Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
We wish to thank the co-authors of the previous version of this chapter, Drs. Courtney C.J. Voelker and Gregory P. Lekovic.
![]() Please access Videos to view the corresponding videos for this chapter.
Please access Videos to view the corresponding videos for this chapter.
The petrous apex is one of the least accessible areas of the skull base. Located in the anterior-superior portion of the temporal bone, surgical approaches to lesions of the petrous apex require detailed knowledge of vital neurovascular structures. The petrous apex is bounded anteriorly by the bony labyrinth and the ascending portion of the internal carotid artery (ICA), posteriorly by the posterior cranial fossa, superiorly by the middle cranial fossa and Meckel cave, and inferiorly by the jugular bulb and the inferior petrosal sinus.
Computed tomography (CT) and magnetic resonance imaging (MRI) of the temporal bone together assist the surgeon in characterizing the lesion preoperatively. CT provides information on the size of the lesion and its relationship to vital structures including the internal auditory canal (IAC), cochlea, vestibular labyrinth, ICA, and jugular bulb. It also indicates whether the border of the lesion is expansile or invasive, which may help differentiate between benign lesions and malignant neoplasms. Magnetic resonance imaging (MRI) of the temporal bone adds the capability of characterizing the substance of the lesion, rather than its effect on bony interfaces, allowing the surgeon to distinguish between mucus, fat, cholesterol granuloma, cholesteatoma, and neoplasm. This combination of CT, with its superior bone imaging algorithms, and MRI, with enhanced tissue imaging capabilities, allows the surgeon to accurately differentiate between benign cystic lesions, normal anatomical variants, and neoplastic lesions of the petrous apex.
Lesions of the petrous apex tend to remain clinically silent until they grow large enough to encroach on the adjacent cranial nerves. Patients with petrous apex lesions present with various symptoms and physical findings. The most widely recognized is Gradenigo syndrome, characterized by the triad of retro-orbital pain, otorrhea, and ipsilateral sixth cranial nerve paresis. The signs and symptoms of noninflammatory or neoplastic lesions may be subtler. Hearing loss and vestibular abnormalities are frequently associated with enlarging lesions of the petrous apex as they compress the vestibulocochlear nerve within the IAC. The facial nerve is relatively resistant to paresis from slowly expansile lesions, but may be involved early with neoplastic lesions of the petrous apex or nonneoplastic lesions compressing the IAC.
While pain may be present with benign or cystic lesions, it is more common in neoplastic lesions. The pain distribution depends on the region involved. The mastoid cavity is innervated by cranial nerve (CN) IX and may radiate pain into the neck. The middle fossa and superior petrosal regions are innervated by CN V and may cause retro-orbital or facial pain. Alternatively, direct compression of the gasserian ganglion may cause numbness in one or more divisions of the trigeminal nerve (CN V). Lesions extending into the posterior fossa may cause pain along the distribution of CN IX, CN X, and the first three cervical nerves. Although 80% of adult mastoid bones are aerated, only 30% of petrous bones have air cells extending to the apex, and 7% may have asymmetric petrous apex pneumatization. , The increasing use of head and neck MRIs makes it imperative that clinicians can differentiate pathology from normal variants in the temporal bone. Table 40.1 summarizes the common lesions of the petrous apex and their associated radiologic findings.
| Magnetic Resonance Imaging | |||||
|---|---|---|---|---|---|
| Lesion | Computed Tomography | T1 Weighting | T2 Weighting | Enhancement | Diffusion Restriction on DWI |
| Retained mucus (petrous apex effusion) | Normal bony architecture, nonenhancing, nonexpansile | Hypointense | Hyperintense | No | No |
| Mucocele | Hypodense, expansile smooth border, nonenhancing | Hypointense to isointense | Hyperintense | No | No |
| Asymmetric pneumatization (petrous apex marrow) | Normal bony architecture, nonenhancing | Hyperintense | Hypointense | No | No |
| Cholesteatoma (epidermoid) | Loss of normal air cells, nonenhancing, isodense with CSF | Hypointense | Hyperintense | No | Yes |
| Cholesterol granuloma | Expansile smooth border, occasionally rim enhancement, isodense with brain | Hyperintense | Hyperintense | No | No |
| Encephalocele | Nonspecific, may show smooth erosion | Hypointense | Hyperintense | No | No |
| Petrous apicitis | Irregular erosion, rim enhancement | Hypointense | Hyperintense | Yes | Yes if abscess |
| Metastatic lesion | Destructive, indistinct border | Isointense—75% | Hyperintense | Yes | No |
| Aggressive bone destruction, calcification | Hypointense—25% | ||||
| Chondroma | Aggressive bone destruction, calcification | Hypointense to isointense | Hyperintense | Yes | No |
| Chondrosarcoma | Aggressive bone destruction, calcification | Hypointense to isointense | Hyperintense | Variable | No |
| Chordoma | Destructive soft tissue mass centered in clivus, calcifications (residual bony trabeculae) | Hypointense | Hyperintense | Honeycomb enhancement pattern, variable | No |
Asymmetric pneumatization is clearly visible on CT, but supplementary MRI may be needed to rule out pathological lesions in a symptomatic ear. Normal bony architecture can be observed on CT, with hyperintensity on T1-weighted MRI and hypointensity on T2-weighted scans because of the large fat content in marrow. MRI and CT are useful to identify encephaloceles of the petrous apex. CT may reveal well-corticated margins suggestive of a dominant air cell. MRI imaging of the fluid within the apex that follows cerebrospinal fluid (CSF) signal on both T1- and T2-weighted imaging may establish the diagnosis of encephalocele and identify a potential site of temporal bone CSF leak. These lesions may also be bilateral or symmetric in appearance. Retained mucus in the air cells also appears with normal bony architecture on CT scans, but is hypointense on T1- and hyperintense on T2-weighted MR images. Cholesteatoma (epidermoid) is usually associated with chronic otitis media, but may alternatively arise from congenital rest cells in the petrous apex. Because of its high water content, cholesteatoma is isodense with CSF on CT, and it is hypointense on T1- and hyperintense on T2-weighted MR images. Diffusion-weighted imaging (DWI) MRI is particularly useful in distinguishing a cholesteatoma (epidermoid) lesion. It is the only lesion that displays diffusion restriction on DWI MRI ( Table 40.1 ). On CT, cholesterol granuloma is an expansile lesion isodense to brain that erodes the bony trabeculae. MRI demonstrates a pathognomonic appearance in most patients, with hyperintensity on both T1- and T2-weighted scans. Additionally, cholesterol granulomas do not attenuate on fluid-attenuated inversion recovery (FLAIR) MRI and do not restrict diffusion on DWI MRI.
The radiological descriptions of other, less common lesions are summarized in Table 40.1 . Differentiating between chordoma, chondroma, and chondrosarcoma of the temporal bone remains difficult, even with the current CT and MRI capabilities. The patient age and lesion origin must be considered when determining the pathology of destructive lesions of the petrous apex.
Most surgical approaches to the petrous apex were developed in the preantibiotic era for drainage of petrous apex abscesses and a cure for Gradenigo syndrome. With the arrival of modern antibiotics, the number of patients exhibiting infectious processes of the petrous apex has declined. However, these same approaches are used to drain cystic lesions of the petrous apex.
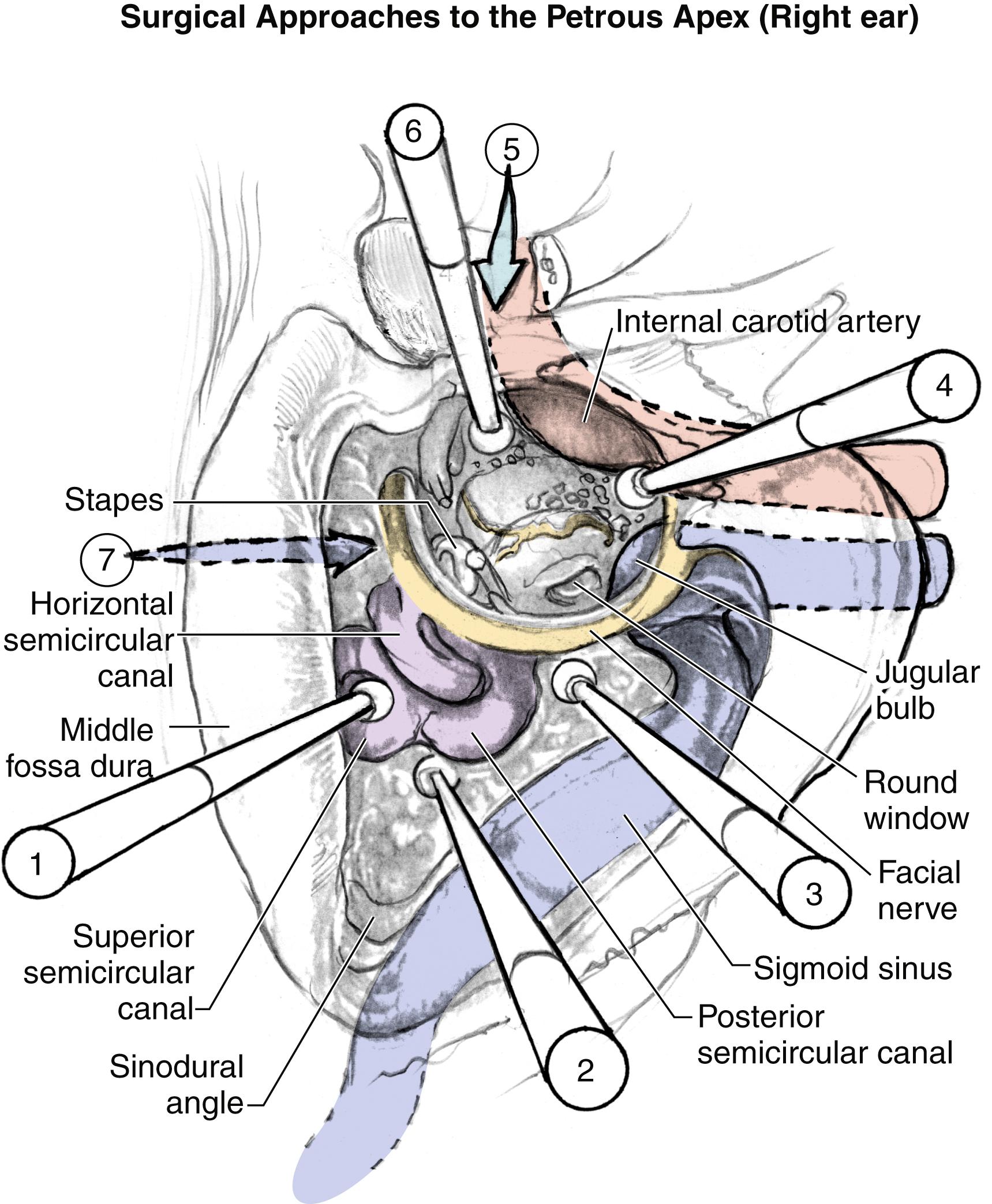
Critical factors in determining a surgical approach to a petrous apex lesion include the hearing level, the size and location of the lesion within the petrous apex, and the neurovascular anatomy. If the patient has serviceable hearing, four main surgical approaches are considered: (1) infralabyrinthine, (2) transcanal infracochlear, (3) transsphenoidal, and (4) middle cranial fossa ( Fig. 40.1 ). Various other approaches are also shown in Fig. 40.1 but are not described in this chapter. The air cell tracts extend superior, inferior, and anterior to the otic capsule, potentially allowing for safe access to the petrous apex. The approaches that follow superior air cell tracts include the middle cranial fossa approach, through the superior semicircular canal, the attic, and the root of the zygomatic arch. The approaches inferior to the inner ear include the infralabyrinthine and the infracochlear approaches. Anterior approaches have been described by Ramadier, Eagleton, and Lempert, who used the triangle between the anterior border of the cochlea, carotid artery, and middle fossa dura ( Fig. 40.1 ). If the hearing is nonserviceable, the translabyrinthine or transotic approaches can be used.
Cholesterol granuloma is the most common cystic lesion of the petrous apex. Compared with other primary petrous apex lesions, cholesterol granulomas were found to be 10 times more common than cholesteatomas and 40 times more common than petrous apex mucoceles. , Cholesterol granuloma is a foreign body giant cell reaction to cholesterol deposits, with associated fibrosis and vascular proliferation. It may develop in any aerated portion of the temporal bone, but most commonly occurs in the mastoid air cells distant to a lesion that prevents normal aeration. The pathogenesis is controversial with two prevailing theories; in the classical obstruction-vacuum theory, cholesterol granuloma of the petrous apex develops when a pathological process or trauma obstructs the air cell tracts to a well-pneumatized petrous apex. Hemorrhage occurs into the air cells, and the subsequent degradation of accumulated hemosiderin to cholesterol in turn incites an inflammatory reaction and progressive granuloma formation. In the exposed marrow theory, exuberant petrous apex pneumatization breaks down the bony partitions separating the air cells and adjacent hypervascular fatty marrow, which herniates into the petrous apex air cells causing recurrent hemorrhage. This blood then coagulates within the air cells occluding the outflow tracts. The cyst wall of cholesterol granulomas is made of fibrous connective tissue. Unlike cholesteatomas, they lack a true epithelial lining and do not produce keratinizing squamous debris. Therefore, the treatment for temporal bone cholesterol granulomas is drainage and reestablishment of adequate aeration to the involved area. Complete removal of the cyst is not necessary.
Solid tumors of the temporal bone and cholesteatoma cause destruction of vital structures as they grow. Therefore, these lesions should be removed when first identified. Drainage procedures inadequately treat these lesions. All reasonable efforts should be made to remove them entirely. Total removal may require sacrificing the CNs and major vascular structures.
The preoperative evaluation of patients is based on exhibited symptoms. Patients presenting with hearing loss are initially evaluated with audiometric testing, including air, bone, and speech reception thresholds and speech discrimination scores. Videonystagmography is performed in patients who complain of imbalance or vertigo. An MRI is obtained in patients with asymmetric hearing or other CN abnormalities.
Patients who exhibit normal hearing but demonstrate other CN deficits that may be referred to the petrous apex may be screened with a high-resolution, thin-section CT scan of the temporal bone or a MRI scan with gadolinium. If abnormalities are identified on imaging, all patients should undergo air, bone, and speech reception thresholds and speech discrimination audiometric testing before surgery to accurately document their hearing levels before any procedure that jeopardizes hearing.
Preoperatively, patients are counseled to expect resolution of pain, if present, and the possibility of improvement in cranial nerve function. Cranial nerves that have been affected for shorter periods appear to have a better prognosis with fewer long-standing deficits than do nerves that were affected longer. In the case of cholesterol granuloma, patients are reminded that this is a drainage procedure with the goal of decompressing the lesion and providing an aerated cavity. The goal is not to remove the entire lesion. Therefore, close, long-term follow-up is necessary. Recurrence of the lesion secondary to inadequate drainage is usually heralded by a return of the preoperative symptoms. Follow-up MRI frequently reveals a cholesterol granuloma cyst that remains full of fluid, but the T1-weighted image is hypointense compared with the preoperative hyperintense image on T1-weighted views. A return of hyperintensity on the T1-weighted image, indicating high protein concentration, suggests inadequate drainage in a symptomatic lesion.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here