Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Adverse drug reactions are unintended and undesired effects of drugs used for prevention, diagnosis, or treatment of disease. In light of the ever-increasing number of medications available, it should come as no surprise that such reactions are extremely common. The incidence statistics vary considerably depending upon the method by which the data are derived and the nature of the population under study. Estimates, however, range from 2% to 7% of hospital inpatients. Although most reactions are mild, they are sometimes severe and a source of considerable morbidity and occasional mortality.
The diagnosis of an adverse drug reaction is frequently problematical, the clinical appearances often being similar, if not identical, to a number of primary dermatoses and infectious conditions (particularly viral exanthems) and, in the context of transplantation patients, graft-versus-host disease (GVHD). The histologic diagnosis can also be extremely difficult, as drug reactions can demonstrate several inflammatory histologic patterns that mimic other dermatoses (i.e., spongiotic, psoriasiform, lichenoid, pityriasiform). The problem is exacerbated in the immunologically compromised patient. Frequently, the diagnostic difficulties are worsened by the multitude of drugs prescribed. The problem is further compounded by the multiplicity of different eruptions that any one particular drug may induce. Contrariwise, a given clinical appearance may be caused by a large number of unrelated drugs.
The prevalence of agents responsible for adverse drug reactions reflects the prescribing tendencies for any given population as much as the relative risks ascribed to any particular drug. It should come as no surprise, therefore, that – in a hospital environment – antibiotics, nonsteroidal anti-inflammatory drugs (NSAIDs), and psychotropic drugs are commonly reported as being the most frequently incriminated. Oral anticoagulants, low-dose aspirin, and digoxin are also frequent causes. In a large hospital survey, penicillin and sulfonamides accounted for over 80% of all adverse drug reactions. Experience in general practice has been much less often documented. In a survey from the Netherlands, sulfonamide-trimethoprim combinations, fluoroquinolones, and penicillin were the most common antibacterials causing drug-related eruptions. In the series of approximately 150 000 patients, 1% developed a reaction.
Adverse drug reactions are mostly nonimmunologically mediated. They develop either as a result of an unwanted but known property of the drug (and hence are entirely predictable) or as a consequence of drug intolerance/idiosyncrasy (and are completely unpredictable). The former are by far the more common, accounting for approximately 80% of all adverse drug reactions. Less often, adverse drug reactions represent a manifestation of an immunological phenomenon, so-called allergic drug reactions. Although in theory the above subdivisions are sharply defined, in many patients the underlying pathogenetic mechanisms are far from clear.
Adverse drug reactions are particularly encountered in certain population groups, for example, the elderly, females, patients with Sjögren syndrome, and those suffering from the effects of immune deficiency including patients receiving immunosuppressive therapy and those suffering from the acquired immunodeficiency syndrome (AIDS).
Adverse drug reactions can be divided into three categories: type A, type B, and type C.
Type A reactions, which are predictable and are related to the pharmacological actions or metabolism of the drug, include :
side effects,
drug toxicity,
drug interactions.
Side effects, which occur with almost all drugs, represent unwanted pharmacological actions. For example, methotrexate, cyclophosphamide, and nitrosourea commonly result in anagen alopecia by inducing Bax protein-mediated apoptosis. Gold may be associated with cutaneous pigmentation (chrysiasis) and penicillamine can be associated with the development of skin laxity and fragility.
Drug toxicity develops as a consequence of the gradual accumulation of a drug or its metabolite (e.g., minocycline or amiodarone deposition with resultant abnormal pigmentation). Delayed toxicity may take months to many years before expression (e.g., arsenical keratoses).
Drug interactions develop when one drug alters the pharmacological efficacy of another that is given concurrently. The effect may enhance or diminish the effect of the drug with resultant toxicity or loss of therapeutic value. Drug interactions are thought to arise when one drug affects clearance of the other as a consequence of several mechanisms including :
alteration in the rate of absorption resulting in diminished drug levels,
alteration in the renal excretion resulting in inappropriately high drug levels,
plasma protein or tissue drug binding site competition resulting in displacement and inappropriately high drug levels,
alterations in hepatic cytochrome P-450-mediated drug metabolism.
The last is believed to be of particular importance and includes increased enzyme synthesis with excessive drug degradation and diminished circulating or tissue levels and inhibition of drug breakdown with increased circulating and tissue levels.
Drug interactions are of particular importance in the elderly, the immunosuppressed, and in those patients receiving multiple medications.
Type B reactions are uncommon and unpredictable. They do not have an allergic pathogenesis and include :
idiosyncratic drug reactions,
exacerbation of a pre-existing condition,
pseudoallergic drug reactions.
Idiosyncratic reactions (drug intolerance) develop as a result of genetic or metabolic influences. They may represent the effects of abnormal or altered hepatic drug metabolism. For example, a lupus erythematosus-like condition is a rare complication of hydralazine therapy in the average population, but the risk is greatly increased in patients who metabolize the drug slowly. Drug-induced lupus erythematosus may also be caused by procainamide, chlorpromazine, isoniazid, methyldopa, penicillamine, minocycline, quinidine, and sulfasalazine. Cefaclor-induced serum sickness-like eruptions and the antiepileptic hypersensitivity syndrome are also believed to result from reactive intermediate metabolic products.
This is a not uncommon problem; for example, lithium, beta blockers, antimalarial drugs, NSAIDs, and tetracycline may precipitate, aggravate, or induce a psoriatic eruption.
Pseudoallergic reactions result from the nonimmunologically mediated release of effector substances such as histamine from tissue-bound mast cells or circulating basophils with resultant urticarial reactions, angioneurotic edema, and anaphylaxis. The complement system can also be activated by similar nonimmune mechanisms, and there is evidence that perturbation of arachidonic acid metabolism may be involved in some cases. Drugs that have been incriminated in such pseudoallergic reactions include radiocontrast media, NSAIDs, acetyl salicylic acid, opium derivatives, codeine, curare, d -tubocurare, polymyxin B, and angiotensin-converting enzyme (ACE) inhibitors.
Type C reactions are rare, immunologically mediated, and develop as a consequence of previous exposure to the drug with resultant allergy. The majority of drugs are of low molecular weight (less than 1000 Daltons) and therefore on their own are incapable of eliciting an immune response. By functioning as haptens and forming conjugates with carrier plasma proteins or cell membrane constituents, they develop immunogenic potential. The ability of the majority of drugs to cause an immune response is therefore dependent on whether it is able to bind to circulating or tissue protein. A number of drugs are likely to induce allergic reactions, including antibiotics, anticonvulsants, chemotherapeutic agents, heparin, insulin, protamine, and biological response modifiers such as interferons (IFNs) and growth factors. A variety of mechanisms may be involved in cutaneous drug-induced hypersensitivity reactions including :
IgE-mediated type 1 reactions,
immune complex-associated type 3 reactions,
type 4 delayed hypersensitivity reactions.
In type 1 reactions, the release of histamine and other chemical mediators from tissue-fixed mast cells results in increased vascular permeability with development of edema in the dermis or deeper tissues. Immediate reactions which develop within an hour or less of drug exposure present as urticaria, angioedema, or anaphylaxis, whereas accelerated reactions which develop from 1 to 72 hours following the administration of the drug are usually urticarial. Urticaria following treatment with penicillin is a typical type 1 reaction. Certain other antibiotics, antisera, and gammaglobulin are also common offenders.
The most common cause of anaphylaxis is penicillin. Other causes include foods, stings, anesthetics, muscle relaxants, latex, contrast material, antibiotics, and allergenic extracts. In addition to histamine, anaphylaxis is mediated through a number of substances including prostaglandin D2, leukotriene C4, interleukin (IL)-4 and IL-13, and tumor necrosis factor alpha (TNF-α).
Type 3 reactions are expressed as urticaria, the Arthus reaction, serum sickness, and leukocytoclastic (allergic) vasculitis. The disease manifests a week or more after exposure to the drug, by which time sufficient circulating antibody has been generated to result in immune complexes of an appropriate size to avoid phagocytosis. Their deposition in the tissues or within blood vessel walls is accompanied by complement fixation and resultant acute inflammatory reaction.
Delayed hypersensitivity reactions are T-lymphocyte mediated and exemplified in acute allergic contact dermatitis. Cytotoxic T-cell-mediated reactions are of importance in many other adverse allergic drug reactions including exanthematous/morbilliform, bullous, and interface variants. Although most delayed hypersensitivity reactions are immune responses to the hapten-carrier complex, recent studies indicate that some drugs may be capable of directly activating the immune system, independent of a covalent drug-peptide complex. Certain medications may directly bind T-cell receptors (TCRs) and major histocompatibility complex (MHC) molecules and trigger the release of cytokines which recruit specific leukocytes. The delayed hypersensitivity reactions may be subclassified based on the cell type recruited: monocytes (type 4a), eosinophils (type 4b), T cells (type 4c), and neutrophils (type IVd). The resultant clinical phenotype may be determined by which cells are involved.
Although the range of drugs that may result in adverse drug reactions is extensive, the variety of clinical responses encountered is fairly limited. Many drugs may cause more than one clinical response, and any given reaction pattern may result from a wide range of drugs. There are, however, a number of clinicopathological responses that are fairly unique to a particular drug and these are dealt with individually, later in this chapter.
Adverse drug reactions may therefore present with a considerable number of clinical manifestations as outlined in Box 14.1 .
Exanthematous reactions
Urticaria, angioedema and anaphylaxis
Serum sickness
Phototoxic/photoallergic eruptions
Hypersensitivity syndrome
Lichenoid drug reactions
Fixed drug eruptions
Erythema multiforme
Stevens-Johnson syndrome/toxic epidermal necrolysis
Pigmentary abnormalities
Vasculitis
Purpura
Granulomatous drug reactions
Erythema nodosum
Drug-induced alopecia
Lupus erythematosus-like drug reactions
Bullous drug reactions
Psoriasiform drug reactions
Pityriasis rosea-like eruptions
Pustular drug reactions
Ichthyosiform drug reactions
Pseudolymphomatous drug reactions
Eczematous drug reactions
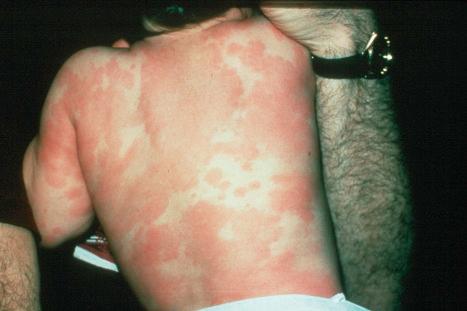
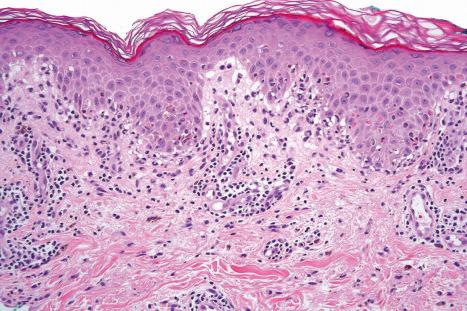
Exanthematous (morbilliform, maculopapular) reactions are the most frequently encountered adverse drug reaction, accounting for 51% to 95% of skin reactions, and mimic a variety of infective conditions including scarlet fever, measles, and rubella ( Figs 14.1 and 14.2 ). Patients present with erythematous macules and papules that may become confluent or gyrate/polycyclic. Pruritus, low-grade fever, and eosinophilia are sometimes present. The eruption is often symmetrical and usually presents on the trunk and extremities or sites of pressure and trauma. The palms and soles are sometimes affected but the mucous membranes are not usually involved.
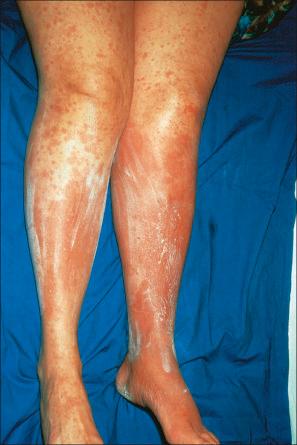
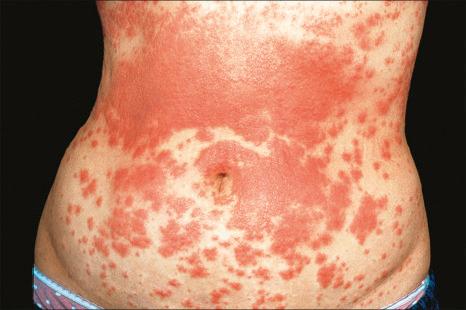
Exanthematous eruptions typically develop within 1–2 weeks of starting the drug. Occasionally, the eruption is delayed and may even present after the treatment has ceased. In more seriously affected patients the eruption can progress to erythroderma (exfoliative dermatitis), in which the erythema becomes generalized and is often accompanied by scaling. Resolution of exanthematous drug reactions is characterized by exfoliation and sometimes is followed by postinflammatory hyper- or hypopigmentation. Penicillin, sulfonamides, ampicillin, amoxicillin, phenylbutazone, isoniazid, barbiturates, phenytoin, carbamazepine, benzodiazepines, gold, and trimethoprim are especially incriminated, but a wide range of medications can induce exanthematous drug eruptions. Patients who suffer from infectious mononucleosis are at risk of developing an exanthematous reaction following therapy with ampicillin or amoxicillin. Dietary supplements can also induce a similar cutaneous eruption.
The pathogenesis of exanthematous drug reactions is not fully understood, although a cytotoxic T-cell-mediated reaction is likely in most cases (see below).
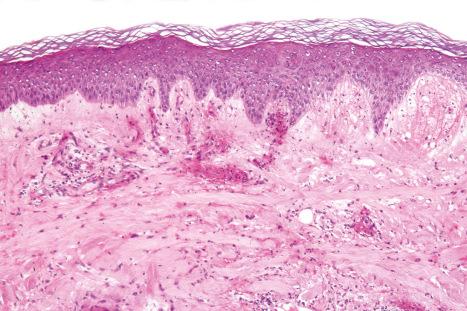
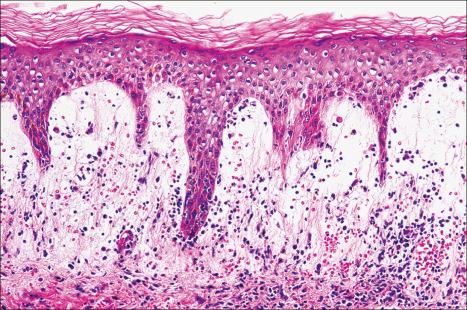
The histologic features are often subtle. Although the epidermis may appear normal, focal parakeratosis is commonly present. The characteristic changes include lymphocytic exocytosis with mild spongiosis, typically accompanied by basal cell liquefactive degeneration and a few dyskeratotic keratinocytes ( Figs 14.3–14.7 ). In some cases, no interface change is histologically apparent. The dermis shows a perivascular infiltrate of lymphocytes and histiocytes with variable numbers of eosinophils. Eosinophils – although often emphasized in the literature as an important feature of drug reactions – can, in our experience, be very scanty or even absent. Sometimes marked edema is seen, particularly if an urticarial element is clinically evident. Red cell extravasation may also be a feature in those lesions that include a purpuric component.
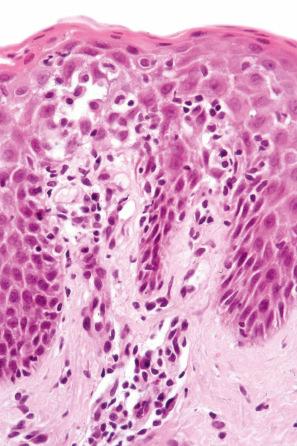
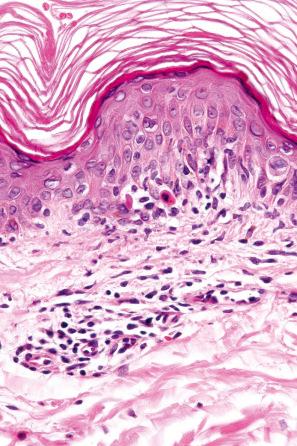
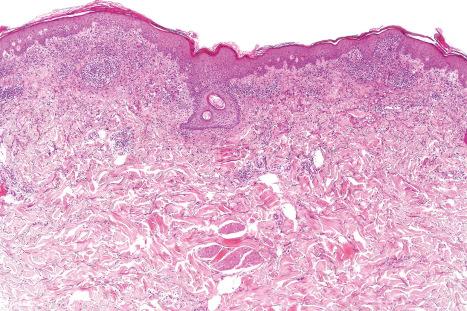
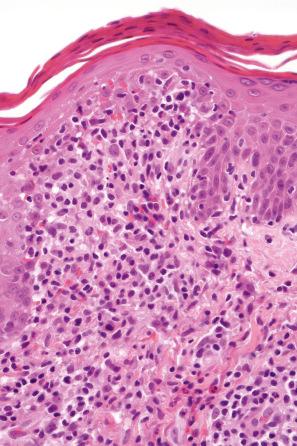
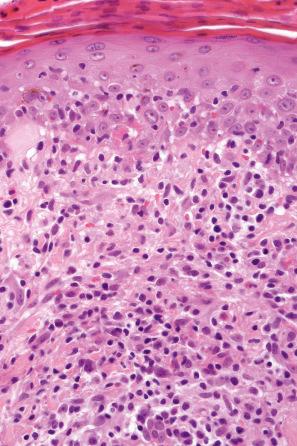
By immunohistochemistry, the lymphocytes are largely CD3+ T-cells with a predominance of CD4+ cells in the superficial perivascular infiltrate. Lymphocytes at the dermal–epidermal junction and within the epidermis consist of approximately equal numbers of CD4+ and CD8+ forms. These latter cells regularly express human leukocyte antigen (HLA)-DR and a subpopulation also expresses CD25. There is an admixture of T-helper Th1 and Th2 cells. Occasionally, the infiltrate is almost entirely composed of the CD4+ lymphocytes and, contrariwise in human immunodeficiency virus (HIV)-positive patients, the infiltrate may consist of CD8+ cells alone. CD1a+ dendritic cells and CD68+ histiocytes are also present. CD56+ natural killer (NK) cells may be identified. Cytotoxic pathways mediated by perforin and granzyme B have been shown to be of particular importance in exanthematous drug reactions. Fas/Fas-L cytotoxic mechanisms are not thought to be of relevance.
The features of drug-induced erythroderma are rather non-specific and include parakeratosis and psoriasiform hyperplasia, sometimes accompanied by mild spongiosis. Eosinophils may be identified within the dermal chronic inflammatory cell infiltrate.
Exanthematous adverse drug reactions are a frequent feature in transplantation patients who are usually taking multiple medications and, therefore, must be distinguished from acute GVHD. In reality, it is difficult, if not impossible, to make this distinction histologically. Previously, the presence of eosinophils was thought to support a drug reaction. However, tissue eosinophils can be seen in both GVHD and drug reactions. It is only when eosinophils are relatively numerous (≥ 16 per 10 high-power fields [HPFs]) can a drug reaction be strongly favored over GVHD. A viral exanthem also commonly enters the differential diagnosis – the histologic findings are often indistinguishable although the presence of eosinophils may favor a drug reaction.
Urticaria is the second most common adverse drug reaction. It is characterized by pruritic, erythematous, and edematous wheals. If accompanied by marked edema involving the deeper dermis and subcutaneous fat, or if the submucosal layers are affected, angioedema results. Urticaria can also be a manifestation of serum sickness and anaphylaxis.
Urticarial reactions may be caused by a large number of drugs. In a large retrospective study from Thailand, 147 different drugs were presumed etiological agents of drug-induced urticaria. Antibiotics, especially cephalosporins, and NSAIDs were most commonly implicated. Other common causes include aspirin, penicillin, ACE inhibitors, and blood products. Drugs that directly stimulate mast cell release of vasoactive substances such as histamine can cause urticaria, and include opiates, curare, vancomycin, and polymyxin B. Radiocontrast media may have a similar effect. Urticarial reactions due to aspirin and NSAIDs are thought to sometimes be a result of abnormal arachidonic acid metabolism.
The pathogenesis of urticarial drug reactions includes IgE-mediated type 1 reactions, immune complex mechanisms, and pseudoallergic phenomena (non-IgE-mediated). Ultrastructural studies have found mast cell tryptase and FXIII in the superficial nerves of patients with drug-induced urticaria, possibly contributing to the symptomotology. There is evidence that chemokines may also play a role.
Histologically, urticaria is characterized by dermal edema and vascular dilatation accompanied by a perivascular infiltrate consisting of lymphocytes and eosinophils ( Fig. 14.8 ). Edema is often difficult to appreciate histologically but may be inferred by separation of collagen bundles. Mast cell degranulation may be present. Vasculitis is not a feature.
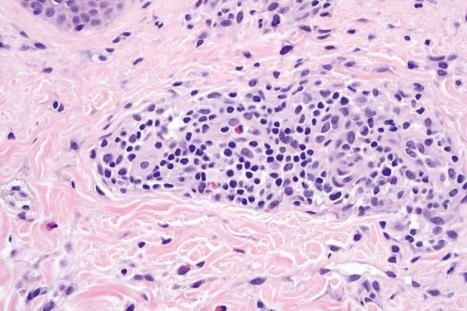
Angioedema is characterized by edema extending into the deeper dermis and subcutaneous fat.
Serum sickness develops within 1–3 weeks after taking the serum or vaccine. It presents with an erythematous maculopapular or urticarial response or with palpable purpura variably accompanied by fever, arthralgia, myalgia, arthritis, lymphadenopathy, glomerulonephritis, myocarditis, and neuritis ( Fig. 14.9 ). The cutaneous manifestations often commence on the sides of the fingers, toes, and hands before becoming more generalized. A wide range of drugs has been implicated in the development of serum sickness-like drug reactions including phenytoin, phenylbutazone, and carbamazepine. Antibiotics are also a common offender, with cefaclor featured most prominently along with other antibiotics such as cefprozil, ciprofloxacin, minocycline, penicillin V, amoxicillin, flucloxacillin, and co-trimoxazole. More recently, therapy with monoclonal antibodies such as rituximab, infliximab, natalizumab, and omalizumab has been associated with serum sickness-like reactions. It has also been seen in transplant patients receiving rabbit antithymocyte globulin treatment.
Serum sickness is thought to represent an immune complex-mediated type 3 reaction although the possibility of direct toxicity against vessel wall, autoimmunity, and cell-mediated cytotoxicity have been proposed as alternative pathogenetic mechanisms. Direct immunofluorescence reveals immunoglobulin and C3 in relation to blood vessel walls.
The histologic features are those of leukocytoclastic vasculitis ( Fig. 14.10 ). Patients with a serum sickness-like drug reaction with histologic findings reminiscent of neutrophilic urticaria have been described.
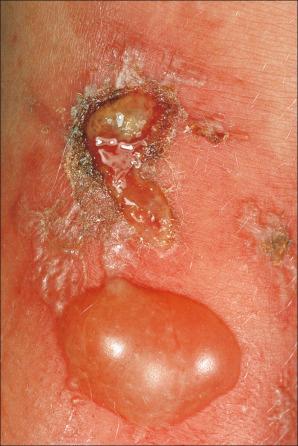
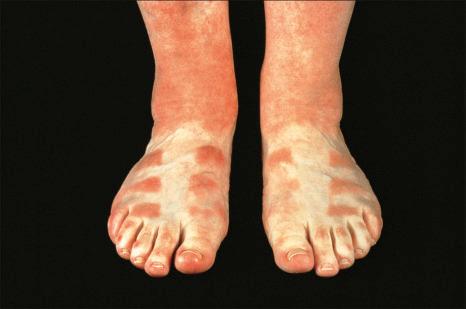
There are two types of photosensitive drug reactions: phototoxic and photoallergic. Phototoxic reactions are more common; however, they are not necessarily mutually exclusive and are not always clinically distinguishable. They are relatively common, representing up to 8% of adverse drug reactions. The clinical appearances of acute phototoxic reactions mimic severe sunburn and include erythema, edema, and blistering with subsequent desquamation and postinflammatory hyperpigmentation ( Figs 14.11 and 14.12 ). Typically, only exposed skin is affected, and it occurs minutes to hours after sun exposure. Phototoxicity has also been associated with onycholysis.
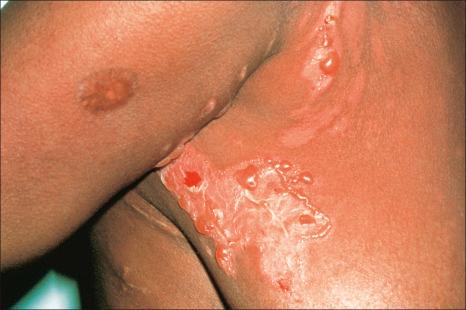
Chronic phototoxicity presents with poikilodermatous features including hyper- and hypopigmentation, epidermal atrophy, and telangiectasia. It is an important feature of the porphyrias and the inherited photodermatoses such as xeroderma pigmentosum, Rothmund-Thomson syndrome, and Bloom syndrome. It rarely results from drug treatment but may follow long-term therapy with psoralen and UVA (PUVA therapy), where there is also an increased incidence of actinic keratosis, basal cell carcinoma, squamous cell carcinoma, and, more rarely, melanoma.
Drugs that are incriminated in acute phototoxic reactions include thiazide diuretics, sulfonamides, tetracycline antibiotics, NSAIDs (including naproxen, diclofenac, and ketoprofen), phenothiazines (particularly chlorpromazine), amiodarone, tars, psoralens, antidepressants, chemotherapeutics, calcium channel blockers, and atovastatin. Phototoxicity has also been described with use of St. John's wort and following photodynamic therapy. Porphyrins are potent phototoxic sensitizers. In this instance, the damage affects the dermal constituents including the vasculature, leaving the epidermis relatively unaffected.
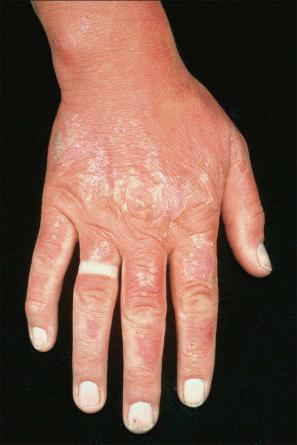
The clinical appearances of photoallergic drug reactions are variable and include eczematous and lichenoid dermatitides ( Figs 14.13 and 14.14 ). Patients may also have erythema multiforme (EM), telangiectasia, hyperpigmentation, and pseudoporphyria. The rash usually develops 24 hours or more after sun exposure. Unlike phototoxic reactions, unexposed skin may also be affected in addition to exposed skin. Typically, the dermatitis resolves after withdrawal of the offending agent. Rarely, a persistent light reaction may occur in which the photodermatitis persists despite removal of the photosensitizing chemical. This usually occurs in the setting of photoallergic contact dermatitis.
The majority of photoallergic reactions are induced by the application of topical medicaments and chemicals (contact photoallergy) including antihistamines, local anesthetics, chlorpromazine, hydrocortisone sunscreens containing p -aminobenzoic acid, and halogenated phenolic compounds in soaps and fragrances (e.g., 6-methylcoumarin and musk ambrette). Photoallergy can also follow systemic administration of drugs including sulfonamides, griseofulvin, phenothiazines, tetracyclines, NSAIDs, chloroquine, thiazides, ACE inhibitors, antidepressants, cholesterol lowering drugs, amiodarone, and some chemotherapeutic drugs. Diagnosis is best confirmed by a photopatch test.
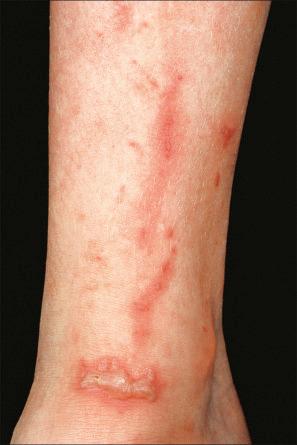
Phytophotodermatitis represents a phototoxic drug reaction due to contact with plants containing furanocoumarins. Patients develop erythema followed by postinflammatory hyperpigmentation. Rarely, blisters may develop ( Fig. 14.15 ). Members of the Umbelliferae, Rutaceae, and Moraceae families are implicated.
Photosensitization has been described as a process whereby ‘a reaction to nonionizing radiation occurs as a consequence of the introduction of a radiation-absorbing reagent (the sensitizer), which induces another substance (the substrate) to undergo chemical change’. There are two basic mechanisms: phototoxic and photoallergic. While an enormous range of drugs has been implicated in photosensitivity reactions, NSAIDs, phenothiazines, amiodarone, antibiotics, and antifungal agents such as griseofulvin appear to be of particular importance.
Two types of phototoxic reactions are recognized: photodynamic and nonphotodynamic.
Photodynamic reactions are oxygen-dependent and result in singlet oxygen or superoxide anions which cause injury to cellular constituents such as cell membranes, cytoplasmic proteins, and nucleic acids.
Nonphotodynamic reactions are oxygen independent and damage DNA or RNA directly.
Many drug reactions are photodynamic, whereas psoralen represents a nonphotodynamic reaction. Phototoxic reactions do not depend on prior exposure to the drug and will affect all patients of the same skin type provided that sufficient bound drug is available for reaction with the appropriate radiation. The action spectrum for phototoxicity is UVA, and less often UVB and visible light.
Photoallergic drug-induced photosensitivity is immunologically mediated and represents a delayed papular, vesicular, or eczematous response. It is a lymphocyte-mediated delayed hypersensitivity type IV reaction. It typically requires previous exposure to the drug or chemical. Only a proportion of patients taking the drug will develop a reaction. Photoallergic reactions are usually induced by UVA.
The histologic appearances of acute phototoxic reactions include conspicuous apoptotic keratinocytes (sunburn cells), which in severe cases may affect the entire epidermis, with variable neutrophil exocytosis, dermal edema, and a perivascular lymphohistiocytic infiltrate with small numbers of neutrophils and eosinophils ( Figs 14.16 and 14.17 ).
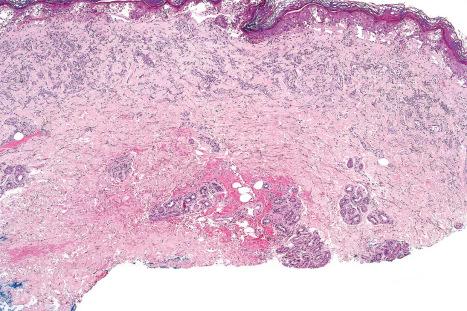
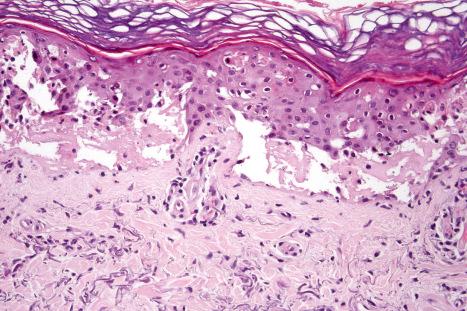
Chronic lesions are characterized by hyperkeratosis, hypergranulosis, variable acanthosis and epidermal atrophy, increased melanin pigmentation, melanocyte hyperplasia, and pigmentary incontinence. Elastosis and telangiectatic vessels may be conspicuous, and in severely affected patients stellate atypical myofibroblasts can be a feature. Epidermal disorganization and dyskeratosis may also be present.
The histologic appearances of drug-induced photoallergic reactions include spongiosis (often with vesiculation, lymphocytic, and eosinophil exocytosis), accompanied by papillary dermal edema and an upper dermal lymphohistiocytic infiltrate with variable numbers of eosinophils.
Drug reaction with eosinophilia and systemic symptoms (DRESS) was previously known as anticonvulsant hypersensitivity syndrome because of its close link with anticonvulsants. Cutaneous eruptions associated with anticonvulsants include erythematous maculopapular lesions, erythroderma, acneiform lesions, hypo- and hyperpigmentation, vasculitis, EM, and toxic epidermal necrolysis (TEN), and can affect up to 19% of patients on anticonvulsants, especially phenytoin. However, in addition to anticonvulsants, DRESS may be triggered by numerous types of medications including allopurinol, azathioprine, numerous antibiotics (especially dapsone, minocycline and sulfonamides), terbinafine, antivirals, antidepressants, antihypertensives, biologics, NSAIDs, and miscellaneous other drugs ( Table 14.1 ). DRESS is now the preferred terminology. Pseudolymphomatous drug reactions may also occur, and these are discussed later in the chapter.
| Drug category | Drug name |
|---|---|
| Anticonvulsant | Carbamazepine, lamotrigine, phenobarbital, phenytoin, valproic acid, and zonisamide |
| Antimicrobial | Ampicillin, cefotaxime, dapsone, ethambutol, isoniazid, linezolid, metronidazole, minocycline, pyrazinamide, quinine, rifampin, sulfasalazine, streptomycin, trimethoprim-sulfamethoxazole, and vancomycin |
| Antiviral Antidepressant Antihypertensive Biologic NSAID Miscellaneous |
Abacavir, nevirapine, and zalcitabine Bupropion and fluoxetine Amlodipine and captopril Efalizumab and imatinib Celecoxib and ibuprofen Allopurinol, epoetin alfa, mexiletine, and ranitidine |
DRESS is typically defined by a triad of pyrexia, exanthematous skin rash, and evidence of systemic involvement. Three different systems for diagnostic criteria have been proposed. In clinical practice, the criteria proposed by Bocquet et al may be the simplest to apply to ensure that the diagnosis is not missed. In this system, the diagnosis requires three criteria: cutaneous drug eruption, hematological abnormalities, and systemic involvement. The hematological abnormalities include eosinophilia ≥ 1.5 × 10 9 /L and atypical lymphocytes. Systemic involvement includes any of the following: lymphadenopathy, hepatitis with liver transaminases ≥ 2× normal, interstitial nephritis, interstitial pneumonitis, or carditis. Lymphadenopathy and hepatitis are, by far, the most common systemic manifestation. Liver involvement is seen in 70–95% of patients. DRESS typically starts 1–8 weeks after initiation of the drug. The syndrome is more common in black patients. There is no sex predilection. Children may be affected. Clinical features include pyrexia, a maculopapular or erythrodermatous eruption, facial or periorbital edema, strawberry tongue, tender lymphadenopathy, myositis, and hepatitis associated with leukocytosis and eosinophilia ( Figs 14.18 and 14.19 ). Less often, the cutaneous manifestations include localized or generalized follicular pustules, EM, and TEN. In patients with the pustular variant, lesions present on the scalp before becoming generalized. Conjunctivitis and/or pharyngitis may also be present. Renal, pulmonary (interstitial pneumonitis), and hematological (atypical lymphocytosis) involvement sometimes occur. Certain drugs are more commonly implicated in specific organ systems. Liver involvement is strongly associated with phenytoin, minocycline, and dapsone. Renal involvement is most commonly associated with allopurinol, carbamazepine, and dapsone. Minocycline is the most common triggering agent for pulmonary involvement, whereas ampicillin and minocycline are the most common drugs linked to cardiac involvement. The prognosis of this syndrome is variable, with most fatalities secondary to hepatic involvement. In patients with hepatitis, the mortality is approximately 10–20%.
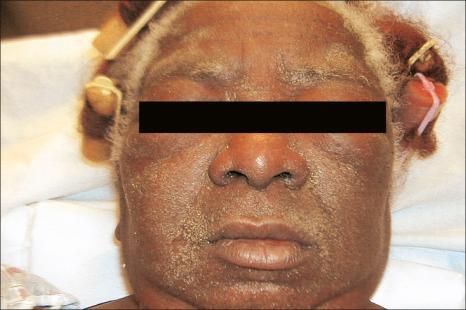
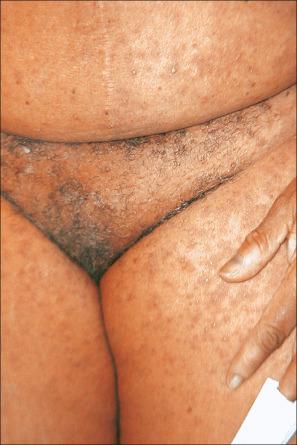
The precise etiology of this syndrome is uncertain but appears to be multifactorial. Patients with mutations in drug detoxification enzymes have an increased risk of DRESS. It is thought to result from an inability to detoxify arene oxide anticonvulsant metabolites due to absence, possibly genetically determined, of specific hydrolases. These genetic differences may help explain familial cases and the increased risk of DRESS in black patients. Immunology may also play a role. Immunosuppressed patients are at increased risk, possibly secondary to reactivation of herpesvirus infections, including reactivation of human herpes virus 6 and 7, Epstein-Barr virus (EBV), and cytomegalovirus. HHV-6 DNA and mRNA has been detected in the skin of patients with DRESS. Clinical manifestations of primary HHV-6 can closely mimic DRESS, suggesting that viral reactivation may play a significant role. Patients with DRESS have increased plasmacytoid dendritic cells (PDC) in the skin and decreased amounts in the circulation, possibly contributing to viral reactivation. Patients have increased levels of IL-5, contributing to eosinophilia. Therefore, it appears to be a complex interactions with biochemical, genetic, and immunological factors that contribute to DRESS.
The histologic features vary from spongiotic dermatitis to those of EM or TEN. Pustular lesions are characterized by a subcorneal pustule associated with follicular infundibular dilatation ( Figs 14.20 and 14.21 ).
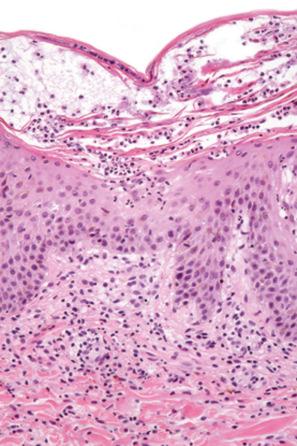
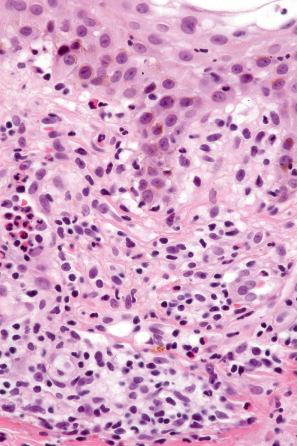
Lichenoid drug reactions account for approximately 5% of adverse drug reactions. Lichenoid drug reactions are clinically similar to lichen planus, although lesions are often larger. Wickham striae are usually not apparent. Mucosal involvement is rare and clinically and histologically resembles oral lichen planus. In contrast to lichen planus, where lesions are characteristically on the flexural surfaces of the forearms, the legs, and the genitalia, in lichenoid drug reactions the trunk and extremities are more often affected ( Figs 14.22 and 14.23 ). The eruption may sometimes be photodistributed and predominantly affect the hands and forearms, although other sun-exposed sites can be involved. The latent period between starting the drug and the onset of the eruption is often long, which can take months or even years. Atypical features including eczematous and psoriasiform lesions are sometimes seen, and bullous or ulcerative variants are occasionally encountered. Postinflammatory hyperpigmentation may be very marked and is often persistent. Scarring alopecia is sometimes present, and some patients may develop anhidrosis.
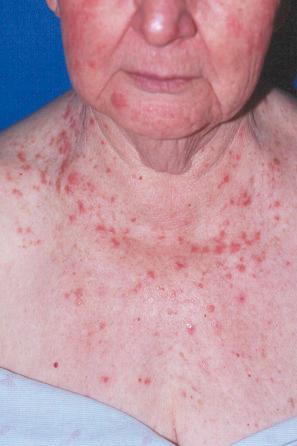
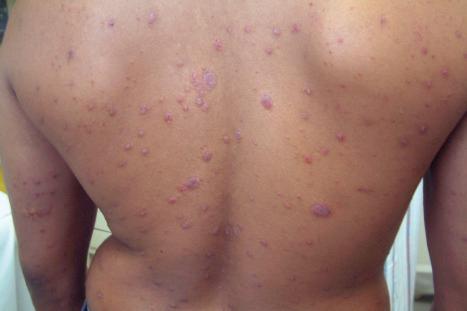
Although many drugs can cause a lichenoid reaction, those of particular importance include gold, antimalarials such as quinine and quinidine, penicillamine, captopril, various beta blockers (e.g., propranolol), lithium, thiazide diuretics, furosemide (frusemide), spironolactone, and ethambutol. More recently, the TNF-α inhibitors (infliximab, adalimumab, and etanercept), imatinib mesylate, and PD-1 and PD-L1 inhibitors have been associated with lichenoid eruptions. Lichenoid drug reactions have also been associated with hepatitis B, influenza, and human papilloma virus vaccinations.
Contact with p -phenylenediamine by workers engaged in the photographic color developing process and hairdressers may also result in a cutaneous lichenoid reaction. This is of two types:
Continuous exposure to small amounts results in the appearance of typical lichen planus.
If exposed to a single large dose, the features are those of a lichenoid contact dermatitis.
Other causes of a contact lichenoid eruption include dental restorative materials, musk ambrette, nickel, and gold.
Captopril and cinnarizine may cause lichen planus pemphigoides-like eruptions (see bullous drug reactions below).
The histologic features are frequently indistinguishable from typical lichen planus, although focal parakeratosis and spongiosis are sometimes present and interface change may be patchy ( Figs 14.24–14.26 ). The epidermis is often thinner and hypergranulosis less marked. Cytoid bodies may be found in the upper granular cell layer or even in the stratum corneum. Sometimes, eosinophils and occasionally plasma cells are found in the dermal infiltrate. Focal interruption of the granular cell layer, exocytosis of lymphoid cells into the upper epidermis, and a perivascular infiltrate in the deeper dermis are said to be additional helpful diagnostic pointers. Photodistributed lichenoid drug reactions are said to more closely resemble idiopathic lichen planus than nonphotodistributed variants. The changes that allow distinction from lichen planus are often seen in hypertrophic lichen planus. However, in lichenoid drug eruptions, prominent hyperplasia is hardly ever present. With some drug reactions, interface changes are present in the absence of a background of epidermal lichenoid features ( Figs 14.27 and 14.28 ). In lichenoid drug reactions secondary to anti-PD1 and anti-PD-L1 medications, spongiosis and epidermal necrosis are more prominent, and there are an increased number of histiocytes in the infiltrate.
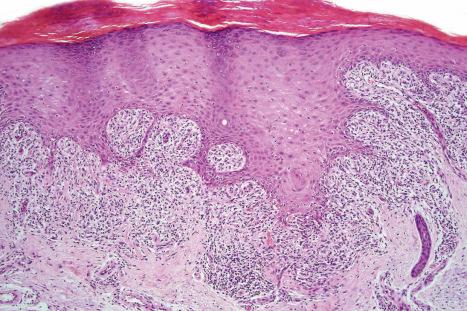
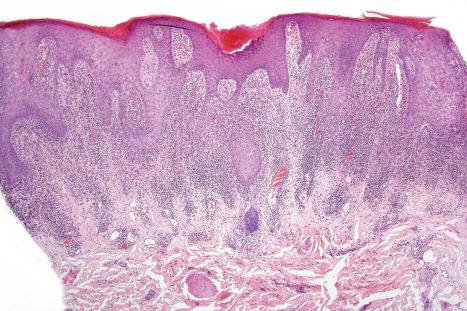
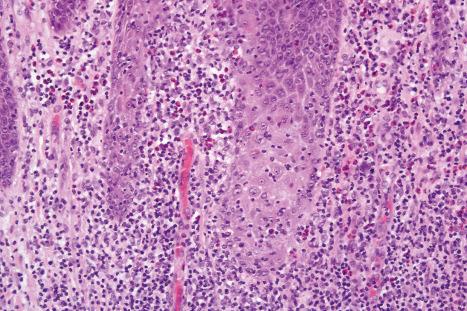
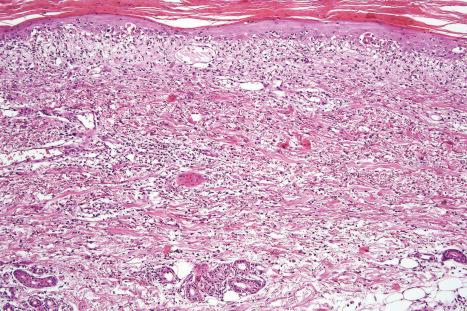
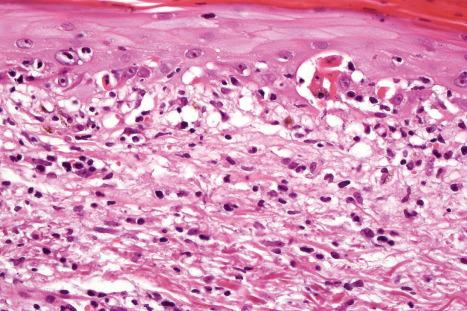
Fixed drug eruptions present as one or more circumscribed erythematous to violaceous or brown plaques that show a predilection for the extremities including the hands, feet, and external genitalia ( Figs 14.29 and 14.30 ). The mucous membranes may be affected, either alone or in association with cutaneous manifestations. Lesions – which may be pruritic or present with a burning sensation – typically recur at the same site on rechallenge with the offending drug. They usually develop within 30 minutes to 8 hours after taking the drug. Vesiculation and blistering are common. Resolution is typically marked by postinflammatory hyperpigmentation varying from brown to brown-violet or even black. In cases with multiple lesions, the multiple pigmented patches result in an appearance described as ‘Dalmatian dog’. Occasionally, the eruption is generalized and resembles TEN. Although the number of drugs that are capable of eliciting a fixed reaction is very large, those that are said to be more commonly incriminated include barbiturates, phenylbutazone, ibuprofen, acetyl salicylic acid, sulfonamides, trimethoprim-sulfamethoxazole, tetracyclines, dapsone, phenolphthalein, and quinine. In a large retrospective study from Singapore, the NSAID drug etoricoxib was responsible for close to 40% of fixed drug eruptions.
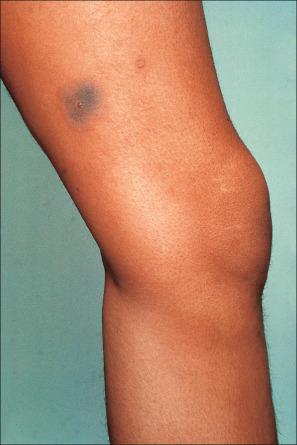
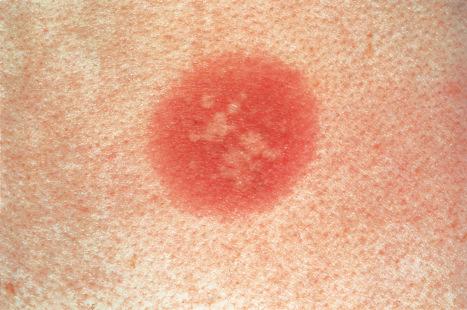
Fixed drug eruption is unique owing to the precise localization of the eruption and its recurrence at the same site on rechallenge. To understand this process, initial research was directed toward identifying the site of cutaneous memory. Autotransplantation experiments in which normal skin was grafted to a previously affected site and vice versa, followed by rechallenge with the causative drug, produced conflicting results. Some workers found that following challenge, grafted normal skin was unaffected, whereas transplanted previously affected skin developed erythema and became symptomatic. Others experienced quite the opposite results.
Immunofluorescence studies have been equally conflicting. While some authors have documented in vivo bound immunoglobulin and complement in the intercellular region of the epidermis or at its basement membrane, the majority of investigations have been negative.
It seems unlikely, therefore, that humoral immunity has a significant part to play in the pathogenesis of fixed drug eruption. Current research is directed toward understanding the role of cellular immunity. On initial exposure, the drug appears to bind to the epidermal keratinocytes (thereby functioning as a hapten) and is presented by Langerhans cells to lymphocytes within the dermis or in local lymph nodes. This stimulates an effector CD8+ lymphocyte population which, on returning to the epidermis, produces various cytokines including IFN-gamma (IFN-γ) and TNF-α, which result in epidermal necrosis. Keratinocyte death is believed to be mediated by both cytolytic pathways (e.g., perforin, granzyme A, and granzyme B) and FAS-mediated apoptosis. Resolution of the disease is thought to be due to recruitment of CD4+ T cells into the epidermis which suppress CD8+ T-cell activation and limit cell destruction, possibly via production of IL-10.
Although the precise mechanism by which memory in fixed drug eruption is achieved is incompletely understood, there is now considerable evidence to suggest that an intraepidermal effector-memory CD8+ T-cell population residing in the epidermis after the initial drug reaction is of particular importance. Such cells are defined immunohistochemically by expression of CD3, CD45RA, TCR-alpha beta, CD11a, and CD11b, and absence of CD27, CD28, and CD62L. It has been demonstrated that they remain in a state of activation (CD69+) in the unchallenged state and, following exposure to the drug, rapidly up-regulate IFN-γ expression and induce FAS and FAS-ligand expression, soon followed by epidermal necrosis. The localization may be related to site-specific, tissue resident T cells that remain after the eruption resolves and subsequently reactivate on re-exposure to the offending agent.
Histologically, the acute fixed drug eruption is characterized by marked basal cell hydropic degeneration, with lymphocyte tagging along the dermal–epidermal junction and individual keratinocyte necrosis ( Figs 14.31–14.33 ). Marked pigmentary incontinence is typical. Subepidermal vesiculation may be a feature of advanced lesions. Lymphocytes, histiocytes, and neutrophils are evident in the upper dermis. Eosinophils may sometimes be prominent. In late lesions, pigmentary incontinence may be the sole histologic finding.
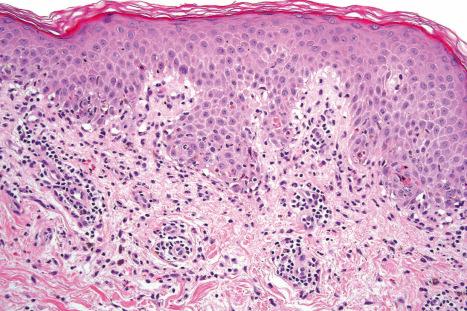
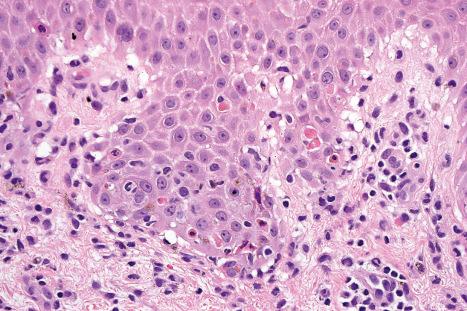
Although infectious agents (herpes simplex virus, Mycoplasma species) are the most common cause of EM, medications, or a combination of medications and viral infections, are implicated in a subset of patients. Drugs with the strongest association include antibiotics, anticonvulsants, and NSAIDs. Sulfonamides, specifically trimethoprim-sulfamethoxazole, carry the highest relative risk. Other causative antibiotics include aminopenicillins, quinolones, cephalosporins, and tetracyclines. The anticonvulsants associated with EM most often are phenobarbital, carbamazepine, phenytoin, and valproic acid. EM due to a combined viral infections and drug exposure has been described with cytomegalovirus and EBV. Despite the known associations, the underlying trigger may be unknown in more than half of patients affected.
In contrast to EM, Stevens-Johnson syndrome (SJS) and TEN are much more strongly associated with medication use. The profile of implicated drugs is similar to that seen in EM (see above). Studies on newer medications cite strong associations for nevirapine and lamotrigine, and weaker but significant associations for sertraline, pantoprazole, and tramadol. In a recent study, allopurinol was the most common trigger, accounting for 20% of cases over a 10-year period.
Cutaneous hyperpigmentation is a frequent complication of drug therapy. It may result from increased melanin synthesis or deposition of the drug or its metabolite within the skin. Heavy metals can also result in skin pigmentation. Most often, however, it results from postinflammatory hyperpigmentation.
Long-term treatment with minocycline might result in usually reversible (types I and II) cutaneous pigmentation. Three clinical variants of cutaneous minocycline pigmentation are generally recognized ( Figs 14.34–14.36 ):
Type I: blue-black macules localized to areas of scarring and inflammation (e.g., facial acne scars),
Type II (most common): blue-black, brown or slate-gray pigmentation on the shins, ankles, and arms,
Type III: generalized muddy-brown pigmentation which may be exacerbated on sunlight-exposed regions.
A fourth variant affecting the lips and possibly representing a fixed drug eruption has been described.
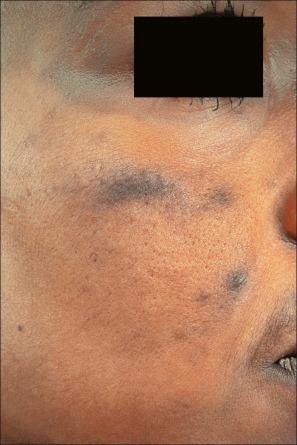
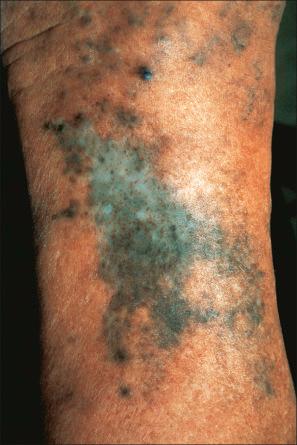
Localized minocycline pigmentation as a complication of cosmetic laser procedures have also been reported.
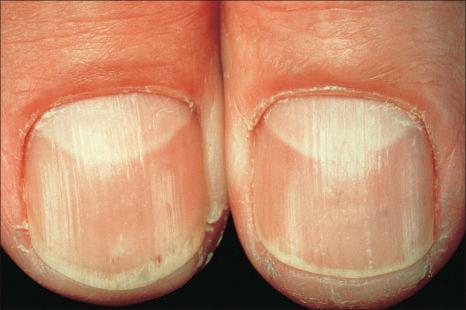
Nail pigmentation most often presents as a persistent slate-gray coloration of the proximal nail bed. Additional features include longitudinal melanonychia, diffuse nail pigmentation, and photo-onycholysis.
Minocycline may involve the teeth (causing a green-gray or blue-gray discoloration), predominantly affecting the middle and occasionally the incisal thirds of the crown. Lesions of the oral mucosa are rare although pigmentation has been described on the buccal mucosa, gingiva, tongue, and lips. The bones underlying the oral cavity (black bone disease) represent the single site most commonly affected by minocycline pigmentation. This is best visualized by inspecting the maxillary and mandibular anterior alveolar mucosa. The hard palate and lingual alveolar bone are also commonly affected.
The conjunctiva, sclera, thyroid (black thyroid), aorta, endocardium, and atherosclerotic plaques may also be involved in minocycline pigmentation. Minocycline pigmentation may also affect the vulvar/vaginal mucosa and may clinically mimic a melanocytic lesion.
Many other tetracyclines including methacycline and tetracycline hydrochloride have also been associated with cutaneous pigmentation.
Amiodarone, which is used primarily in the treatment of cardiac arrhythmias, is associated with a phototoxic/photosensitivity reaction in up to 50% of patients. In addition, cutaneous golden-brown to slate-gray or blue/violaceous pigmentation predominantly affecting the exposed surfaces including the face and the backs of the hands may develop, especially in those receiving high doses over a protracted period of time ( Fig. 14.37 ). Pigmentation is also sometimes seen in the sclera and cornea.
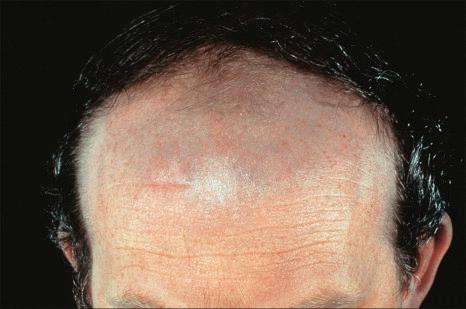
Antimalarials also result in abnormal skin pigmentation. Mepacrine (quinacrine) typically produces a yellow coloration although localized blue-black mucocutaneous lesions have been described ( Figs 14.38 and 14.39 ). Chloroquine and hydroxychloroquine cause yellow-brown to gray pigmentation. Sun-exposed skin is predominantly affected, although mucosal pigmentation may also occur.
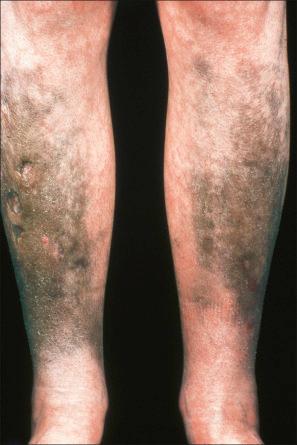
In addition to causing photosensitivity and contact dermatitis, chlorpromazine therapy (particularly when protracted and in high doses) can result in cutaneous pigmentation, especially on sun-exposed skin such as the face, dorsum of the hands, and the neck. Patients may present with a golden brown, tanned appearance while others develop a slate-gray, bluish, or purple appearance. The cornea and lens of the eye can also be involved.
Long-term treatment with imipramine may result in photodistributed hyperpigmentation affecting the face, neck, ‘V’ of chest, arms, and hands ( Fig. 14.40 ). The coloration varies from golden brown to blue-gray or slate-gray. The irises may also darken.
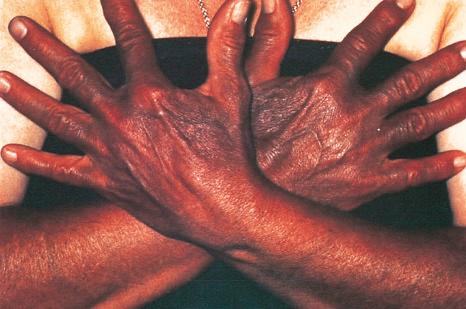
Photodistributed blue-gray pigmentation has been documented following treatment with desipramine.
Heavy metals including gold, silver, and mercury can all result in cutaneous pigmentation (see below).
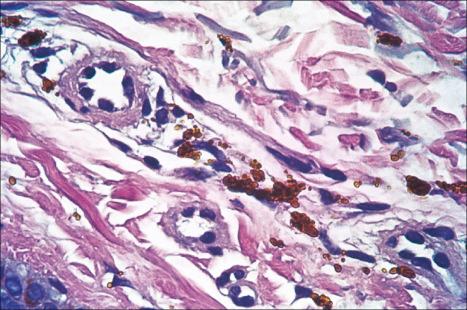
The histologic features of minocycline pigmentation are variable. In types I and II variants, golden brown to brown-black granules are found predominantly within macrophages distributed mainly around the vasculature and sweat gland coils ( Fig. 14.41 ). The pigment, which fluoresces yellow under ultraviolet light, stains positively with both Masson-Fontana and Perls Prussian blue reactions in type II variants ( Figs 14.42 and 14.43 ). The pigment is periodic acid-Schiff (PAS) negative. In contrast, in type I, the pigment only stains with Perls reaction. It is believed to represent minocycline or its breakdown product chelated with hemosiderin, ferritin, or iron. Calcium, sulfur, and chlorine are also present, but melanin is absent. Melanocytes and the epidermis show no increase in melanin pigmentation in types I and II variants. Type III hyperpigmentation is characterized by an increase in epidermal basal cell melanin pigmentation. The Perls stain is negative. Minocycline pigmentation of the subcutaneous fat has been described in the clinical setting of type II disease. Histologically, there is pigment within macrophages and giant cells in the subcutaneous fat, with positive staining for Masson-Fontana and variable staining with Perls reaction. One study also described green-gray nonrefractile globules within macrophages in the fat.
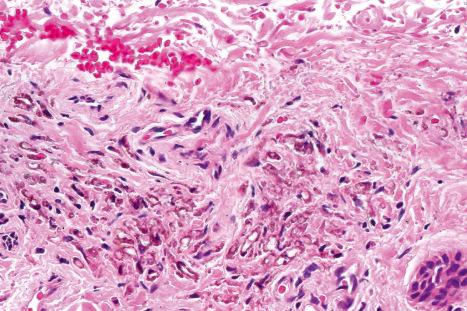
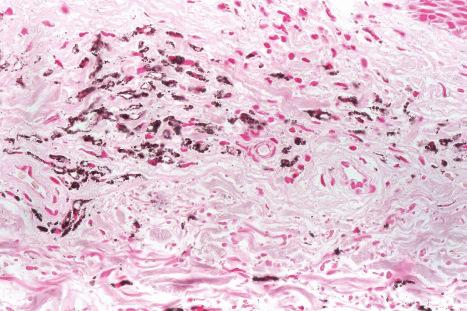
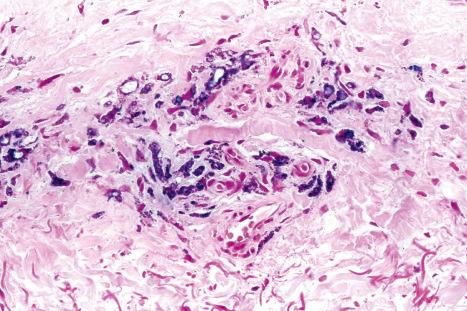
Histologically, amiodarone pigmentation is characterized by macrophages containing PAS-positive, yellow-brown lipofuscin-like granules located in a perivascular distribution ( Figs 14.44 and 14.45 ). Melanin pigmentation of the epidermis is not increased; indeed, its absence in involved skin has recently been documented. By electron microscopy, the granules are located within lysosomes. Lamellar myelin bodies may also be identified. Similar inclusions may be found in the hepatocytes, Kupffer cells, pulmonary macrophages, and neutrophils.
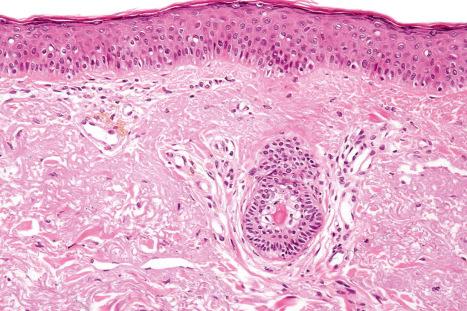
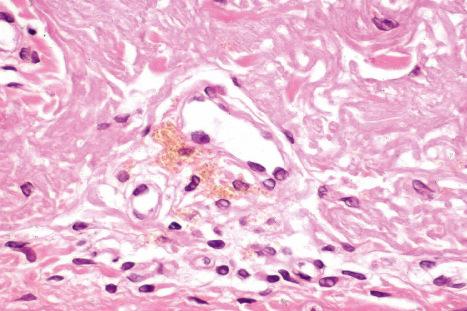
In mepacrine (quinacrine) pigmentation, yellow-brown pigment is found within the cytoplasm of histiocytes throughout the dermis. The pigment is weakly positive with the Perls Prussian blue reaction for iron and is Masson-Fontana negative. The histologic findings in hydroxychloroquine-related pigmentation have been described as yellow-brown granular deposits within macrophages and extracellularly. These granules are nonrefractile and stain positively with Masson-Fontana and are negative for iron.
Histologically, chlorpromazine hyperpigmentation is characterized by golden brown macrophage-bound granules surrounding the superficial vasculature. The granules are positive with the Masson-Fontana reaction but do not stain with Perls Prussian blue. Ultrastructurally, the pigment is lysosome bound and present in endothelial cells, fibroblasts, Schwann, and smooth muscle cells, in addition to macrophages. Increased melanin also contributes to the cutaneous pigmentation.
Histologically, imipramine and desipramine hyperpigmentation contain Masson-Fontana positive golden brown granules within the upper dermis, lying both free and within macrophages ( Fig. 14.46 ). Perls Prussian blue is negative. Ultrastructurally, histiocytes contain melanosomes in addition to lysosomal-bound electron-dense granules.
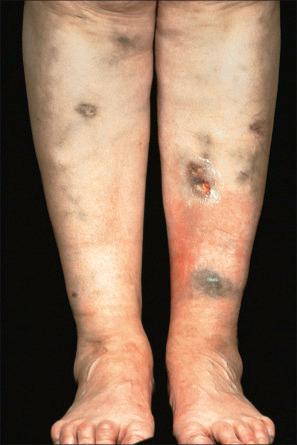
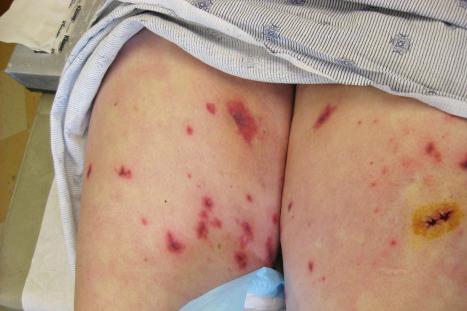
Adverse drug reactions are a common cause of vasculitis, accounting for up to 30% of vasculitides. In a study of hospitalized patients with adverse medication reactions, approximately 3% were vasculitis in nature. An immune complex mechanism mostly represents the pathogenesis in the majority of cases. A wide range of drugs has been implicated, including anti-infective agents, cancer chemotherapeutic agents and adjuvants, psychoactive and cardiovascular drugs, diuretics, anticoagulants, beta-adrenergic receptor agonists, and anticonvulsants. The more important agents include trimethoprim, penicillin, sulfonamides, NSAIDs, and aspirin. Other implicated drugs include cimetidine, clarithromycin, coumadin, furosemide (frusemide), hydralazine, ibuprofen, iodides, phenacetin, phenothiazines, procainamide, rifampin, propylthiouracil, and streptokinase ( Figs 14.47 and 14.48 ). Anti-TNF-α therapies have been linked to vasculitic reactions. Vasculitis has also been associated with ustekinumab. As discussed in the chapter on vasculitis, levamisole tainted cocaine is another cause of drug-induced vasculitis.
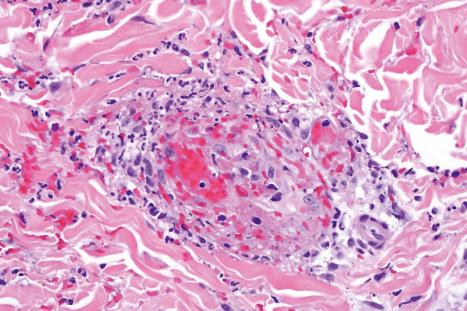
Granulomatous vasculitis has been described following treatment with chlorothiazide, allopurinol, phenytoin, and carbamazepine.
Purpura may be a manifestation of an adverse drug reaction. Causes include NSAIDs, diuretics, meprobamate, zomepirac sodium, ampicillin, pseudoephedrine, linezolid, and lidocaine/prilocaine cream. Purpuric eruptions at friction sites have been associated with angiotensin II receptor inhibitors.
The histologic features are those of red cell extravasation in the absence of changes of vasculitis ( Fig. 14.49 ).
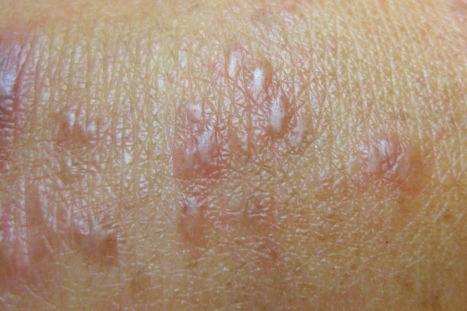
Interstitial granulomatous drug reactions are rare and have been associated with a number of drugs including calcium channel blockers, ACE inhibitors, beta blockers, lipid-lowering agents, diuretics, NSAIDs, antihistamines, anticonvulsants, and antidepressants. IL-1 inhibitor (anakinra), adalimumab, sorafenib, ganciclovir, thalidomide, desmopressin, allopurinol, febuxostat, and quetiapine have also been associated with an interstitial granulomatous reaction. This reaction pattern may also be related to a variety of systemic illnesses including rheumatoid arthritis, hepatobiliary disease, diabetes mellitus, Crohn disease, and chronic infections such as hepatitis C, herpes simplex/varicella-zoster, EBV, and HIV. Patients present with erythematous to violaceous, nonpruritic, irregular and sometimes annular papules or plaques predominantly affecting the inner arms, inner thighs, and the groins ( Fig. 14.50 ). Erythroderma in a case of anticonvulsant-induced hypersensitivity syndrome has also been described. A patient with an allopurinol triggered granulomatous drug eruption also had coexisting DRESS syndrome.
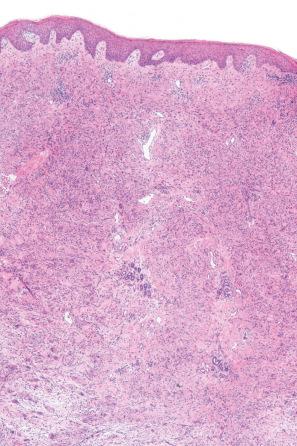
Histologically, the eruption is characterized by an interstitial infiltrate of lymphocytes, histiocytes, eosinophils, plasma cells, and multinucleate giant cells, sometimes associated with increased dermal mucin and showing more than a superficial resemblance to interstitial granuloma annulare ( Figs 14.51 and 14.52 ). Fragmentation of collagen fibers and elastic tissue is commonly evident and phagocytosis of connective tissue debris by giant cells is typically seen ( Figs 14.53 and 14.54 ). Discrete granulomata may also be identified and granulomatous vasculitis has been documented. Flame figures and Churg-Strauss-like granulomata have also been described. Atypical lymphocytes with hyperchromatic, irregular, and variably enlarged nuclei showing epidermotropism are present in up to 50% of cases. In rare cases, the granulomatous drug eruption can have epidermotropism and positive clonality mimicking granulomatous mycosis fungoides. The changes of interface dermatitis, sometimes with an associated lichenoid infiltrate, are found in the majority of cases.
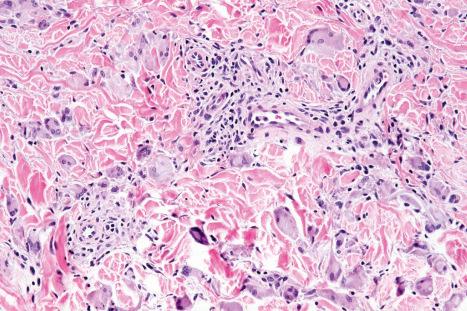
Interstitial granulomatous drug reactions must be distinguished from granuloma annulare and systemic disease-associated lesions as described above. Granuloma annulare is not usually associated with interface dermatitis, and necrobiosis and mucin deposition are not typical features of granulomatous drug reactions. Systemic disease-associated granulomatous dermatitis is usually associated with vasculitic and/or thrombotic phenomena. In cases where significant lymphoid atypia is present, cutaneous T-cell lymphoma enters the differential diagnosis. When elastophagocytosis is marked, granulomatous slack skin or an elastolytic granuloma may be important diagnostic considerations. Correlation with clinical history and resolution with withdrawal of the triggering agent is critical in confirming the diagnosis.
This topic is discussed in the chapter on panniculitis.
Drug-induced alopecia is usually reversible, predominantly nonscarring, and affects females more often than males.
Anagen effluvium, in which hair growth stops due to cessation of mitotic activity, is a common feature of anticancer therapy and often develops days or a few weeks after starting the drug. The scalp and beard areas, which contain a high percentage of anagen follicles, are particularly affected. It especially complicates combination chemotherapy and is likely to be severe. Although all anticancer drugs may be associated with some degree of alopecia, particular offenders include bleomycin, cyclophosphamide, dactinomycin, daunorubicin, doxorubicin, 5-fluorouracil, ifosfamide, and vindesine. There is some variation in drug effect. Some cause alopecia in all individuals whereas others affect only a minority of patients. In addition to traditional chemotherapeutics, targeted chemotherapeutics, especially vismodegib, sorafenib, and vemurafenib, are associated with alopecia. TNF-α antagonists and endocrine-based chemotherapies are also associated with alopecia.
Telogen effluvium, in which hairs are transformed into the telogen phase, develops several months after commencing therapy. Anticoagulants, including heparin and warfarin and dextran sulfate, cause telogen effluvium in up to 50% of patients. Other causes include antithyroid drugs such as iodine, thiouracil and carbimazole, oral contraceptives, lithium, IFNs, retinoids, and pramipexole.
Alopecia areata may develop as a consequence of TNF-α inhibitors such as adalimumab, etanercept, and infliximab. Case reports of alopecia areata have also been reported secondary to acitretin, vandetanib, and ustekinumab.
Drug-induced systemic lupus erythematosus was first described as a complication of hydralazine. It has also been reported in association with procainamide, quinidine, sulfasalazine, chlorpromazine, penicillamine, methyldopa, carbamazepine, acebutalol, isoniazid, captopril, propylthiouracil, and minocycline. Therapy with TNF-α inhibitors has been associated with a lupus-like syndrome. Imiquimod can also induce a lupus-like reaction that histologically and clinically resemble subacute cutaneous lupus erythematosus. With the exception of hydralazine and procainamide (high risk) and quinidine (medium risk), the other associations are low risk. Laboratory investigations reveal anti-Ro, anti-La, and antinuclear antibodies (ANAs) in the majority of cases. Subacute cutaneous lupus erythematosus-like features have been described following treatment with terbinafine. Localized cutaneous lupus lesions occurring at the site of IFN injection have been reported. Cutaneous lesions including malar erythema, discoid lesions, photosensitivity, oral ulceration, and alopecia are rare. Anti-TNF-α therapy-induced lupus may result in a higher incidence of cutaneous lesions, hypocomplementemia, and positive serology for double-stranded DNA as compared to other medications. Symptoms typically develop months to years after initiating therapy. Diagnostic criteria have been defined as follows :
continuous treatment with the suspected drug for 1 month or longer,
common presenting symptoms include arthralgias/arthritis, myositis, serositis, malaise and fever,
antihistone antibodies frequent, particularly IgG anti-([H2A-H2B]-DNA),
most importantly, clinical improvement within days or weeks after stopping the suspected drug.
The histologic features of cutaneous lesions are indistinguishable from those seen in the idiopathic forms.
Blisters may develop within the setting of an adverse drug reaction either as a consequence of severe spongiosis or marked interface change or they may reflect drug-related autoimmune bullous disorders ( Fig. 14.55 ). In this section, only the last are discussed. These include drug-induced linear IgA disease, bullous pemphigoid, epidermolysis bullosa acquisita, pemphigus variants, and drug-induced pseudoporphyria. Many alleged drug reactions are single case reports, particularly in patients taking multiple medications. It is often difficult to determine which associations are therefore coincidental and which are genuine. In occasional reports, recrudescence following re-exposure to the offending agent has been documented.
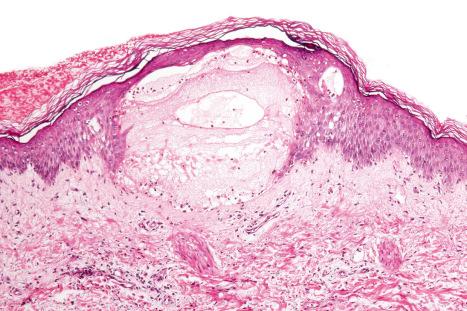
The precise mechanism of drug-induced blistering is unknown, although multiple factors have been suggested :
Direct toxicity to basement membrane constituents or intercellular junctions with resultant autoantibody production.
The drug may function as a hapten.
The drug may be antigenically similar to a basement membrane or intercellular junction constituent.
Perturbation of the immune system with inappropriate production of antibasement membrane antibodies.
Drug-induced abnormality of cell membrane calcium metabolism.
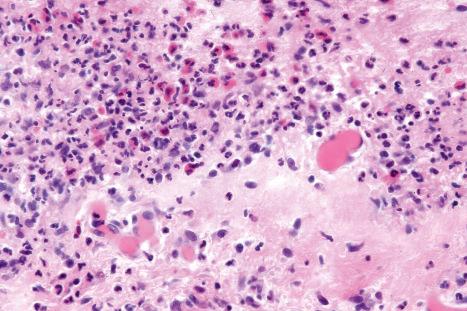
Drug-induced linear IgA disease is most often associated with intravenous therapy with vancomycin. Similar eruptions, however, have also been described following treatment with trimethoprim-sulfamethoxazole, penicillin, phenytoin, somatostatin, lithium, amiodarone, captopril, cefamandole, ceftriaxone, ciclosporine, IL-2, IFN-alpha, penicillin, amoxicillin, vigabatrin, diclophenac, ketoprofen, buprenorphine, infliximab, piperacillin-tazobactam, ustekinumab, and following vaccination for human papilloma virus. Cutaneous manifestations of vancomycin-induced linear IgA disease include pruritic, erythematous, urticarial, targetoid, and bullous lesions with a predilection for the trunk, extremities, palms, and soles ( Figs 14.56 and 14.57 ). Morbilliform lesions have also been described. Mucosal involvement is present in up to 40% of cases. As such, there can be considerable clinical overlap with EM and TEN. Laryngeal involvement has been documented. Drug-induced linear IgA disease tends to be more severe than spontaneous forms.
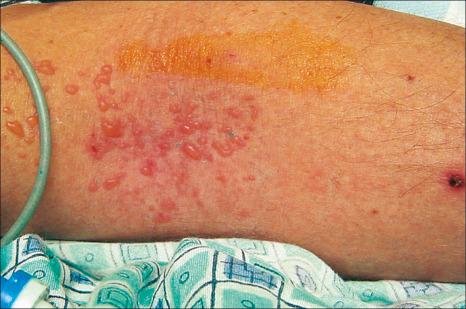
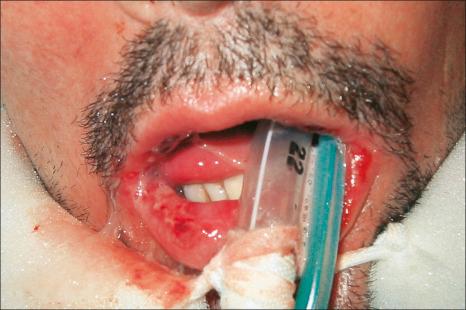
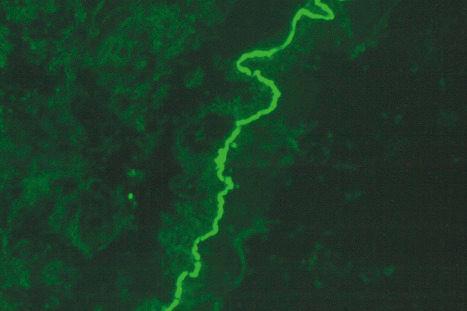
By definition, linear IgA deposition along the basement membrane region is present in all cases ( Fig. 14.58 ). C3 is seen in approximately 20% of cases. Linear IgG may also very exceptionally be present, although distinction from drug-induced bullous pemphigoid can then be problematical. Such cases may, in fact, represent examples of drug-induced IgA-mediated bullous pemphigoid. Circulating IgA antibasement membrane zone antibodies are found in 25% of cases. Split skin studies predominantly localize to the floor of the blister cavity. By immunoelectron microscopy, the results are heterogeneous, IgA having been detected within the lamina lucida, lamina densa, and in the sublamina densa. Western immunoblotting has detected a number of antigens including a 230-kD antigen (bullous pemphigoid antigen 1), a 97-kD antigen (an anchoring filament protein), and also a 250-kD antigen corresponding to type VII collagen.
Histologically, a neutrophil-rich subepidermal blister is seen in the majority of cases, but sometimes eosinophils are conspicuous ( Figs 14.59–14.61 ).
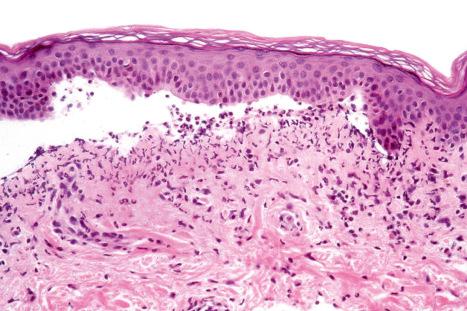
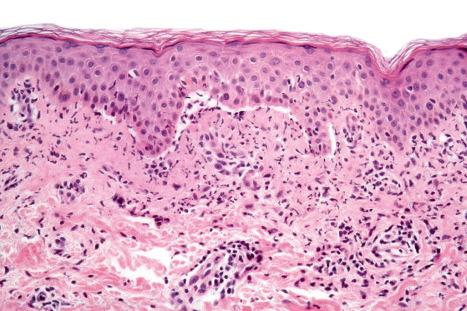
A variety of drugs including captopril, ciprofloxacin, chloroquine, furosemide (frusemide), ibuprofen, mefenamic acid, nifedipine, penicillamine, penicillins, phenacetin, sulfasalazine, spironolactone, enoxaparin, dipeptidyl peptidase IV inhibitors, metronidazole, mammalian target of rapamycin (mTOR) inhibitors, and levofloxacin have been incriminated in alleged drug-induced bullous and mucosal pemphigoid. Of these, antirheumatics, cardiovascular drugs, and antimicrobial drugs are the most important. Penicillamine is among the most commonly incriminated (mucosal more than bullous), usually in rheumatoid arthritis patients. Furosemide (frusemide) is believed to be an important cause of drug-induced bullous pemphigoid, although recently this has been challenged, with the author suggesting that diagnoses of pseudoporphyria or epidermolysis bullosa acquisita may be more appropriate. The ACE inhibitors, captopril and enalapril, have both been associated with immunologically proven bullous pemphigoid. The penicillins including amoxicillin and procaine penicillin G are the most frequently implicated antibiotics.
Clinically, drug-induced bullous pemphigoid is similar to idiopathic disease although the lesions are often polymorphic, mimicking other drug-induced bullous dermatoses such as EM, eczematous dermatitis, and porphyria cutanea tarda. In drug-induced disease, mucous membranes are often involved, thereby blurring the distinction between bullous and mucosal variants of pemphigoid. In some patients, there appears to be overlap between bullous pemphigoid and pemphigus vulgaris.
Drug-induced variants are characterized by linear IgG and C3 along the basement membrane region on direct immunofluorescence. By indirect immunofluorescence, the antibodies bind to the epidermal side (roof) of split skin. Western immunoblotting has demonstrated that the antibodies react with both the 230- and 180-kD bullous pemphigoid antigens.
Histologically, drug-induced variants are similar to typical bullous pemphigoid, being characterized by an eosinophil-rich subepidermal blister.
Drug-induced epidermolysis bullosa acquisita has been described following treatment with granulocyte-macrophage colony-stimulating factor (GM-CSF) and in a patient receiving vancomycin and gentamicin therapy. In both patients, the blisters were subepidermal and eosinophil rich; direct immunofluorescence disclosed linear IgA and IgG deposits at the basement membrane region. Split skin indirect immunofluorescence in the former patient labeled the floor of the blister cavity and by immunoelectron microscopy the deposits were localized to the lamina densa and the sublamina densa region. In the latter patient, IgG antibodies against type VII collagen were recognized by an enzyme-linked immunosorbent assay (ELISA). An epidermolysis bullosa acquisita-like reaction has also been described after treatment with penicillamine. Histologic features included a pauci-inflammatory subepidermal blister. Linear deposition of C3, IgG, and IgM are seen on direct immunofluorescence, and IgG labels to the base of the blister on salt-spilt direct immunofluorescence.
A significant number of cases of vancomycin-induced linear IgA disease are characterized by antibodies which label the floor of split skin on indirect immunofluorescence. Many of these might represent examples of drug-induced epidermolysis bullosa acquisita. An epidermolysis bullosa acquisita-like blistering dermatosis has been described with penicillamine therapy, but this has not been confirmed with immunofluorescent or molecular data.
Pemphigus may be related to a wide range of drugs, either directly as a causative factor or indirectly as a precipitating or triggering factor. The range of drugs is quite wide but many belong to the thiol group of compounds (characterized by the presence of an -SH group) including penicillamine, captopril, bucillamine, and thiopronine. Thiol-induced acantholysis is mediated by both immune and direct biochemical mechanisms. Penicillamine most often induces pemphigus in the setting of rheumatoid arthritis. Although foliaceus is most commonly encountered, vulgaris, erythematosus, and herpetiform variants may occur. Other drugs that contain sulfur, which can also form -SH groups, include gold compounds, penicillins, rifampicin, and cephalosporins. Topical application of imiquimod has been reported to cause pemphigus foliaceus and vegetans. Additionally, both pemphigus foliaceus and vulgaris lesions have been described as a consequence of radiotherapy. Clinically, drug-induced pemphigus can resemble vulgaris, foliaceus, erythematosus, and vegetans variants, the first being most often encountered. In the older literature, foliaceus variants were typical but with a change in prescribing habits to non-thiol-related drugs, vulgaris-type cases are more frequently seen. In most cases of imiquimod-induced pemphigus foliaceus, disease is localized to the sites of application, although there is one report of both localized and distant lesions. In the setting of radiotherapy, all patients developed pemphigus at the site of radiation. One patient also had distant lesions.
Histologically, drug-induced and idiopathic variants are indistinguishable. Intercellular IgG and circulating antibodies are variable in drug-induced pemphigus, although in a series of 10 patients all had positive direct immunofluorescence and 80% had circulating antibodies. Such antibodies may recognize desmoglein 3 and/or 1.
Pseudoporphyria is a rare blistering disease that clinically and histologically mimics porphyria but which develops in the setting of normal porphyrin metabolism ( Fig. 14.62 ). There are many causes including drugs, chronic renal failure usually in the setting of dialysis, excessive sun exposure, and UVA. It has also been described following excessive use of sunbeds. The most common medications include diuretics such as furosemide (frusemide) and NSAIDs, particularly naproxen. Other drugs include isotretinoin and the oral contraceptive. More recently imatinib, sunitinib, voriconazole, and diclofenac have been implicated.
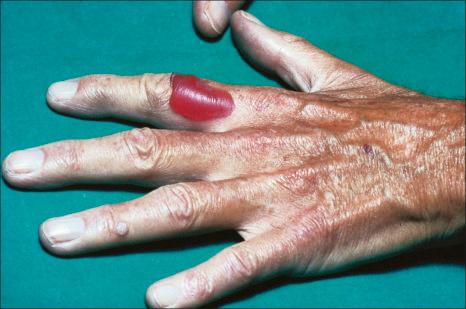
The pathogenesis of pseudoporphyria is unknown, but it may (at least in some patients) represent a phototoxic photosensitivity reaction.
Histologically, pseudoporphyria is identical to porphyria cutanea tarda. Typical lesions are characterized by a subepidermal cell-free blister, typically with preservation of the dermal papillae (festooning) and thickening of the vessel walls in the superficial dermis.
By immunofluorescence, immunoglobulin (most commonly IgG) is present at the dermal–epidermal junction and also outlining the superficial dermal vasculature.
Psoriasis and psoriasiform dermatoses can be caused by a number of drugs including lithium, beta blockers, NSAIDs, synthetic antimalarials, and tetracycline. Drug-induced disease may have variable presentations including :
exacerbation of pre-existing psoriasis,
induction of new lesions in uninvolved psoriatic skin,
precipitation of psoriasis de novo,
resistance to treatment.
Lithium-induced psoriasis varies from exacerbation of pre-existing psoriasis to development of new disease. Manifestations vary from plaque disease to generalized pustular psoriasis, palmoplantar pustulosis, scalp psoriasis, and psoriatic erythroderma. Latency varies from 1 week to years or more.
Beta blockers (e.g., propranolol, oxprenolol, pindolol, alprenolol, and the now discontinued practolol), antimalarials (e.g., chloroquine and hydroxychloroquine) and NSAIDs (e.g., indomethacin, phenylbutazone, oxyphenylbutazone, and ibuprofen) can also induce psoriasiform eruptions or result in exacerbations and flares.
The increasing use of TNF-α inhibitors is associated with new-onset or exacerbated psoriasiform eruptions. TNF-α inhibitors-induced psoriasis may be seen in approximately 1% of patients on the medication. Most patients develop palmoplantar pustulosis or plaque-type psoriasis. The average time to onset of lesions is 6–10 months, although the range is variable. It is speculated that inhibition of TNF-α increases levels of type 1 IFNs which result in psoriasiform lesions.
A large number of other drugs have been linked with exacerbation of psoriasis or development of new disease. These include the antifungal agent terbinafine, antibiotics such as penicillin and tetracycline, digoxin, amiodarone, IFN-α, recombinant IFN-α, erlotinib, and imiquimod.
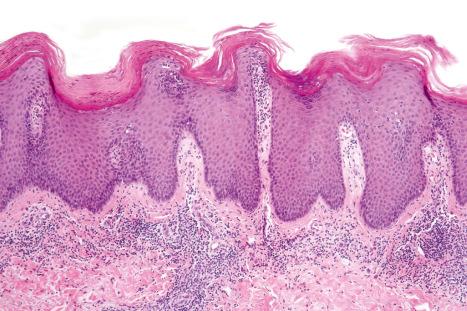
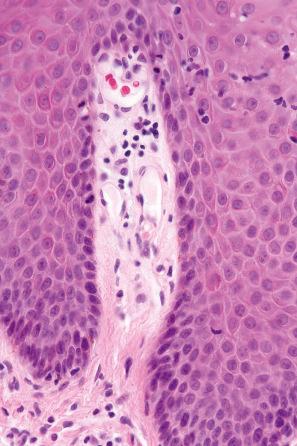
The histologic features overlap lichen simplex chronicus and psoriasis. Occasionally, they are indistinguishable from psoriasis vulgaris ( Figs 14.63–14.65 ) or pustular psoriasis. Psoriasis secondary to TNF-α inhibitors may also demonstrate lichenoid inflammation and spongiosis.
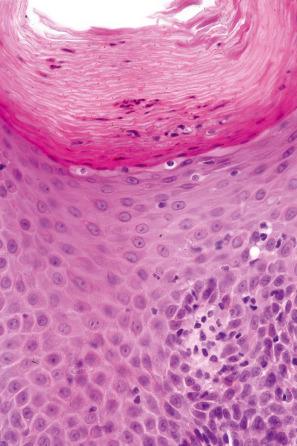
Pityriasiform drug reactions may be particularly caused by ACE inhibitors and gold. Less often, terbinafine, omeprazole, benfluorex, arsenicals, bismuth compounds, isotretinoin, naproxen, acetaminophen, barbiturates, and bacille Calmette-Guérin (BCG) therapy have been implicated. The tyrosine kinase inhibitors imatinib and ponatinib have been associated with pityriasiform dermatitis. Patients present with small erythematous lesions accompanied by a peripheral scale, which may follow Langer lines (cleavage lines), giving rise to the typical ‘fir tree’ appearance. The trunk and extremities are predominantly affected.
Histologically, pityriasiform drug reactions are typically characterized by patchy parakeratosis, focal spongiosis with lymphocytic exocytosis, and a superficial perivascular lymphocytic infiltrate, sometimes associated with red cell extravasation ( Fig. 14.66 ) On occasions, clinically typical pityriasiform drug eruptions may demonstrate a more psoriasiform histology.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here