Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Maternal transmission of Toxoplasma gondii in the first trimester causes the greatest damage to the fetus, while infections later in pregnancy are more readily transmissible to the fetus.
No neonate should be discharged from a birth hospital without documentation of maternal syphilis testing. If maternal syphilis testing is positive, adequacy and timing of maternal treatment, comparison of infant and maternal serology, and infant’s examination must be taken into consideration.
Symptoms of congenital tuberculosis are protean but should be considered in any ill neonate whose mother is from a TB-endemic area.
Symptoms of congenital malaria are nonspecific, though fever is universal. Parasitemia with Plasmodium falciparum , Plasmodium vivax , Plasmodium ovale , or Plasmodium malariae has been reported with congenital malaria and should be considered in any infant whose mother is from a malaria-endemic region.
Toxoplasmosis in the fetus and newborn is due to maternal infection with the parasite Toxoplasma gondii . This obligate protozoan parasite is ubiquitous in nature. While the cat is the definitive host, T. gondii can infect most warm-blooded animals. Human seroprevalence varies both geographically and by socioeconomic status. In the United States, seroprevalence studies obtained from National Health and Nutrition Examination Survey (NHANES) in 2009–2010 show a continuous decrease in seroprevalence, being around 9% for women of childbearing age compared to prior surveys conducted in 1988–1994 and 1999–2004. Accordingly, over 90% of women of childbearing age in the U.S. are susceptible to primary infection with T. gondii during pregnancy, with risk of transmission to the fetus. Seroprevalence is increased for those born outside the United States, living below the poverty level, and with a lower level of education. Global data on seroprevalence show the highest rates in Latin America, parts of Eastern/Central Europe, the Middle East, parts of Southeast Asia, and Africa.
However, seroprevalence rates differ considerably from one country to another, from one region of a country to another, and even from one ethnic group to another in the same region. These widely disparate seroprevalence rates among different adult populations throughout the world have been explained by differences in eating and sanitation practices that contribute to acquisition of infection. Eating goods that are undercooked, raw, cured, dried, as well as meat, raw oysters, clams/mussels, or unwashed raw fruits and vegetables, and drinking unpasteurized goat’s milk, working with meat, having three or more kittens, and even certain climactic conditions have been associated with higher risks of infection. Waterborne transmission, particularly from untreated well water, has been noted.
Older studies through immunoglobulin (Ig) M screening of newborn blood specimens collected on filter paper showed the prevalence of congenital infection in Massachusetts and New Hampshire to be 0.08 per 1000 births. Higher rates are found in other countries, ranging from 1 to 20 per 10,000 births, but are difficult to compare due to different methodology. Few countries have routine screening of pregnant women. In Massachusetts, a case-control study involving 14 years of newborn screening for congenital toxoplasmosis found that the mother’s birth outside the United States, particularly in Cambodia and Laos, as well as the mother’s educational level and higher gravidity, were strongly predictive of congenital infection. With approximately 4 million live births annually in the United States, there are an estimated minimum 400 babies born each year with congenital toxoplasmosis, although the true incidence is likely much higher as the majority of infected newborns are asymptomatic at birth and therefore not diagnosed with maternal newborn screening.
T. gondii exists in three forms:
Sporozoite contained within oocysts that are in the cat’s intestinal tract and are shed in cat feces
A tachyzoite or endozoite that is the proliferative form and was formerly referred to as a trophozoite
A tissue cyst that has an intracystic form termed cystozoite or bradyzoite
Nonfeline mammals or birds ingest infective oocysts from contaminated soil. Tissue cysts then accumulate in the organs and skeletal muscle of these animals. The possible routes of transmission from animal to human are direct contact with infected cat feces, ingestion of undercooked meat containing infective cysts, raw shellfish, and ingestion of fruits, vegetables, or water that have been in contaminated soil. Rarer methods of transmission can be from infected blood transfusions or organ transplantation. Congenital infection results from placental infection and subsequent hematogenous spread to the fetus.
Infection of the fetus occurs because of maternal primary infection during pregnancy or, rarely, just before conception. Reactivation of latent T. gondii infection during pregnancy does not lead to fetal infection, except among immunocompromised women such as those infected with human immunodeficiency virus (HIV) or those receiving chemotherapy or other immunosuppressive therapy as with systemic lupus erythematosis. Even under those circumstances, the risk is low. In addition, maternal reinfection with a new, more virulent strain can result in congenital toxoplasmosis. Severity of disease is related to both host and parasite factors as well as stage of pregnancy.
Infection of the fetus occurs transplacentally during maternal parasitemia. Placental infection is an important intermediary step, and up to 16 weeks may elapse between placental infection and subsequent infection of the fetus. This time delay has been termed the prenatal incubation period and explains the success of therapeutic intervention during pregnancy. Infections in twins show similar clinical manifestations in monozygotic twins, whereas discrepancies in clinical findings are common in dizygotic twins.
Overall, approximately 40% of infants born to mothers who acquired toxoplasmosis during pregnancy are congenitally infected with T. gondii . The rate of vertical transmission varies according to the trimester in which the mother became infected, with fetal infection rates increasing as pregnancy advances. Only 15% of infants are infected with maternal infection occurring in the first trimester, whereas transmission rates are 30% and 60% with maternal infection in the second and third trimesters, respectively. The severity of clinical manifestations is greatest, however, when maternal infection is acquired early in pregnancy. Maternal infection in the first trimester results in severe disease in as many as 40% of infected fetuses, and in stillbirth or perinatal death in an additional 35% of infants. Conversely, maternal infection in the third trimester is rarely if ever associated with severe fetal disease or stillbirth, and approximately 90% of infants in such situations have subclinical infection.
Postnatally, transmission of T. gondii can occur from transfusion of blood or blood products or from transplantation of organ or bone marrow from a seropositive donor with latent infection. Although the organism has been detected in human milk, transmission by breastfeeding has not been documented.
Most newborns with congenital toxoplasmosis are asymptomatic with clinically apparent disease only present in approximately 10% to 25% of infected infants, although thorough evaluation may demonstrate eye or neurologic abnormalities in approximately 20% of cases. The clinical manifestations of toxoplasmosis are often indistinguishable from those seen with other congenital infections, such as cytomegalovirus or congenital syphilis. Approximately one-third of infants have a generalized form of toxoplasmosis that principally involves organs of the reticuloendothelial system. The abnormalities include temperature instability, hepatosplenomegaly, jaundice, pneumonitis, generalized lymphadenopathy, rash, chorioretinitis, anemia, thrombocytopenia, eosinophilia, and abnormal cerebrospinal fluid (CSF) indices ( Table 35.1 ). The other two-thirds of infected infants principally manifest neurologic disease.
| Finding | INFANTS WITH FINDINGS (%) | |
|---|---|---|
| Neurologic Disease ⁎ (108 Cases) | Generalized Disease † (44 Cases) | |
| Chorioretinitis | 94 | 66 |
| Abnormal cerebrospinal fluid | 55 | 84 |
| Anemia | 51 | 77 |
| Convulsions | 50 | 18 |
| Intracranial calcification | 50 | 4 |
| Jaundice | 29 | 80 |
| Hydrocephalus | 28 | 0 |
| Fever | 25 | 77 |
| Splenomegaly | 21 | 90 |
| Lymphadenopathy | 17 | 68 |
| Hepatomegaly | 17 | 77 |
| Vomiting | 16 | 48 |
| Microcephaly | 13 | 0 |
| Diarrhea | 6 | 25 |
| Cataracts | 5 | 0 |
| Eosinophilia | 4 | 18 |
| Abnormal bleeding | 3 | 18 |
| Hypothermia | 2 | 20 |
| Glaucoma | 2 | 0 |
| Optic atrophy | 2 | 0 |
| Microphthalmia | 2 | 0 |
| Rash | 1 | 25 |
| Pneumonitis | 0 | 41 |
* Infants with otherwise undiagnosed central nervous system diseases in the first year of life.
† Infants with otherwise undiagnosed nonneurologic diseases during the first 2 months of life.
Central nervous system involvement is the hallmark of congenital T. gondii infection. Chorioretinitis, intracranial calcifications ( Fig. 35.1 ), and hydrocephalus are the most characteristic findings, occurring in approximately 86%, 37%, and 20% of symptomatic infants, respectively. This constellation of findings has been referred to as the classic triad of congenital toxoplasmosis; its presence should alert the clinician to the diagnosis. Intracranial calcifications may be single or multiple, but typically are generalized and located in the caudate nucleus, choroid plexus, meninges, and subependyma; they also may occur periventricularly, as in cytomegalovirus infection. Calcifications are visualized best by computed tomography (CT) but are often detected on ultrasonography as well. Intracranial calcifications may resolve with appropriate antimicrobial therapy. Hydrocephalus may be the only manifestation of disease; it results from the extensive periaqueductal and periventricular vasculitis with necrosis that causes obstruction of the ventricular system. Ventriculoperitoneal shunting is often required. Abnormalities of the CSF are common; characteristically, they consist of lymphocytic pleocytosis and a markedly elevated protein content. Microcephaly, when present, indicates severe brain injury. Hypothermia and hyperthermia may occur secondary to hypothalamic involvement. T. gondii has been detected in the inner ear and mastoid, with the associated inflammation resulting in deafness. An ascending flaccid paralysis with myelitis has also been reported.
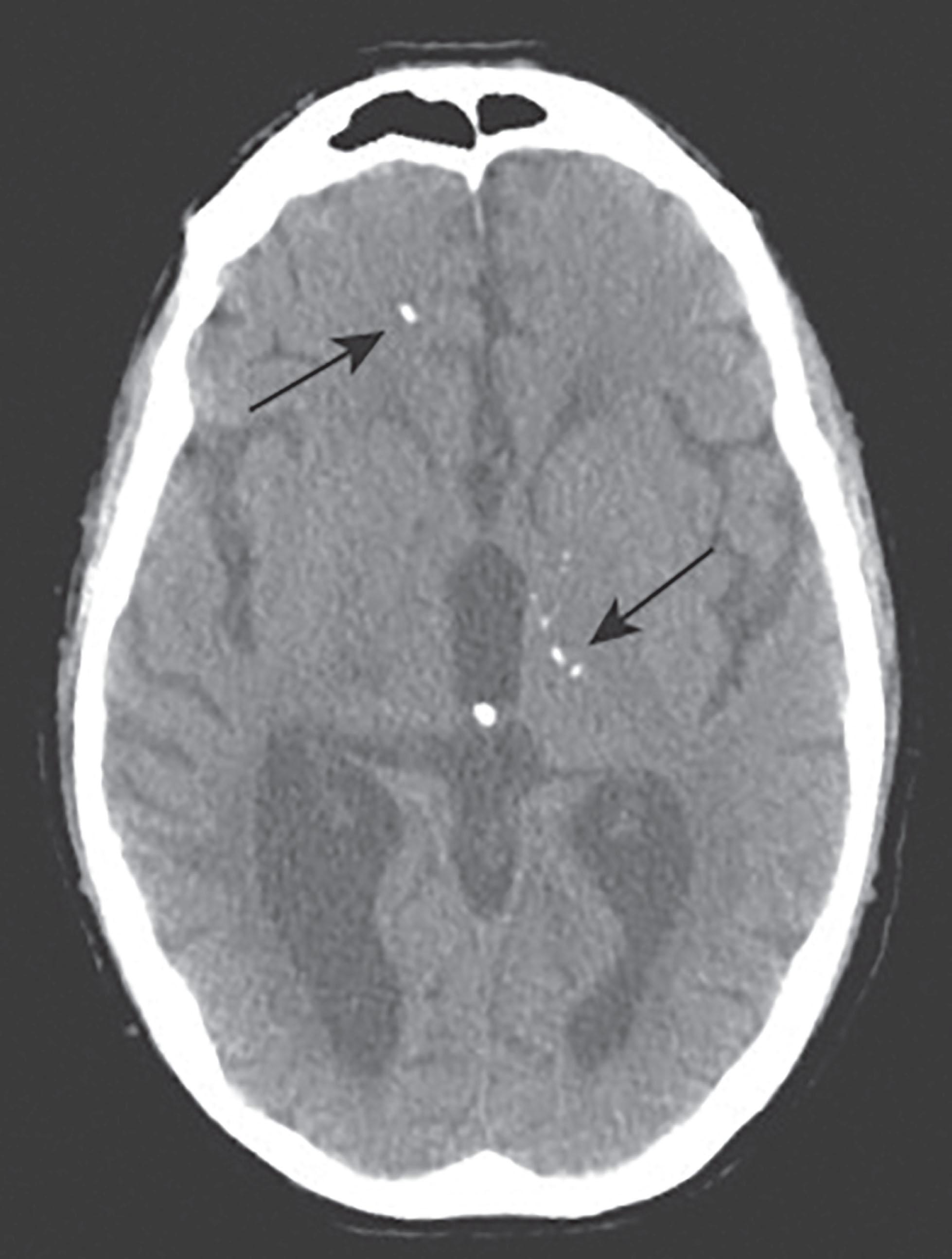
Chorioretinitis secondary to congenital toxoplasmosis can manifest at any age, and is often delayed into the second and third decades of life. It usually manifests as strabismus in infants. Defects in visual acuity are more common in older children who had never received treatment. Typically, the eye lesion consists of a focal necrotizing retinitis that is often bilateral with involvement of the macula and even the optic nerve. Complications include blindness, iridocyclitis, and cataracts.
Other less common manifestations of congenital toxoplasmosis are nonimmune hydrops fetalis, myocarditis, nephrotic syndrome, and immunoglobulin abnormalities with both hypergammaglobulinemia and hypogammaglobulinemia described. Bony abnormalities consisting of metaphyseal lucencies similar to those seen in congenital syphilis have also been reported. A variety of endocrine abnormalities may occur, including hypothyroidism, diabetes insipidus, precocious puberty, and growth hormone deficiency.
Attempts to diagnose congenital toxoplasmosis occur either during pregnancy as part of a surveillance protocol, due to maternal disease or abnormal fetal findings, or, after birth, due to concern for infection. The testing methods vary depending upon the scenario and can be complicated. While commercial laboratories are able to perform some assays, reference laboratories are needed for more intensive evaluation and can be very useful with the availability of their consultative services such as the Palo Alto Medical Foundation Toxoplasma Serology Laboratory (PAMF-TSL; Email: toxolab@pamf.org, telephone: 650-853-4828, http:www.pamf.org/serology/ ).
Serologic assays for measurement of antibodies to T. gondii in serum and body fluids are the most widely used methods of diagnosing toxoplasmosis in a pregnant woman and the fetus or newborn, but can be difficult to interpret. Most commercial laboratories can test for Toxoplasma IgG and IgM, although Toxoplasma IgM assays performed in these laboratories are fraught with low sensitivity and, importantly, false positive results. The more commonly used tests that detect T. gondii -specific IgG antibodies are IgG enzyme-linked immunosorbent assay (ELISA), indirect immunofluorescent antibody test, and direct agglutination. The Sabin-Feldman dye test is considered the gold standard but requires live organisms, and, accordingly, is performed at reference laboratories. If a pregnant woman is known to be seronegative prior to pregnancy and followed serially, then these IgG assays are useful to document seroconversion during pregnancy and risk of the fetus. However, most countries, including the United States, do not have routine prenatal screening, in which case it is important to decipher whether a positive test represents recent infection or chronic infection without a risk to the fetus. Reference laboratories can perform a differential agglutination test to differentiate acute from chronic maternal infection. This test compares the IgG serologic titer obtained with the use of formalin-fixed tachyzoites (HS antigen) with those obtained with acetone- or methanol-fixed tachyzoites (AC antigen). The latter preparation contains stage-specific T. gondii antigens that are recognized by IgG antibodies only during early infection. An additional assay to assist in ruling out maternal infection acquired in the first 3 months of pregnancy is the IgG avidity test performed by the ELISA technique. This test is based on the principle that, although the antibody-binding avidity or affinity for an antigen is initially low after primary antigenic stimulation, IgG antibodies that are present from previous antigenic stimulation are usually of high avidity. Therefore, a high-avidity result in the first trimester would exclude an infection acquired in the previous 12 weeks. Finally, an enzyme-linked immunofiltration assay has been developed that allows some discrimination between IgG antibodies of maternal origin and IgG synthesized by the fetus, as well as identification of antibody subtypes in infected neonates.
Tests that detect T. gondii -specific IgM are (1) the double-sandwich IgM ELISA, which has a sensitivity of 75% to 80% and a specificity of 100%; (2) the IgM immunosorbent agglutination assay (ISAGA), which is the most sensitive test but should not be performed on umbilical cord blood because even small quantities of maternal IgM antibodies contaminating the specimen will yield a false-positive result; and (3) the IgM immunofluorescent antibody test. The last test is not recommended because it has a much lower sensitivity than either the IgM ELISA or IgM and it has poor specificity secondary to rheumatoid factors and antinuclear antibodies, contributing to false-positive results. Other tests used in reference laboratories include a T. gondii- specific IgA ELISA and IgA immunofiltration assay; a T. gondii- specific IgE immunofiltration assay; and IgG, IgM, and IgA immunoblotting tests, which are generally used in comparing samples from the infant and the mother to help determine true infection. Interferon gamma release assays on infants have been used to diagnose congenital infection, but are not available commercially.
Polymerase chain reaction (PCR) analysis has been used successfully to detect T. gondii DNA in amniotic fluid, placenta, CSF, brain, urine, and fetal and infant blood. PCR performed on amniotic fluid obtained by amniocentesis is the preferred method of confirming in utero infection. False-negative results have been reported, however, and interlaboratory variability in performance of PCR assays has been documented. PCR performed on neonatal CSF is recommended for the evaluation of possible central nervous system involvement.
Isolation of T. gondii from body fluids and tissues provides definitive evidence of infection. The organism can be isolated from placenta, amniotic fluid, fetal blood obtained by cordocentesis, umbilical cord blood, infant peripheral blood, and CSF by means of intraperitoneal and subcutaneous inoculation into laboratory mice. Mouse inoculation may require as long as 4 to 6 weeks for demonstration of the parasite. Although it is not a practical method, isolation of the organism should be attempted to confirm a diagnosis and is available at the PAMF-TSL reference laboratory. In addition, tissue culture has been used to isolate T. gondii from amniotic fluid.
Histopathologic examination of the placenta and tissues, obtained at postmortem examination or by biopsy from stillborns or infants, should be performed because the specimens may demonstrate the presence of tachyzoites. In addition, tachyzoites have been demonstrated in CSF, ventricular fluid, and aqueous humor by specialized staining techniques.
Because most adults with acquired T. gondii infection are asymptomatic, evaluation of the pregnant woman and fetus is usually prompted by either seroconversion or an elevated maternal Toxoplasma spp. IgG titer. The latter may reflect chronic past infection; therefore, the acuity of the maternal infection is determined serologically with the HS-AC differential agglutination test where agglutination titers to formalin-fixed tachyzoites (HS antigen) are compared with titers against acetone- or methanol-fixed tachyzoites (AC antigen). In general, an acute pattern demonstrates high AC and HS titers, while a nonacute pattern demonstrates high AC titers and low HS titers. This method can differentiate an acute from a remote infection in pregnant women, whereas IgM and IgA antibodies detectable by ELISA or ISAGA are elevated for prolonged periods. If recent maternal infection is documented by an acute pattern on the HS-AC test, seroconversion, or rising IgG antibody titers, the fetus should be evaluated by ultrasonography, and amniotic fluid should be tested for specific Toxoplasma spp. DNA with PCR. PCR has supplanted the need for cordocentesis, and a positive result confirms fetal infection. Postnatally, serologic testing of paired maternal and infant sera should be performed at a reliable laboratory that will include assays for Toxoplasma spp. IgG, IgM ISAGA and IgA antibodies. Waiting until the newborn is 10 days of age avoids contamination with maternal blood and false positive IgM and IgA results. Subinoculation of placental tissue, amniotic fluid, and umbilical cord blood into mice should be considered. If results of these tests suggest possible infection, the newborn should be evaluated fully with complete blood cell count and platelet determination, liver function tests, CSF evaluation (including tests for IgG and IgM antibodies and PCR), cranial ultrasound or CT imaging of the head, ophthalmologic examination, and hearing evaluation. The presence of neonatal IgM antibody in serum or CSF, or a positive PCR in blood or CSF, indicates congenital infection. In addition, at-risk infants should undergo serologic follow-up to detect rising serum IgG titers during the first year of age, or persistence of IgG antibody beyond 12 to 15 months of age, when most maternal IgG antibody has disappeared. Uninfected infants show a continuous decline in T. gondii IgG titer which usually is gone by 7 months of age with no detectable IgM or IgA antibodies.
Low IgG titers and an HS-AC differential agglutination test that indicate remote maternal infection do not require further evaluation of the mother or infant unless the mother is severely immunosuppressed. Because fetal infection has occurred during chronic T. gondii infection in very immunosuppressed women such as those with poorly controlled HIV infection, their infants should be evaluated serologically at birth for evidence of congenital infection. It has been suggested that HIV-infected pregnant women who have low CD4 + T lymphocyte counts-and who are seropositive for T. gondii antibody-receive prophylaxis to prevent fetal infection. However, insufficient data currently are available to recommend that such therapy be given routinely for this indication. Nevertheless, if such women previously have had toxoplasma encephalitis, prophylaxis with pyrimethamine, sulfadiazine, and leucovorin (folinic acid) should be considered.
Fetuses and infants younger than 1 year who are infected with T. gondii should receive specific therapy effective against this congenital pathogen, even if they have no clinical signs of disease. Compared with untreated historical controls, the outcome is improved substantially by neonatal treatment. The effectiveness of maternal and fetal treatment is less clear. Spiramycin has been used in pregnant women with acute toxoplasmosis to reduce transplacental transmission of T. gondii . If fetal infection is confirmed after the 17th week of pregnancy, however, treatment with pyrimethamine, sulfadiazine, and folinic acid is recommended. Prenatal treatment of congenital toxoplasmosis is believed to reduce the clinical severity of infection in the newborn while shifting the disease to a more subclinical form. This may ameliorate the long-term neurologic complications that are commonly seen among infants who have clinical manifestations in the neonatal period. A meta analysis of the effectiveness of prenatal treatment of toxoplasmosis infection found no evidence that such treatment significantly decreased clinical manifestations of disease in infected infants. However, other studies in Brazil, Germany, and France suggest efficacy.
Neonatal treatment has resulted in reductions in sensorineural hearing loss, as well as neurodevelopmental and visual handicaps. Table 35.2 shows the recommended guidelines for the treatment of congenital toxoplasmosis. In infants with congenital toxoplasmosis, the treatment consists of pyrimethamine, sulfadiazine, and folinic acid (leucovorin). The optimal duration of therapy is not known, although prolonged courses of at least 1 year are preferred. Currently most experts recommend combined treatment until the patient is 1 year old. Accessing pyrimethamine in the United States has recently become difficult due to extreme cost escalation. Assistance with access is available through http://www.daraprimdirect.com (phone number: 1-877-258-2033).
| Condition | Therapy | Dose (Oral Unless Specified) | Duration |
|---|---|---|---|
| Pregnant woman with acute toxoplasmosis | Spiramycin for first 21 weeks of gestation or until term if fetus not infected | 3 g/day, divided twice a day without food | Until fetal infection documented or excluded at 21 weeks; if fetal infection documented, replaced with pyrimethamine, leucovorin, and sulfadiazine (see below) |
| Pregnant women-fetal infection confirmed (amniotic fluid PCR+) | Pyrimethamine (if fetal infection confirmed after 18th week of gestation or if infection acquired in last few weeks of gestation) and | Loading dose: 100 mg/day in two divided doses for 2 days followed by 50 mg/day | Until delivery |
| Sulfadiazine and | 3 g/day divided in | Until delivery | |
| Folinic acid | 5–20 mg/day | Until delivery | |
| Congenital Toxoplasma gondii infection in infant | Pyrimethamine and | Loading dose: 2 mg/kg/day for 2 days; then 1 mg/kg/day for 6 months; then 1 mg/kg/day on Monday, Wednesday, and Friday each week | 1 year |
| Sulfadiazine and | 100 mg/kg/day in 2 daily divided doses | 1 year | |
| Leucovorin (folinic acid) † | 5–10 mg 3 times weekly | 1 year | |
| Corticosteroids (prednisone) ‡ | 1 mg/kg/daily in 2 daily divided doses | Until resolution of elevated (≥1 g/dL) CSF protein or active chorioretinitis that threatens vision |
Complete blood cell and platelet counts must be monitored closely while the patient is receiving therapy because granulocytopenia, thrombocytopenia, and megaloblastic anemia can occur. These parameters usually improve once a higher dose of folinic acid (leucovorin) is administered or pyrimethamine and sulfadiazine are discontinued temporarily. The indications for adjunctive therapy with corticosteroids such as prednisone (0.5 mg/kg twice per day) are CSF protein concentration 1 g/dL or higher and chorioretinitis that threatens vision; corticosteroid treatment is continued until either condition resolves. Current therapies are not effective against encysted bradyzoites and, therefore, may not prevent reactivation of chorioretinitis and neurologic disease.
Maternal toxoplasmosis acquired during the first and second trimesters has been associated with stillbirth and perinatal death secondary to severe fetal infection in approximately 35% and 7% of cases, respectively. Among infants born with congenital toxoplasmosis, the mortality rate has been reported to be as high as 12%. In addition, infants with congenital toxoplasmosis are at high risk for ophthalmologic, neurodevelopmental, and audiologic impairments, including mental retardation (87%), seizures (82%), spasticity and palsies (71%), and deafness (15%). Of neonates with subclinical infection, long-term follow-up reveals eye or neurologic disease in as many as 80% to 90% by the time they reach adulthood. While data from the United States National Collaborative Treatment Trial show that severity is influenced by host and parasite factors, treatment of neonates with congenital toxoplasmosis early and for 1 year resulted in more favorable outcomes than were reported for untreated infants or infants who were treated for only 1 month.
Pregnant women whose serologic status for T. gondii is negative or unknown, as well as women who are attempting to conceive, should be educated on the prevention of congenital toxoplasmosis through avoidance of at-risk behaviors that may expose them to cat feces or encysted bradyzoites in raw meat. Instructions to wear gloves when changing cat litter boxes or gardening and to wash hands after such activities should be given. Daily changing of cat litter will also decrease the chance of infection, because oocysts are not infective during the first 1 to 2 days after passage. In addition, keeping domestic cats inside, and feeding them commercially prepared foods rather than undercooked meats or wild rodents, reduces the likelihood of their becoming infected and capable of transmitting the infection to a pregnant woman. Oral ingestion of T. gondii can be prevented by either cooking meat to well done, smoking it, or curing it in brine, and by washing kitchen surfaces and utensils that come into contact with raw meat. Vegetables and fruits should be washed, and hands and kitchen surfaces should be cleaned after handling fruits, vegetables, and raw meat. Flies and cockroaches may serve as transport hosts for T. gondii , so their access to food must be prevented.
Routine serologic screening of women during pregnancy has been an effective means of prevention in such countries as France and Austria, where the incidence of congenital toxoplasmosis is high. No such screening is currently in existence in the United States. However, high-risk women, including those who are immunocompromised, should be screened early in pregnancy. Neonatal screening for IgM antibody has also been advocated so that asymptomatic infants can be detected and treated before neurologic symptoms develop. This strategy, however, has been hampered by the lack of readily available and reliable IgM test kits. Moreover, such screening will not detect the approximately 25% of infected neonates who lack anti- Toxoplasma spp. IgM antibody. Further study involving cost analyses is needed to define the best preventive strategy for congenital toxoplasmosis in specific populations, regions, and countries.
Congenital syphilis, a result of fetal infection with the spirochete Treponema pallidum , remains a major public health problem worldwide. While adults acquire the infection sexually, infants are infected mostly in utero by a transplacental route or possibly during delivery by contact with a genital lesion of an infected mother. When maternal infection is detected during pregnancy, congenital syphilis is both preventable and treatable. However, if infected infants are not identified in a timely fashion, they may experience lifetime consequences. Accordingly, the Centers for Disease Control and Prevention (CDC) recommend that no mother or newborn be discharged from the hospital without the maternal serologic status for syphilis having been documented at least once during the pregnancy, and preferably again at delivery if the mother is at increased risk or lives in a community with high prevalence of syphilis infection.
The incidence of congenital syphilis mirrors the rates of primary and secondary syphilis in women. Overall, congenital syphilis disproportionately affects infants of black women whose prenatal care was inadequate or lacking. Less prenatal care has been associated with increased risk of fetal death that most often occurs by 31 weeks’ gestation. From 1999 to 2013 in the United States, neonatal mortality secondary to congenital syphilis was 12/1000 live births, with a case fatality rate of 6.5%. Of the 418 reported deaths, 82% were stillbirths and 89% of the mothers had untreated or inadequately treated syphilis.
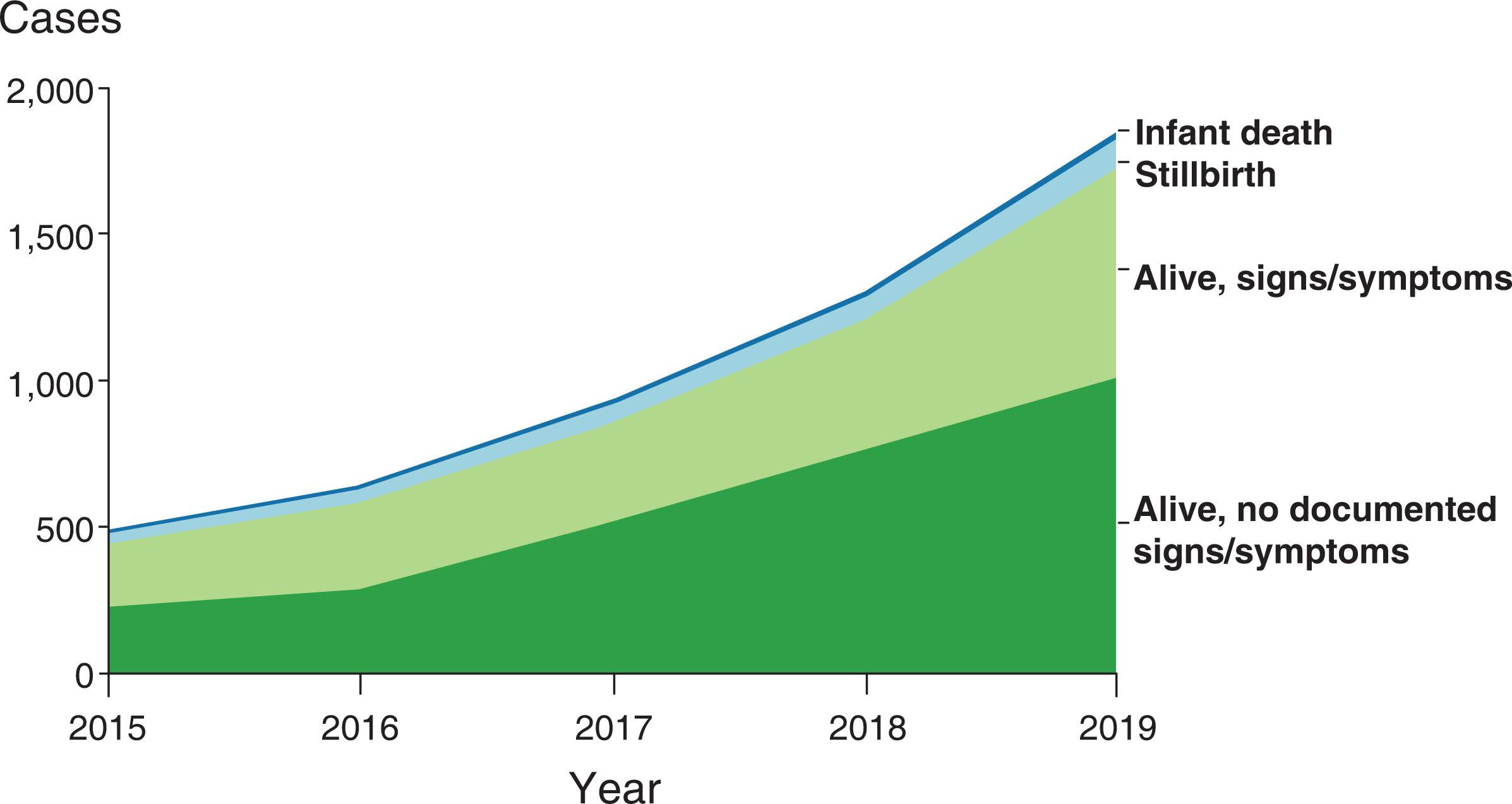
During the pre-penicillin era of the 1930s and 1940s in the congenital syphilis clinic of the Harriet Lane Home (Baltimore, MD), 60 to 80 infants and children attended each week for arsenic therapy. Many more were lost to follow-up before completing their 2- to 3-year course of treatment. It was unusual if fewer than three or four new cases were discovered in the general outpatient department each week. Subsequently, the frequency of congenital syphilis declined for several decades, only to increase dramatically in the late 1980s and early 1990s. This increase was fueled by the crack cocaine epidemic with women exchanging drugs for sex with multiple and anonymous partners. At the same time in 1988, the CDC surveillance case definition for reporting cases of congenital syphilis was broadened to include all liveborn and stillborn infants who had reactive serologic tests for syphilis, irrespective of clinical findings, and those delivered to women with untreated or inadequately treated syphilis. This change resulted in a fourfold increase in reported cases of congenital syphilis when compared to the previously used Kaufman criteria that included only symptomatic infants (reviewed in). For unclear reasons, maternal syphilis subsequently declined, and the rate of congenital syphilis decreased from 1991 to 2005 but increased slightly during 2005 to 2008. From 2008 to 2012, the overall rate of congenital syphilis again decreased from 10.5 to 8.4 cases per 100,000 live births, reflecting decreasing trends in primary and secondary syphilis among women in the United States. Since 2012, rates of congenital syphilis increased significantly (8.4 cases per 100,000 live births in 2012 to 48.5 cases per 100,000 live births in 2019). In 2019, there were 1870 cases of congenital syphilis that included 94 stillbirths and 34 infant deaths ( Fig. 35.2 ). The recent increase in syphilis has been attributed to the ongoing opioid epidemic in the United States.
Worldwide, congenital syphilis remains a major cause of fetal and neonatal mortality with more newborns affected by congenital syphilis than by any other neonatal infection. The World Health Organization estimated that, globally, nearly 1 million pregnant women were actively infected with syphilis in 2016, resulting in an estimated 660,000 cases of congenital syphilis, and half of them have infants with adverse outcomes such as stillbirth or prematurity, nonimmune hydrops, and death. The global burden of congenital syphilis is confounded further by the high prevalence of infection with the human immunodeficiency virus (HIV), as syphilis is a known risk factor for acquisition of HIV.
The causative agent for syphilis is T. pallidum , a thin, corkscrew-shaped, flagellated, highly motile spirochete. T. pallidum is able to invade the fetal compartment at any time during gestation, although the risk of fetal infection increases as the stage of pregnancy advances. Spirochetes have been detected in fetal tissue from spontaneous abortion as early as 9 and 10 weeks’ gestation and recovered from amniotic fluid at 14 weeks of pregnancy. Vertical transmission is related directly to the maternal stage of syphilis, with early syphilis resulting in significantly higher transmission rates than late latent infection. Ingraham reported in 1950 that among 251 women with syphilis of less than 4 years’ duration, 41% of their infants were born alive and had congenital syphilis, 25% were stillborn, 14% died in the neonatal period, 21% had low birth weight but no evidence of syphilis, and 18% were normal full-term infants. In contrast, only 2% of infants born to mothers with late latent disease had congenital syphilis. In 1952, Fiumara and colleagues reported that untreated maternal primary or secondary syphilis resulted in 50% of infants having congenital syphilis while the other 50% were stillborn, premature, or died in the neonatal period. With early and late latent infection, 40% and 10% of infants, respectively, had congenital syphilis. These data are supported by a more recent study of Sheffield and colleagues in which mothers with primary, secondary, early latent, and late latent infection had transmission rates of 29%, 59%, 50%, and 13%, respectively.
Because T. pallidum enters the fetal bloodstream directly, the primary stage of infection is completely bypassed. There is no chancre and no local lymphadenopathy. Instead, there is widespread hematogenous spread to all organs and tissues, including the liver, spleen, pancreas, intestine, kidney, skin, mucous membranes of the lips, nose and anus, bones and cartilage, and the central nervous system. Invasion of the lung results in a characteristic “pneumonia alba” that is seen more frequently in developing countries.
Microscopically, the tissue alterations consist of interstitial fibrosis and perivascular inflammation with plasma and round cell infiltration, with visualization of spirochetes by silver or fluorescent staining. Gumma formation is infrequent in neonates, while extramedullary hematopoiesis involving the liver, spleen, dermis, kidneys, and other organs is common.
The placenta of infants with congenital syphilis often is large, thick, and pale. Histopathologic features include villous enlargement, acute villitis, and erythroblastosis. Intense inflammation of the umbilical cord results in a “barber's pole” appearance where the edematous portions have a spiral striped zone of red and pale blue discoloration, interspersed with streaks of chalky white. Histologically, the umbilical cord exhibits abscess-like foci of necrosis within Wharton’s jelly and umbilical vessels. Placental and umbilical cord histopathology should be performed on every case of suspected syphilis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here