Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Our individual sex has a major role in determining the physical attributes of our bodies, the structure of our brains, our behavioral tendencies, and our self-concept. Understanding how biological sex differences arise has both informed and been informed by the advances in the molecular dissection of genes responsible for conditions collectively termed differences of sex development (DSD). New genes have been identified, allowing for rapid diagnosis, understanding of the pathophysiology, and prediction of future fertility.
In parallel with the progress in the biology of sexual development, the management of individuals with DSD has started a gentle revolution. From a practice based on the opinions of a few leaders in the field, the clinical approach to DSD has slowly entered the era of evidence-based medicine, under the pressure of patient advocacy groups, who have been highly instrumental in establishing a dialogue between healthcare providers and the families. As a result, physicians are beginning to understand the difficulties involved in defining normalcy, as well as the ethical dilemmas of acting medically on children before they reach the age of consent.
In 2005, a consensus on the management of “intersex” disorders emerged, bringing together experts from a variety of fields (endocrinology, genetics, surgery, psychology, advocacy). The consensus statement provided guidelines in all aspects of management of “intersex” conditions, including genital surgery, the requirement of a multidisciplinary team and of mental health professionals on that team, and the diagnostic approach. This chapter reflects the advances made in the field as well as the recommendations of the consensus statement.
One area that changed considerably is the nomenclature. The term disorders of sex development was proposed, as defined by “congenital conditions in which development of chromosomal, gonadal or anatomical sex is atypical.” This broad definition replaced the word intersex, which has social connotations and reflects a concept of sexual identity rather than a clear biological concept. It includes individuals who do not necessarily have ambiguous external genitalia, such as those with complete androgen insensitivity or Turner syndrome. The revised nomenclature (summarized in Table 17.1 , with an example of DSD classification in Table 17.2 ) was guided by the following principles. First, although a modern categorization should integrate the important progress in molecular genetics, it should not overemphasize one particular aspect of the biology of sex (for instance, gonadal sex) and should accommodate the spectrum of phenotypic variations. Second, terms should be as precise as possible and should reflect the genetic etiology when available. Finally, the new nomenclature should be understandable by patients and families and should be psychologically sensitive. In particular, gender labeling in the diagnosis should be avoided, and use of the words hermaphrodite, pseudohermaphrodite, and sex reversal should be abandoned because they either are confusing or have a negative social connotation that may be perceived as harmful by some individuals and families.
| Current | Previous |
|---|---|
| DSD | Intersex |
| 46,XY DSD | Male pseudohermaphrodite, undervirilization of an XY male, undermasculinization of an XY male |
| 46,XX DSD | Female pseudohermaphrodite, virilization of an XX female, masculinization of an XX female |
| Ovotesticular DSD | True hermaphrodite |
| 46,XX testicular DSD | XX male or XX sex reversal |
| 46,XY complete gonadal dysgenesis | XY sex reversal |
| Sex Chromosome DSD | 46,XY DSD | 46,XX DSD |
|---|---|---|
| 45,X (Turner syndrome and variants) 47,XXY (Klinefelter syndrome and variants) 45,X/46,XY (mixed gonadal dysgenesis, ovotesticular DSD) 46,XX/46,XY (chimeric, ovotesticular DSD) |
Disorders of gonadal (testicular) development:
|
Disorders of gonadal (ovarian) development:
|
Androgen synthesis or action:
|
Androgen excess:
|
Since the rollout of the new nomenclature, there have been a number of discussions about the specific use of the word “disorder.” For example, interAct Advocates for Intersex Youth, a nonprofit organization devoted to the advocacy for the legal and human rights of children with intersex traits, has criticized the use of the “disorder” terminology. In a Global DSD Update issued 10 years after the Consensus Statement, some positive aspects of the original DSD nomenclature were highlighted (e.g., improved access to healthcare and insurance, distinction from other conditions such as gender dysphoria), but it also warned against the negative connotation and potential stigma associated with the word “disorder.”
The word “difference” has become an alternative, and academic publications, as well as some advocacy organizations, have used it increasingly. We will use the DSD nomenclature in this chapter; however, we will take the DSD acronym as meaning “differences of sex development.”
Another development has been the advent of next-generation genomic sequencing and its increasing use in clinical settings, mostly in the form of exome sequencing, with some forays in clinical whole genome sequencing and RNA sequencing. The mapping of all human genes through the Human Genome Project has helped identify the causative genes at several known DSD loci and reclassification of many syndromes after it was uncovered they shared a common genetic etiology. The availability of exome sequencing in the clinical realm has also ushered in a change in paradigm in DSD diagnosis. With exome sequencing and targeted DSD gene panels available at decreasing cost and with turnaround time similar to that of some endocrine tests (4 weeks for exome at some testing centers, and down to 1 week or less for urgent cases), prioritizing genetic testing as the first-tier diagnostic tool for the assessment of DSD patients is quickly becoming a reality in clinical practice. ,
Human sex development occurs in two main steps during embryonic development:
Sex determination : Expression of the Y-chromosome gene SRY triggers the transformation of the bipotential gonad into a testis in males. The absence of this signal in females results in the activation of pathways leading development into an ovary.
Sex differentiation : The developing testis secretes hormones that trigger differentiation of Wolffian structures, phallus growth, and testicular descent. The absence of testosterone and anti-Müllerian hormone (AMH) in females allows for the development of Müllerian structures and typical female external genitalia.
DSD can result from anomalies at any stage of these complex developmental pathways.
Sex determination refers to the developmental decision that directs the orientation of the bipotential, undifferentiated embryo into a sexually dimorphic individual. In mammals, this decision occurs during the development of the gonads. In 1947, physiologist Alfred Jost performed a series of elegant experiments showing that all mammalian embryos that are castrated early in development and reimplanted into the uterus develop into females, regardless of their genetic sex. , Unilateral grafting of a testis in the embryo resulted in ipsilateral masculinization of structures, demonstrating that testis-specific factors trigger development of male internal genitalia ( Fig. 17.1 ). This study established the current paradigm of sex determination and differentiation in placental mammals: genetic sex determines gonad development, and gonadal sex, in turn, governs anatomical sex.

At conception, genetic sex is determined based on whether one inherits an X or a Y chromosome from one’s father. , Sex determination is defined by the developmental choice of the bipotential and undifferentiated gonad to become either testis or ovary. Following this sex determination decision, the process of sex differentiation begins and the testes start producing the male hormones testosterone (T) and AMH, which are responsible for male internal and external genitalia development, while the lack of these hormones allows the development of female internal and external genitalia ( Fig. 17.2 ).

The molecular mechanisms of mammalian sex determination are still incompletely understood. About half of human DSD cannot be explained at the molecular level yet suggesting the existence of a number of unknown sex-determining genes or mechanisms. The next sections outline the known molecular mechanisms affecting sex determination and sex differentiation.
This paradigm for sex development established by Jost led to the search for a sex-determining gene that was a testis-determining factor. When the karyotypes of patients with Klinefelter syndrome (who are male) and Turner syndrome (who are female) were determined to be 47,XXY and 45,X, respectively, it became clear that the Y chromosome was sex-determining and that the testis-determining factor had to be located on the Y chromosome.
In the early 1990s, a series of elegant experiments found SRY to be the mammalian testis-determining gene. Positional cloning located a 35-kb fragment of the Y chromosome translocated to the X chromosome, explaining the presence of male gonadal tissue in individuals with 46,XX testicular and ovotesticular DSD. , Further analysis defined a conserved sequence within this region. In mice, Sry gene expression analysis revealed a male-specific increase in transcript consistent with the earliest known divergence of male and female gonadal development. Furthermore, XX mice transgenic for a 14-kb fragment containing Sry developed testes and the full male phenotype. This was followed up with the demonstration that deletions and mutations in SRY / Sry result in XY phenotype/karyotype incongruence (formerly known as sex reversal) in both humans and mice.
In humans, 8 weeks after conception the bipotential gonad progresses toward testis organogenesis in the presence of SRY and can be recognized on ultrasound by week 10. Factors expressed earlier than SRY prime the bipotential gonadal ridge to differentiate toward the male pathway. The mitogen-activated protein (MAP)-kinase signaling pathway is important in promoting SRY expression within the developing gonad and mutations in this pathway result in 46,XY DSD with partial or complete gonadal dysgenesis (GD). Chromobox-2 (CBX2) has also been shown to be required for Sry expression in mouse sex determination and a mutation was found in a 46,XY woman. ,
SRY expression in the Sertoli cell precursors initiates testis determination by activating downstream effectors such as SOX9 ( SRY -related high-mobility group [HMG]-box 9). Sertoli cell proliferation and organization into tubular cords are directed by SOX9 and NR5A1/ SF1 (steroidogenic factor-1, encoded by the NR5A1 gene). At 9 weeks of gestation, after the organization of discrete tubules, Sertoli cells begin the secretion of AMH. , NR0B1/ DAX-1 is required in a single copy for testes development as point mutations result in hypogonadotropic hypogonadism, whereas two copies in XY fetuses result in XY GD by repressing testis organogenesis. Another gene involved in sex determination is Desert Hedgehog (DHH) , which up-regulates NR5A1/ SF1 and in turn regulates Leydig cell proliferation. ,
The cellular fate of ovarian and testicular tissues is not sealed at the point of sex determination. In murine models of sex determination, cellular fates of the testis or ovary are actively maintained throughout adult life by DMRT1 (Doublesex and mab-3 related transcription factor 1) and FOXL2 (Forkhead box L2), respectively. , This lack of finality in the gonadal cell fate determination indicates that cell fate is carefully cultivated throughout adulthood, and if the factors important in maintaining cell fate are mutated, it may lead to decreased secondary sex characteristics and/or infertility.
Relative to testis organogenesis, there is less information on the genes involved in ovary organogenesis. Studies suggesting that ovary development does not require a large number of genes are incomplete in that they look at time points that are important in testis determination but may not be critical in ovarian determination. , In fact, several autosomal genes required for proper ovarian development, including WNT-4 , RSPO-1 (R-spondin-1), and FST (follistatin), have been clearly identified and the number of genes linked to ovarian dysgenesis and premature ovarian failure, in isolated or syndromic form, has exploded in the past few years , and new mechanisms involving noncoding RNA have been discovered. At least 25 genes are now listed in the Online Mendelian Inheritance in Man ( OMIM.org ) database under the POF (primary ovarian failure, a term now replaced with primary ovarian insufficiency, POI) or OGD (Ovarian Dysgenesis, Hypergonadotropic) categories (see Table 17.3 ). In addition, ovarian insufficiency is part of several syndromes; for example, six genes are now known to cause Perrault syndrome, a recessive condition where deafness is associated with ovarian dysgenesis in women ( Table 17.4 ).
| Gene | Condition | Variants | DSD Classification | ||||
|---|---|---|---|---|---|---|---|
| Name | Chr Location | Inheritance | Name | OMIM# | Types | XX or XY | Mechanism |
| AKR1C2 | 10p15.1 | AR | SRXY8 | 614279 | M, fusion | XY | Gonadal dysgenesis |
| AMH | 19p13.3 | AR | PMDS | 261550 | FS, N | XY | Sex differentiation (other) |
| AMHR2 | 12q13.13 | AR | PMDS | 261550 | FS, Sp, M, Del | XY | Sex differentiation (other) |
| AR | Xq12 | XLR, de novo | CAIS, PAIS | 300068 | Del, M, N, Sp, FS | XY | Androgen action |
| BMP15 | Xp11.22 | XL | ODG2/POF4 | 300510 | M, N | XX | Ovarian development |
| BNC1 | 15.25.2 | AD | POF16 | 618723 | FS | XX | Ovarian development |
| CBX2 | 17q25.3 | AR | SRXY5 | 613080 | M | XY | Gonadal dysgenesis |
| CYP11A1 | 15q24.1 | AR | P450scc deficiency | 613743 | M, FS, Del | XY | Androgen synthesis |
| CYP11B1 | 8q24.1 | AR | CAH | 202010 | M, N, FS | XX | Androgen excess |
| CYP17A1 | 10q24.32 | AR | CAH, 17,20 lyase deficiency | 202110 | Sp, Del, FS, M, N, rearrangement | XY | Androgen synthesis |
| CYP19A1 | 15q21.2 | AR | CAH, aromatase deficiency | 613546 | fusion, del, M, N, intronic | XX | Androgen excess |
| CYP21A2 | 6p21.33 | AR | CAH | 201910 | Del, dup, ins, intron, M | XX | Androgen excess |
| DHH | 12q13 | AR | SRXY7 | 233420 | FS, N, M | XY | Gonadal dysgenesis |
| DHX37 | 12q24.31 | AD | SRXY11 | 273250 | M | XY | Gonadal dysgenesis |
| DIAPH2 | Xq21.22 | XLD | POF2A | 300511 | translocation | XX | Ovarian development |
| DMRT1 | 9p24.3 | de novo | SRXY4 | 154230 | 9p24 deletion | XY | Gonadal dysgenesis |
| ERCC6 | 10q11.23 | AD | POF11 | 616946 | M, N | XX | Ovarian development |
| ESR2 | 14q23.2 | AD | ODG8 | 618187 | M | XX | Ovarian development |
| FANCM | 14q21.2 | AR | POF15 | 618096 | N | XX | Ovarian development |
| FIGLA | 2p13.3 | AD | POF6 | 612310 | FS, M | XX | Ovarian development |
| FMR1 | Xq27.3 | XL | POF1 | 311360 | premutation | XX | Ovarian development |
| FOXL2 | 3q22.3 | AD | POF3 | 608996 | FS, del, M | XX | Ovarian development |
| FSHR | 2p16.3 | AR | ODG1 | 233300 | M | XX | Ovarian development |
| GDF9 | 5q31.1 | AR | POF14 | 618014 | FS, promoter | XX | Ovarian development |
| HFM1 | 1p22.2 | AR | POF9 | 615724 | N, M | XX | Ovarian development |
| HSD3B2 | 1p12 | AR | CAH | 201810 | FS, N, M | XY | Androgen synthesis |
| HSD17B3 | 9q22.32 | AR | HSD17B3 deficiency | 264300 | M, Sp, FS, N | XY | Androgen synthesis |
| MAP3K1 | 5q11.2 | AD | SRXY6 | 613762 | M, Sp | XY | Gonadal dysgenesis |
| MCM8 | 20p12.3 | AR | POF10 | 612885 | M, Sp, FS | XX | Ovarian development |
| MCM9 | 6p22.31 | AR | ODG4 | 616185 | Sp, N | XX | Ovarian development |
| MRPS22 | 3q23 | AR | ODG7 | 618117 | M | XX | Ovarian development |
| MSH5 | 6p21.33 | AR | POF13 | 617442 | M | XX | Ovarian development |
| NOBOX | 7q35 | AD | POF5 | 611548 | M, N | XX | Ovarian development |
| NR0B1 | Xp21.2 | XL | SRXY2 | 300018 | duplication | XY | Gonadal dysgenesis |
| XLR | Adrenal hypoplasia congenita | 300200 | del, M, N | XX | Androgen excess | ||
| NR2F2 | 15q26.2 | AD | SRXX5, with cardiac defect | 618901 | FS, Del | XX | Testicular or ovotesticular DSD |
| NR5A1 | 9q33.3 | AD | POF7 | 612964 | N, M, Del, FS | XX | Ovarian development |
| AD | SRXX4 | 617480 | R92W variant | XX | Testicular or ovotesticular DSD | ||
| AD | SRXY3 | 612965 | N, M, Del, FS | XY | Gonadal dysgenesis | ||
| NUP107 | 12q15 | AR | ODG6 | 618078 | M | XX | Ovarian development |
| POF1B | Xq21.1 | XLR | POF2B | 300604 | M | XX | Ovarian development |
| POR | 7q11.23 | AR | CAH, CytP450 oxidoreductase deficiency | 613571 | Sp, M, FS, Del | XY | Androgen synthesis |
| XX | Androgen excess | ||||||
| PSMC3IP | 17q21.2 | AR | ODG3 | 614324 | FS, Del | XX | Ovarian development |
| SOHLH1 | 9q34.3 | AR | ODG5 | 617690 | FS, N | XX | Ovarian development |
| SOX3 | Xq27.1 | de novo | SRXX3 | 300833 | CNV, promoter | XX | Testicular or ovotesticular DSD |
| SOX9 | 17q24.3 | de novo | SRXX2 | 278850 | dup XXSR upstream region | XX | Testicular or ovotesticular DSD |
| AD | SRXY10 | 616425 | del XYSR upstream region | XY | Gonadal dysgenesis | ||
| AD | Campomelic dysplasia with GD | 114290 | N, M, FS, Sp, Del, rearrangement | XY | Gonadal dysgenesis | ||
| SRY | Yp11.2 | YL | SRXX1 | 400045 | translocation | XX | Testicular or ovotesticular DSD |
| YL | SRXY1 | 400044 | M, N, FS, Del | XY | Gonadal dysgenesis | ||
| SRD5A2 | 2p23.1 | AR | 5α-reductase deficiency | 264600 | FS, M, N, Sp, Del | XY | Androgen synthesis |
| STAG3 | 7q22.1 | AR | POF8 | 615723 | FS, Sp, N | XX | Ovarian development |
| StAR | 8p11.23 | AR | Lipoid CAH | 201710 | FS, Sp, M, N | XY | Androgen synthesis |
| SYCE1 | 10q26.3 | AR | POF12/SPGF15 | 616947 | N | XX | Ovarian development |
| WNT4 | 1p36.12 | AD | CGD or ovoT DSD | __ | duplication | XY | Gonadal dysgenesis |
| AD | Müllerian hypoplasia + hyperandrogenism | 158330 | M | XX | Testicular or ovotesticular DSD | ||
| WT1 | 11p13 | AD | Frasier | 136680 | Sp | XY | Gonadal dysgenesis |
| AD | Denys–Drash | 194080 | Exons 8 & 9 | XY | Gonadal dysgenesis | ||
| AD | T DSD, OvoT DSD | __ | M, FS in ZF4 domain | XX | Testicular or ovotesticular DSD | ||
| ZFPM2 | 8q23.1 | AD | SRXY9 | 616067 | M | XY | Gonadal dysgenesis |
| Syndrome | Gonadal Phenotype | Associated Traits | OMIM # | Gene | Locus |
|---|---|---|---|---|---|
| Genital Phenotype Seen Only in XY Individuals | |||||
| 3MC3 syndrome ∗ (formerly Malpuech) | UT, possible ectopic testis | Hypertelorism, facial clefting, short stature, eye anomalies, hearing loss, DD μPh, bifid scrotum, UT, penoscrotal hypospadias |
248340 | COLEC10 | 8q24.12 |
| 9p deletion syndrome | GD, Gba | Trigonocephaly, hypertonia, DD. Hypoplastic or ambiguous genitalia. | 158170 | DMRT1 + others | 9p |
| GD, immature T w/o germ cells, Gba | 9p24.3 del. syndrome. Genital phenotype only. Female external genitalia, uterus. One case with OvoT. | 154230 | DMRT1 | 9p24.3 | |
| Aarskog-Scott syndrome | Aka, faciogenital dysplasia: short stature, hypertelorism, brachydactyly, shawl scrotum | 305400 | FGD1 | Xp11.21 | |
| ACOGS | UDT | agenesis of corpus callosum, cardiac, ocular, and genital syndrome. Undescended testes, micropenis. | 618929 | CDH2 | 18q12.1 |
| Axenfeld-Rieger | NL? | Hypodontia, malformations of anterior chamber of eye and iris, face dysmorphism. Hypospadias, ambiguous genitalia in XY due to pituitary dysfunction. | 180500 | PITX2 | 4q25 |
| Cabezas. INTELLECTUAL DEVELOPMENTAL DISORDER, X-LINKED, SYNDROMIC, CABEZAS TYPE; MRXSC | small testes | Short stature, abnormal gait, ID, prominent lower lip and other dysmorphic features. Hypogonadism in 85%, Gynecomastia in 33%. Delayed puberty | 300354 | CUL4B | Xq24 |
| Camp(t)omelic dysplasia | cGD or ovoT in XY | Congenital bowing of long bones, hypoplastic scapulae, hypoplastic pedicles of thoracic vertebrae. Female external genitalia. | 114290 | SOX9 | 17q24.3 |
| Cornelia de Lange syndrome | UT | Facial dysmorphism, low hairline, arched eyebrows, synophrys, growth retardation, DD. Hypoplastic male genitalia. | CDLS1 122470 CDLS5 300882 |
NIPBL HDAC8 | 5p13.12 Xq13 |
| Cystic fibrosis | Normal T | Congenital bilateral absence of the vas deferens in XY | 602421 | CFTR | 7q31.2 |
| Dubowitz syndrome | UT | Short stature, microcephaly, mild DD with behavioral problems, eczema, distinctive facies, high-pitched voice. Hypospadias. | 233370 | UNK | UNK |
| GD with nephropathy (also see WAGR syndrome below) | Female external genitalia. Kidney sclerosis, Wilms tumor, nephrotic syndrome are overlapping traits in: | WT-1 | 11p13 | ||
| cGD | Denys-Drash syndrome | 194080 | |||
| cGD, Gba | Frasier syndrome | 136680 | |||
| small T | Meacham syndrome † | 608978 | |||
| cGD | Nephrotic syndrome type 4 | 256370 | |||
| GUBS | Ovaries cGD (streak) not found |
Brain malformations. Spectrum: UT, μPh, hypospadias or clitoromegaly, with labial fusion Müllerian anomalies. Presence of Müllerian structures and female genitalia. | 618820 | PPP1R12A | 12q21 |
| IMAGe syndrome | UDT | Intrauterine growth retardation, Metaphyseal dysplasia, Adrenal aplasia congenita, Genital anomalies. Small penis, hypogonadotropic hypogonadism. | 614732 | CDKN1C | 11p15.4 |
| Lenz-Majewski hyperostotic dwarfism | UT | Progressive skeletal sclerosis, growth retardation, progeroid appearance, palate and teeth defects Hypospadias, chordee |
151050 | PTDSS1 | 8q22.1 |
| Noonan syndrome (see LEOPARD syndrome below) | UT in ∼50% of XY | Facial dysmorphism, short stature, webbed neck, cardiac anomalies (Turner-like features) | 163950 | PTPN11 (NS1, LPDS1) LZTR1 (NS2 AR, NS10 AD) KRAS (NS3) SOS1 (NS4) RAF1 (NS5) NRAS (NS6) BRAF (NS7, LPDS3) RIT1 (NS8) SOS2 (NS9) MRAS (NS11) RRAS2 (NS12) MAPK1 (NS13) |
12q24.13 |
| Pallister-Hall syndrome | UT | Hypothalamic hamartoblastoma, postaxial polydactyly, imperforate anus, IUGR, renal anomalies μPh, UT in XY |
146510 | GLI3 | 7p14.1 |
| Proud syndrome | UT | “Agenesis of corpus callosum with abnormal genitalia”, Lissencephaly. Hypospadias | 300004 | ARX | Xp21.3 |
| Rubinstein Taybi syndrome | UT in 80%–100% of XY | Poor postnatal growth, DD, microcephaly, broad thumbs and toes, facial dysmorphism, hirsutism Shawl scrotum, hypospadias Genital involvement is not described in association with E300 variants. |
180849 | CREBBP EP300 |
16p13.3 22q13.2 |
| SIDDT | dysplastic, UT | Visceroautonomous dysfunction leading to sudden death in infancy. Ambiguous genitalia and other malformations. | 608800 | TSPYL1 | 6q22.1 |
| Testicular anomalies with or without congenital heart disease | UT, Testicular calcifications | Ambiguous genitalia, spectrum of cardiac malfomations | 615542 | GATA4 N-terminal Zinc Finger | 8p23.1 |
| TKCR syndrome | Oligospermia | Torticollis, keloids, cryptorchidism, renal dysplasia UT, infertility in XY | 314300 | UNK | Xq28? |
| VACTERL/VATER | NL? | Vertebral defects, anal atresia, tracheoesophageal fistula with esophageal atresia, radial and/or renal dysplasia Hypospadias in XY |
192350 | UNK | UNK |
| X-linked MR with hypotonic facies | UT | μPh, hypoplastic scrotum in individuals raised as males | ATRX | Xq21.1 | |
| With α-thalassemia | 301040 | ||||
| Juberg-Marsidi syndrome | 309580 | ||||
| Yunis-Varon syndrome | UT | Cleidocranial dysplasia, neural loss, finger anomalies. Hypospadias, micropenis in XY |
216340 | FIG4 | 6q21 |
| Genital Phenotype Seen Only in XX Individuals | |||||
| BPE syndrome type I | POF | Blepharophimosis, ptosis, epicanthus inversus | 110100 | FOXL2 | 3q22.3 |
| Generalized glucocorticoid resistance | NL? | Hypertension, hypoglycemia, metabolic alkalosis, anxiety. Hyperandrogenism causing male-pattern baldness, hirsutism, Infertility, irregular periods in some. | 615962 | NR3C1 | 5q31.1 |
| Herlyn-Werner-Wunderlich syndrome | NL? | Uterus didelphys, blind hemivagina with ipsilateral renal agenesis. Also called OHVIRA = obstructed hemivagina and ipsylateral renal anomaly. | — | UNK | UNK |
| Leukodystrophy with ovarian failure | POF | Progressive leukoencephalopathy | 615889 | AARS2 | 6p21 |
| Mayer-Rokitansky-Küster-Hauser (MRKH) syndrome | Normal ovaries | Müllerian agenesis or anomalies. Primary amenorrhea. Associated skeletal anomalies |
277000 | UNK Contiguous genes deletion |
UNK 22q11.2? 17q12? |
| + Hyperandrogenism (rare) | 158330 | WNT4 | 1p36 | ||
| MURCS association | Absent ovary NL ovarian function |
Müllerian aplasia, renal aplasia, cervicothoracic somite dysplasia | 601076 | UNK | UNK |
| Ovarioleukodystrophy | Ovarian failure | Leukoencephalopathy with vanishing white matter; primary or secondary amenorrhea. Elevated gonadotropin | 603896 | EIF2B2 EIF2B4 EIF2B5 |
14q24.3 2p23.3 3q27.1 |
| Palmoplantar hyperkeratosis with squamous cell carcinoma | Hypoplastic T or OvoT, hyperplasia of Leydig cells, seminoma | Clitoral enlargement, premature menopause in ovoT DSD. Hypospadias, gynecomastia, μPh. Low T, high FSH in T-DSD | 610644 | RSPO1 | 1p34.3 |
| Perrault syndrome | Streak gonads in XX | Sensorineural deafness with primary amenorrhea, infertility, ovarian dysgenesis, POF in XX. + Progressive neurological disease in Type II | 233400 614926 614129 615300 616138 617565 |
HSD17B4 HARS2 CLPP LARS2 TWNK ERAL1 |
5q23.1 5q31.3 19p13.3 3p21.31 10q24.31 17q11.2 |
| SRXX5, with cardiac defect | T or OvoT | BPES, congenital diaphragmatic hernia. Undescended testes or ambiguous genitalia | 618901 | NR2F2 (COUP-TFII) | 15q26.2 |
| Genital Phenotype Seen in XX or XY Individuals | |||||
| Bardet-Biedl syndrome | Small T in XY Small ovaries in XX |
Retinal dystrophy, renal abnormalities, polydactyly, DD, obesity, mood disorders Small testes and genitalia in XY Menstrual irregularities, vaginal atresia, Müllerian anomalies in females |
209900 | BBS1-BBS20 20 genes & modifiers |
Various |
| Beckwith Wiedemann | UDT in males | Overgrowth (hemihypertrophy, macroglossia), hypoglycemia, predisposition to tumors. Overgrowth of external genitalia in males and females | 130650 | ICR1 CDKN1C KCNQ10T1 | 11p15.5 |
| Borjeson-Forssman-Lehmann syndrome | Hypogonadism, small testes in XY | Severe DD, epilepsy, hypometabolism, obesity, prominent ears Small external genitalia and prostate, postpubertal gynecomastia in XY; delayed puberty, amenorrhea in XX |
301900 | PHF6 | Xq26.2 |
| Cardiomyopathy, dilated with hypergonadotropic hypogonadism ‡ | POI in XX Primary T failure in XY |
Cardiac valve insufficiency, ± skeletal anomalies, ±DD. Elevated LH, FSH | 212112 | LMNA | 1q22 |
| CHARGE syndrome § | HH | Coloboma, heart defects, choanal atresia, DD, ear malformations. HH of central origin, with delayed puberty in XX and XY Micropenis, UT in 50% of XY, genital anomalies in 25% of XX |
214800 | CHD7 | 8q12 |
| Cardiac Urogenital Syndrome (CUGS) | UT in XY | UDT, penoscrotal hypospadias, ambiguous genitalia or Swyer in XY and males. Aplasia or hypoplasia of Müllerian structures in XX. 94% have Cardiac anomalies, 75% urogenital, 63% diaphramatic, 44% lung hypoplasia | 612280 | MYRF | 11q12.2 |
| Popliteal pterigial syndrome | UT in XY NL? in XX |
Cleft lip/palate, webbing of the intercrural pterygium Bifid scrotum, UT in XY. Hypoplastic labia in XX. |
119500 | IRF6 | 1q32.2 |
| Fragile-X syndrome | Normal T in affected XY POI in XX carriers |
DD, macroorchidism (with normal testicular function) in XY POI in XX carriers of premutation |
300624 | FMR1 | Xq27.3 |
| Fraser syndrome | UT in XY | Cryptophthalmos, syndactyly, DD UT, μPh, hypospadias in males Clitoromegaly, labial, and Müllerian anomalies in females |
219000617666617667 | FRAS1 FREM2 GRIP1 |
4q21.21 12q14.3 13q13.3 |
| Hand-foot-genital syndrome | ? | Fully penetrant, bilateral, first and other digit and toe anomalies. Hypospadias in males, Müllerian anomalies in females with variable penetrance. | 140000 | HOXA13 | 7p15.2 |
| HH with or without anosmia | HH | Incl. Kallmann syndrome | 23 loci | ||
| Oligogenic inheritance with combined variants at different loci | 147950 | FGFR1 | 8p11.23 | ||
| + X-linked form | 308700 | KAL1/ANOS1 | Xp22.31 | ||
| HH with obesity | small testes and ovaries | Obesity, hyperphagia. Gynecomastia, small penis in males. Primary amenorrhea in females. | 614962 | LEP | 7q32.1 |
| Johanson-Blizzard syndrome | UT in XY NL? in XX |
Nasal alar hypoplasia, pancreatic achylia, congenital deafness, hypothyroidism. μPh, UT, hypospadias in XY; clitoromegaly, vaginal malformations in XX | 243800 | UBR1 | 15q15.2 |
| LEOPARD syndrome | Absent, hypoplastic ovaries in XX UT in XY |
Lentigines, ECG anomalies, ocular defects, pulmonary stenosis, “abnormal genitalia,” retardation of growth, deafness Oligodysmenorrhea, delayed puberty in XX. Hypospadias in XY. |
|||
| LPRD1—hypospadias, UT | 151100 | PTPN11 | 12q24.1 | ||
| LPRD2—UT in 75% of XY | 611554 | RAF1 | 3p25.2 | ||
| LPRD3—unk genital involvement | 613707 | BRAF | 7q34 | ||
| McKusick-Kaufman syndrome | UT in XY NL? In XX |
Postaxial polydactyly and congenital heart malformation Hydrometrocolpos in XX Hypospadias, UT, μPh in XY |
236700 | MKKS (=BBS6) | 20p12.2 |
| Meckel Syndrome | UT in XY | XX and XY: Small or ambiguous genitalia. XX: Separated vagina, uterine abnormalities. Renal, CNS, liver malformations ± postaxial polydactyly. | 249000 | MKS1 and 12 other | 17q22 |
| Methemoglobinemia and ambiguous genitalia | UDT in XY | Disturbed pubertal development in both XX and XY. Bifid scrotum, micropenis, hypospadias in XY. | 613218 | CYB5A | 18q22.3 |
| OEIS complex/cloacal anomaly spectrum | NL? | Omphalocele, exstrophy of the cloaca and bladder; imperforate anus, spinal defects. Epispadias, UT in XY; labioscrotal malformations in XY & XX; bifid uterus in XX. | 258040 | UNK | UNK |
| Opitz GBBB syndrome | UT, ectopic T, in XY | Hypertelorism with hypospadias and esophageal abnormality, DD | 300000 (GBBB1) | MID1 | Xp22.2 |
| Anomalies of scrotum, meatus, ureter in XY. Labial and hymen anomalies in rare XX. | 145410 (GBBB2) | SPECCIL | 22q11.23 | ||
| Prader-Willi syndrome | HH | Hypotonia, DD, short stature, obesity, small hands and feet UT, μPh, hypoplastic scrotum in XY Hypoplastic labiae, delayed and reduced menstruation in XX |
176270 | Contiguous gene syndrome incl. SNRPN, NDN | 15q11.2 |
| Renal cysts and diabetes syndrome (RCAD) | NL? | In males: hypospadias, epidydimal cysts, asthenospermia, atresia of vas deferens. In females, Vagina aplasia, bicornuate or rudimentary uterus. CAKUT. Renal cysts or hypoplasia. MODY | 13792s0 | HNF1B/TCF2 | 17q12? |
| Robinow syndrome | UT in XY NL? In XX |
Mesomelic limb shortening associated with facial and genital abnormalities μPh ±UT in XY; small clitoris and labia in XX |
268310 618529180700 616331 616894 |
ROR2 NXN WNT5A DVL1 DVL3 |
9q22.31 17p133p14.3 1p36.33 3q27.1 |
| Smith-Lemli-Opitz syndrome | Testis OvoT Ovary Streak Absent |
Multiple congenital malformations + DD Cholesterol metabolism defect Hypospadias, bifid scrotum, UT, or female external genitalia in XY Delayed menarche, irregular menses in XX |
270400 | DHCR7 | 11q13.4 |
| Townes-Brocks syndrome | UT in XY NL? In XX |
Malformations of anus, ears, thumbs, kidneys, heart (ToF VSD). Uterus and vaginal anomalies; UT; urethrostenosis. | 107480 617466 |
SALL1 ‖ DACT1 |
16q12.1 14q23.1 |
| WAGR syndrome | Streak Ovaries Gba UT in XY |
Wilms tumor, Aniridia, Genital anomalies, DD hypospadias, UT in XY ambiguous genitalia, Müllerian anomalies in XX | 194072 | WT1+ PAX6 deletion | 11p13 |
∗ 3MC includes 4 rare AR syndromes: Malpuech, Michaels, Carnevale, Mingarelli. Genital involvement only in 3MC3.
§ Allelic disorder and overlapping phenotype with HH5 (Kallmann syndrome; OMIM# 612370 ).
‖ Goldenhar syndrome (hemifacial microsomia; OMIM # 164210 ; genetic etiology unknown) has overlapping features also seen in families with SALL1 mutations.
‡ Includes Najjar and Malouf syndromes.
† Meacham syndrome includes retention of Müllerian structures, testis linked to Fallopian tubes, double vagina, absent uterus, cardiac and diaphragm malformations.
The type of internal ( Fig. 17.3 ) and external ( Fig. 17.4 ) genitalia is decided by different testicular and ovarian factors governing sex differentiation. The bipotential ductal system consists of the Müllerian and Wolffian ducts, which give rise to female and male internal genitalia, respectively.

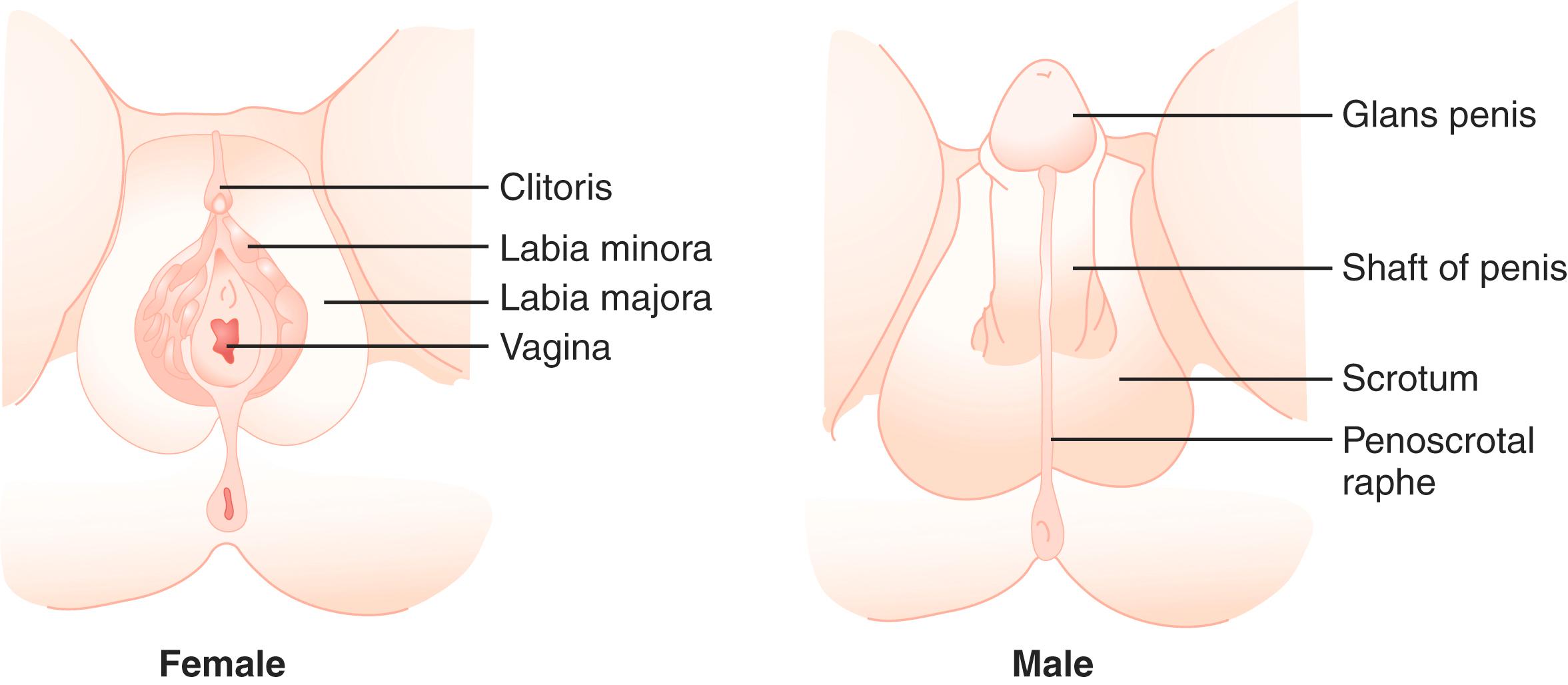
Hormones secreted from testes are essential to the development of male internal and external genitalia. Normally developed testes have both Sertoli cells and testicular cords. Sertoli cells secrete AMH, causing Müllerian duct regression. At the same time, Leydig cells, the steroidogenic cells of the testes, secrete testosterone and INSL3 (insulin-like 3), which promote the development of Wolffian structures (epididymis, vas deferens, and seminal vesicles) and mediate transabdominal descent of the testes to the internal inguinal ring, respectively. Dihydrotestosterone (DHT), a more potent androgen converted from testosterone by the enzyme 5α-reductase, mediates the development of the male external genitalia. Except for phallic growth and inguinoscrotal descent in the third trimester, male sexual differentiation is essentially complete by week 14 of gestation.
In females, the absence of testicular tissue and its associated hormones allows Müllerian structure development (Fallopian tubes, uterus, upper vagina). The regression of Wolffian ducts (occurring at around 12 weeks of gestation in humans) is actively induced by COUP-TFII (encoded by the NR2F2 gene), which suppresses a mesenchyme-epithelium cross-talk responsible for the maintenance of the Wolffian ducts. Finally, in the absence of DHT, no virilization of external genitalia occurs.
DSD can result from anomalies in any of the processes described above. They can present as an isolated urogenital atypia but can also be part of a large number of multiorgan syndromes, underscoring the need for an accurate molecular diagnosis to seek and manage associated comorbidities. ( Fig. 17.5 ) illustrates some of the most well-known syndromic conditions, including main extragenital affected organs, inheritance patterns, and causative genes when known. Gonadal and external genitalia phenotypes associated with syndromic forms of DSD are further detailed, with their genetic cause, in Table 17.4 (see Hutson et al. 2014 for classification of syndromic DSD by the affected biological process).
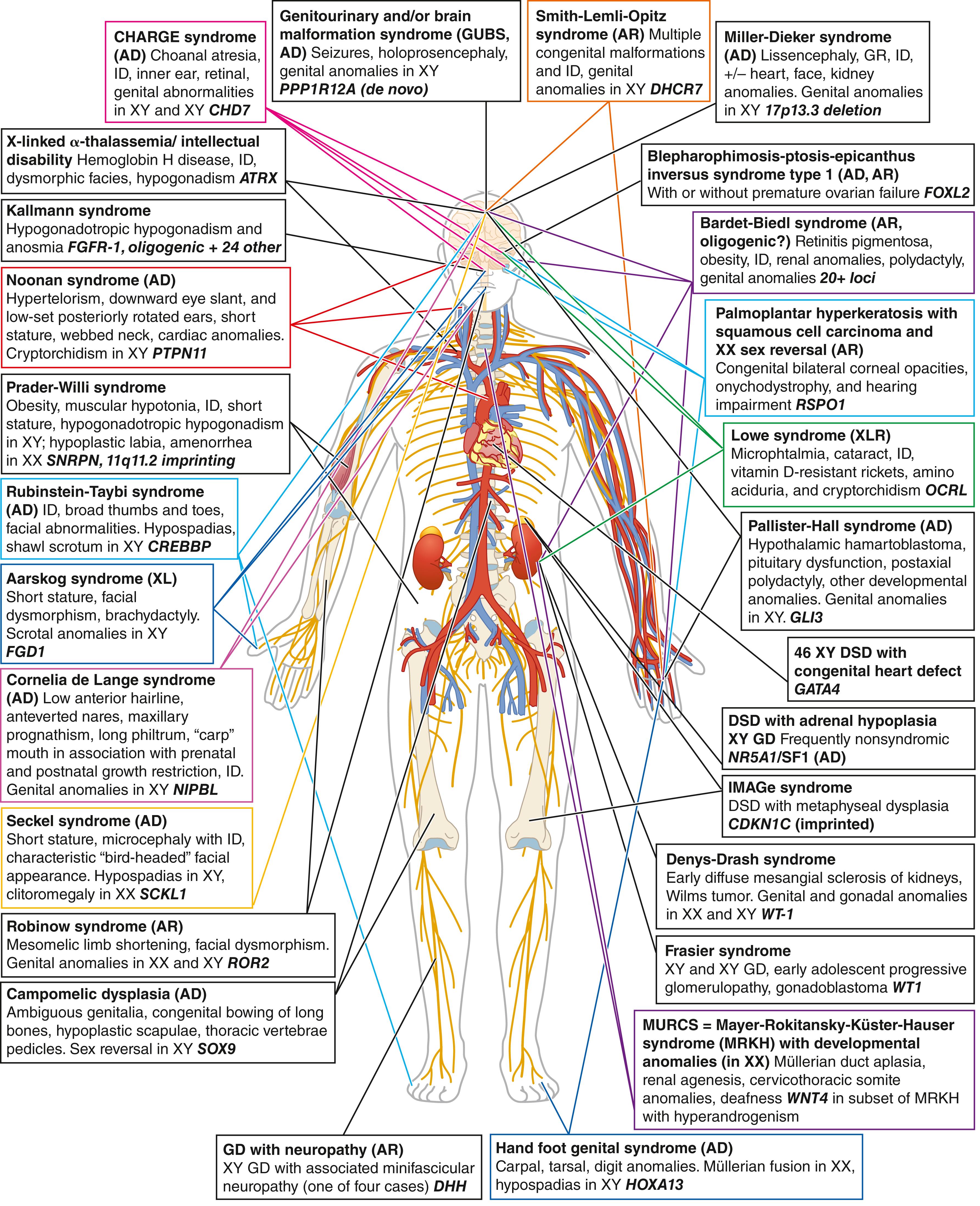
Conditions associated with dysgenetic gonads result from events affecting sex determination, while conditions that can cause ambiguous genitalia but are not associated with dysgenetic gonads, such as androgen insensitivity syndrome (AIS) or congenital adrenal hyperplasia (CAH), are considered differences in sexual differentiation. Overview of the anatomical and biochemical diagnostic differences in DSD, including both disorders of sex determination and disorders of sex differentiation, are presented in Table 17.5 . An exhaustive algorithm for diagnostic of over 50 conditions presenting with ambiguous genitalia was also recently designed.
| DSD | Biochemical Changes | Differentiating Features | Major Genetic Diagnostic Criteria | |
|---|---|---|---|---|
| 46,XY disorders of gonadal development | 46,XY pure/partial/mixed gonadal dysgenesis | Complete/partial/mixed ↑FSH, LH Nml to ↓ AMH No ↑ with hCG NmL adrenal hormones and precursors Pure ↓↓; T, DHT, E2 ↓↓ AMH Partial/mixed ↓ T, DHT, E2 ↓ T, DHT, E2 |
Presence of partial testicular function (T, AMH) points toward partial or mixed GD. However, histologic examination of testes after prophylactic gonadectomy can differentiate between pure, partial, and mixed GD. There is little genotype-phenotype correlation; however, the presence of mosaicism 45,X/46,XY is often associated with mixed GD. | Sequencing to assess for mutations in known DSD genes. Variants in NR5A1 / SF1 or SRY provide a definitive genetic diagnosis. If partial/mixed GD suspected, patient should be tested for mosaicism. CMA should be performed to look for copy number variants. |
| Testicular regression | ↑ FSH, LH ↓↓ T, DHT, E2 ↓↓ AMH No ↑ with hCG |
Complete absence of gonad or fibrosis of gonad (as opposed to streak or dysgenetic testes in GD patients). Completely virilized male phenotype. | Fibrous nodules on laparoscopic exam (not streak or dysgenetic testes). | |
| Disorders of sex differentiation: disorders in androgen synthesis and action | Lipoid CAH: StAR deficiency P450scc deficiency |
↑ Renin ↓ Aldo ↑ K ↓ Na ↓ All adrenal hormones ↓17-OHP |
Presence of lipid vacuoles in adrenals on histology. P450scc deficiency does present with enlarged adrenals. No HTN and hyperkalemia differentiates from CYP 17A1 deficiency. | Presence of lipid-filled vacuoles on histology; sequencing to assess for mutations in known DSD genes. Variants in the STAR or CYP11A1 genes give a definitive diagnosis. |
| 3βHSD type II deficiency | ↑ Renin ↓ Aldo, F ↑ K ↓ Na ↑ Ratio Δ 5 -17-pregnenolone: cortisol ↓ Δ4A, T |
Baseline and ACTH-stimulated ratios of Δ 5 -17-pregnenolone:cortisol consistently distinguished between affected and nonaffected patients. | Sequencing to assess for mutations in known DSD genes. Variants in the HSD3B2 gene provide a definitive genetic diagnosis. | |
| 17α-hydroxylase/17,20-lyase deficiency | ↓ Renin ↓ Aldo, F ↓17-OH progesterone ↑ LH, FSH ↑ Progesterone, DOC, B ↑ Na ↓ K ↓ DHEA-S, Δ4A, T; no response to hCG stim |
Hypertension and hypokalemic alkalosis in the presence of low 17-OH progesterone. Isolated 17,20-lyase deficiency would have decreased levels of sex steroids with normal mineralocorticoids and normal glucocorticoids. | Sequencing to assess for mutations in known DSD genes. Variants in CYP17A1 or CYB5A genes provide a definitive diagnosis. | |
| P450 oxido-reductase deficiency | ↑ 17OH-progesterone ↑ Progesterone ↓ F, DHEA-S ↓ Δ4A, T |
Hypertension, in the presence of elevated 17-OH progesterone (differentiates from CYP17 deficiency); sometimes presence of Antley-Bixler skeletal malformations. | Sequencing to assess for mutations in known DSD genes. Variants in the POR gene provide a definitive diagnosis. | |
| 17βHSD type 3 deficiency | Nml to ↑ Δ4A↑ ratio Δ4A/T (> 15) ↓ T, DHT |
Differentiated from 5α-reductase type 2 by levels and ratios of serum hormones. | Sequencing to assess for mutations in known DSD genes. Variants in the HSD17B3 gene provide a definitive diagnosis. | |
| Leydig cell hypoplasia | ↑ LH Nml FSH ↑ AMH ↓ T, DHT, E 2 ↓ hCG response Nml Δ4A/T ratio |
To differentiate LCH from GD, AMH is used as a marker of testicular function. | Sequencing to assess for mutations in known DSD genes. Deletions, insertions, and point mutations in the LHCGR gene provide a definitive diagnosis. | |
| 5α-reductase type 2 deficiency | Nml FSH, LH Nml T, E 2 ↓ DHT↑ ratio T/DHT (> 30) |
Development of male secondary sex characteristics in puberty with fine and sparse facial hair. These can be differentiated clinically from HSD17B3 deficiency and AIS by the lack of gynecomastia during puberty. | Sequencing to assess for mutations in known DSD genes. Point mutations, deletions, insertions, and parental isodisomy in the SRD5A2 gene provide a definitive diagnosis. | |
| Mutations in the backdoor pathway to androgen activity | Nml ratio T/DHT (< 30) Nml AMH Nml cortisol ↓ DHEA-S after hCG ↑ 17OH-pregnenolone |
Similar in phenotype to 17βHSD and 5-alpha reductase deficiency but can be differentiated by serum hormone levels. | Sequencing to assess for mutations in known DSD genes. Variants in the AKR1C2 ( ± AKR1C4 ) may provide a diagnosis. | |
| Androgen insensitivity | Nml FSH, LH (PAIS) ↓ FSH, LH (CAIS) Nml to ↑ AMH Nml Δ4A, T, DHT; nml ratios ↑ ↑ hCG response |
Female phenotype with breast development at puberty, with sparse pubic and axilla hair. | Sequencing to assess for mutations in known DSD genes. Variants in AR gene are diagnostic. | |
| Persistent Müllerian duct syndrome | Nml hormonal profile | Presence of both Müllerian and Wolffian derivatives, usually discovered incidentally. | Compound heterozygote or homozygote variants in the AMH or AMHR2 genes are diagnostic. | |
| 46,XX disorders of sex determination | Testicular/ovo-testicular DSD | ↑ FSH, LH ↓ T, DHT Nml AMH No ↑ with hCG |
Only way to differentiate between 46,XX testicular and ovotesticular DSD is by complete gonadal histologic examination looking for the presence of ovarian tissue with follicles. | FISH for presence of SRY or SOX9 . CMA to identify copy number variants upstream of SOX9 or SOX3 . Search for XX/XY mosaicism (∼a third of ovotesticular DSD). Sequencing to identify diagnostic variants in SRY (∼10% of ovotesticular DSD), RSPO1, R92W variant in NR5A1 , and others. |
| XX ovarian dysgenesis | ↑ FSH, LH No ↑ with hCG No AMH |
Full female phenotype with amenorrhea and lack of secondary sex development. | Sequencing to assess for mutations in 20+ known ovarian dysgenesis genes. CMA may also identify gene duplications or deletions. | |
| Disorders of androgen excess | 21α-hydroxylase deficiency | ↑ Renin ↓Aldo, DOC, F↑ K ↓ Na↑ 17OH-progesterone↑ DHEA-S, Δ4A, T | 17-OH progesterone elevated in the absence of hypertension, which is typical in 11βHSD1 deficiency. | Homozygous or compound heterozygous variants in the CYP21A2 gene are diagnostic. Panel of most common variants in CYP21A2 may not identify variants in all ethnicities and reflex to full sequence is recommended. Knowledge of the mutations can inform genetic counseling. |
| 11βHSD1 deficiency | ↓ Aldo, F, Renin↑ DOC↑ K ↓ Na↑ 17OH-progesterone↑ DHEA-S, Δ4A, T | Differentiate from CYP21 deficiency by presence of hypertension and hypokalemic alkalosis. | Sequencing to assess for mutations in known CAH genes. Variants in CYP11B1 gives a definitive diagnosis. | |
| P450 aromatase deficiency | ↑ 16OH-Δ4A (maternal)↑ FSH, LH, Δ4A, T↓Estrone, E 2 | The presence of maternal virilization during pregnancy and XX virilization which stops after delivery. | Sequencing to assess for mutations in known CAH genes. Mutations in the CYP19A1 gene gives definitive diagnosis. |
Below we will describe the presentation and management of the different types of conditions associated with DSD:
Sex chromosome DSD, associated with an abnormal sex chromosome complement.
46,XY DSD, which includes disorders of gonadal development (complete, partial, or mixed gonadal dysgenesis, where sex determination pathways are affected), as well as disorders of the sex differentiation pathways, including disorders of androgen action (complete or partial androgen insensitivity, AIS) and disorders of androgen synthesis.
46,XX DSD, which include disorders of androgen excess (congenital adrenal hypoplasia, CAH), Testicular and ovotesticular DSD (with male or ambiguous genitalia and presence of testicular tissue), Ovarian dysgenesis (primary or secondary amenorrhea in phenotypic females, e.g., POI), and developmental syndromes such as Mayer Rokitansky Künster Hauser (MRKH).
Table 17.3 presents a list of causative genes, including chromosomal location, pattern of inheritance, types of causative mutations, associated conditions, and affected pathways.
Sex chromosome aneuploidies, in mosaic and nonmosaic forms, result in recognizable syndromes such as Turner syndrome (45,X and associated karyotypes) and Klinefelter syndrome (47,XXY and variants).
Sex chromosome DSDs are defined by aneuploidy of the sex chromosomes, X and Y. In disorders of aneuploidy (e.g., Trisomy 21), maternal errors in meiotic nondisjunction during meiosis-I account for the majority of cases. However, in Klinefelter syndrome (47,XXY), maternal and paternal errors are equally likely, and advanced maternal age is not a risk factor for Turner syndrome.
In 1938, a group of females with short stature, primary amenorrhea, and a lack of secondary sexual characteristics was described by Turner. Turner syndrome is classified as a sex chromosome DSD in the DSD Consensus Statement. There is, however, a growing discussion about whether Turner syndrome is part of the DSD family. The European Society for Paediatric Endocrinology has a separate Turner working group, for example. It is a complex discussion with different perspectives, especially when patients carry a mosaic karyotype that includes a Y chromosome. There have also been nomenclature discussions trying to limit the category of “Turner syndrome” to patients with a female phenotype and the recent clinical guidelines for care published by an international group address only this group. However, many chromosomal variants exist, mainly in the form of 45,X/46,XY mosaics, with a spectrum of genital phenotypes. This creates potential complications by creating different categories of individuals with a 45,X/46,XY karyotype—some deemed as “Turner syndrome,” some not— while the underlying molecular features are the same. For the purpose of this chapter, we will consider Turner syndrome and variants as part of the DSD umbrella, as defined by the presence of 45,X cells, without specific phenotypic or gender requirements.
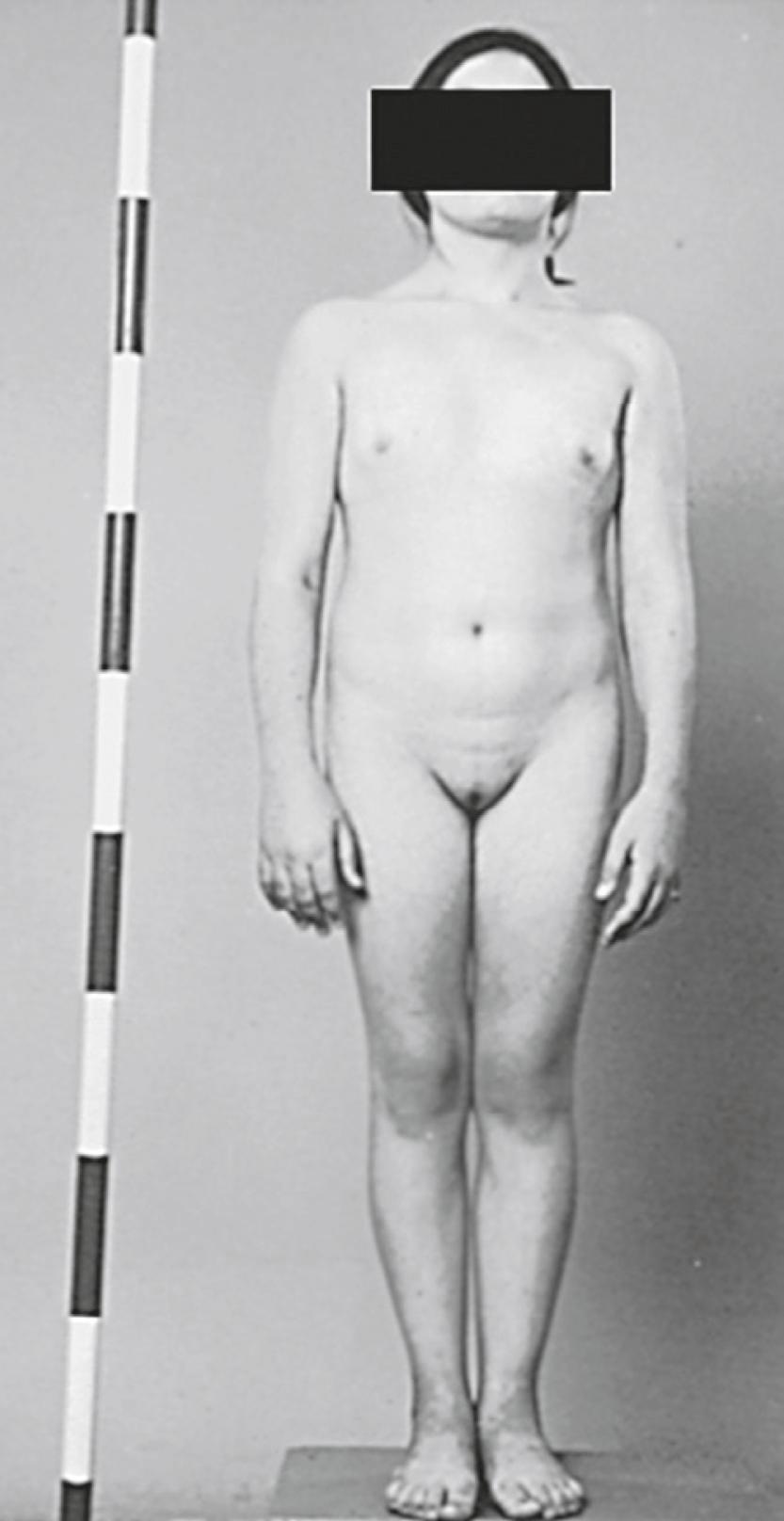
The majority of individuals with Turner syndrome carry a 45,X karyotype, and the remaining have a 46,XX karyotype with a deletion in part of one X chromosome or have various mosaics involving 45,X, 46,XX, and 46,XY cells or more complex combinations. In 100% of patients with 45,X and 80% of mosaic patients, the universal feature is short stature ( Fig. 17.6 ), with an average height of less than 148 cm. The genetic locus for short stature in both Turner syndrome and idiopathic short stature lies in the PAR1 pseudoautosomal region of the X chromosome. This region is deleted in individuals with short stature caused by the lack of one copy of the short stature homeobox gene (SHOX) .
The classic Turner 45,X karyotype is thought to be one of the most common human chromosomal abnormalities and is estimated to occur in 0.8% of all zygotes. However, less than 3% of these zygotes survive to term, and this karyotype is commonly found in spontaneous abortions. The incidence of 45,X karyotypes is approximately 1 in 2500 live newborn phenotypic females. ,
The gonads of Turner syndrome females do not develop normally, having reduced growth and follicle formation in utero . A normal number of eggs develop in girls with Turner syndrome, but, for unknown reasons, they disappear prematurely. The gonads appear as streaks of white tissue next to the Fallopian tubes and are called streak gonads, which on histology have primitive connective tissue stroma without primary follicles. Patients with Turner syndrome have fewer follicles and therefore less estrogen secretion from granulosa cells than typical females, resulting in delayed puberty.
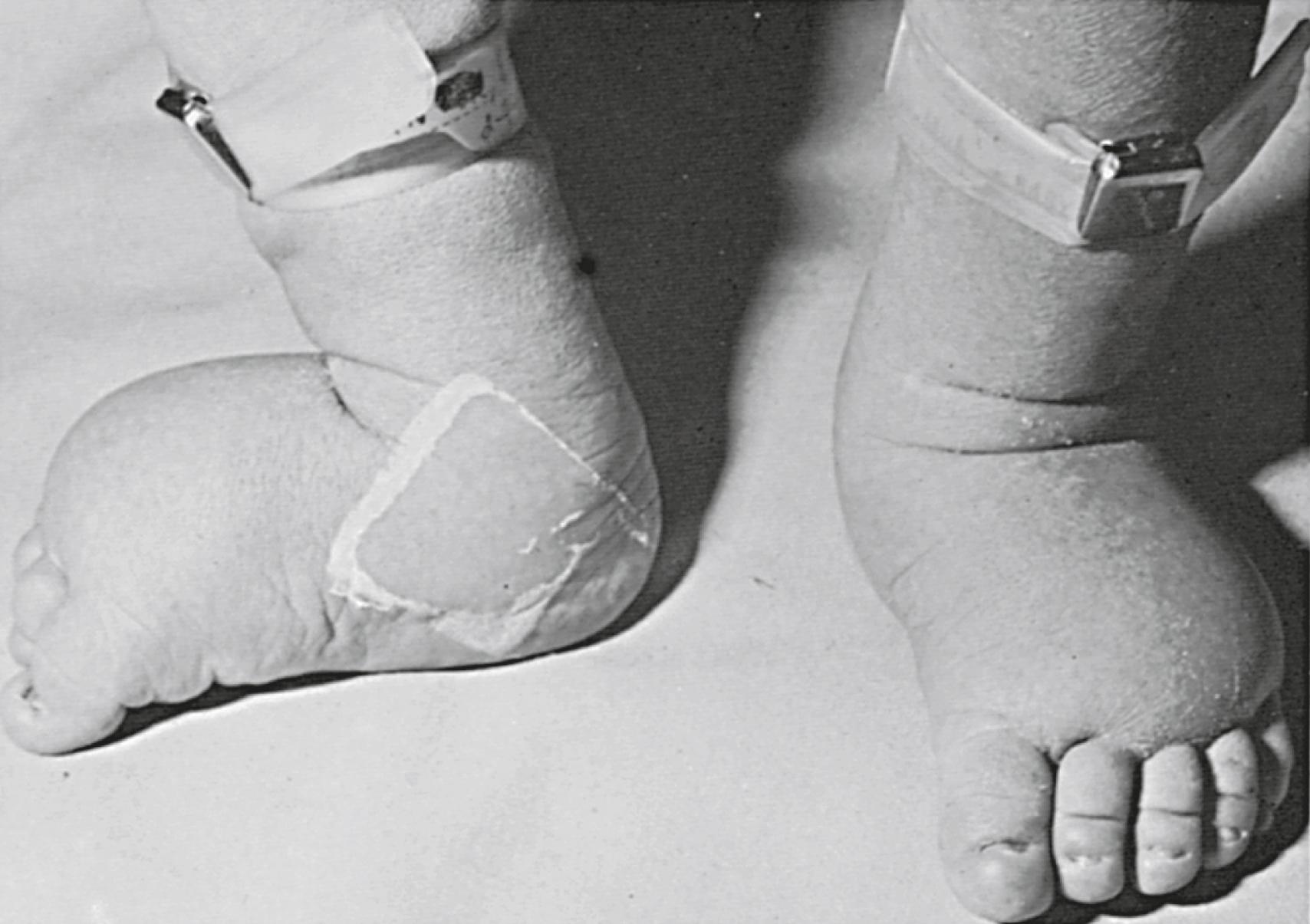
In the prenatal period, fetuses with a 45,X karyotype and its variants frequently have intrauterine growth restriction. Suspicion for Turner syndrome should be raised if the presence of large septate cystic hygromas, nuchal thickening, short femur, total body lymphangiectasia, or cardiac defects is detected by ultrasound. At birth, Turner syndrome babies often have low birth weights; 30% of babies will present with lymphedema of the upper and lower extremities ( Fig. 17.7 ), an extension of the lymphangiectasia in utero , which disappears in the first few months of life. Additionally, the presence of a webbed neck (pterygium colli) (see Fig. 17.6 ) and dysmorphic features including a high-arched palate, low-set prominent ears, low posterior hairline, epicanthal folds, micrognathia, hypoplastic nail beds, or hypoplastic fourth and fifth metacarpals at birth should raise the suspicion for 45,X DSD. Turner syndrome patients may have increased carrying angle of the arms (cubitus valgus), shield-like chest with wide-set nipples, hearing loss, and a higher frequency of cardiovascular disease such as coarctation of the aorta. Furthermore, the incidence of renal anomalies is between 30% and 50%, the most common finding being a horseshoe kidney, followed by abnormal vasculature.
During adolescence, the most common presentation is short stature, amenorrhea, and lack of secondary sexual characteristics. Patients may also have any of the other characteristics mentioned above. 45,X women have normal intelligence. Depending on the degree of GD, up to 30% of Turner patients undergo some degree of spontaneous puberty.
Diagnosis is made by karyotype, which will show in a 45,X, or mosaic variant of cells. If the karyotype appears normal but the presentation is suggestive of Turner syndrome, fluorescent in situ hybridization (FISH), or chromosomal microarray study should be done because there may be a cryptic deletion in the pseudoautosomal region in one of the X chromosomes. Typically patients with 45,X or 46,XX with a chromosomal abnormality in one X chromosome have more prominent phenotypic features than those who are a mosaic.
For most patients with 45,X DSD, the greatest concern is for short stature and secondary sexual development. The treatment of both of these should be carefully titrated. For cases diagnosed in childhood, the use of low-dose anabolic steroids coupled with growth hormone has been shown to increase final adult height with increased beneficial effects compared to those who started therapy late. Optimal duration and timing of the steroid treatment are still under investigation. At the onset of the age of puberty, high-dose estrogen and progesterone should be given to promote the development of secondary sex characteristics. Estrogens and progesterone should be taken throughout life to prevent complications, such as osteoporosis.
In young patients diagnosed with Turner syndrome, spontaneous puberty is a good indicator of the presence of functional ovarian tissue. Overall, it is estimated that 2% to 5% of patients with Turner syndrome have the potential for spontaneous pregnancy.
A frequent question from families concerns fertility. Despite progress in egg preservation techniques, which could provide hope for fertility in women with Turner syndrome, pregnancies (conceived with either autologous or donated oocytes) carry a high risk of life-threatening cardiovascular complications (aortic dissection, severe hypertension). A complete cardiac assessment is recommended prior to consideration of pregnancy, as well as close monitoring during pregnancy and in the postpartum period. Yet, a growing number of centers have developed programs of fertility preservation for Turner syndrome and novel approaches that are still considered experimental such as in vitro maturation of immature oocytes and in vitro activation (IVA) of immature follicles, are promising.
For optimal management of all conditions associated with Turner syndrome and variants, patients should be referred to multidisciplinary centers specializing in their care when available.
Klinefelter syndrome is typically associated with hyper gonadotropic hypogonadism and infertility. In utero , the XXY fetal testis has the normal complement of primordial germ cells. However, these germ cells degenerate during childhood, possibly due to a defect in Sertoli cells and germ cell communication during testes maturation. In theory, the etiology of the nondisjunction in Klinefelter can be from maternal meiosis I or II or paternal meiosis II, and thus each situation should contribute to 33% of cases. However, nearly 50% of Klinefelter patients show a paternal origin, with some studies showing increased frequency of diploid XY sperm with advanced paternal age.
Klinefelter syndrome is largely undiagnosed in the general population. In early life, Klinefelter may be diagnosed in boys with behavioral disorders, abnormally small testes, and long legs ( Fig. 17.8 ). The presence of only long lower extremities distinguishes Klinefelter syndrome from the other forms of eunuchoidism that result in equally long upper and lower extremities. In patients with Klinefelter syndrome, IQ typically falls in the normal range; however, it tends to be below that of siblings. , Most patients present in adolescence with small firm testes and hypogonadism with varying degrees of androgen deficiency. In later life, many males present at infertility centers with azoospermia.
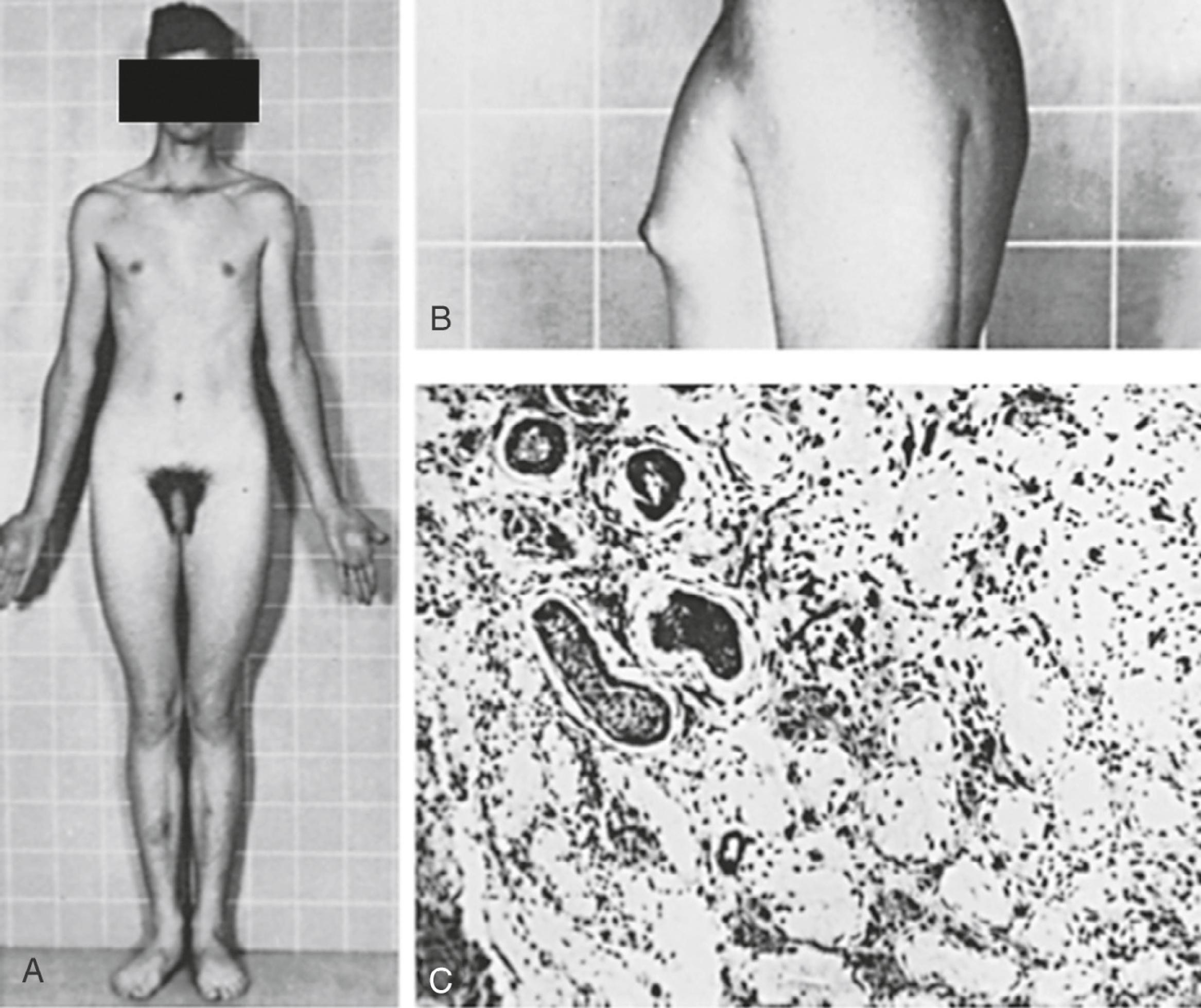
Diagnosis of Klinefelter syndrome is performed by lymphocyte karyotype or chromosomal microarray. Some mosaic cases will only be detected by karyotype of skin fibroblasts and occasionally of testicular biopsy specimens.
The European Society of Andrology recently published guidelines for the management of Klinefelter syndrome. Early detection allows for early intervention for cognitive and behavioral traits. Current studies are underway exploring the role of androgen replacement therapy in childhood, to help with cognitive and behavioral disabilities. Replacement of androgens also allows for the development of masculine secondary sex characteristics, improved self-esteem, and increased libido, strength, and bone mineral density. Typically, androgen replacement therapy is started if puberty does not start on time or is not maintained. However, recent randomized clinical trials tested whether low doses of androgens (Oxandrolone) given as early as 4 years of age modified the clinical outcome. Positive effects on measures of cardiometabolic health in prepubertal boys with KS were observed, albeit accompanied by lower HDL cholesterol and advanced bone age. Positive effects were shown on visual motor functions and psychosocial function (anxiety, depression, social problems) but not on cognitive function or on hyperactive or aggressive behaviors. Another trial starting at 6 weeks of age (Testosterone cypionate 25 mg intramuscularly monthly for three doses versus no treatment) showed changes in body composition by 5 months of age, with adiposity of untreated infants 15% greater than that of male controls.
With regard to fertility, testicular mosaicism is an important factor in determining spermatogenesis and the potential for fertility. The vast majority of 47,XXY men are azoospermic but, since the advent of surgical testicular sperm extraction (TESE) and intracytoplasmic sperm injection (ICSI) technology, have the potential to be fertile. The success rates of TESE are similar to those in other causes of nonobstructive azoospermia if it is performed between 16 and 35 years of age.
It should be noted that there is a higher rate of sex chromosomal hyperploidy and autosomal aneupoloidy in the sperm of Klinefelter patients. However, the risk of passing sex chromosome aneuploidy to the offspring remains unclear. Regardless, preimplantation genetic diagnosis (PGD) or fetal karyotype can identify chromosomal abnormalities in the progeny. Using testicular sperm extraction technology, sperm can be successfully extracted from the testes and injected into a donor oocyte, with a reported fertility success of up to 50%, a dramatic change from the near complete historical infertility of Klinefelter patients.
46, XY DSDs include gonadal dysgenesis (GD), abnormal biosynthesis of androgens, abnormal androgen action on their receptor.
46,XY GD results from abnormal testis development in utero. The absence of secreted hormones results in a spectrum of genital phenotypes from that of a typical female presenting in adolescence with primary amenorrhea to ambiguous external genitalia. Known causative genes include SRY , SOX9 , NR5A1 /SF1, MAP3K1, and many others. Patients need to be monitored for risk of gonadoblastoma.
46,XY disorders of androgen biosynthesis affect sex differentiation and include steroidogenic acute regulatory protein (StAR), HSD3B2, CYP17A1, P450 oxidoreductase (POR), HSD17B3, and steroid 5α-reductase (SRD5A2) deficiencies. Although all of these are often difficult to distinguish clinically, an early genetic diagnosis is critical because they have different natural histories requiring different management.
46,XY disorders of androgen action are caused by mutations in the androgen receptor (AR) resulting in complete or partial Androgen Insensitivity Syndrome (CAIS or PAIS).
XY GD is a result of abnormal testis development in utero . There are three types of GD, complete (or pure), partial, or mixed, all of which can be differentiated by the extent of normal testicular tissue within the gonad and karyotype of the individual. In complete GD, individuals have intraabdominal, bilateral, fibrous streaks that do not secrete AMH or testosterone. Phenotypically, pure XY GD individuals are unambiguously phenotypic females (previously known as Swyer syndrome) but usually possess hypoplastic Müllerian structures internally.
Given the primary importance of SRY in human testis determination, it is surprising that mutations in SRY only account for approximately 15% of all 46,XY DSDs with complete GD. , This suggests the existence of many other genes involved in primary human sex determination. SRY is a transcription factor, but its mechanism of action remains incompletely understood. For instance, it is still unclear whether it acts as an activator or repressor, and its only confirmed target is SOX9, another transcription factor from the Sox (Sry-box) family of genes. This interaction is mediated by the testis-specific Sox9 enhancer, TESCO, and by TESCO-independent mechanisms. To date, there are more than 50 verified mutations within the SRY gene. Mutations that result in streak gonads primarily occur within the HMG box and cause reduced DNA binding, , alterations in DNA bending, or prevent the nuclear import of the SRY protein. Larger cytogenetic deletions of Yp that include SRY have also been implicated in XY GD. , ,
Steroidogenic factor 1 (SF1, encoded by the gene NR5A1 ) is an orphan nuclear receptor necessary for adrenal and gonadal development. The genital phenotypic spectrum associated with variants in SF1/ NR5A1 is large, including complete or partial GD and azoospermia in 46,XY individuals and testicular or ovotesticular DSD and POI in 46,XX individuals (see Table 17.3 ), suggesting that modifier genes may be responsible for the variability. At least 15% of isolated XY GD can be attributed to SF1 haploinsufficiency. NR5A1 variants are also a frequent cause of 46,XY partial GD (see below), and rare NR5A1 mutations with XY GD and adrenal hypoplasia congenita have been reported, reflecting the role of SF1/ NR5A1 also in adrenal development. , Together, mutations or deletions of SRY and NR5A1 account for approximately a third of XY GD, indicating that other mechanisms critical to the testes determination cascade still remain to be discovered.
Interactions between NR5A1 /SF1 and the transcription factors GATA-binding protein 4 (GATA4) and friend of GATA 2 (FOG2) appear to be necessary for SRY expression in developing testes , and, in mice, the Gata4 protein has been shown to interact with Nr5a1 /Sf-1, Fog2, and/or Wt1 to regulate the downstream cascade of genes critical for urogenital development, such as Sry , Sox9 , Cyp19a1 , Hsd3b2 or Star. Very rare instances of chromosomal rearrangements leading to disruption of the FOG2/ ZPMF2 gene , presented with GD in XY individuals. Exome sequencing of patient cohorts led to reports of large numbers of variants in these two genes in XY DSD cohorts. However, reanalysis and more accurate measurement of the level of activity of the variant proteins have led to the reclassification of many of these variants as likely benign and the role of GATA4 in human DSD has been difficult to pinpoint. So far, only variants in a very specific domain of GATA-4 (N-terminal Zinc Finger domain) have been shown to be associated with DSD, in a condition termed Testicular Anomalies with or without Congenital Heart Disease (TACHD, OMIM # 615542 ).
Mutations in genes involved in testis sex determination (SOX9, WT-1, DHH, DMRT1) and duplications of putative “anti-testis” genes (WNT-4, DAX1 /NR0B1) have also been shown to be responsible for a minority of all XY GD. , , Research exome sequencing continues to identify other genes, such as that MAP3K1 , or DHX37 , as causative genes, which need to be included in sequencing panels for diagnosis.
Like many developmental disorders, there is a wide range of phenotypic variability in patient presentation. When testes dysgenesis does not involve the entirety of both gonads and the external genital phenotype is ambiguous rather than typical female, the condition is called partial GD . The internal genitalia display varying degrees of Wolffian and Müllerian development, which correspond with the proportion of the gonads that is dysgenetic or “streak.” The medical literature is often confusing in regard to the difference between “partial” and “mixed” GD, which are often used interchangeably. Partial XY GD refers to intermediate stages of dysgenetic testes, between streak gonads and normal testes, and usually has a 46,XY, nonmosaic karyotype. Mixed XY GD typically refers to a situation in which one gonad is a streak gonad while the contralateral gonad is partially dysgenic or a normal testis. Mosaicism for 45,X/46,XY is the most frequent cause of mixed GD, although a minority have a 46,Xi (Yq) karyotype. The evidence of the variable phenotypic spectrum of individuals with 45,X/46,XY mosaicism was reported in a series of 10 45,X/46,XY patients in which four individuals were undervirilized males with bilateral testes, three were diagnosed with mixed GD and genital ambiguity, and three were diagnosed with Turner syndrome. Although not proven, there is preliminary evidence that the percentage of normal testicular tissue and phenotypic “maleness” are correlated with an increase in the proportion of gonadal Y chromosome. ,
Familial cases of complete and partial 46,XY GD , , , have been reported. In some of these cases, SRY mutations present with phenotypic variability in which the father and male relatives are phenotypically normal and fertile, while their XY offspring have GD. , , From these cases, it is clear that autosomal genetic modifiers influence the sex determination cascade and ultimate phenotypic appearance of the individual. There are also cases of fathers who are mosaic for mutant and normal SRY transmitting the mutation to their XY female daughters. , Presumably, the normal father’s mosaicism reflects a postzygotic mutation event.
Besides SRY , variants in NR5A1 may explain ∼20% of PGD, with DHX37 and DHH a few each but the genetic cause of about half of 46,XY GD remains unknown.
Complete XY GD presents as a phenotypic female with normal or tall stature, bilateral streak gonads, delayed puberty, amenorrhea, small or normal Müllerian structures, and without signs of Turner syndrome. If patients are not diagnosed at birth (e.g., when in utero karyotype does not match the phenotypic sex at birth), most patients will be diagnosed during adolescence due to pubertal delay and primary amenorrhea. Patients can rarely present with an abdominal or pelvic mass, which is often a gonadoblastoma. Occasionally, XY GD can be part of a constellation of other symptoms, outlined in Table 17.4 .
Individuals with partial or mixed 46,XY GD typically present at birth with varying degrees of masculinization of external and internal genitalia. Depending on the percent of testicular tissue, dysgenetic testes can be found anywhere along the line of testes descent, from the abdomen to the scrotum. However, the streak gonad in mixed 46,XY GD is always abdominally located. Patients with partial 46,XY GD usually have female external genitalia with some degree of virilization, such as clitoromegaly or a bifid scrotum ( Fig. 17.9 ). Uterus and Fallopian tubes are usually well formed but occasionally may be hypoplastic. In mixed GD, the development of Wolffian and Müllerian structures and the virilization of external genitalia correlate with the degree of development of the ipsilateral testis resulting in asymmetric virilization of the external or internal genitalia and unilateral cryptorchidism. Pelvic ultrasound or MRI can often detect the presence or absence of male or female internal genitalia and, in the case of mixed XY GD, asymmetry in the development of the Müllerian and Wolffian structures. Although rare, there are reported cases of patients presenting with premature adrenarche in an otherwise unambiguous female, due to a testosterone-producing gonadal tumor.
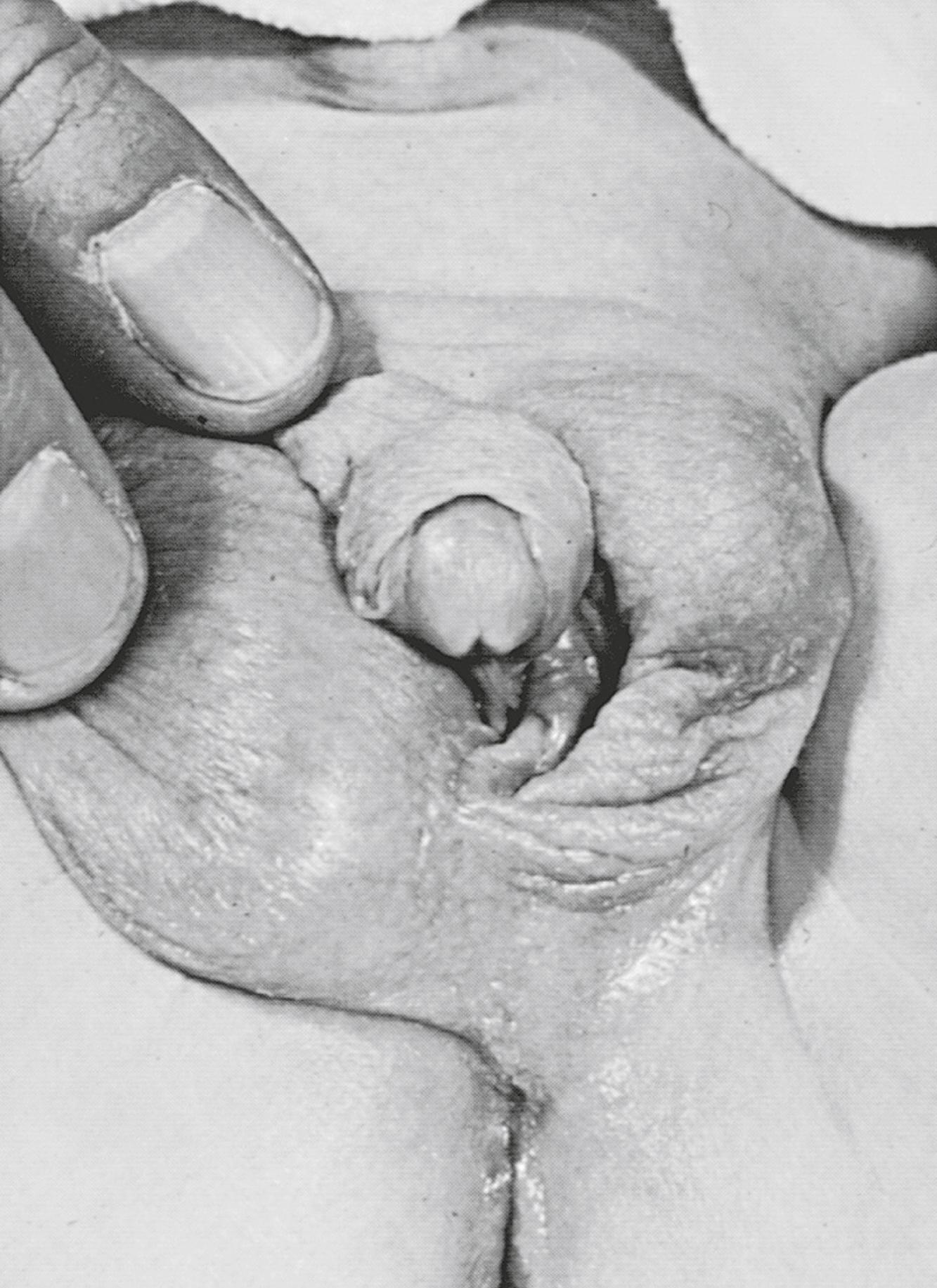
Isolated 46,XY pure GD is considered in an adolescent with primary amenorrhea and sexual immaturity with a full female external phenotype, while partial or mixed GD should be considered more likely in the differential diagnosis of a 46,XY patient with ambiguous genitalia. The biochemical changes in complete, partial, and mixed GD are outlined in Table 17.5 .
The major criteria for the diagnosis of pure, partial, or mixed GD are the appearance and histology of both gonads. Therefore, the ultimate diagnosis of any of the forms of GD, particularly the distinction between mixed and partial GD, requires a biopsy of both gonads. Because the risk of gonadoblastoma in these patients is elevated, a precise diagnosis of the type of GD is usually determined after prophylactic or therapeutic gonadectomy. In pure 46,XY GD both gonads are streak gonads. Partial GD is defined by bilateral dysgenetic gonads, whereas mixed GD typically has one streak gonad. Karyotype of peripheral leukocytes shows 46,XY in pure and partial GD, and mosaic 45,X/46,XY is frequent in mixed GD. However, if there is a streak gonad on one side and a normal testis contralaterally, but the peripheral karyotype is 46,XY, cryptic mosaicism can often be revealed within the gonad.
Once a presumptive diagnosis is made in 46,XY patients, FISH for SRY or for Yp can be performed. Only a minority of cases of XY GD can be explained by complete or partial SRY deletion or mutation, or NR5A1 /SF1 mutation. Sequencing of the SRY or NR5A1 /SF1 open reading frames for mutations is only positive in up to 30% of 46,XY pure GD. Certain isolated and syndromic forms of XY GD can be diagnosed molecularly with cytogenetics, chromosomal microarrays, or sequencing of known genes (see Table 17.5 ). For example, 46,XY DSD in conjunction with congenital heart disease may increase the suspicion of a GATA4 mutation. However, next-generation sequencing is now available in the clinical realm to replace this sequential guesswork and should be prioritized as a diagnostic tool to identify etiology.
In addition, whole-genome chromosomal microarray studies have identified many rare copy number variants associated with patients with 46,XY GD and 46,XX testicular and ovotesticular DSDs, including deletions and duplications in and around SOX3 , GATA4 , WWOX , and DMRT1 . , Deletions or duplications in the upstream promoter region of SOX9 are associated with isolated and familial cases of 46,XY GD and 46,XX testicular DSDs (see Table 17.3 ). ,
If a mutation or translocation involving SRY is found, the father should be tested for a possible familial mutation because SRY mutations can result in a full spectrum of phenotypes, from 46,XY fertile males to ambiguous individuals to females with partial or complete GD. Due to potential autosomal modifiers, it is very difficult to predict the risk of recurrence of a GD phenotype in XY offspring.
The major concern in the treatment of patients with XY GD is the risk of gonadoblastoma, a mixed germ-cell, sex-cord tumor. The risk of gonadoblastoma formation in XY GD increases with age and has been estimated to be as high as 30% by 30 years of age. , , Due to the high risk of gonadoblastoma formation, patients typically undergo prophylactic or therapeutic gonadectomy in the first decade of life. Patients are followed with regular ultrasounds every 6 months starting at age 2 until gonadectomy can be performed.
In individuals with XY complete GD, genitals and gender identity are unambiguously female. There are no reported cases of gender dysphoria. In patients with partial or mixed GD, the degree of genital masculinization can be used for initial gender assignment. However, issues of gender identity do arise in partial and mixed GD and are outlined further in the management section.
To complete secondary sex development, sex steroid replacement should be initiated at puberty. Sex steroid replacement is essential not only for the development of secondary sex characteristics but also for growth spurts and normal accrual of bone mineral density. Height, weight, and bone density should be monitored regularly. Furthermore, a dual-energy X-ray absorptiometry (DEXA) scan for bone density should be performed prior to induction of puberty with exogenous hormone replacement, and yearly thereafter for the first 2 years. As long as the first three DEXA scans are reported as normal, they can be done every 2 to 3 years.
Because a normal uterus is often present, hysterectomies are not common to preserve childbearing potential with in vitro fertilization methods. Despite this, for unclear reasons, most patients do not carry successful pregnancies. Mixed XY GD individuals with more masculinization of the external genitalia are often raised as males and consequently require lifetime testosterone therapy, through intramuscular injections or transdermal patches or gel.
Testicular regression syndrome (TRS), also known as vanishing testes , results from an insult after initial testes determination, most likely vascular thrombosis or testicular torsion. Internal and external genitalia are variably developed, which presumably depends on when the loss of fetal testes occurred. , , , Rarely, patients can present in adulthood as females with primary amenorrhea. , Typically TRS is characterized by primitive epididymis and the spermatic cord in the presence of a fibrous nodule with hemosiderin deposition rather than the streak or dysgenetic gonads seen in 46,XY GD. , , The presence of spermatic cord structures, the absence of Müllerian derivatives, and the typical male external phenotype suggest viable testes existed early in development, and imply a late fetal or early neonatal regression. Diagnostic criteria are outlined in Table 17.5 .
The incidence of TRS has been estimated at 5% of males presenting with cryptorchidism and as high as 12% of cryptorchid patients older than 1 year. , Correct diagnosis of TRS versus 46,XY GD is essential because of the significant malignant potential of abdominal or dysgenetic testes. Mutational analysis of SF1 /NR5A1 as a cause of TRS did not find any association between mutations in NR5A1 and TRS and the etiology of TRS remains mostly unknown. However, recent reports have identified variants in the gene DHX37 in cases clinically diagnosed as both GD and TRS, underscoring the difficulty of the differential diagnosis. , Although the condition is thought to be rather frequent, optimal management remains unclear. No reports of testicular germ cell tumors in TRS have been published, suggesting that excision of the remnant gonad may be unnecessary.
46,XY DSDs, with phenotypes ranging from typical females to undervirilized males, can also be a result of defects in steroidogenic enzymes ( Fig. 17.10 ) (see Chapter 4 ). The biosynthesis of testosterone is essential for the development of secondary sex characteristics in XY individuals, including differentiation of Wolffian structures, inguinoscrotal testes descent, and masculinization of external genitalia after conversion to DHT. Additionally, mutations in Sertoli cell products, such as AMH or its receptor, can result in incomplete regression of female internal genitalia. Mutations in the synthesis and action pathways of testosterone, or upstream regulators of testosterone production, can all cause incomplete virilization or Müllerian regression in the XY male. However, in these individuals, the testes are of normal size.

Mutations in genes encoding proteins involved in the initial enzymatic regions of steroidogenesis, such as steroidogenic acute regulatory protein (StAR) and cytochrome P450 side chain cleavage (P450scc, also known as CYP11A1) , , results in global silencing of adrenal and gonadal steroidogenesis. The accumulation of cholesterol in the adrenals and gonads ultimately results in primary adrenal and gonadal failure. StAR protein transports cholesterol across the inner and outer mitochondrial membranes of steroidogenic cells. P450scc catalyzes the initial reaction in all steroidogenic tissues, the conversion of cholesterol to pregnenolone (see Fig. 17.10 ). , On adrenal histology, patients with mutations in StAR protein or P450scc have lipid vacuoles. However, patients with STAR mutations have adrenal hyperplasia, whereas patients with P450SCC mutations typically do not exhibit adrenal enlargement.
Congenital lipoid adrenal hyperplasia (lipoid CAH) usually presents with a salt-wasting adrenal crisis at birth, which is usually fatal if not immediately diagnosed and treated; however, there are reports of delayed presentation. All XY individuals present with a female phenotype or, in cases of partial defect, some degree of genital ambiguity. XX individuals only exhibit the features of adrenal insufficiency at birth.
46,XY patients have testes, no Müllerian structures (resulting in a blind vaginal pouch) due to the presence of AMH, and partial or absent Wolffian derivatives due to a lack of testosterone biosynthesis. The spectrum of external genital phenotypes ranges from female to ambiguous external genitalia. Testes can be in the abdomen, inguinal canal, or labia. Additionally, intrauterine glucocorticoid deficiency results in elevated adrenocorticotropic hormone (ACTH) levels, which manifests clinically as generalized hyperpigmentation at birth.
Partial deficiency of StAR protein may, in 46,XX individuals, result in spontaneous puberty, menarche, and anovulatory menses because their ovaries are able to produce estrogen through StAR-independent pathways. , , However, at puberty, XX females develop multiple cysts in their ovaries, possibly from anovulation. Because the ovaries do not produce steroids until puberty, they are spared from the cholesterol-induced damage, which occurs in the adrenals from birth.
Definitive diagnosis of congenital lipoid adrenal hyperplasia is done by sequencing, with identification of variants in StAR or CYP11A1 . Characteristic biochemical abnormalities are outlined in Table 17.5 .
Patients should be given physiologic replacement doses of glucocorticoid and mineralocorticoids. At the onset of puberty, patients should be given sex hormones in concordance with phenotypic sex.
This rare variant of CAH can have a wide spectrum of phenotypes, from classical presentation (salt-wasting, with a range of female external genitalia to ambiguous genitalia in genetic XY individuals) to nonclassical (no salt-wasting and later presentation). Rare defects in adrenal and gonadal 3β-hydroxysteroid dehydrogenase (HSD3B2) affect the synthesis of three major adrenal steroid hormones: cortisol, aldosterone, and testosterone synthesis. The resulting phenotype is adrenal insufficiency in XX and XY but genital ambiguity only in XY.
Biosynthesis of adrenal and sex hormones requires cytochrome P450 17α-hydroxylase/17,20-lyase (P450C17) encoded by the CYP17A1 gene. Here, a single protein, P450C17, catalyzes two steps in the steroidogenic pathway (see Fig. 17.10 ), both of which are essential for sex steroid and glucocorticoid synthesis. Mutations in this gene ultimately result in decreased cortisol synthesis with shunting of steroidogenesis toward a mineralocorticoid precursor that has mineralocorticoid activity. Isolated cases of 17,20-lyase deficiency are caused by mutations in Cytochrome b5 (CYB5A), a gene that promotes the specific allosteric interaction between CYP17A1 and POR. ,
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here