Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
As the role of fine-needle aspiration biopsy (FNAB) in the evaluation of focal liver lesions has evolved, it has presented new challenges. , Advances in dynamic imaging modalities have obviated the need for tissue confirmation in clinically classic cases of hepatocellular carcinoma (HCC). Ultrasound surveillance and serum α-fetoprotein (AFP) assessment of high-risk patients have improved the detection of small nodules (≤2 cm in diameter). Accurate cytohistological characterization of small, well-differentiated hepatocellular nodules on limited tissue samples is extremely challenging, but it has important therapeutic implications.
Much progress has been made in the molecular characterization of tumors by genomic, microRNA, and proteomic methods. Promising new tools for tumor diagnosis are available for predicting the prognosis and progression of disease and for personalized, molecularly targeted anticancer therapies and chemopreventive strategies. Because FNAB is the least invasive tool for tissue procurement, it is predicted that it will become a point of care in the management algorithm of HCC for diagnostic, therapeutic, and prognostication purposes.
Percutaneous (transabdominal) FNAB performed with ultrasound or CT guidance is a safe, efficacious, and cost-effective outpatient procedure for the diagnosis of focal liver lesions. Aspiration can also be performed during laparotomy or laparoscopy under palpation or direct vision. Endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) is the latest diagnostic and staging tool and is used primarily for left hepatic lobe lesions and hilar or perihilar masses in which the needle traverses the wall of the gastrointestinal tract. Factors influencing the choice of guidance system include the size and location of the lesion, operator expertise and preference, and availability of imaging technologies.
Ultrasonography provides rapid localization, flexible patient positioning, and imaging of the lesion without radiation. It is typically used for initial guidance, particularly when there are multiple lesions or large, relatively superficial lesions. CT allows optimal resolution of small lesions or lesions that are not visible with ultrasound, accurate localization of the needle tip immediately before sampling, improved definition of tissue components and vascularity, and more precise demonstration of the anatomic relationships of a given lesion.
EUS-FNA is a safe, accurate, and versatile technique, but it is highly operator dependent. It allows concurrent sampling of pancreas and liver lesions, which can confirm primary and metastatic lesions in one diagnostic encounter. The technique is useful for small and deep-seated left lobe lesions that are below CT or magnetic resonance imaging (MRI) resolution or lesions that are not easily accessible by percutaneous FNAB. It improves staging of liver metastases, is good for early detection of multifocal HCC in cirrhosis, and can assess the accurate number of lesions (e.g., intrahepatic staging of HCC) for transplantation eligibility. ,
Percutaneous FNAB techniques include individual puncture , coaxial biopsy, and tandem needle biopsy techniques. Multiple aspirations (as many as four passes) usually can be performed with minimal morbidity. The needle size is between 20 and 22 gauge. Aspiration needles with a guillotine mechanism enable microbiopsy cores to be procured at the same sitting. Concomitant core-needle (18-gauge) biopsies may also be performed.
FNAB is the diagnostic procedure of choice for focal liver lesions, especially to confirm a suspected malignancy ( Box 46.1 ). It is particularly advantageous for advanced malignancies and patients who are poor surgical candidates. Another indication is drainage of a cyst or abscess for culture and therapeutic ablation. Early affirmative diagnosis leads to cost savings in terms of further investigational tests and hospitalization.
Evaluating a mass or cystic lesion
Draining a cyst or abscess
Uncorrectable bleeding diathesis
Lack of safe access route
Uncooperative patient
Intestinal obstruction (for endoscopic ultrasound-guided fine-needle aspiration biopsy)
Bleeding
Needle tract seeding
Tumor recurrence and post-transplantation tumor recurrence
Contraindications for percutaneous FNAB include an uncorrectable bleeding diathesis, lack of a safe access route (i.e., biopsy through a vascular structure), and an uncooperative patient for whom awkward positioning or maintenance of strict breath control is necessary to ensure proper needle placement. For EUS-FNA, gastrointestinal obstruction is an absolute contraindication because of the risk of perforation.
Complications from FNAB are uncommon. There may be bleeding, which is mostly associated with cirrhosis and coagulopathy but may relate to the size of needle used, particularly in vascular lesions and in large superficial tumors not covered by normal parenchyma (see Box 46.1 ). Needle tract seeding is rare, with an incidence of 0.003% to 0.009% for malignancy in general and 0.003% to 5% for HCC; if a small (22-gauge), noncutting needle is used, the rate for HCC is approximately 0.11%. , A higher incidence of HCC recurrence in the transplanted liver has been reported in transplantation cases with preoperative FNAB. Death, usually resulting from bleeding, is rare, with a reported mortality rate of 0.018%.
There is much debate regarding the use of preoperative FNAB for the diagnosis of HCC. Several reasons are cited for opposing FNAB :
Sufficiently sensitive advances in dynamic imaging modalities for establishing an HCC diagnosis , , , ,
Risk of needle tract seeding , ,
Risk of intraperitoneal bleeding
Risk of intraprocedural hematogenous dissemination, which results in a higher incidence of tumor and posttransplantation recurrence ,
Adoption of a wait-and-watch policy for hepatocellular nodules that are less than 1 cm in diameter ,
Indeterminate cytohistological reports rendered for well-differentiated hepatocellular nodules ,
Several reasons are cited for favoring FNAB , :
Serum AFP has a low sensitivity (<50%). ,
Use of the coaxial biopsy technique to reduce the risk of seeding
Decreased costs of long-term imaging surveillance
Avoids futile transplantation in false-positive imaging cases
Allays patient anxiety after a liver nodule has been detected on imaging
Confirms HCC, which increases eligibility for liver transplantation
Immediate institution of anti-HCC therapy when the lesion is less than 2 cm in diameter
Excludes other types of primary liver carcinomas
According to the European Association for the Study of the Liver (EASL) 2000 Conference and the American Association for the Study of Liver Disease (AASLD) guidelines, nodules more than 2 cm in diameter in cirrhotic livers are diagnosed as HCC if they show an intense arterial profile with contrast washout in delayed venous phase on one dynamic imaging modality. , Nodules between 1 and 2 cm in cirrhotic livers require concurrence of two coincidental imaging modalities; otherwise, biopsy is recommended. For nodules less than 1 cm in diameter, the EASL guidelines recommend a wait-and-watch policy with surveillance every 3 months. Approximately 68% of nodules less than 1 cm in diameter in cirrhotic livers are HCCs. In skilled hands and with an expert reader, ultrasound-guided FNAB of hepatic nodules less than 1 cm in diameter can yield correct a diagnosis in 90% of cases. The conundrum is to balance the risk of unnecessary surgery (2.5%) against the risk of seeding. , , ,
The types of small tissue samples from liver FNAB include direct smears, needle rinses, core imprints, cell blocks, microbiopsies, and core-needle biopsies. Smears may be air-dried and stained with Diff-Quik or May-Grünwald-Giemsa stain or fixed in 95% alcohol and stained using the Papanicolaou method. Rapid touch preparations (imprints) can be performed to assess the adequacy of tissue cores. If well-fixed, adequately smeared slides are difficult to obtain, the aspirate can be expressed into a preservative and submitted as a liquid-based specimen for processing by the ThinPrep or SurePath method.
Cell blocks are made from needle rinses and tissue fragments. , Particulate material should be quickly retrieved from the glass slides before staining for subsequent formalin-fixed, paraffin-embedded cell block preparation. Histological sections allow for architectural appraisal, special stains, and immunohistochemistry. Immunocytochemistry may be necessary if only smears are available.
The prerequisites for optimal results with FNAB are listed in Box 46.2 . The sensitivity of FNAB varies according to the method used (i.e., blind vs. guided aspiration, number of passes, and operator skill), characteristics of the lesion (size, location, consistency, and heterogeneity), and standard of cytopathology service (e.g., quality of smears, combined cytohistological analysis, ancillary testing, reader expertise). On-site cytology service may provide rapidly stained smears for evaluation of sample adequacy, retrieval of particulate material for cell block preparation, and triage of specimens for culture, flow cytometry, electron microscopy, and other ancillary tests, including molecular studies. , , Imprint smears of core-needle biopsies should be thoroughly screened on-site and the corresponding material separately labeled per pass for subsequent correlation. In the absence of an on-site service, it is imperative that the aspirator learns how to make proper smears and that a tissue management protocol is clearly understood by the patient care team.
Dedicated and experienced aspirator and reader team
Image-guided representative sampling for heterogeneous focal liver lesions
On-site cytology service to assess specimen adequacy, retrieve material for cell blocks, and triage specimen for microbiological studies, flow cytometry, electron microscopy, molecular analysis, and other ancillary tests
Combined cytohistological approach
Judicious use of immunohistochemistry
Clinicopathological correlation
The sensitivity and specificity of percutaneous FNAB for detection of liver malignancy are about 90% (range, 67% to 100%) and 100%, respectively. , , The positive and negative predictive values and overall accuracy of FNAB diagnosis for liver malignancy were reported in one large study to be 100%, 59.1%, and 92.4%, respectively. EUS-FNA also has a high sensitivity (82% to 94%) and specificity (90% to 100%) for malignancy. , , , Similar to percutaneous FNAB, EUS-FNA leads to a better diagnostic yield with metastases compared with well-differentiated hepatocellular tumor nodules. False positives are rare. False-negative diagnoses are most often the result of a sampling error because of inexact needle localization related to small masses, large areas of necrosis, fibrosis, or a prominent inflammatory rim. The supplemental nature of FNAB and concomitant core-needle biopsy improves overall accuracy, especially for benign neoplasms and for heterogeneous carcinomas and poorly differentiated neoplasms that require ancillary studies. ,
Liquid-based cytology of liver aspirates shows good correlation with conventional smears and reduces false-negative diagnoses. Advantages include more representative sampling and better cell preservation. Disadvantages include no on-site assessment of adequacy or triage of specimens, no air-dried smears for Giemsa preparations, removal of background elements, high cost, and the need for familiarity with cytological artifacts and recognition of diagnostic pitfalls to avoid misinterpretation. In our experience (unpublished data) with the ThinPrep method, the cytological features of HCC appear to be rather nondifferentiated. Cytoarchitectural features, such as arborizing trabecular structures, pseudoacinar rosettes, and peripheral or transgressing endothelium, are less evident or completely lost. Tumor aggregates tend to be tighter, three-dimensional, and smaller. Cells are usually smaller with nondescript shapes and denser cytoplasm, and they often have frayed borders.
Focal liver lesions range from cystic and inflammatory or infectious entities to benign or malignant primary and metastatic neoplasms. HCC is well known for its heterogeneity. Intrahepatic cholangiocarcinoma (CC) frequently shows nondescript features of adenocarcinoma. The phenotypic landscape is further muddied by the recently redefined combined hepatocellular-cholangiocarcinoma (cHCC-CCA), intermediate cell carcinoma, and cholangiolocarcinoma (CLC). , The liver is a common depository for metastases. Given these permutations, primary liver carcinomas can mimic many tumors and vice versa. , The diagnostic issues are listed in Box 46.3 .
Differentiating well-differentiated hepatocellular nodules from reactive hepatocytes
Distinguishing various types of benign, well-differentiated hepatocellular nodules
Differentiating early, well-differentiated hepatocellular carcinoma from benign hepatocellular nodules
Recognizing hepatocellular carcinoma and its variants from their mimics
Recognizing the components of combined hepatocellular-cholangiocarcinoma as part of a combination (not collision) tumor
Differentiating poorly differentiated hepatocellular carcinoma from intrahepatic cholangiocarcinoma
Differentiating intrahepatic cholangiocarcinoma from metastatic adenocarcinoma
Differentiating poorly differentiated primary liver carcinomas from metastases
Determining the histogenesis of nonhepatocellular tumors
Determining the primary site of origin of malignant nonhepatocellular tumors
Differentiating benign and malignant cystic lesions
Recognizing inflammatory or infectious lesions that may mimic tumors
A stepwise algorithmic approach to evaluating liver aspirates includes full clinicopathological correlations , , , ( Box 46.4 ):
Evaluate clinical information. A patient may be seen with one or more focal liver lesions in the following scenarios: routine medical checkup, screening or surveillance of chronic liver disease, , follow-up of a known cancer case, investigation of a symptomatic patient, or investigation of a pediatric lesion. Relevant clinical findings, liver function profile, serology, and tumor markers should be obtained. , ,
Evaluate radiological information. Focal liver lesions may be cystic or solid and single or multiple. The presence of preexisting or chronic hepatobiliary disease and associated radiological findings should be determined. Advances in dynamic contrast-enhanced imaging modalities have increased the sensitivity and specificity for detection of classic HCC. , , The radiological recognition of combination tumors requires a high index of suspicion, especially when there is discordance between tumor markers and imaging appearances. Siderotic nodules are readily detected on T2- or T2∗-weighted MRI. ,
Evaluate cytohistological findings. Smears are scanned at low power to establish the pattern and architecture before close inspection of cellular details as listed in Box 46.5 . The operator should ascertain whether the lesion is (1) cystic (true cyst or pseudocyst) and benign or malignant or (2) solid (hepatocellular or nonhepatocellular) and benign or malignant. The diagnostic algorithms for evaluation of cystic and solid lesions of the liver are provided in Boxes 46.6 and 46.7 , respectively.
Cellularity
Architectural features
Cellular composition
Background
Size of cells
Shape of cells
Spatial relationship (e.g., degree of cohesion, crowding, polarity)
Cytoplasm, nucleus, nucleus-to-cytoplasm ratio
Inclusions, intracytoplasmic and intranuclear
Pigments
Vasculature
Background
Cystic lesion with epithelial lining
Cuboidal or low columnar epithelium: solitary bile duct cyst, fibropolycystic disease, and obstructive dilation of bile duct
Ciliated epithelium: ciliated foregut cyst
Biliary, mucinous, or oncocytic (with or without papillae)
With ovarian-like stroma
Mucinous cystic neoplasm with low-, intermediate-, or high-grade intraepithelial neoplasia
Mucinous cystic neoplasm with associated invasive carcinoma
Without ovarian-like stroma
Biliary intraductal papillary neoplasm with low-, intermediate-, or high-grade intraepithelial neoplasia
Biliary intraductal papillary neoplasm with an associated invasive carcinoma
Intrahepatic cholangiocarcinoma associated with cystic change, arising from malignant transformation of preexisting cystic disease, or associated with cystically dilated bile ducts
Metastases
Cystic lesion without epithelial lining
Neoplastic (including tumor-like lesions)
Cavernous hemangioma
Mesenchymal hamartoma
Cystic degeneration in benign or malignant tumors
Undifferentiated embryonal sarcoma
Nonneoplastic
Laminated wall: hydatid cyst
Inflammation and necrosis: pyogenic, fungal, or amebic abscess, granulomas, necrotizing eosinophilic granuloma, hydatid cyst
Hemorrhagic cyst: cystic hematoma
Focal fatty change
Large regenerative nodule
Dysplastic nodule (low or high grade)
Siderotic nodule (regenerative, dysplastic)
Focal nodular hyperplasia
Hepatocellular adenoma
Hepatocellular carcinoma, variants and special types
Hepatoblastoma
Combined hepatocellular-cholangiocarcinoma
Glandular pattern: bile duct hamartoma, bile duct adenoma (peribiliary gland hamartoma), intrahepatic cholangiocarcinoma, combined hepatocellular-cholangiocarcinoma, cholangiolocarcinoma, metastases
Squamous, including adenosquamous, pattern: Intrahepatic cholangiocarcinoma, metastases
Mucinous pattern: mucinous cystic neoplasm, intrahepatic cholangiocarcinoma, metastases
Papillary pattern: intraductal papillary neoplasm, intrahepatic cholangiocarcinoma, metastases
Clear cell pattern: angiomyolipoma, metastases (renal cell carcinoma, adrenocortical carcinoma)
Oncocytic cell pattern: angiomyolipoma, metastases (renal cell carcinoma, adrenocortical carcinoma), melanoma
Small cell pattern: neuroendocrine tumor, lymphoma, metastases (small cell carcinoma, lobular carcinoma of breast, melanoma), inflammatory pseudotumor
Large cell pattern: neuroendocrine carcinoma, metastases (undifferentiated carcinoma)
Spindle cell pattern: cavernous hemangioma, infantile hemangioma, solitary fibrous tumor, inflammatory pseudotumor, angiomyolipoma, gastrointestinal stromal tumor, sarcomatoid carcinoma (including primary liver carcinomas and metastatic sarcomatoid renal cell carcinoma), sarcoma (primary and metastatic, including angiosarcoma, Kaposi’s sarcoma, carcinosarcoma, hepatobiliary rhabdomyosarcoma, synovial sarcoma, and leiomyosarcoma)
Giant cell pattern: sarcomatoid or giant cell carcinoma (primary liver carcinomas and metastases), sarcoma (primary and metastases)
Others: nodular hematopoiesis, epithelioid hemangioendothelioma, germ cell tumors (teratoma, yolk sac tumor)
Evaluate ancillary studies. The initial cytomorphological impression is often crucial because the small amount of material may limit the utility of immunohistochemical and other ancillary tests.
Correlate all previous steps. Clinicopathological correlation is mandatory to arrive at a final definitive diagnosis. An “indeterminate” diagnosis may be rendered for very-well-differentiated hepatocellular nodules. Information obtained from all of the previous steps, including a complete immunohistochemical workup, may help establish a generic morphological diagnosis for nonhepatocellular lesions but cannot provide the specific organ of origin.
Evaluate clinical information
Evaluate radiological information
Evaluate cytohistological findings
Evaluate ancillary studies
Correlate all previous steps
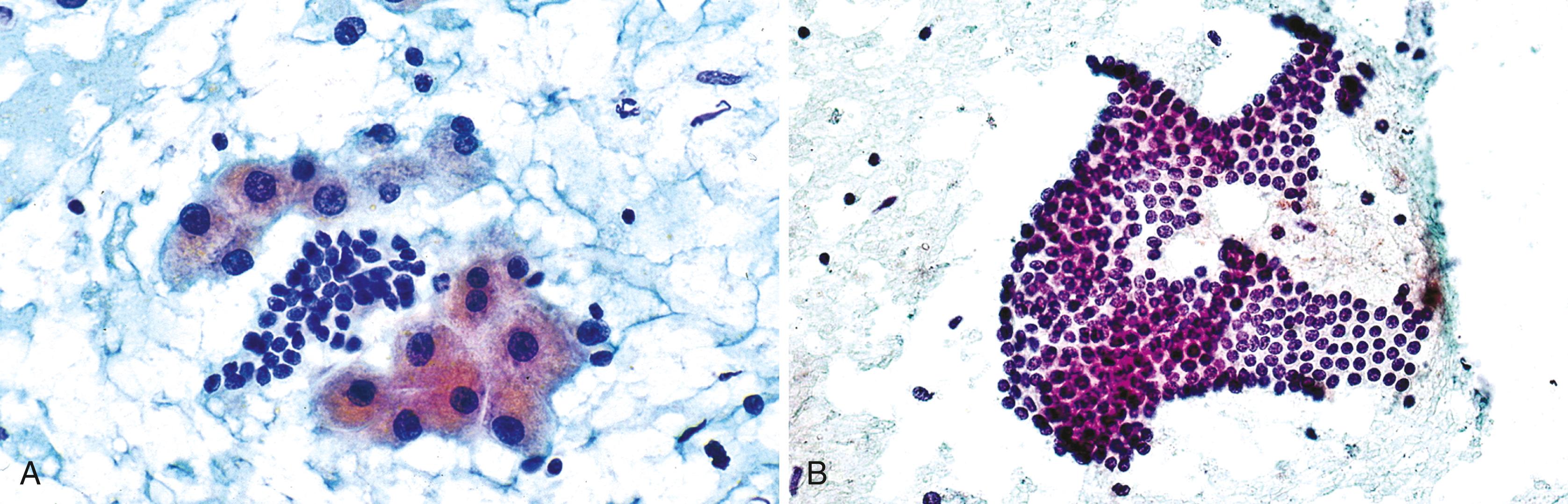
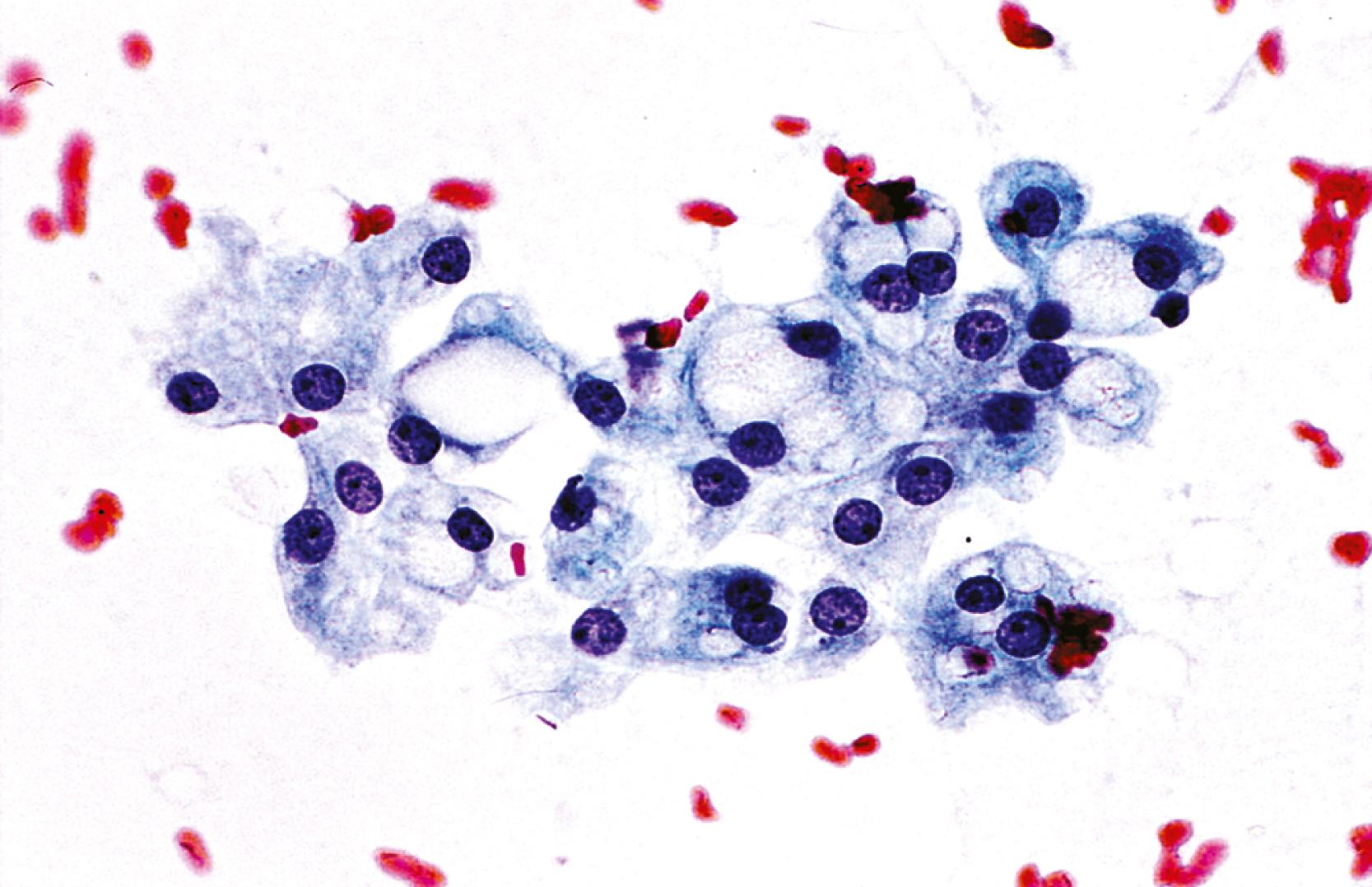
Aspirate smears of normal liver typically show a polymorphous cell population dominated by hepatocytes, occurring singly in short rows or small clusters and intermingling with scattered bile duct epithelial cells and sparse sinusoidal endothelial cells and Kupffer cells ( Fig. 46.1A ). Normal hepatocytes are large, polygonal cells with abundant granular cytoplasm and a centrally placed, round nucleus with a well-delineated nuclear membrane, granular chromatin pattern, and distinct nucleolus. Hepatocytes normally exhibit variations in cell and nuclear size (i.e., sibling polymorphism). The nucleus-to-cytoplasm ratio (N:C ratio) is approximately 1:3. Bile duct epithelial cells are smaller than hepatocytes and appear as flat, monolayered, honeycomb sheets of glandular epithelium (see Fig. 46.1B ). They may also reveal an on-edge picket-fence arrangement or small acinar structures. The cells have minimal pale cytoplasm and equidistant, round to oval, bland nuclei without nucleoli. Bile duct epithelium can be differentiated from bile ductular epithelium.
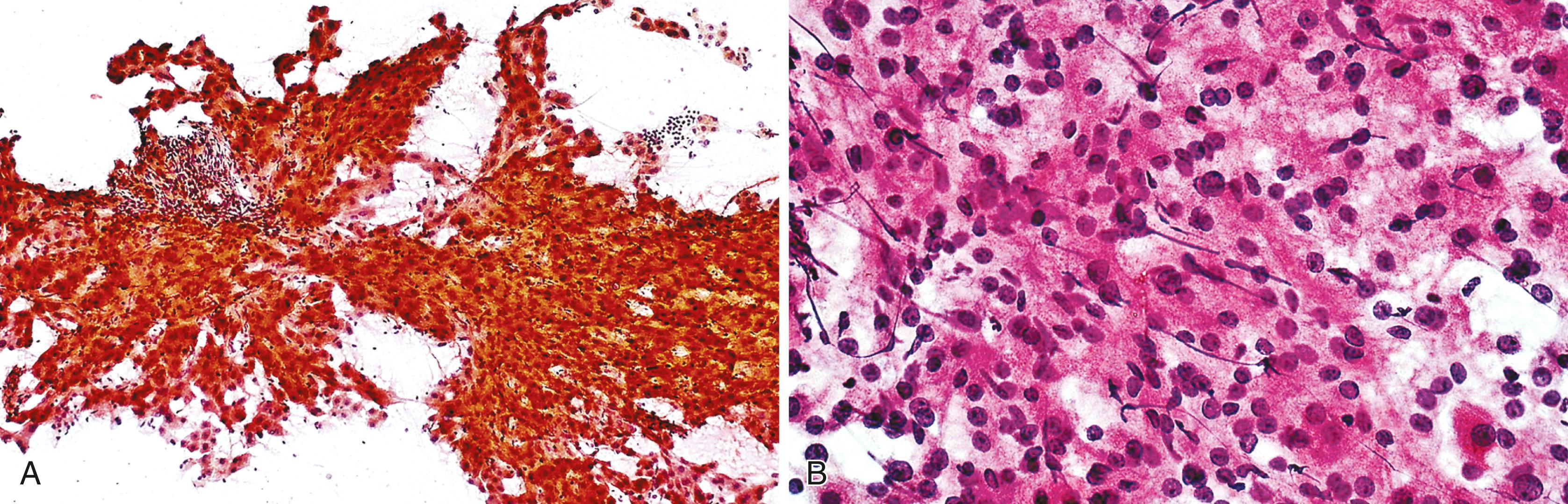
Elongated endothelial cells and comma-shaped Kupffer cells are almost indistinguishable from each other, especially when they appear as nuclear streaks. The thickness of liver cords can be appreciated in large sheets of cells and in fragments of liver parenchyma by focusing on the nuclear streaks in different planes of magnification ( Fig. 46.2 ).
Lipofuscin is a fine, golden, granular, relatively nonrefractile pigment with Papanicolaou stain and appears black with Giemsa stain ( Fig. 46.3 ). This predominantly perinuclear pigment is common in aspirates of adult livers, and its absence in the context of a mass lesion is considered suspicious for a hepatocellular neoplasm. One potential diagnostic pitfall is that lipofuscin stains similar to melanin with Fontana-Masson stain, and the sample may be mistaken for metastatic melanoma.
Bile pigment varies in color, texture, size, and density, but it is best recognized by its coarse, irregular, rather amorphous, nonrefractile appearance. In Giemsa-stained preparations, bile appears as a purplish-black pigment within the cytoplasm of hepatocytes or as ropey strands within bile canaliculi ( Fig. 46.4 ). Hall stain may be used to confirm the pigment as bile.
Iron or hemosiderin may be present in hepatocytes, bile duct epithelial cells, and Kupffer cells and appears as a coarse, golden-brown, refractile pigment with Papanicolaou stain and brown-black with Giemsa stain ( Fig. 46.5 ). Malignant hepatocytes typically lose their ability to retain iron. In the setting of hemochromatosis, Perl (Prussian blue) stain is a helpful ancillary tool to highlight nonstaining tumor cells. ,
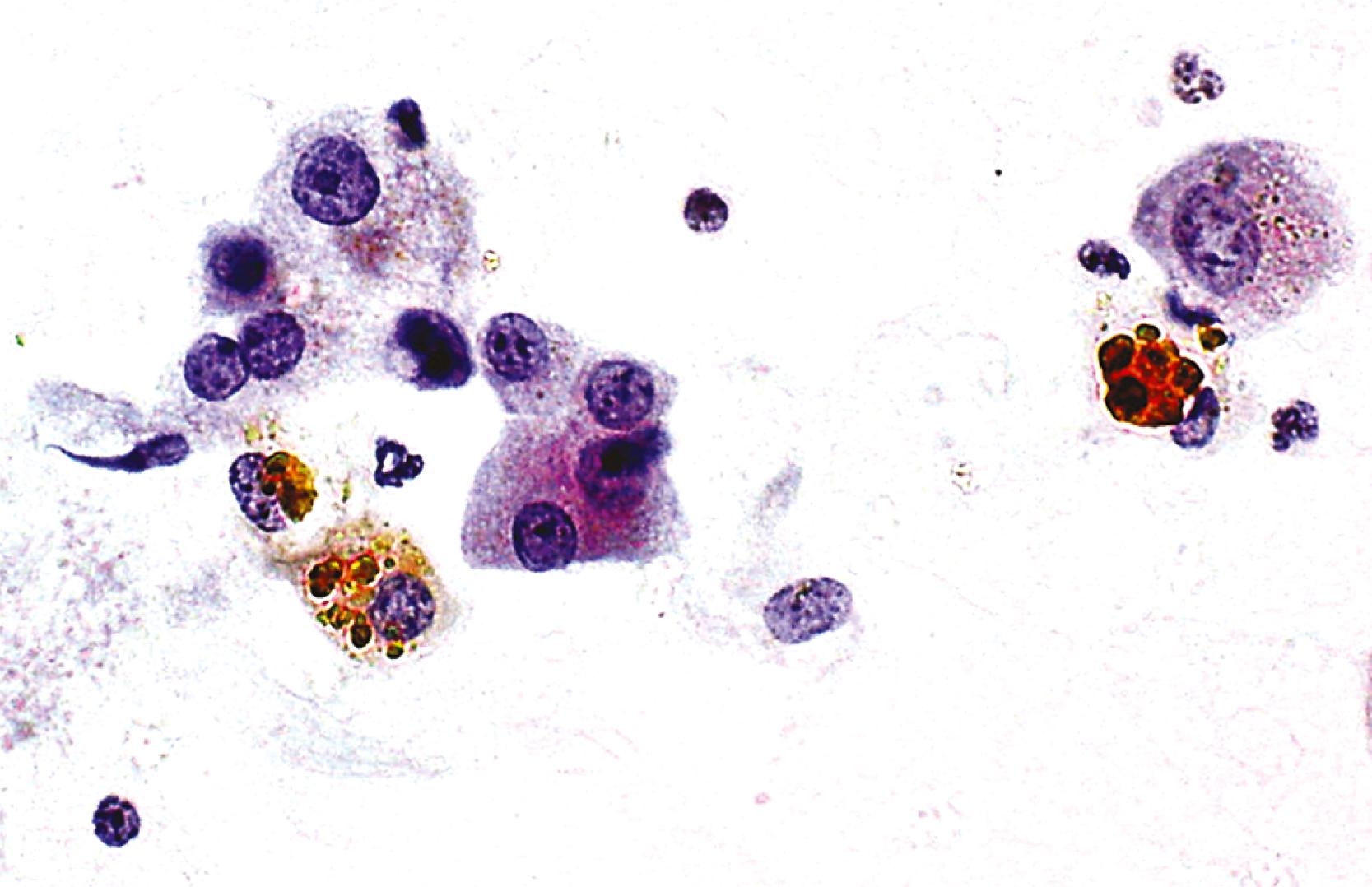
Steatosis is common in the liver, and the examiner should be aware of focal fatty changes. , Cytologically, fat may be a single, large, intracytoplasmic vacuole in a cell with an eccentric nucleus (i.e., macrovesicular steatosis) ( Fig. 46.6 ) or multiple, small vacuoles in a cell with a central nucleus (i.e., microvesicular steatosis). Fat is best appreciated in Giemsa-stained preparations, which often reveal abundant oil globules in the background as a result of ruptured hepatocytes.
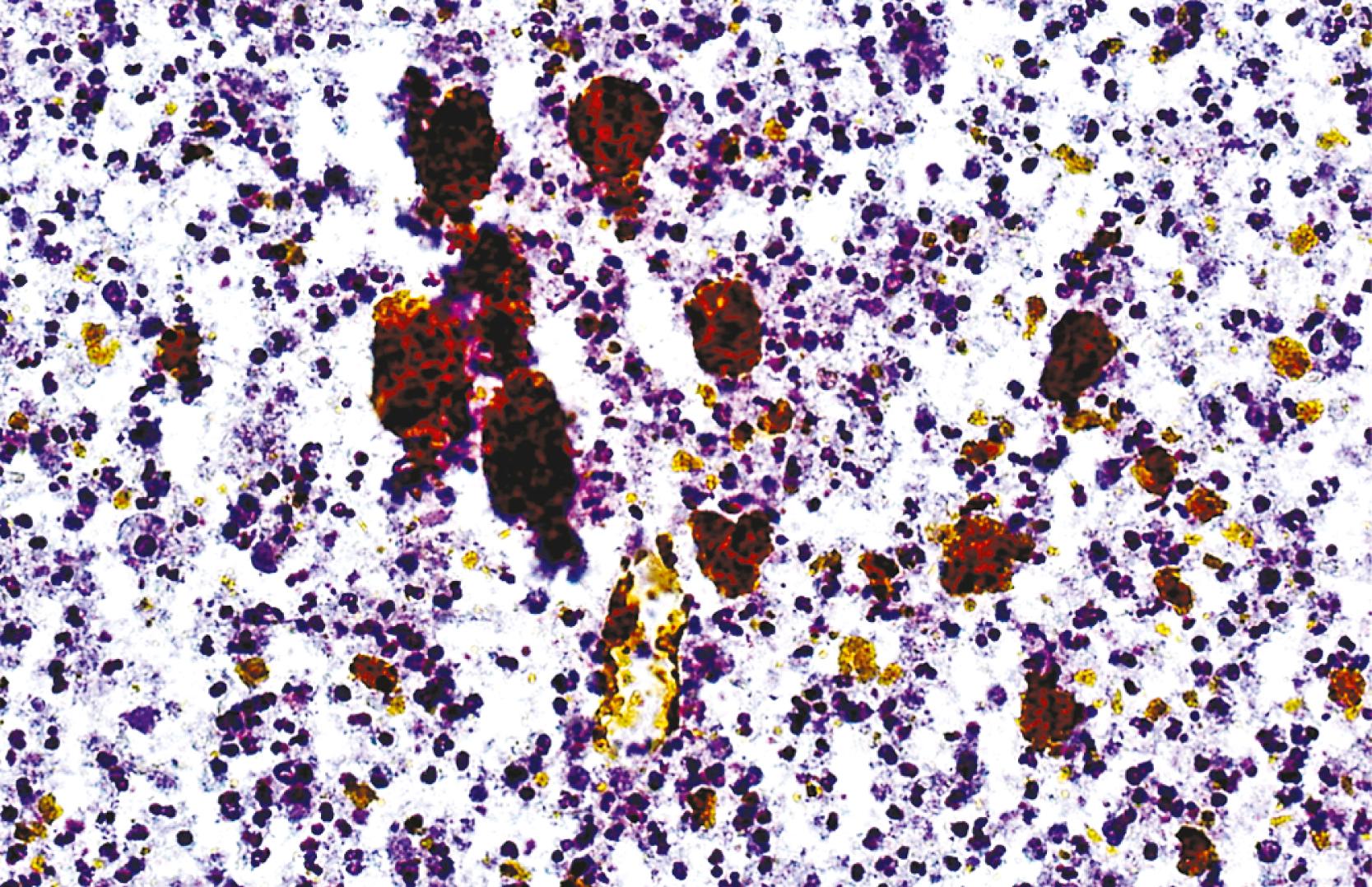
Liver abscess formations are suspected radiologically by the characteristic double target sign on CT scan. The most common is pyogenic abscess, in which case aspiration is performed for tissue confirmation, culture, and drainage. , Smears are dominated by acute inflammatory cells and cellular debris ( Fig. 46.7 ). Cultures are helpful for identification of bacterial organisms. Special stains, such as Gram stain, are not usually helpful, but uniquely characteristic organisms may be readily identified on smears. For example, Actinomyces has sulfur granules ( Fig. 46.8 ).

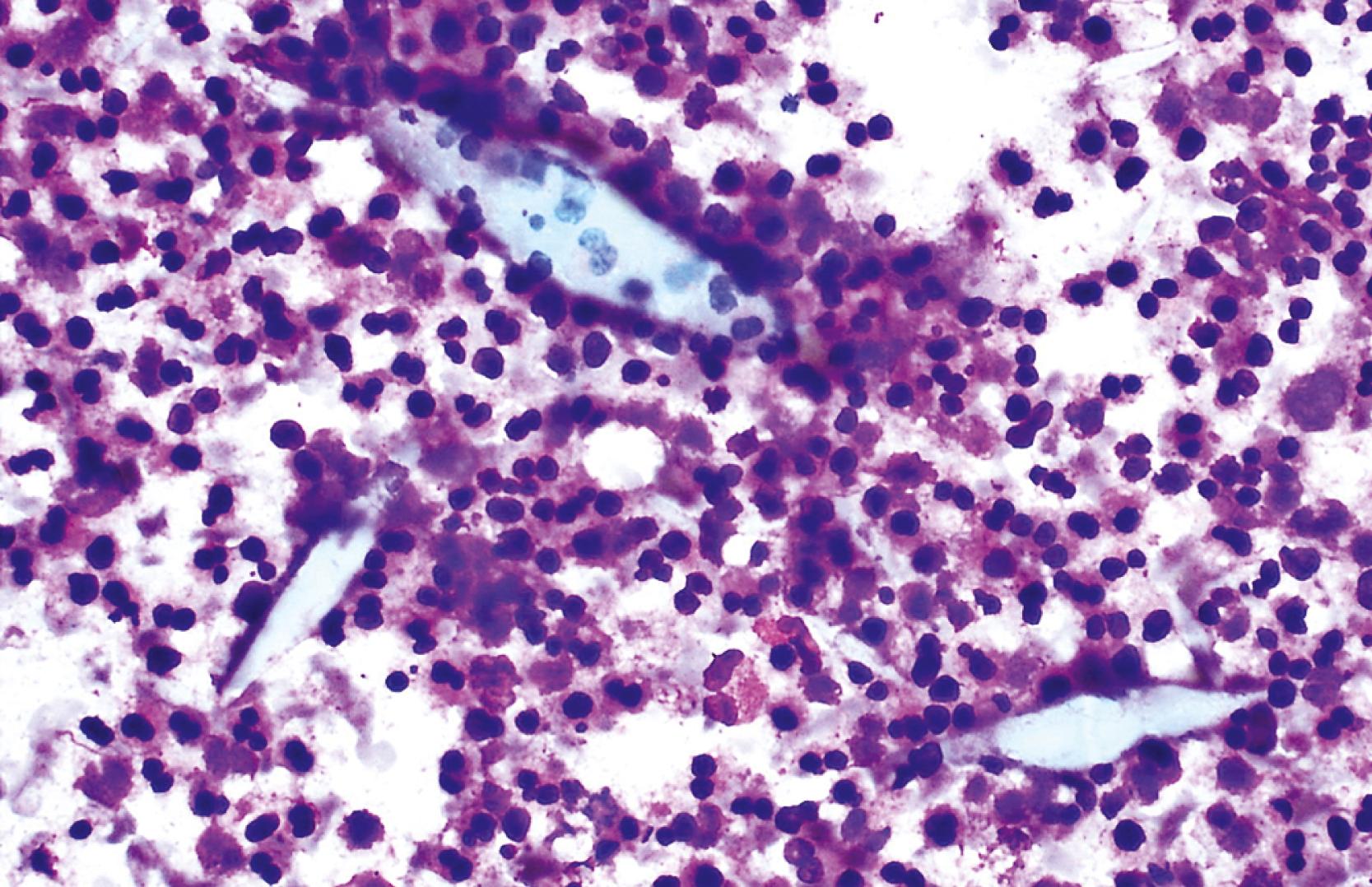
Smears from suspected fungal abscesses should be stained with Gomori methenamine silver and periodic acid–Schiff (PAS) stains. Amebic abscesses tend to be paucicellular with abundant amorphous granular debris, and trophozoites are rare. Necrotizing eosinophilic granulomas contain abundant eosinophils and possibly Charcot-Leyden crystals. Parasitic or fungal organisms should be sought ( Fig. 46.9 ).
A potential diagnostic pitfall is that necrotic tumors may be associated with fever, simulating abscesses clinically and radiologically, or they may become secondarily infected. If clinically warranted, aspiration cytology is recommended to avoid delay in instituting appropriate therapy.
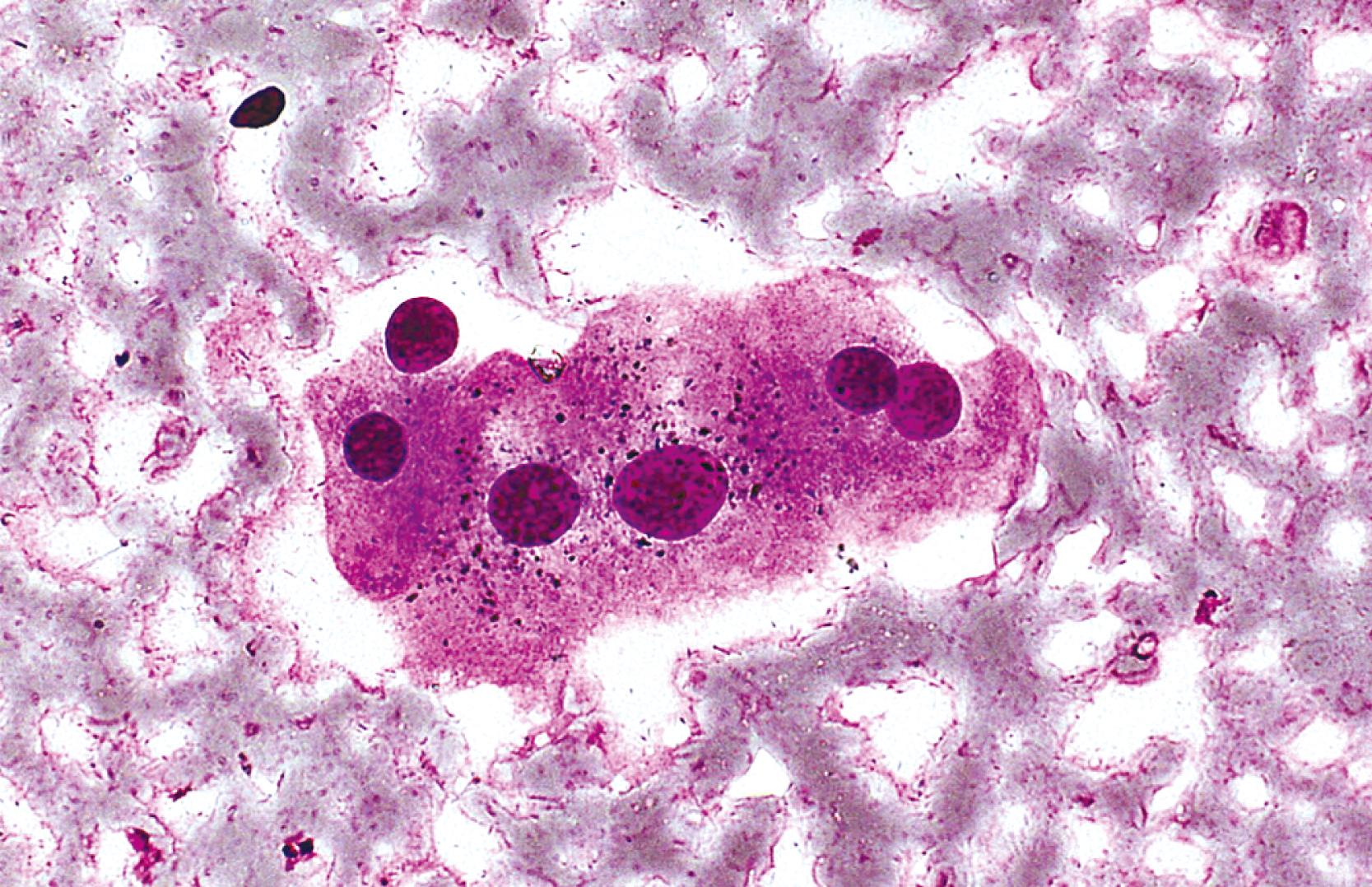
Hydatid disease leads to cyst formation, which may become secondarily infected (see Cystic Lesions of the Liver). Echinococcosis is usually ruled out with clinical and imaging studies and the findings of a negative test result for specific echinococcal antibodies by serum enzyme immunosorbent assay performed before percutaneous needle aspiration. , In the event that a hydatid cyst is aspirated, the pathologist should search for protoscolices or hooklets of Echinococcus granulosus in the fluid or pus ( Fig. 46.10 ).

Granulomatous inflammation may be caused by fungal or acid-fast organisms, but the presence of granulomas is not necessarily diagnostic of an infection. Cytologically, granulomas are composed of clusters of epithelioid histiocytes that have oval to elongated, sometimes twisted nuclei and visible but indistinct cytoplasm. Multinucleated giant cells may be present ( Fig. 46.11 ). The characteristics of the necrotic background should also be evaluated. Gomori methenamine silver, PAS, and Ziehl-Neelsen staining may be performed on smears but are easier to perform on tissue sections.

Inflammatory pseudotumors are potential clinical, radiological, and cytohistological pitfalls because they mimic tumors. They may be lymphoplasmacytic, fibrohistiocytic, xanthogranulomatous, or granulomatous in nature ( Fig. 46.12 ). Activated lymphoid cells may be mistaken for lymphomas, atypical reactive hepatocytes for HCC, atypical reactive biliary epithelium for adenocarcinoma, atypical reactive stroma for sarcoma, and foamy histiocytes for amebic trophozoites. Immunoglobulin G4 (IgG4)–related disease may rarely manifest as a fibroblastic mass with lymphoplasmacytic infiltrates, eosinophils, IgG4-positive plasma cells, dense fibrosis of large bile ducts, and obliterative phlebitis.

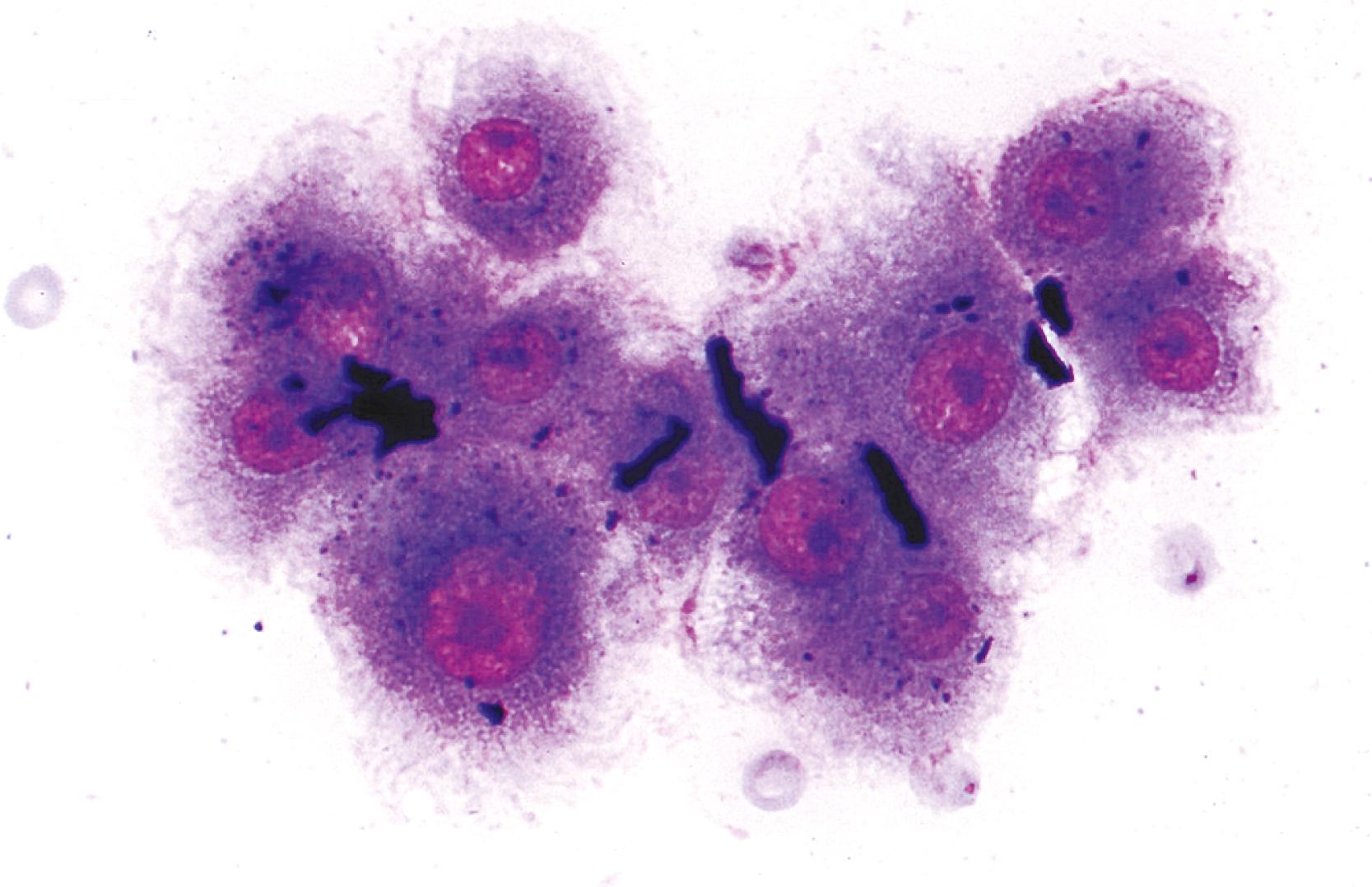
Nodular hematopoiesis is a rare diagnostic pitfall. Hematopoietic cells (especially megakaryocytes) may mimic inflammatory or malignant cells when perceived out of context. ,
The type of cyst contents should be evaluated because the presence of mucin is a helpful diagnostic clue. The next step is to establish the presence and type of epithelial lining and whether it is neoplastic or malignant. Box 46.6 provides a diagnostic algorithm for cystic lesions of the liver.
In cases of bile duct cyst, simple hepatic cyst, and fibropolycystic disease, the fluid may contain isolated cells or scant strips or sheets of cuboidal to low columnar epithelium without cytological atypia ( Fig. 46.13 ).

Aspirates of foregut cysts are characterized by ciliated columnar cells and detached ciliated tufts. The background usually contains amorphous debris and rare macrophages. The differential diagnosis includes cystic neoplasms.
Mucinous cystic neoplasms and intraductal papillary neoplasms have overlapping cytomorphological features. The cytodiagnosis may indicate a benign cyst, papillary neoplasm, or adenocarcinoma. FNAB alone cannot differentiate the various types of hepatobiliary cysts. , Generic terms such as cystic mucinous neoplasm or neoplastic mucinous cyst may be applied if imaging studies are not available for correlation.
Mucinous cystic neoplasms typically occur as single or multiloculated, mucinous, epithelial-lined cysts surrounded by ovarian-type stroma. The cyst fluid may be watery or viscous. Diagnostic aspirates likely contain degenerated cuboidal to columnar epithelial cells with bland nuclear features ( Fig. 46.14 ). The neoplastic epithelial cells may be arranged singly, on edge, in sheets, or as small papillary tufts. The background usually shows mucin with chronic inflammatory cells, histiocytes, and debris. The degree of cellularity varies and tends to increase with higher grades of intraepithelial neoplasia. With malignant transformation, features of adenocarcinoma with or without mucin and cellular debris are often evident. The definitive diagnosis requires correlation with clinical information, imaging studies, and histopathological confirmation of ovarian-type stroma surrounding the cyst. Stromal components are not usually present with cyst content aspiration, but forceps biopsy of the cyst wall may sample the stroma, which can be confirmed with immunohistochemical stain for the estrogen receptor. The cyst fluid may be assayed for carcinoembryonic antigen and CA 19-9 to distinguish neoplastic from nonneoplastic liver cysts ( Box 46.8 ). , ,

Degenerated mucin-secreting cuboidal to columnar epithelium with bland nuclear features; arranged singly, on edge, in sheets, or as small papillary tufts
Background usually shows mucin with chronic inflammatory cells, histiocytes, and debris
Mesenchymal stromal components are usually absent
Variable cellularity, depending on grades of intraepithelial neoplasia
Features of adenocarcinoma seen with malignant transformation
Assay of cyst fluid for carcinoembryonic antigen and CA 19-9
Biliary intraductal papillary neoplasm is a fusiform or cystic (unilocular or multilocular) luminal dilation in communication with the biliary tract. The affected intrahepatic bile ducts are typically filled with a noninvasive papillary or villous biliary neoplasm covering delicate fibrovascular stalks, and mucin may be seen. Aspirates are typically hypercellular, comprising broad and often double-layered sheets of ductal cuboidal or columnar epithelium with a distinctive three-dimensional, complex-branching papillary configuration ( Fig. 46.15 ). The sheets of cells do not form a honeycomb pattern but are crowded. Phenotypes include pancreaticobiliary, intestinal, gastric, and oncocytic forms. The epithelium shows intracytoplasmic mucin vacuoles and occasional signet ring cells. The nuclei show variable abnormalities. Psammoma bodies may be identified. Intraductal papillary neoplasms with associated invasive carcinoma have the highest degree of cellularity and pleomorphism ( Box 46.9 ) (see Metastases).

Hypercellular smears
Broad, double-layered sheets of ductal columnar epithelium with three-dimensional, complex-branching papillae
Pancreaticobiliary, intestinal, gastric and oncocytic phenotypes
Crowded sheets of cells with variable nuclear atypia
Intracytoplasmic mucin vacuoles, including few signet ring cells
Psammoma bodies
Features of adenocarcinoma seen with malignant transformation
Pitfall: Endobiliary cancerization by metastatic colorectal adenocarcinoma
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here