Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
If you think your ex utero life is complicated, wait till you see what goes on in utero! Understanding development of the central nervous system (CNS) aids in understanding congenital brain and spine anomalies and coexistence of multiple anomalies. Although there is some overlap, to put things simply, the CNS goes through six major developmental events that include (1) primary neurulation (3 to 4 weeks of gestation); (2) promesencephalic development (2 to 3 months of gestation); (3) neuronal proliferation (3 to 4 months of gestation); (4) neuronal migration (3 to 5 months of gestation); (5) organization (5 months of gestation to postnatal years); and (6) myelination (birth to postnatal years). Above mentioned termination periods of each event means that the malformation of that particular event may have its onset before the event is over.
Neurulation is a series of inductive events that takes place in the dorsal aspect of the embryo and results in development of the brain and spinal cord. Primary neurulation involves events that are related to formation of the brain and spinal cord exclusive of its most caudal region. The first fusion of neural folds occur in the lower medulla. Closure generally proceeds rostrally and caudally, although it is not a simple zipper-like process. Disorders of primary neurulation are cranioschisis, anencephaly, myeloschisis, encephalocele, myelomeningocele, and Chiari II malformations. Various malformations of neural tube closure are accompanied by axial skeleton, meningovascular and dermal covering abnormalities.
Secondary neurulation occurs later than primary neurulation with sequential processes of canalization and retrogressive differentiation. Disorders of secondary neurulation result in malformations of the lower sacral and coccygeal segments of the neural tube affecting the conus medullaris, filum terminale, and cauda equina. Lipomas of the filum, short thickened tethered fila and tethered cords, and caudal regression syndrome with sacral agenesis are also listed under this category.
The primary inductive relationship is between the notochord-prechordal mesoderm and forebrain and takes place ventrally in the rostral end of the embryo, thus some authors use the term ventral induction . Formation of the face and forebrain occur during this event, so malformations of the brain are usually accompanied by facial anomalies (such as cyclopia and probosci). The spectrum of malformations can be as severe as aprosencephaly, to perhaps clinically occult callosal malformations. Three major sequential events (and related malformations) are promesencephalic development (atelencephaly), premesencephalic cleavage (holoprosencephaly), and midline promesencephalic development (agenesis of corpus callosum, agenesis of septum pellucidum, septooptic dysplasia, hypothalamic dysplasia).
Cerebellar development also occurs at the 5- to 15-week period of fetal development. Just as there is cerebral hemispheric development from neural tissue derived from the germinal matrix around the lateral ventricles, so too is cerebellar development dependent on the germinal matrix about the fourth ventricle. Because hemispheric development occurs before vermian development, and the superior vermis forms before the inferior vermis, it is rare to see hemispheric anomalies without vermian maldevelopments and superior vermian lesions without associated inferior vermian anomalies. Inferior vermian hypoplasia may be seen in isolation. Those entities associated with vermian dysgenesis include the Dandy-Walker malformation, Joubert syndrome, and rhombencephalosynapsis.
Disorders of hemispheric development include hemispheric hypoplasia, but this is a rare bird more commonly seen with other supratentorial and vermian anomalies.
All neurons and glia are derived from the ventricular and subventricular zones of the germinal matrix. Disorders of neuronal proliferation can result in small or large brain (microcephaly or macrocephaly). Keep in mind the neurocutaneous syndromes when assessing macrocephaly or hemimegalencephaly...stay tuned, more on this later in the chapter.
The neurons migrate from the ventricular and subventricular zones of the subependyma to their final residence for life. Initially neurons migrate by translocation of the cell body followed by two basic varieties of cell migration: radial and tangential. Radial migration leads to projection neurons of the cerebral cortex and deep nuclei in the cerebrum, and Purkinje cells in the cerebellum. Tangential migration leads to interneurons of the cerebral cortex and internal granule layer of the cerebellar cortex. The layering of the neurons are inside out: early arriving neurons are deep whereas late arriving neurons are in the superficial aspect of the cortex. Disorders of neuronal migration include schizencephaly, lissencephaly (pachygyria), polymicrogyria, heterotopia, and focal cerebrocortical dysgenesis. Tip: Commisural anomalies (e.g., corpus callosum agenesis/dysgenesis) and septum pellucidum anomalies can accompany these disorders, so keep a look out!
A series of complex processes take place: subplate neuron differentiation; alignment, orientation and layering of cortical neurons; establishment of synaptic contacts; cell death; proliferation and differentiation of glia. Primary disorders are mental retardation, autism, and syndromes such as Fragile X, Rett, Down, and Angelman. Potential disturbances such as those seen in premature infants (germinal matric hemorrhage spectrum, periventricular leukomalacia) can occur.
The structure of myelin is rich in lipid and protein. In the CNS, myelin is primarily found in white matter, although it is also present in gray matter, but in smaller quantities. Myelination of the brain begins during the fifth fetal month, progresses rapidly for the first 2 years of life, and then slows markedly after 2 years. In contrast, fibers to and from the association areas of the brain continue to myelinate into the third and fourth decades of life. In general, myelination has a predictable maturation, with progressing from caudal to cephalad, posterior to anterior, and central to peripheral directions ( Box 8-1 ). As an example, myelination progresses from the brain stem to the cerebellum and basal ganglia, and then to the cerebral hemispheres. In a particular location, the dorsal region tends to myelinate before the ventral regions. The process of myelination also relates to functional requirements such that the somatosensory system in neonates myelinates earlier than motor and association pathways. A rapid growth of the myelinated white matter volume is observed between birth and 9 months of age.
Posterior to anterior
Central to peripheral
Caudal to cephalad
T1-weighted imaging to T2-weighted imaging
Immature to mature
Anatomic magnetic resonance imaging (MRI) sequences (T1- and T2-weighted) are very helpful in assessment of myelination. There is reduction in T1 and T2 relaxation times with continued white matter maturation that corresponds with reduction in tissue water as well as the interaction of water with myelin lipids. On T1-weighted imaging (T1WI), most of the white matter in the newborn brain is hypointense compared with gray matter, and the appearance is similar to that of T2-weighted imaging (T2WI) in adults. Some of the exceptions include the dorsal brain stem pathways (medial lemnisci, medial longitudinal fasciculi) and the posterior limb of the internal capsules, which are myelinated at birth and show hyperintensity on T1. In general, the white matter myelinates earlier on T1WI than on T2WI for reasons that have to do with water content versus lipid content. Diffusion tensor imaging (DTI) can visualize and quantify neural tracts relating to their myelination: fractional anisotropy (FA) increases with age (myelination), whereas apparent diffusion coefficient (ADC), axial diffusivity (AD), and radial diffusivity (RD) decreases, with a turning point around 6 years of age. Note that developmental stages differ by topography and tract of interest, and changes on T1WI and T2WI differ. Myelination can be assessed using these milestones as outlined in Table 8-1 . After 2 years of age, delayed myelination can be detected only when quite severe.
| Anatomic Structure | T1WI | T2WI |
|---|---|---|
| PLIC (posterior portion) | 36 GW | 40 GW |
| Median longitudinal fasciculus | 25 GW | 29 GW |
| Superior cerebellar peduncles | 28 GW | 27 GW |
| Middle cerebellar peduncles | Birth | Birth-2 mo |
| PLIC (anterior portion) | Birth-1 mo | 4-7 mo |
| Anterior limb internal capsule | 2-3 mo | 5-11 mo |
| Cerebellar white matter | Birth-4 mo | 3-5 mo |
| Splenium of corpus callosum | 3-4 mo | 4-6 mo |
| Genu of corpus callosum | 4-6 mo | 5-8 mo |
| Occipital white matter (central) | 3-5 mo | 9-14 mo |
| Frontal white matter (central) | 3-6 mo | 11-16 mo |
| Occipital white matter (peripheral) | 4-7 mo | 11-15 mo |
| Frontal white matter (peripheral) | 7-11 mo | 14-18 mo |
| Centrum semiovale | 2-4 mo | 7-11 mo |
It is useful to separate congenital disorders of the brain into those involving the supratentorial structures ( Box 8-2 ) and those involving the infratentorial structures ( Box 8-3 ), because radiologists think in terms of anatomy rather than embryology. Naturally, disorders can affect both spaces, but as an initial classification scheme, this may be helpful. In addition to supratentorial and infratentorial compartments, separating the lesions into those that are cystic and those that are solid can provide some help in arriving at a differential diagnosis. Clinical information is always useful in distinguishing among various congenital abnormalities, because several disorders also have associated cutaneous, ocular, or metabolic abnormalities.
Disorders of the forebrain development
Holoprosencephaly
Commissural anomalies
Septooptic dysplasia
Pituitary anomalies
Cortical malformations
Proliferation disorders
Primary microcephaly with simplified gyral pattern
Hemimegalencephaly
Migrational Disorders
Lissencephaly
Pachygyria
Subcortical band heterotopia
Nodular heterotopia
Cobblestone brain
Focal cortical dysplasia
Hamartomas
Organizational disorders
Polymicrogyria
Schizencephaly
Anomalies limited to the cerebellum
Dandy-Walker malformation
Blake pouch cyst
Mega cisterna magna
Rhombencephalosynapsis
Other
Isolated vermian hypoplasia
Arachnoid cyst in the posterior fossa
Anomalies involving the cerebellum and brain stem
Joubert syndrome
Pontocerebellar hypoplasia
Cerebellar disruptions
Chiari malformations I-III
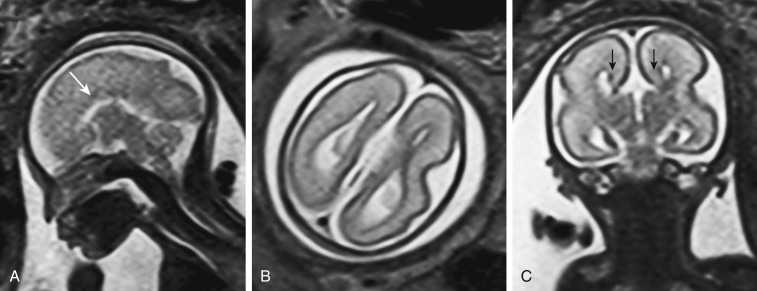
Holoprosencephaly refers to a constellation of disorders characterized by failure of cleavage or differentiation of the prosencephalon, resulting in a fused appearance of the prosencephalonic structures ( Fig. 8-1 ). These abnormalities are associated with rather severe mental retardation, microcephaly, hypotelorism, and abnormal facies. The finding of a solitary median maxillary central incisor is also indicative of holoprosencephaly. The olfactory bulbs and tracts are usually absent with lack of development of olfactory sulci and flat gyri recti. The range of this disorder is subclassified as alobar, semilobar, and lobar holoprosencephaly (with decreasing severity; Table 8-2 ). However, you should be aware that the term holoprosencephaly represents a continuum of forebrain malformations, and that no clear distinction exists between categories.
| Feature | Lobar | Semilobar | Alobar |
|---|---|---|---|
| Facial deformities (cyclopia) | None | None to minimal | Yes |
| Falx cerebri | Anterior tip missing or dysplastic | Partially formed | Absent |
| Thalami | Separated | Partially fused | Fused |
| Interhemispheric fissure | Formed | Present posteriorly | Absent |
| Dorsal cyst | No | Yes, if thalamus is fused | Yes |
| Frontal horns | Yes, but unseparated | No | No |
| Septum pellucidum | Absent | Absent | Absent |
| Vascular | Normal | Normal except rudimentary deep veins | Azygous anterior cerebral artery, absent venous sinuses and deep veins |
| Splenium of corpus callosum | Present | Present without genu or body | Absent |
| Third ventricle | Normal | Small | Absent |
| Occipital horns | Normal | Partially formed | Absent |
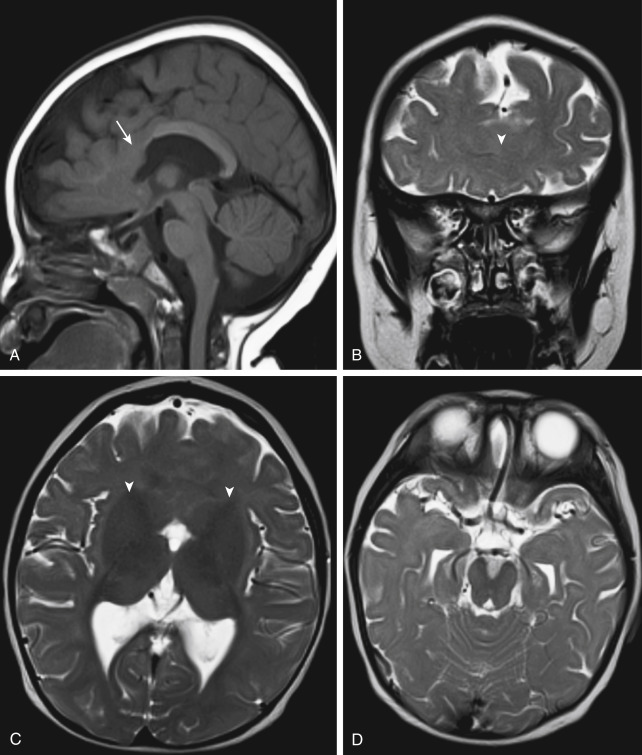
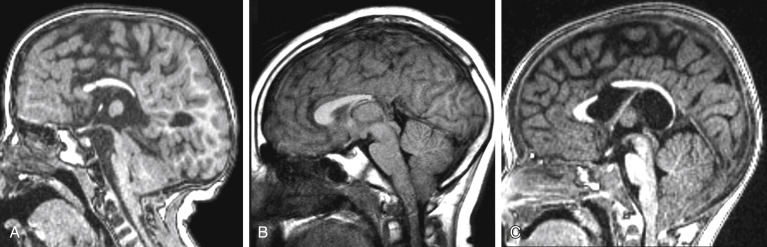
Lobar holoprosencephaly presents with near complete cleavage/separation of the frontal lobes (see Fig. 8-1 ). Formation of the frontal horns of the lateral ventricles can be dysplastic but present. Sylvian fissures are near normal but formation of an interhemispheric fissure and cerebral falx is incomplete. The deep cerebral nuclei are fully formed. Tip: A fully formed third ventricle, at least partially present frontal horns, and presence of the posterior body and splenium of corpus callosum classifies the anomaly under lobar holoprosencephaly.
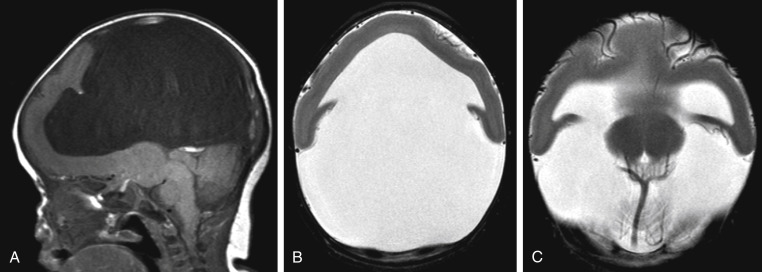
The most severe form is alobar holoprosencephaly ( Fig. 8-2 ), also the most common one, yet rarely encountered in clinical practice because most infants are stillborn. No interhemispheric fissure, falx, or significant separation of the hemispheric structures is identified. A crescentic/horseshoe-shaped monoventricle continuous with a large dorsal cyst usually occupies most of the cranium. The basal ganglia and thalami are fused, and the septum pellucidum and corpus callosum are absent. The anterior cerebral arteries in these cases are nearly always azygous.
Between the two extremes of lobar and alobar holoprosencephaly is semilobar holoprosencephaly, in which there is partial development of the falx and the interhemispheric fissure (with partial separation of the lateral ventricles). The basal ganglia and thalami are at least partially fused.
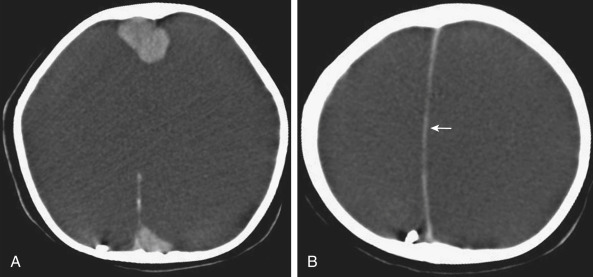
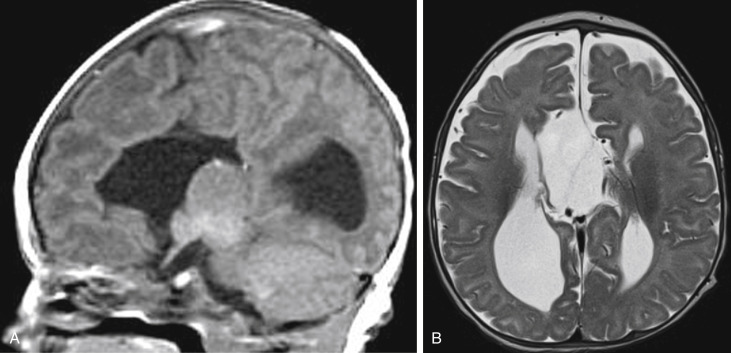
Common differential diagnoses include hydranencephaly and severe congenital hydrocephalus. If no cortical mantle is discernible around the dilated cerebrospinal fluid (CSF) space centrally, the diagnosis is hydranencephaly, especially if all that can be seen is a nubbin of occipital or posterior temporal cortex remaining with a falx present ( Fig. 8-3 ). If you see a well-formed falx, cortical mantle, and separated ventricles with a septum pellucidum, suggest severe hydrocephalus. If the thalami are fused, septum pellucidum or falx is absent, a cortical mantle is seen, and the ventricles have lost their usual shape, diagnose alobar holoprosencephaly. Holoprosencephalies can be associated with a number of clinical syndromes ( Box 8-4 ).
Caudal agenesis
DiGeorge syndrome
Fetal alcohol syndrome
Kallmann syndrome
Maternal diabetes
Trisomy 13, 15, 18
The corpus callosum is the largest of the three midline commissures; others are the anterior commissure and the hippocampal commissure. Agenesis of the corpus callosum is one of the most commonly observed features in the malformations of the brain and is a part of many syndromes. The classic, more descriptive callosal segments include the lamina rostralis, genu, body, isthmus, and splenium. However, from a functional and developmental anatomical point of view, the corpus callosum is divided into two segments by the isthmus: a prominent anterior frontal segment that carries the fibers of the frontal cortex and white matter, and a smaller posterior splenial segment that carries the fibers of the primary visual as well as the posterior parietal and medial occipitotemporal cortices and white matter. This helps understanding of two important concepts: (1) myelination proceeds posterior to anterior reflecting the fact that myelination of the primary cortical areas connected through the isthmus and splenium precedes myelination of the body, genu, and rostrum, and (2) in holoprosencephalies, the posterior callosum is present, because the temporal and occipital lobes are separated in those cases.
The septum pellucidum forms in close association with the anterior corpus callosum. The cavum is not truly apparent before 20 gestational weeks. This is important to understand partial agenesis/dysgenesis of the genu of the corpus callosum in cases with septooptic dysplasia. The interhemispheric glial bridge provides a support for the first pioneer axons to cross at about gestational weeks 12 to 13. The corpus callosum develops within a very short time during week 13 and at week 14 it is virtually complete, although still short. The shape of the corpus callosum is essentially final by week 20; however, its sagittal cross-sectional area is only 5% of what it will be in a mature brain ( Fig. 8-4 ). The corpus callosum enlarges together with the connectivity and the tangential growth of the cortex.
It has long been assumed that the corpus callosum develops from front to back, and the partial commissural agenesis, most commonly posterior splenial agenesis, would be lesser forms of agenesis. It is now understood that by determining the missing portions, one cannot reliably predict whether the partial agenesis is a sequela of destructive event versus a developmental anomaly. One needs to evaluate all interhemispheric commissures, the isthmus, the connection of anterior segment to isthmus, and the connection of the posterior segment to isthmus to conclude whether or not the corpus callosum is developmentally anomalous.
Sagittal midline T1WI and/or T2WI are ideal in this evaluation. In the most typical partial agenesis, the entire corpus callosum is present, but short in anteroposterior (AP) diameter. Alternatively along the spectrum of callosal dysgenesis, the anterior and posterior segments may be present without the connecting isthmus (no connection of the fornix to the splenium), the splenium may be hypoplastic, or the corpus callosum can be completely absent ( Fig. 8-5 ).
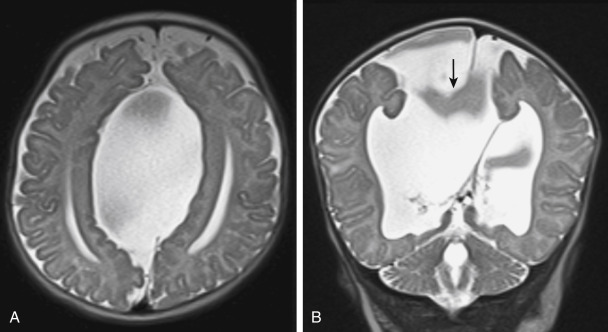
In fact, in classic corpus callosum agenesis, the commissural fibers are not completely genetically absent but rather heterotopic. The noncrossing fibers make an angle and form a parasagittal bundle in the medial aspect of the lateral ventricles, forming the Probst bundles. The lateral ventricles are parallel, frontal horns are pointed and crescentic in shape and occipital horns are enlarged (colpocephaly), with a high-riding third ventricle (if no corpus callosum, there is nothing to hold the third ventricle in place). The cingulate gyrus is everted, and the cingulate sulcus does not form resulting in radiating/disorganized appearance of the sulci in the interhemispheric region ( Box 8-5 ; Fig. 8-6 ).
Pointed, crescent-shaped frontal horns
Colpocephaly
High-riding enlarged third ventricle
Incomplete development of hippocampal formation
Interhemispheric cyst or lipoma
Medial impingement of Probst bundle on ventricles
No cingulate sulcus
Radially oriented fissures (eversion of cingulate gyrus) into the high-riding third ventricle
Septum pellucidum absent or widely separated
Other midline abnormalities may be associated with agenesis of the corpus callosum, the most common being interhemispheric cysts or a midline lipoma (both of which are related to meningeal dysplasia). Interhemispheric cysts may be communicating ( Fig. 8-7 ) or noncommunicating with the ventricles; these cysts are important to identify especially in those cases with ventriculomegaly. As expected, the cyst would follow the same density/signal intensity as the CSF, unlike the lipoma, which has fat density/signal intensity ( Fig. 8-8 ).

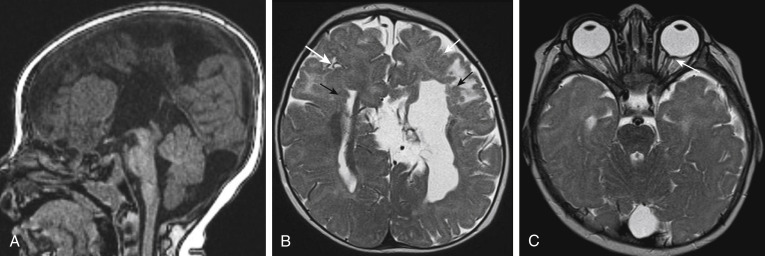
Callosal development is closely associated with development of the cortex so associations of dysgenesis with heterotopias, agyria, pachygyria, holoprosencephaly, septooptic dysplasia, cephaloceles, Chiari I and II malformations, and Dandy-Walker syndrome are not uncommon. Trisomy 13, 15, and 18, fetal alcohol syndrome, Meckel syndrome (occipital encephalocele, microcephaly, polycystic kidneys, polydactyly) are also associated with agenesis of the corpus callosum (and also with holoprosencephaly). Aicardi syndrome is characterized by the triad of agenesis of the corpus callosum, infantile (or even neonatal) spasms usually without typical hypsarrhythmia (abnormal interictal pattern seen on EEG in patients with infantile spasms), and severe neurological and mental impairment. The imaging features are partial/total callosal agenesis, a marked asymmetry between the hemispheres, an interhemispheric cyst, polymicrogyria, periventricular or subcortical nodular heterotopias, and choroid plexus cysts or papillomas, posterior fossa cyst, choroidal ocular lacunae and ocular colobomata ( Fig. 8-9 ).
Interhemispheric lipomas occur secondary to meningeal dysplasia believed to result from an abnormal differentiation of the meninx primitiva. These are usually associated with dysplastic vessels and may calcify. Many (up to 50%) are associated with agenesis of the corpus callosum. The most common sites of lipomas are the interhemispheric fissure (50%), the quadrigeminal plate cistern, the pineal region, hypothalamic region, and the cerebellopontine angle cistern. These lesions typically do not grow and are symptomatic only because of other associated congenital anomalies and/or mass effect on neighboring structures. Treatment tends to be conservative because vessels often course through the lipoma, making surgical removal that much harder. Lipomas have attenuation values on computed tomography (CT) that are in the negative range, usually −30 to −100 Hounsfield units (HU), and are isodense to subcutaneous fat. High signal intensity on T1WI and bright on fast spin echo T2WI, the fat of the lipoma can be corroborated by the presence of a chemical shift artifact along the frequency encoding direction. This causes one edge of the lesion to be highlighted as very bright on T2WI and the other edge along the frequency-encoded axis to be darkened. Alternatively, a fat-suppression pulse sequence can verify the fat by demonstrating signal diminution after it is applied.
Septooptic dysplasia (de Morsier syndrome) consists of hypoplasia of the optic nerves and hypoplasia/absence of the septum pellucidum along with hypothalamic-pituitary axis abnormality. Seizures may coexist. Effects on the visual pathway may range from blindness to normal vision, nystagmus to normal eye movements. Absence of the septum pellucidum causes a squared-off appearance to the frontal horns of the lateral ventricles ( Fig. 8-10 ). MRI appearances are categorized in two groups, one with a high incidence of malformations of cortical development (especially schizencephaly and heterotopias), and partial absence of the septum pellucidum; the second with overlapping features of mild lobar holoprosencephaly with complete agenesis of the septum pellucidum. Hypoplasia of the anterior falx can be seen in the second group. When the septum pellucidum is partially absent, usually the anterior portion is present. This is best seen on coronal magnetic resonance (MR). Agenesis of the corpus callosum and white matter hypoplasia may be associated with this abnormality. In general, patients with septooptic dysplasia demonstrate small hypoplastic optic nerves and a small optic chiasm resulting from the dysplastic optic pathways. In some cases that dysplasia may be limited to the optic disc and the nerves/chiasm may not be small. Pituitary abnormalities such as ectopic posterior pituitary gland can be seen.
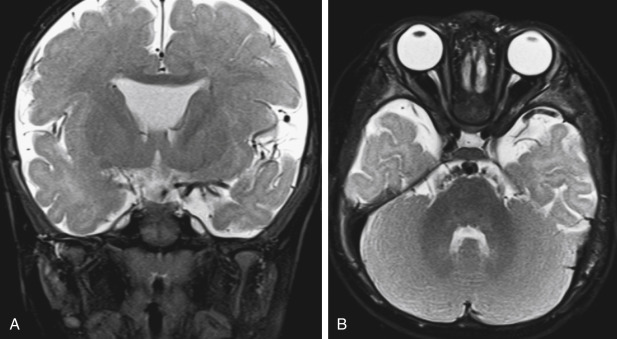
Rathke cleft cysts are embryologic remnants of Rathke pouch, the neuroectoderm that ascends from the oral cavity to the sellar region to form the pituitary’s anterior lobe and pars intermedia. These cysts are lined with a single layer of cuboidal or columnar epithelial cells and may arise within the sella, the suprasellar region, or most commonly both. They are not very common lesions. The cysts can compress normal posterior or anterior pituitary tissue to cause symptoms of hypopituitarism, diabetes insipidus, headache, and visual field deficits but patients are usually asymptomatic. The cysts are well-defined masses that are located between the adenohypophysis and neurohypophysis. Signal characteristics may vary on MRI: high or low signal intensity on T1WI (depending of amount of proteinaceous contents), high signal intensity on T2WI, and lack of postcontrast enhancement are common ( Fig. 8-11 ). These can appear hypodense on CT. Intracystic, yellow waxy solid nodules have recently been reported in Rathke cysts containing cholesterol and/or mucinous proteins probably accounting for the bright signal on T1WI in some cysts. The differential diagnosis is a craniopharyngioma or hemorrhagic pituitary gland; first check out the location—if the lesion is between the adeno- and neurohypophysis and lacks calcification, solid component, and enhancement, then favor Rathke cleft cyst.
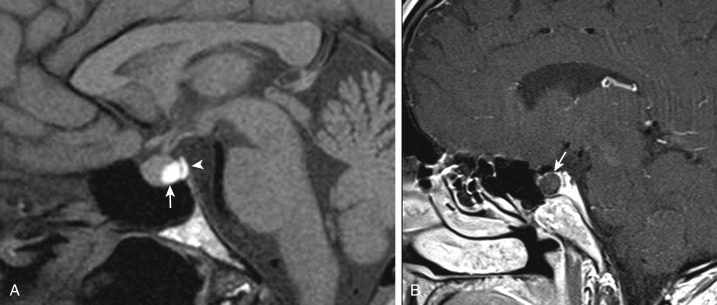
These children are microcephalic because of the reduced proliferation of neurons and glia in the germinal zones or increased apoptosis. The head circumference is 3 or more standard deviations below the norm. Neuroimaging shows the primary and secondary sulcations; however, the tertiary sulcations are not formed. The primary sulci are shallow grooves on the surface of the brain that become progressively more deeply infolded and that develop side branches, designated secondary sulci. Gyration proceeds with the formation of other side branches of the secondary sulci, referred to as tertiary sulci. The volume of the white matter in the cerebral hemispheres is reduced. This is a heterogenous group of disorders classified under six subgroups of increasing severity of neuroimaging findings. Groups 5 and 6 have the simplest looking gyral pattern and are named microlissencephaly ( Fig. 8-12 ).
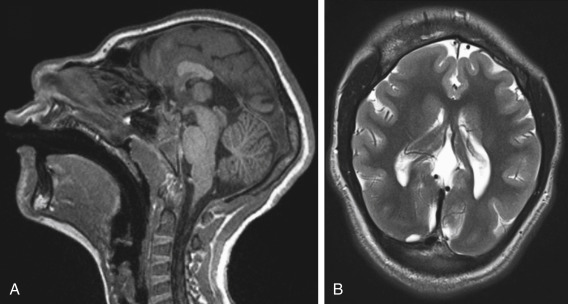
Hemimegalencephaly is hamartomatous overgrowth of all or part of a cerebral hemisphere with defects in neuronal proliferation, migration, and organization, and thus this seems to be an abnormal stem cell proliferation disorder. This is a heterogenous disorder which can occur in isolation, or more commonly associated with cutaneous abnormalities and hemihypertrophy. Hemimegalencephaly may occur in a variety of syndromes including epidermal nevus syndrome, Proteus syndrome, neurofibromatosis type 1 (NF-1), Soto syndrome, tuberous sclerosis, and Klippel-Trenaunay-Weber syndrome, to name a few ( Box 8-6 ). These children are typically macrocephalic at birth. Patients have seizures, hemiplegia, developmental delay, and abnormal skull configurations.
Neurofibromatosis type 1
Dyke-Davidoff-Masson syndrome/Sturge-Weber association
McCune Albright syndrome
Soto syndrome
Tuberous sclerosis
Klippel-Trenaunay-Weber syndrome
Proteus syndrome
Epidermal nevus syndrome
On MRI and CT, part or all of the affected cerebral hemisphere appears moderately to markedly enlarged. Typically, the gyri are broadened, thickened, and dysplastic; however, various degrees of poly/pachygyria and dysplasia of the cortex can be seen. White matter volume is increased and usually reveals heterogeneous signal either because of heterotopic gray matter or delayed myelination. Characteristically, the ipsilateral ventricle is enlarged with dysmorphic appearance of the frontal horn ( Fig. 8-13 ). This unique feature of ventricular dilatation on the side of the enlarged hemisphere separates congenital hemimegalencephaly from other infiltrative lesions. Interestingly, the radiological appearance may change over time. Patients with hemimegalencephaly of one hemisphere in infancy have been reported to develop atrophy of the affected hemisphere at 1 year of age. Reduced tracer uptake has been noted on iodoamphetamine single-photon emission computed tomography (IMP-SPECT), which has been attributed to episodes of status epilepticus. Although rare, associated enlargement and dysplasia of the cerebellum and brain stem can be seen, a condition named as total hemimegalencephaly.

Anatomical or functional hemispherectomy may be indicated in cases with intractable seizures, if the contralateral hemisphere is normal. Therefore, careful evaluation of the contralateral hemisphere is critical in these patients.
The term lissencephaly means “smooth brain” with an observed paucity of gyral and sulcal development of the surface of the brain. Agyria is a global absence of gyri along with thickened cortex and is the same entity as “complete lissencephaly,” whereas “pachygyria” refers to partial involvement in a hemisphere. Multiple genetic mutations have been identified in lissencephaly, and one of the most well-known is a defect of LIS1 gene at locus 17p13.3. Miller-Dieker syndrome also has a mutation in chromosome 17, and presents with a characteristic facies and lissencephaly on imaging. Mutation in the DCX gene (also known as XLIS ) in chromosome 22 is known as X-linked lissencephaly and accounts for about 75% of lissencephaly mutations. Appendicular and oropharyngeal spasticity develop with maturation of the CNS. The X-linked gene doublecortin (XLIS) predisposes to lissencephaly in boys and band heterotopia in girls (protected by two X chromosomes).
LIS1 and DCX mutations have similar neurological presentations, namely hypotonia at birth. Classic lissencephaly has characteristic neuroimaging features: smooth brain surface, diminished white matter and shallow/vertically oriented Sylvian fissures (thus the figure of eight or hour-glass appearance on axial images). The thin outer cortex is separated from the thick deeper cortical layers by a zone of white matter, called as “cell-sparce zone.” The trigones and occipital horns of the ventricles are dilated likely because of underdevelopment of the calcarine sulcus. The brain stem is small, likely secondary to lack of normally formed corticospinal and corticobulbar tracts ( Fig. 8-14 ).

Agyria/pachygyria typically results from abnormal neuronal migration. Compare this with polymicrogyria, which is a malformation of cortical development and involves interruptions in late neuronal migration and cortical organization. Keep in mind that the sulci in polymicrogyria are abnormal and do not correspond to any normal described in textbooks, whereas the sulci in pachygyria are normal in their location and can be identified neuroanatomically, although shallow and less in number. Patients with congenital cytomegalovirus (CMV) infection have high rates of pachygyria ( Fig. 8-15 ). As opposed to hemimegalencephaly, white matter volume is decreased. The gray matter appears thicker because of poor sulcation. Abnormal myelination may coexist.
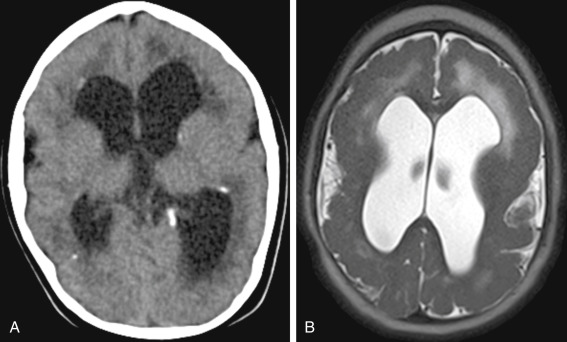
Heterotopia is disorganized brain tissue, and it is usually gray matter that is located in the wrong place. Heterotopias form when migration of the neuroblasts from the periventricular region to the pia is thwarted, possibly because of damage to the radial glial fibers, which orient migrating neurons. The classification of heterotopias is usually divided into two varieties; nodular and band types.
The “band heterotopia” (double cortex) may present at any age, but usually in childhood with variable degrees of developmental delay and mixed seizure disorders. Seizures are usually on the milder side. The DCX mutation is the most common genetic abnormality with a strong female preponderance (>90%). As the name implies, band heterotopia is a homogenous band of gray matter located between the cortex and white matter, separated from the ventricles and cortex by normal appearing white matter. The band heterotopia may be complete or partial. Partial frontal lobe bands are more common in females whereas posterior involvement is more common in males. The overlying cortex may be normal or malformed. Look for gray, white, gray, white, ventricle as the pattern for band heterotopias—it is the second gray that is the rub ( Fig. 8-16 ).

Under nodular heterotopias, you will find subependymal and subcortical variants. Patients with subependymal heterotopias can be divided in two groups: the larger group of patients have symmetric and fewer subependymal heterotopias usually confined to the trigones and temporal/occipital horns ( Fig. 8-17 ). These are rarely familial but can be seen in cases with Chiari II malformations, callosal anomalies and cephaloceles. The smaller group of patients may have familial either X-linked or autosomal recessive patterns of inheritance. Mutations in several genes (such as Filamin -1 gene on the long arm of the X chromosome) may cause subependymal heterotopias. Subependymal heterotopias appear as smooth, ovoid masses that are isointense to gray matter on all imaging sequences (see Fig. 8-17 ). The long axis of the heterotopias is parallel to the ventricular wall and the heterotopias do not show evidence of enhancement on postcontrast images nor do they have perilesional edema. They can grow exophytically, extending into the ventricle, and if big enough may have mass effect on the ventricle (see Fig. 8-17 ). Hyperintensity on T1WI may be due to dystrophic microcalcifications and these may show hyperdensity at CT.
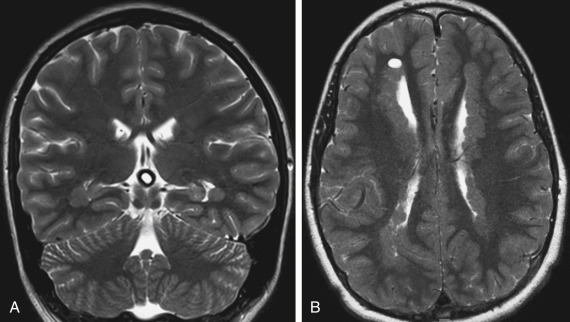
Patients with subcortical heterotopias often have abnormal sulcation patterns superficial to the heterotopia. The hemisphere ipsilateral to the site of the subcortical heterotopia may be smaller with thinning of the overlying cortex. Subcortical heterotopias may be nodular or curvilinear in shape. The nodular variety of subcortical heterotopias are usually identified in a periventricular or subcortical location, whereas the diffuse (or laminar) heterotopias are seen more commonly in or close to the cortex. Imaging features are similar to that of subependymal heterotopias. Encephaloceles, holoprosencephaly, schizencephaly, Chiari malformations, and agenesis of the corpus callosum may coexist with gray matter heterotopias. If you see one, actively search for other anomalies!
Congenital muscular dystrophies (CMD) are included in this group of malformations, characterized by hypotonia at birth, generalized muscle weakness, joint contractures, and other CNS anomalies. Fukuyama type was the first described in this group, an autosomal recessive condition prevalent in Japan. Walker-Warburg syndrome and muscle-eye-brain disease have brain and ocular anomalies.
There is considerable overlap in pathologic and imaging findings. Walker-Warburg syndrome patients have cobblestone lissencephaly, congenital hydrocephalus, severe congenital eye malformations (microphthalmus, retinal dysplasia, persistent hypoplastic primary vitreus, optic nerve hypoplasia), and may have occipital encephaloceles. The cortex is thick with very few sulci. Cobblestone lissencephaly refers to the quite distinctively irregular gray-white matter junction possible reflecting the disorganized neurons interrupting the white matter. More severe cases have pontine hypoplasia with a distinctive kink at the mesencephalic-pontine junction because of cerebellar hypoplasia/dysplasia. Fukuyama Congenital Muscular dystrophy is characterized by frontal polymicrogyria and temporooccipital cobblestone cortex. In addition, the cerebellum is dysplastic and with subcortical cysts. Muscle-eye-brain disease shows similar features to that of above two described entities; however, the severity is somewhat intermediate.
Cortical dysplasia may be a source of seizures and motor deficits. The findings may be very subtle, manifested as mild focal volume loss, thickening of cortex, abnormal sulcation, and blurring of gray-white matter junction ( Fig. 8-18 ). The CSF cleft overlying a cortical dimple is a specific sign of cortical dysgenesis. The signal intensity of the dysplastic cortex and underlying white matter may change with age; therefore, multiple studies may be required to find the focal cortical dysplasia (FCD) in young children. Cortical dysplasia can be associated with other conditions such as neuroglial tumor (dysembryoplastic neuroepithelial tumors) and hippocampal sclerosis.
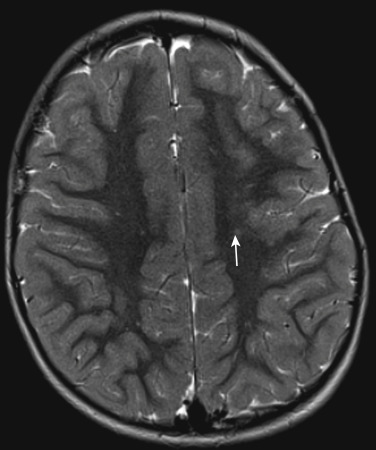
FCD with balloon cells (a histopathologic term) consists of a cortex with abnormal lamination, abnormal cells (dysplastic neurons and balloon cells), and focal signal abnormality that extends from the gray-white matter junction to the ventricular surface (which led to the term transmantle cortical dysplasia). Cortical thickening, hyperintense subcortical white matter on T2WI (likely because of hypomyelination and astrogliosis), and radial bands from the ventricle to the cortex are seen ( Fig. 8-19 ). These bands may be of white or gray matter signal intensity. The gray-white junction is blurred.
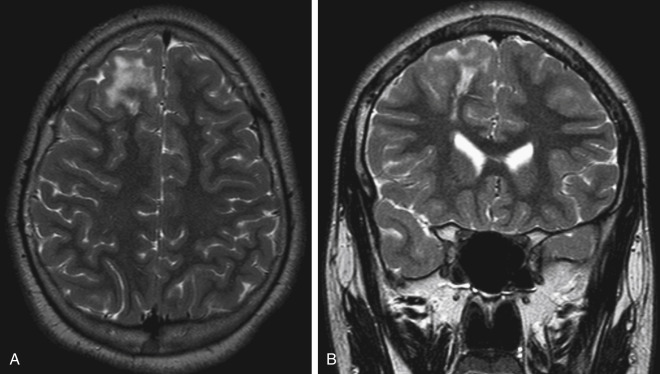
A frontal lobe location favors a balloon cell dysplasia, whereas a temporal lobe (especially medial temporal lobe) location is more suggestive of a neoplasm. Rarely, one may see anomalous venous drainage from areas of cortical dysplasia. Both radiologic and histologic findings may overlap with cortical tubers seen in tuberous sclerosis, therefore genetics and pediatric neurology consult may be advisable in these cases.
Hamartomas represent an abnormal proliferation of disorganized but mature cells, usually a combination of neurons, glia and blood vessels in an abnormal location. Whereas the heterotopias are due to anomalous neuronal migration, hamartomas are a nonneoplastic proliferation of brain tissue. There is a propensity for hamartomatous formation in the hypothalamus typically located between the mammillary bodies and the tuber cinereum of the hypothalamus ( Fig. 8-20 ). Boys are more commonly affected than girls. These patients typically present with precocious puberty (before 2 years of age in boys and slightly later in girls) and gelastic (laughing spells) seizures. However, occasionally, visual disturbances may be present, because the hypothalamic hamartoma involves the optic pathways.
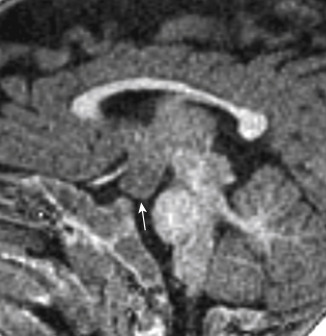
Because the tissue that makes up a hamartoma is essentially normal brain substance, the hamartoma is isodense with the gray matter on CT. On MR, the hamartoma is isointense to gray matter on T1WI and variable on T2WI. The lesion is identified as a bulbous protrusion of the hypothalamic region in the midline. The hamartomas have a normal blood-brain barrier and are not expected to show enhancement on either CT or MR. Occasionally, mass effect may be associated with the hamartoma as evidenced by displacement of the inferior portion of the third ventricle.
After the hypothalamic region, the next most common location for hamartomas is the cerebral cortex-subcortical region. Occasionally, hamartomas may be seen in a periventricular location. Fetus in fetu refers to duplication of brain structures, usually seen as an extraaxial frontal region mass. The signal intensity approaches that of normal brain. Case studies of ectopic brain in the nasopharynx or pterygopalatine fossa have also been reported.
Polymicrogyria is a malformation of cortical development which results from interruptions in later neuronal migration and neuronal organization. Histologically, there is derangement of the six-layered lamination of the cortex with an associated derangement of sulcation. Therefore, in polymicrogyria, no normal sulci are seen. Patients may present with developmental delay, focal neurologic signs/symptoms or epilepsy at any age. CMV infection, in utero ischemia, or chromosomal mutations can be associated with polymicrogyria. It can be focal, multifocal or diffuse; it can be unilateral or bilateral. Posterior sylvian fissure and frontal lobe are common locations for this sneaky abnormality.
On imaging, one can see excessive numbers of small, disorganized cortical convolutions. The cortex appears thickened. The white matter thickness is normal (remember that it is increased in hemimegalencephaly and smaller in agyria). To distinguish pachygyria from polymicrogyria, check the cortex; the cortex is less thick with polymicrogyria than with true pachygyria (>5 mm thick). On thin section images polymicrogyria is “bumpier” than pachygyria. Cortex may appear buckled or with an inward folding. Another distinguishing feature between pachygyria and polymicrogyria is the possible presence of abnormal deep white matter in the latter ( Fig. 8-21 ). Keep in mind that the degree of myelination affects the imaging appearance. In unmyelinated areas, the polymicrogyria looks thin, whereas in myelinated areas the thickness may reach 5 mm or more with a smoother looking outer cortex. The polymicrogyria tends to involve the frontal and parietotemporal lobe, whereas pachygyria tends to involve the temporooccipital lobes. An association with developmental venous anomalies (anomalous venous drainage) is noted with polymicrogyria as with other dysplastic cortices.
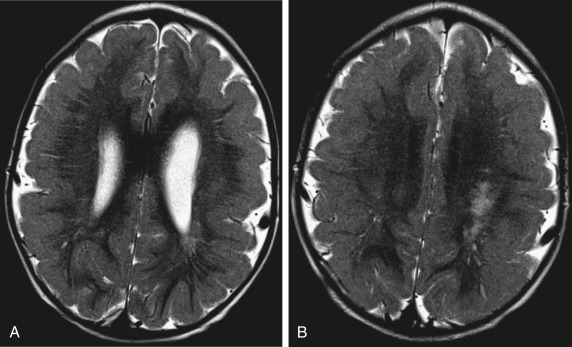
Congenital bilateral perisylvian (opercular) syndrome is recognized as an entity in which there is polymicrogyria involving the opercular cortex associated with abnormal sylvian fissure sulcation ( Fig. 8-22 ). The inheritance pattern is heterogenous. Patients with congenital bilateral perisylvian syndrome disorder have seizures, congenital pseudobulbar paresis, and developmental delay. The abnormal sylvian fissure may have cortical thickening on either side of it. Schizencephaly may also be present.
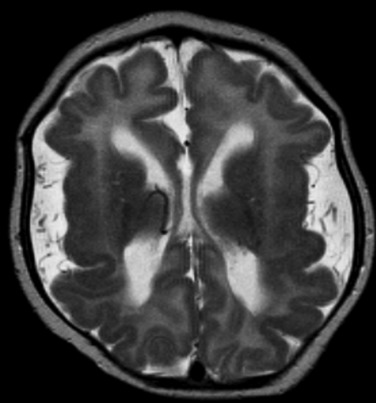
Bilateral symmetrical frontoparietal polymicrogyria syndrome and bilateral medial parietooccipital polymicrogyria syndrome have also been described.
Schizencephaly is gray matter lined cleft that extend from the pial covering of the cortex to the ependymal lining of the lateral ventricle. They are difficult to classify, because the malformation likely occurs during neuronal cell proliferation, migration, and organization. Give the toss-up, we left them in the organization section. Both genetic and acquired causes are considered in the etiology.
The abnormality can be unilateral or bilateral, with closed lips or open lips. Bilateral involvement (∼%40) is associated with seizures, worse developmental delay, and developmental dysphasia. Motor dysfunction is more common with frontal lobe schizencephaly, open lipped varieties, and wider gaps in the open lips.
The cleft has a dysplastic gray matter lining with abnormal lamination that looks like polymicrogyria and is usually seen in the supratentorial space (near the precentral and postcentral gyrus) coursing to the lateral ventricles. Interestingly, the common locations follow that of polymicrogyria. Schizencephaly occurs most commonly in the frontal (44%), frontoparietal (30%), and occipital (19%) lobes. The gray-white matter junction is irregular along the clefts. The lips of the cleft may be opposed (“closed lipped”) or gaping (“open lipped”) ( Fig. 8-23 ). The closed lipped variety may be missed if the clefts are tightly apposed, but a dimple at the ventricle-cleft interface should suggest the diagnosis. The gyral pattern of the cortex adjacent to the schizencephaly is usually abnormal.
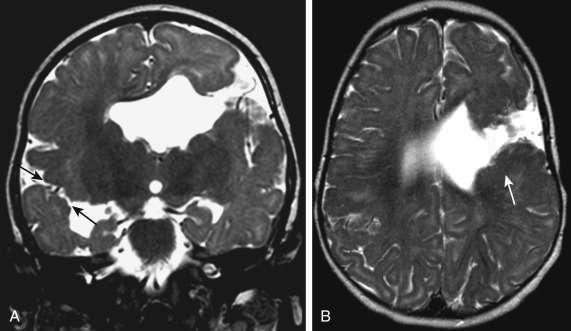
Optic nerve hypoplasia is seen in one third of the patients. Optic nerve hypoplasia with high absence of septum pellucidum can result in some patients getting categorized under septooptic dysplasia.
Schizencephaly is often associated with FCD, gray matter heterotopias, agenesis of the septum pellucidum (∼80%), and pachygyria. If bilateral clefts are present, then the septum pellucidum is absent. The lining of the cleft with dysplastic gray matter, best seen on MR, is the differential point in this lesion and distinguishes it from ischemic/encephalomalacic abnormalities, which are usually lined by white matter ( Fig. 8-24 ). Prenatal open lip clefts may become closed lip postnatally in 50% of cases. The inner surface of the cleft is pia-lined and communicates with the ependyma of the ventricle, which differentiates the lesion from an enlarged sylvian fissure because of volume loss such as those seen in premature infants.
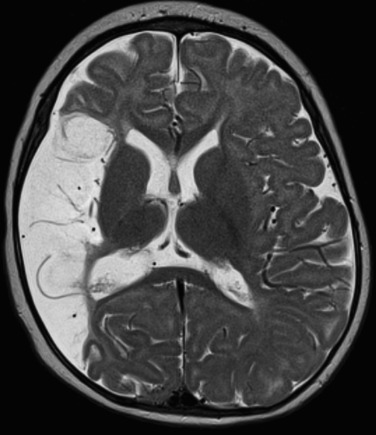
Anencephaly is failure of anterior neural tube closure. Anencephaly ( Fig. 8-25 ) is now a prenatal diagnosis because most women are screened relatively early in pregnancy for elevated levels of serum a-fetoprotein, a marker of neural tube defects. The diagnosis by obstetric ultrasound (US) is made when the cranial vault is seen to be small, with only the fetus’s face and posterior fossa well seen. Only a nubbin of tissue is seen at the skull base on ultrasound, and amniotic fluid α-fetoprotein levels are elevated. These babies die soon after delivery. An association with spinal dysraphism exists.
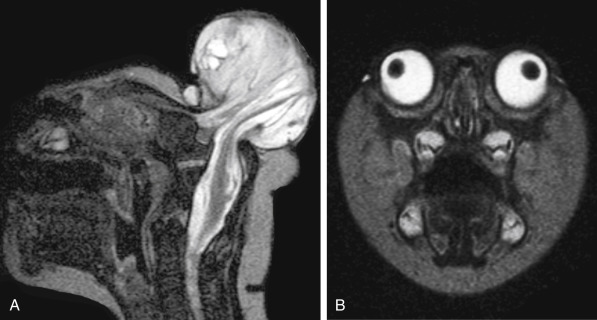
Inherited (genetic) or acquired (disruptive) causes are identified in congenital abnormalities of the posterior fossa. It is important to differentiate the disruptive causes, since the chance of recurrence in other offspring is very limited. Disruptive abnormalities most commonly result from prenatal infections, hemorrhage and ischemia. If one identifies a unihemispheric abnormality of the cerebellum, disruptive causes should be considered.
For practical purposes, posterior fossa malformation are classified based on the neuroimaging pattern: (1) predominantly cerebellar, (2) cerebellar and brain stem, (3) predominantly brain stem, and (4) predominantly midbrain. We will focus on the first two in this classification because they cover the most common malformations of the posterior fossa. Evaluation of vermis is of great importance, so make good use of your mid-sagittal images! Always pay attention to the size and form of the fourth ventricle, and everything else around it.
The cerebellum consists of the vermis and two cerebellar hemispheres. The cerebellum may be hypoplastic (small volume), dysplastic (abnormal foliation and architecture of the cerebellar white matter), or a combination of both, involving the entire cerebellum, limited to the vermis only, or limited to the cerebellar hemispheres only.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here