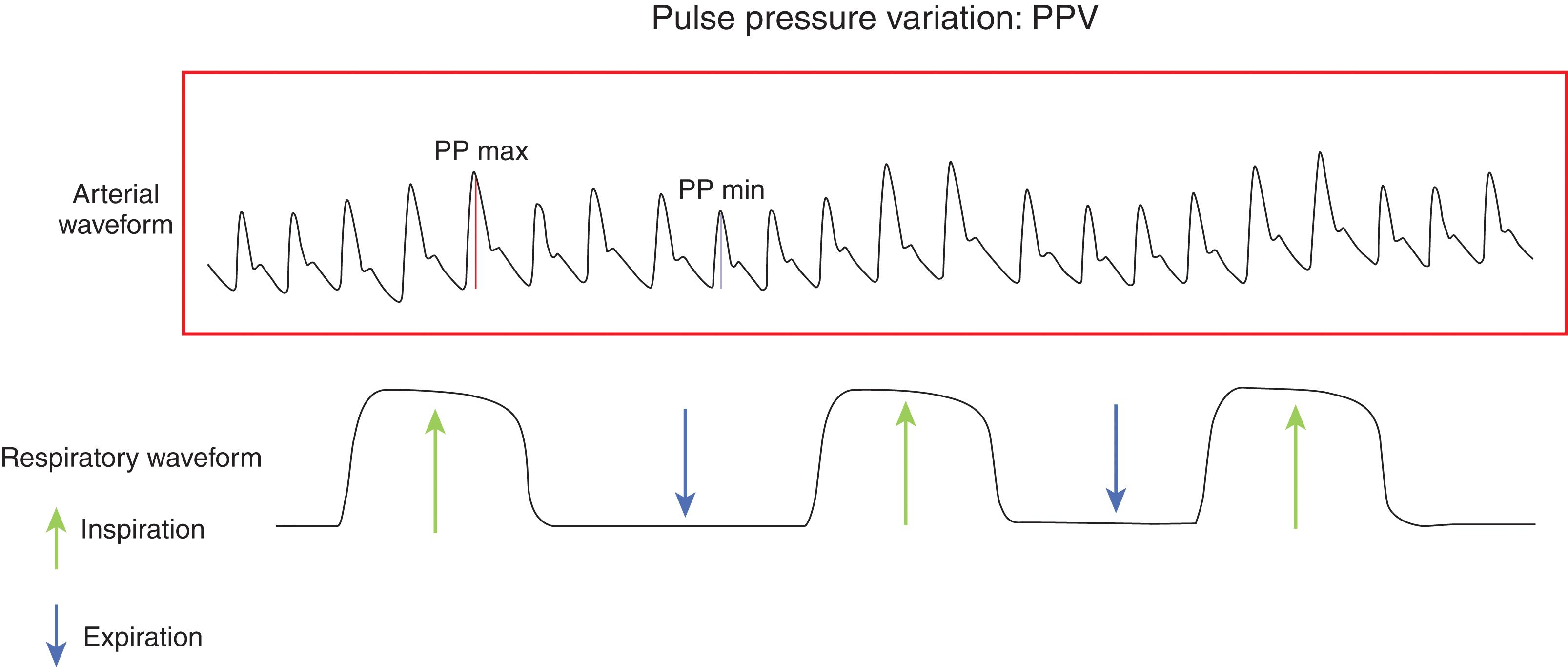
For decades we have been using catheters (central venous pressure [CVP], pulmonary artery [PA] catheters) to measure static measures of preload and volume responsiveness; these are single point in time measurements, such as CVP and filling pressures, and have been replaced by dynamic measures that take advantage of heart and lung interactions to predict volume responsiveness.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here