Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Epidermodysplasia verruciformis (EV) is a genetic lifelong disease associated with specific human papillomaviruses (HPV) and multiple skin cancers. Immunosuppressed patients have an increased risk of developing EV lesions.
Bowenoid papulosis presents as wart-like papules usually affecting the genitals of young sexually active adults. HPV genotype is detected in most lesions of bowenoid papulosis with HPV-16 being the most commonly identified.
Progression of bowenoid papulosis to invasive squamous cell carcinoma is relatively uncommon but may occur, especially in immunocompromised hosts.
Verrucous carcinoma is a locally aggressive low-grade malignancy with minimal atypia and is often associated with HPV infection. The lesions rarely metastasize but are associated with local morbidity.
Cutaneous neoplasms are common in HIV-infected patients. As these individuals are now living longer as a consequence of administration of HAART, these are becoming a more important ongoing problem in management.
As we gain increasing knowledge into the pathophysiology of cutaneous malignant neoplasms, it becomes increasingly evident that many are driven by virally mediated mechanisms. These processes, especially in HPV-induced condyloma near the anogenital region, can lead to squamous cell carcinoma (SCC). Human papillomavirus is a small double-stranded DNA virus that is phylogenetically divided into five genera, the most relevant in the skin being the alpha and gamma papillomavirus. Most alpha HPVs infect the mucosal environment surrounding the anogenital region along with the adjacent skin and include types 6 and 11, found in 90% of genital warts. The beta papillomavirus genus refers to the group related to epidermodysplasia verruciformis, with types 5 and 8 related to skin cancer. The viral genome of HPV encodes proteins E6 and E7 that modulate cellular regulatory proteins. E6 binds to and enhances the ubiquitination and degradation of the global tumor suppressor p53 . E7 inactivates the retinoblastoma tumor suppressor gene, which is responsible for halting progression through the cell cycle.
HIV infection and the concomitant immunodeficiency lends itself to increased rates of cutaneous neoplasm. Some are specific to HIV patients, such as Kaposi sarcoma. Others are related to the prolonged immunodeficiency from the infection. Since the introduction of highly active antiretroviral therapy (HAART), the lifespan for HIV-infected patients has expanded, resulting in many patients suffering from common cutaneous malignancies that are endemic to the normal population, albeit at a higher rate.
Epidermodysplasia verruciformis (EV) serves as a unique model for the study of HPV-mediated oncogenesis. EV is a rare, mainly autosomal recessive, virally mediated genetic disease associated with mutations in the EVER1 and EVER2 genes (band 17q25). The disease is characterized by pityriasis versicolor-like macular and planar verruca-like lesions over sun-exposed areas that develop early in life and transform into squamous cell carcinoma later in adulthood. Although patients are infected with multiple beta-HPV types, only a select few (HPV-5 and HPV-8 mainly) are associated with the development of cutaneous malignancies. In addition, immunosuppressed patients are at increased risk of developing EV lesions that resolve with improvement of the immune status. As such, the genodermatosis and its associated genes lend a unique perspective on inherent anti-HPV pathways and virally mediated skin oncogenesis.
EV was first described as a genodermatosis by Lewandowsky and Lutz in 1922 and as a model of viral cutaneous oncogenesis in 1972. The oncogenic EVHPV types were discovered in 1978. Genetic analysis of the 17q25.3 region led to the discovery of the EVER1 and EVER 2 genes.
The disease is rare in the general population. The largest series to date involves fewer than 200 patients, mainly in Eastern Europe and Latin America.
A large proportion of patients are born to consanguineous parents, with the proportion approaching one-third. The mode of inheritance for the vast majority is autosomal recessive. An X-linked recessive mode of inheritance has also been reported. The classically associated EVER1 and EVER2 genes, also termed TMC6 and TMC8 , encode TMC (transmembrane channel-like) proteins that localize to the endoplasmic reticulum with features of integral membrane proteins. The exact function in prevention of persistent infections has not yet been revealed. EV is associated commonly with specific benign and malignant strains of beta HPVs that share nucleotide homology. Of about 20 characterized EVHPVs, high-risk HPV-5 and HPV-8 are predominantly associated with malignant lesions.
The pathogenesis of EV has not clearly been elucidated. It has been hypothesized that the EVER genes play a role in immune response to the infection or control of the infection within keratinocytes. A cell-mediated immunity defect relating to inhibition of natural cytotoxicity and T lymphocyte proliferation to HPV infection has been shown. The exact role that HPV plays in the development of cutaneous carcinoma is also unclear. Some have suggested that EVHPV infection is a cofactor in the pathogenesis of skin carcinoma, with cancers occuring more commonly in sun-exposed areas and higher viral loads discovered in actinic keratosis than in squamous cell carcinoma for EV patients.
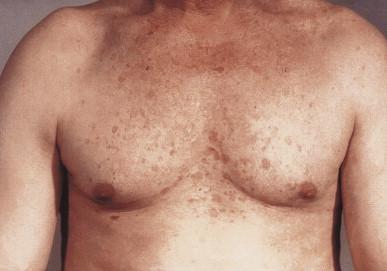
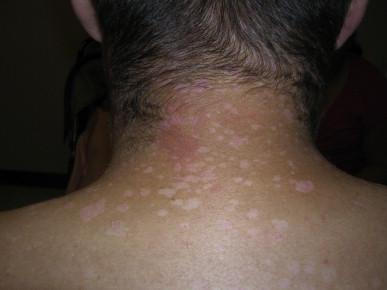
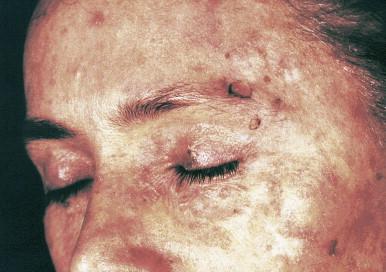
The first cutaneous lesions appear at the age of 5–8 years, usually as flat plane warts localized mainly on the face and hands, spreading progressively all over the body. Somewhat later, red plaques, located usually on the neck and trunk, appear ( Fig. 19.1 ). Skin changes are polymorphous. There can also be brown, red and achromic plaques and pityriasis versicolor-like lesions ( Fig. 19.2 ). The plane wart-like lesions can display a Koebner phenomenon. Malignant transformation occurs in about half of the cases and usually in the third decade. The preferential localization of neoplasms is the forehead and temporal areas, with premalignant lesions resembling actinic keratosis. The rate of progression depends on the viral type of EVHPVs and the extent of sun exposure ( Fig. 19.3 ). Cancers – although persistent – are usually only locally destructive with low metastatic potential. The disease is characterized by a lifelong persistence of cutaneous disease, usually with no involvement of internal organs, mucous membranes or lymph nodes. The overall prognosis is good.
The diagnosis of EV is based on clinicopathological correlation along with virological findings. EV-like lesions can be found incidentally in normal skin and in immunosuppressed patients and histological findings by themselves do not constitute a diagnosis. In the correct clinical setting and with physical findings, the diagnosis can be confirmed by skin biopsy. This demonstrates highly characteristic cytopathic effects of bluish-purple color to ballooned keratinocytes in the epidermis ( Fig. 19.4A ). Characterization of mutations of specific genes EVER1 and EVER2 provides confirmation of the diagnosis. As the strain of EVHPVs causing the condition cannot be elucidated by histology alone, PCR analysis of paraffin-embedded tissue can confirm the presence of an oncogenic or non-oncogenic strain. Southern blot analysis is highly specific for HPV typing ( Fig. 19.4B ). The clinical differential diagnoses include tinea versicolor, planar warts, and widespread macular seborrheic keratoses. The extent of the disease, clinical history, prevalence in sun-exposed portions of the body, and early appearance of the disease in cases with genetic predisposition are clues to diagnosis.
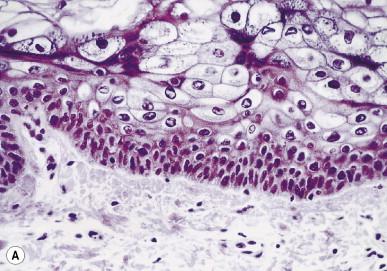
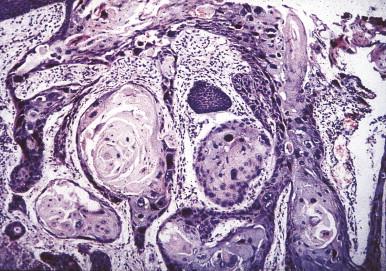
The classic histological presentation of EV on low-power histological evaluation is that of a planar wart-like papule with hyperkeratosis, hypergranulosis and acanthosis. The keratinocytes possess a blue-gray pallor with perinuclear halos and keratohyaline granules. They are arranged in columns or nests, with the pallor being most conspicuous in the superficial granular layer. Premalignant lesions appear clinically and histologically similar to actinic keratosis with atypical keratinocytes in the epidermis as well as in adnexal epithelium. As the lesions progress to frank carcinoma, the keratinocytes lose typical EV findings and appear more as traditional SCC in situ and invasive SCC ( Fig. 19.5 ).
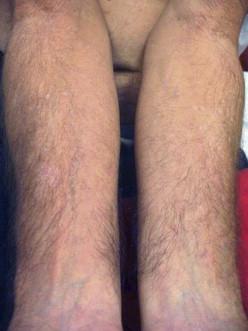
No therapy for EV secondary to a genetic defect is definitive. Treatment includes preventive modalities, especially sun protection and avoidance. For patients with EV secondary to immunosuppression ( Fig. 19.6 ), reconstitution of the immune system is key, although complete resolution of EV lesions is unlikely. Various modalities have been employed in case reports and case series for treatment of malignant lesions. including surgery, systemic retinoids, interferon-alpha, imiquimod, cimetidine, 5-fluorouracil, and photodynamic therapy. Cidofovir has been shown to be unsuccessful in one case report. For neoplasms in a localized region, auto-transplantation from uninvolved skin has been reported with some success. Radiotherapy should be avoided.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here