Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Anemia is defined as a reduction in hemoglobin concentration, hematocrit, or red cell mass by more than two standard deviations below the mean for age and sex for the normal population. The normal ranges are also affected by geographic ancestry; African Americans have lower hemoglobin concentration on average (by ~0.5 g/dL) than people of European ancestry. As a result, 2.5% of the general population and up to 10% of African Americans will be classified as anemic, especially if race-specific normal ranges are not used. Also note that children with cyanotic congenital heart disease, chronic respiratory insufficiency, arteriovenous pulmonary shunts, or hemoglobinopathies that alter oxygen affinity can be functionally anemic with hemoglobin levels in the normal or reference range.
Anemia can be an isolated abnormality or be a part of multiple cell line abnormalities (red cells, white cells, and platelets). Abnormalities of two- or three-cell lines may indicate one of the following:
bone marrow involvement (e.g., infections, aplastic anemia, leukemia, toxicity from medications);
autoimmune disorders (e.g., connective tissue disease, Evans syndrome);
sequestration (e.g., hypersplenism) or intravascular trapping and destruction (e.g., thrombotic microangiopathy).
Anemia can be classified based on morphology (e.g., size and shape of RBCs) or physiology (i.e., mechanism of anemia). Physiologically, anemia can be categorized into:
disorders of decreased red cell formation: this can be due to failure of erythropoiesis in which there is an absolute erythroblastopenia and reticulocytopenia (e.g., marrow failure diseases) or due to ineffective erythropoiesis in which the marrow has many erythroblasts but with defective maturation (e.g., thalassemias);
disorders of erythrocyte destruction (hemolysis);
loss of red blood cells (hemorrhage).
Table 3.1 presents an etiologic classification of anemia and the diagnostic features.
The blood smear is an essential first step in the diagnosis of anemia. Anemia can be classified based on RBC morphology and RBC size into microcytic [decreased mean corpuscular volume (MCV)], normocytic (normal MCV), and macrocytic (high MCV). The mean corpuscular hemoglobin (MCH) and MCH concentration (MCHC) are calculated values and have complementary diagnostic value. The MCH usually parallels the MCV. The MCHC is a measure of cellular hydration status. A high value (>35 g/dL) due to membrane loss is characteristic of spherocytosis, and a low value is commonly associated with iron deficiency. Fig. 3.1 delineates the causes of anemia based on peripheral blood morphology, and Table 3.2 shows the cause of anemia based on MCV and red cell distribution width (RDW), an index of the variation in red cell size (anisocytosis).
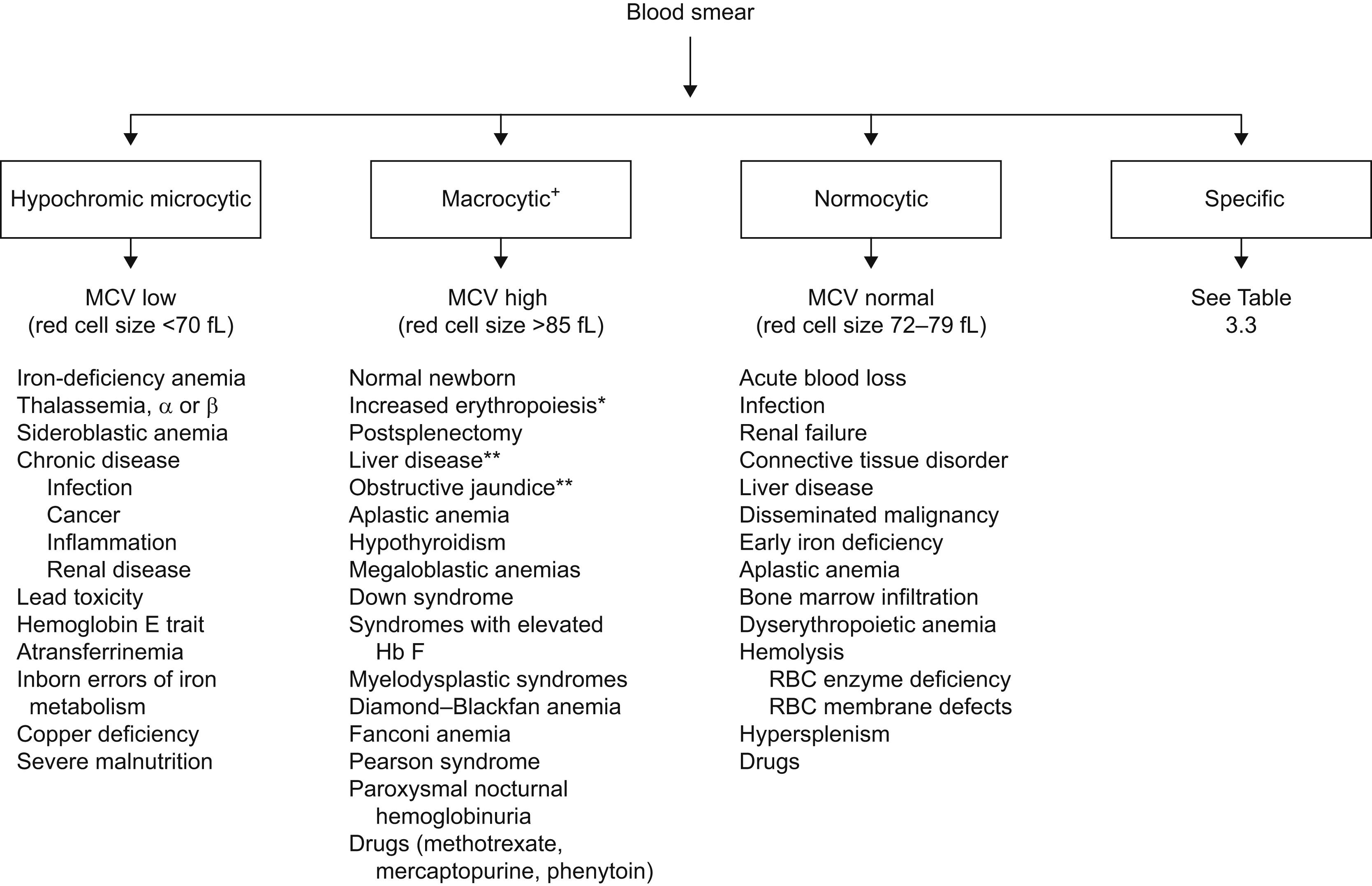
| MCV low | MCV normal | MCV high | |
| RDW normal | Microcytic homogeneous | Normocytic homogeneous | Macrocytic homogeneous |
| Heterozygous thalassemia | Normal | Inherited bone marrow failure syndromes | |
| Chronic disease | Chronic disease | "Preleukemia" or myelodysplastic syndrome | |
| Chronic liver disease | |||
| Chemotherapy | |||
| Chronic myelocytic leukemia | |||
| Hemorrhage | |||
| Hereditary spherocytosis | |||
| RDW high | Microcytic heterogeneous | Normocytic heterogeneous | Macrocytic heterogeneous |
| Iron deficiency | Early iron or folate deficiency | Folate deficiency | |
| HbS/β-thalassemia | Mixed deficiencies | Vitamin B 12 deficiency | |
| Hemoglobin H | Hemoglobinopathy (e.g., Hb SS) | Immune hemolytic anemia | |
| Red cell Fragmentation disorders | MyelofibrosisSideroblastic anemia | Cold agglutinins | |
In addition, the blood smear may show specific morphologic abnormalities suggestive of red cell membrane disorders (e.g., spherocytosis, stomatocytosis, or elliptocytosis) or hemoglobinopathies (e.g., sickle cell disease and thalassemia). Table 3.3 lists the differential diagnosis of anemia based on the specific red cell morphological abnormalities.
Target cells— Increased surface/volume ratio (generally does not affect red cell survival)
Spherocytes— Decreased surface/volume ratio, hyperdense (>MCHC)
Acanthocytes (spur cells)— Cells with 5–10 spicules of varying length; spicules irregular in space and thickness, with wide bases; appear smaller than normal cells because they assume a spheroid shape
Echinocytes (burr cells)— 10–30 spicules equal in size and evenly distributed over RBC surface; caused by alteration in extracellular or intracellular environment
|
| Pyknocytes— Distorted, hyperchromic, contracted RBC; can be similar to echinocytes and acanthocytes |
Schistocytes— Helmet, triangular shapes, or small fragments. Caused by fragmentation upon impact with abnormal vascular surface (e.g., fibrin strand, vasculitis, artificial surface in circulation)
Elliptocytes— Elliptical cells, normochromic; seen normally in less than 1% of RBCs; larger numbers occasionally seen in a normal patient
Teardrop cells— Shape of drop, usually microcytic, often also hypochromic
Stomatocytes— Has a slit-like area of central pallor
Nucleated red blood cells— Not normal in the peripheral blood beyond the first week of life
Blister cells —Red cell area under membrane free of hemoglobin, appearing like a blister
Basophilic stippling— Coarse or fine punctate basophilic inclusions that represent aggregates of ribosomal RNA
Howell–Jolly bodies— Small, well-defined, round, densely stained nuclear-remnant inclusions; 1 mm in diameter; centric in location
Cabot’s ring bodies— Nuclear-remnant ring configuration inclusions
Heinz bodies— Denatured aggregated hemoglobin
|
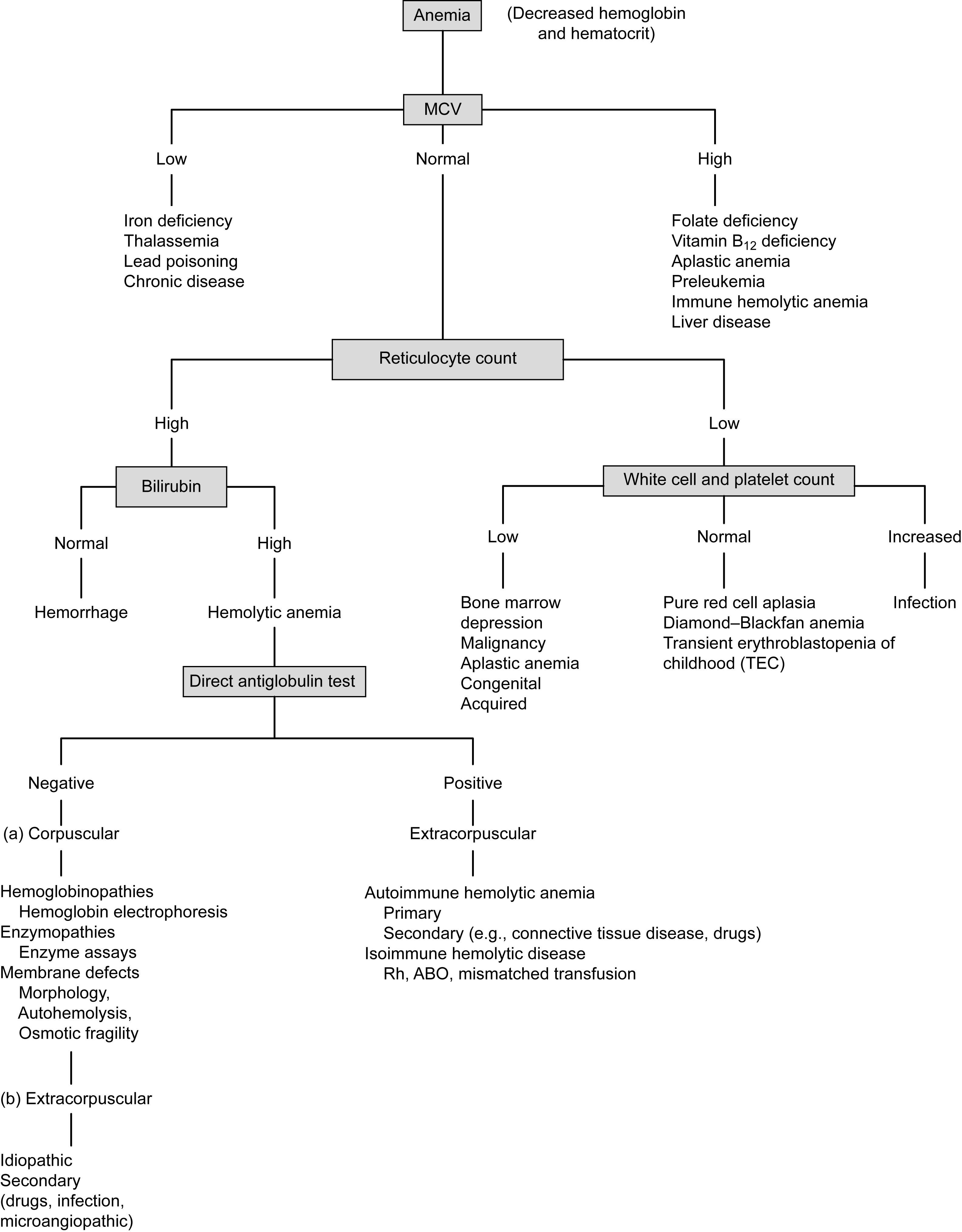
The reticulocyte count is an important indicator of the physiology of anemia ( Fig. 3.2 ). An elevated reticulocyte count suggests blood loss or hemolysis, while a normal or decreased count suggests impaired red cell formation. With acute blood loss or sequestration, the elevation of the reticulocyte count may take many hours to become apparent and several days (4–5) to reach its maximum. The reticulocyte count must be interpreted in the context of the degree of anemia, using either the absolute reticulocyte count or reticulocyte index, to assess the adequacy of erythropoiesis. In patients with bleeding or hemolysis, the reticulocyte index should be at least 3%, whereas in patients with anemia due to decreased production of red cells, the reticulocyte index is <3% and frequently <1.5%.
| Etiologic classification | Diagnostic features |
|---|---|
|
|
|
Hypochromic, microcytic red cells; low MCV, low MCH, low MCHC, high RDW, a low serum ferritin, high FEP |
|
Macrocytic red cells, high MCV, high RDW, megaloblastic marrow, low serum, and red cell folate, high homocysteine and normal methylmalonic acids |
|
Macrocytic red cells, high MCV, high RDW, megaloblastic marrow, low serum B 12 , decreased gastric acidity, high homocysteine, and high methylmalonic acids |
|
Clinical scurvy |
|
Kwashiorkor |
|
Hypochromic red cells, sideroblastic bone marrow, high serum ferritin |
|
Clinical hypothyroidism, low free T 4 , high TSH |
|
|
| Amegakaryoctic thrombocytopenic purpura with absent radii (TAR) | Limb abnormalities, thrombocytopenia, absent megakaryocytes |
|
|
| Congenital red cell aplasia (Diamond–Blackfan anemia) | Absent red cell precursors |
| Acquired red cell aplasia (TEC) | Absent red cell precursors |
|
|
| Congenital neutropenias | Neutropenia, recurrent infection |
|
|
|
|
| Fanconi anemia | Multiple congenital anomalies, chromosomal breakage |
| Familial without anomalies | Familial history, no congenital anomalies |
| Dyskeratosis congenita | Mucosal and cutaneous abnormalities |
|
|
| Idiopathic | No identifiable cause |
| Secondary | History of exposure to drugs, radiation, household toxins, infections (parvovirus B19, HIV) associated with immunologic disease |
|
|
|
|
|
Bone marrow: morphology, cytochemistry, immunologic markers, cytogenetics, molecular features |
|
VMA, imaging studies, skeletal survey, bone marrow |
|
|
|
Evidence of systemic illness |
|
kidney and liver function tests |
|
Clinical evidence |
|
Rheumatoid arthritis |
|
Clinical evidence |
|
Hypochromic anemia, ring sideroblasts |
|
|
|
Splenomegaly, jaundice |
|
Morphology, osmotic fragility |
|
Enzyme assays |
|
|
|
|
|
|
| Qualitative (e.g., sickle cell)Quantitative (e.g., thalassemia) | Hb electrophoresisQuantitative HbF, HbA 2 content |
|
|
|
Direct antiglobulin test (Coombs’ test) |
|
Decreased C 3 , C 4 , CH 50 , positive ANA |
| Idiopathic | Direct antiglobulin test, antibody identification |
| Secondary | |
| Immunologic disorder (e.g., lupus) | |
| One-cell line (e.g., red cells) | Anemia—direct antiglobulin test positive |
| Multiple cell line (e.g., white blood cells, platelets) | Evans syndrome: neutropenia—autoimmune neutropenia, thrombocytopenia—ITP |
|
|
a RDW—coefficient of variation of the RBC distribution width (normal between 11.5% and 14.5%).
Table 3.4 lists various laboratory studies helpful in the investigation of a patient with anemia.
The investigation of anemia entails the following steps:
Detailed history . Table 3.1 lists the various causes of anemia with the associated diagnostic laboratory and clinical features.
Complete blood count to establish whether anemia is isolated or part of a multilineage abnormality (abnormality of red cell count, white blood cell count, and platelet count). Determination of the morphologic characteristics of the anemia based on blood smear ( Fig. 3.1 and Table 3.3 ) and consideration of the MCV ( Fig. 3.2 and Table 3.2 ) and RDW ( Table 3.2 ) and white blood cell and platelet morphology .
Reticulocyte count as a reflection of erythropoiesis ( Fig. 3.2 ).
Determination of whether there is evidence of a hemolytic process by:
consideration of the clinical features suggesting hemolytic disease ( Table 3.5 ),
|
laboratory demonstration of the presence of hemolysis ( Table 3.4 ), and
determination of the precise cause of the hemolytic anemia by special hematologic investigations ( Table 3.4 ).
Bone marrow aspiration and biopsy , if required, to examine erythroid, myeloid, and megakaryocytic morphology to determine whether there is normoblastic, megaloblastic, or sideroblastic erythropoiesis and to exclude marrow pathology (e.g., aplastic anemia, leukemia, and benign or malignant infiltration of the bone marrow) ( Fig. 3.3 ).
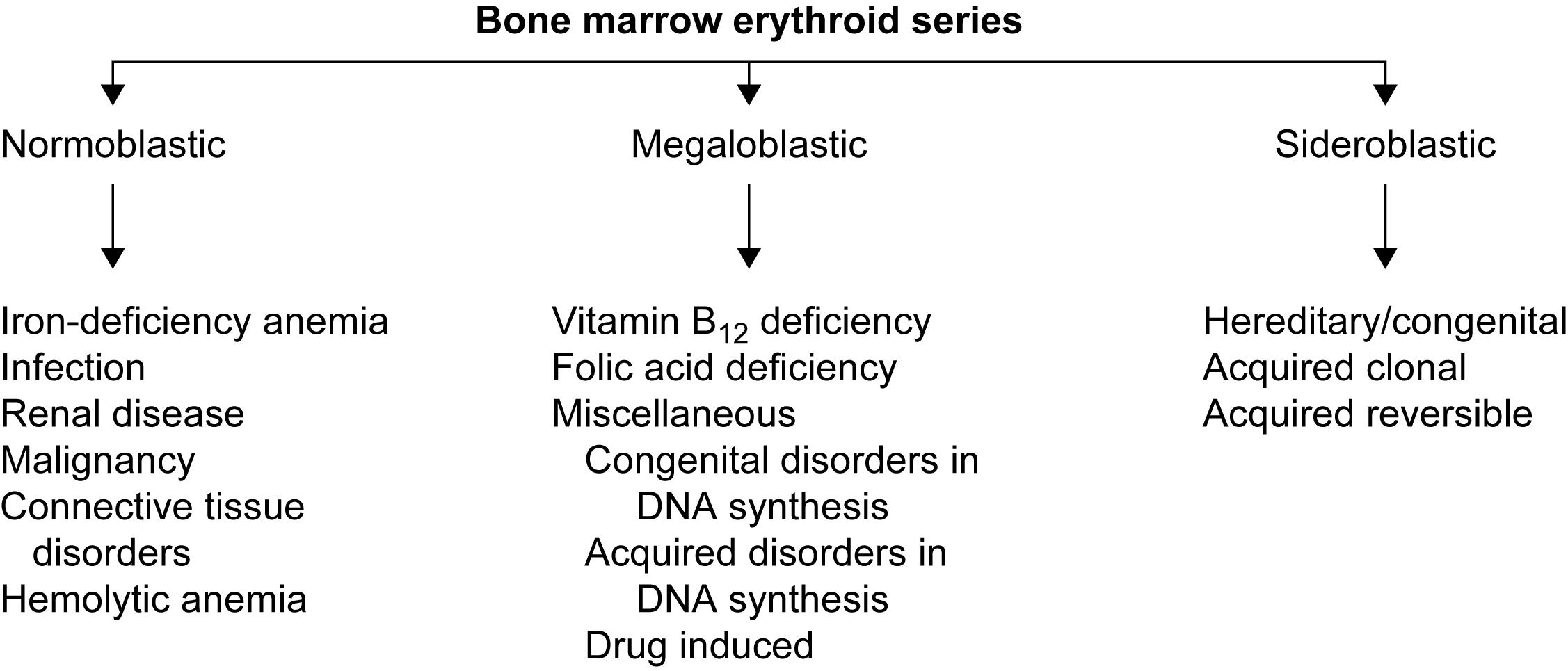
Determination of underlying cause of anemia by additional tests ( Table 3.4 ).
Usual initial studies
Suspected iron deficiency
Suspected vitamin B 12 or folic acid deficiency
Suspected hemolytic anemia
Suspected aplastic anemia or leukemia
Other tests often used especially to diagnose the primary disease
|
Anemia during the neonatal period can be caused by:
hemorrhage : acute or chronic;
hemolysis : congenital hemolytic anemias or due to immune hemolytic anemias; and
hypoplasia : failure of red cell production in inherited bone marrow failure syndromes, for example, Diamond–Blackfan anemia (pure red cell aplasia) or congenital infections.
Table 3.6 lists the causes of anemia in the newborn.
|
Blood loss may occur during the prenatal, intrapartum, or postnatal periods. Prenatal blood loss may be transplacental, intraplacental, or retroplacental or may be due to a twin-to-twin transfusion.
Fetal red blood cells can be demonstrated in the maternal circulation in up to 50% of pregnancies; however, a clinically significant hemorrhage (>30 mL) is seen in only 1–2% of pregnancies. Significant fetomaternal hemorrhage is commonly seen following procedures such as diagnostic amniocentesis or external cephalic version. Also, intraplacental blood loss from the fetus may occur when there is a tight umbilical cord around the neck or body or when there is delayed cord clamping. Retroplacental bleeding from placental abruption is diagnosed by ultrasound or intraoperatively. Fetomaternal blood loss may be acute or chronic. Table 3.7 lists the characteristics of acute and chronic blood loss in the newborn.
| Characteristic | Acute blood loss | Chronic blood loss |
|---|---|---|
| Clinical | ||
| Acute distress; pallor; shallow, rapid, and often irregular respiration; tachycardia; weak or absent peripheral pulses; low or absent blood pressure; no hepatosplenomegaly | Marked pallor disproportionate to evidence of distress. On occasion signs of congestive heart failure may be present, including hepatomegaly | |
| Venous pressure | Low | Normal or elevated |
| Laboratory | ||
| Hemoglobin concentration | May be normal initially; then drops quickly during the first 24 h of life | Low at birth |
| Red cell morphology | Normochromic and normo- or macrocytic | Hypochromic and microcytic, anisocytosis, and poikilocytosis |
| Serum iron | Normal at birth | Low at birth |
| Course | Prompt treatment of anemia and shock | Generally uneventful |
| Treatment | Intravenous fluids or packed red blood cells. If indicated, iron therapy | Iron therapy. Packed red blood cells on occasion |
Fetomaternal hemorrhage is diagnosed by demonstrating fetal red cells by flow cytometry using an antibody against HbF (fetal hemoglobin)] in the maternal circulation. Less commonly, an older, differential acid elution technique (Kleihauer–Betke method) may still be used. Diagnosis of fetomaternal hemorrhage may be missed in situations in which red cells of the mother and infant have incompatible ABO blood groups. In such instances the infant’s incompatible red blood cells are rapidly cleared from the maternal circulation by maternal anti-A or anti-B antibodies. In these cases an increase in maternal immune anti-A or anti-B titers in the weeks after delivery, if measured, may be a diagnostic clue. The optimal timing for demonstrating fetal cells in maternal blood is within 2 hours of delivery and no later than 24 hours following delivery. These techniques are not reliable when maternal HbF is raised for other reasons (e.g., maternal thalassemia, sickle cell anemia, or hereditary persistence of HbF). In the presence of these conditions, other techniques based on differential agglutination have been used, but the fetomaternal hemorrhage is usually a clinical diagnosis (a diagnosis of exclusion) in such cases.
Significant twin-to-twin transfusion occurs in at least 15% of monochorionic twins. Velamentous cord insertions are associated with increased risk of twin-to-twin transfusion. The hemoglobin level differs by 5 g/dL and the hematocrit by 15% or more between individual twins compared to discrepancy ≤3.3 g/dL in cord blood hemoglobin between dizygotic twins. The donor twin is anemic, pale, and smaller and may have evidence of oligohydramnios and show evidence of congestive heart failure and shock. The recipient is polycythemic and larger, with evidence of polyhydramnios, and may show signs of hyperviscosity syndrome: hypoglycemia, central nervous system injury, hypocalcemia, disseminated intravascular coagulation, hyperbilirubinemia, and congestive heart failure.
Hemorrhage may occur during birth as a result of various obstetric accidents, malformations of the umbilical cord or the placenta, or a bleeding disorder (e.g., coagulation factor deficiency or thrombocytopenia; Table 3.6 ).
Postnatal hemorrhage may occur from a number of sites and may be internal (enclosed) or external. Hemorrhage may be due to:
traumatic deliveries (resulting in intracranial or intraabdominal hemorrhage)
coagulation factor deficiencies (see Chapter 13 : Disorders of Coagulation)
congenital—hemophilia or other coagulation factor deficiencies
acquired—vitamin K deficiency, disseminated intravascular coagulation
thrombocytopenia (see Chapter 12 : Disorders of Platelets)
congenital—Wiskott–Aldrich syndrome, Fanconi anemia, thrombocytopenia absent radius syndrome
acquired—neonatal alloimmune thrombocytopenia, maternal immune thrombocytopenia, sepsis
rare causes—neonatal adenovirus infection, fetal cytomegalovirus infection, vascular malformations
When hemoglobin is catabolized in a resorbing hematoma, hyperbilirubinemia may develop after several days.
The clinical and laboratory manifestations of hemorrhage depend on the volume of the hemorrhage and the rapidity with which it occurs.
Anemia—pallor, tachycardia, and hypotension (if severe, e.g., ≥20 mL/kg blood loss). Nonimmune hydrops can occur in severe anemia.
Liver and spleen not enlarged (except in chronic transplacental bleed).
Jaundice absent (except after several days in entrapped hemorrhage).
Laboratory findings:
reduced hemoglobin (as low as 2 g/dL has been observed),
increased reticulocyte count,
polychromatophilia,
nucleated RBCs raised,
fetal cells in maternal blood (in fetomaternal bleed), and
direct antiglobulin test (DAT) negative.
Severely affected
Transfusion of packed red blood cells.
Crossmatch blood with the mother. If unavailable, use group O Rh-negative blood or intravenous fluids, temporarily for shock, while awaiting available blood.
Mild anemia due to chronic blood loss
Ferrous sulfate (4–6 mg elemental iron/kg body weight per day) for 3 months.
Hemolytic anemia in the newborn is usually associated with an abnormally low hemoglobin level, an increase in the reticulocyte count, and unconjugated hyperbilirubinemia. The degree of reticulocytosis may be less than anticipated depending on the age of neonate or infant, because any hemolytic anemia will be superimposed on the physiologic anemia of infancy. The hemolytic process is often first detected as a result of investigation for jaundice during the first week of life. The causes of hemolytic anemia in the newborn are listed in Table 3.6 .
Congenital erythrocyte defects involving the red cell membrane, hemoglobin, and enzymes are listed in Table 3.6 and discussed in Chapter 7 , General Considerations of Hemolytic Diseases, Red Cell Membrane, and Enzyme Defects, and Chapter 8 , Extracorpuscular Hemolytic Anemia. Any of these conditions may occur in the newborn and manifest clinically with:
hemolytic anemia (low hemoglobin, reticulocytosis, increased nucleated red cells, morphologic changes);
unconjugated hyperbilirubinemia; and
DAT negative.
The cause of this condition has not been clearly defined and should only be contemplated when other established causes of pyknocytes in the blood have been excluded such as glucose-6-phosphate dehydrogenase (G6PD) deficiency, pyruvate kinase deficiency, microangiopathic hemolytic anemia, neonatal hepatitis, vitamin E deficiency, neonatal infections, and hemolysis caused by drugs and toxic agents. It is a congenital but not a constitutional disorder; that is, one is born with it (congenital) but it is not an ongoing, lifelong, or necessarily genetic condition (not constitutional). Infantile pyknocytosis is characterized by:
Hemolytic anemia—DAT negative (nonimmune).
Distortion of as many as 50% of red cells–dense, contracted cells (pyknocytes) with several to many spiny projections (up to 6% of cells may be distorted in normal infants). Abnormal morphology is extracorpuscular in origin, and transfused red blood cells from normal donors can acquire pyknocytosis.
Disappearance of pyknocytes and hemolysis by the age of 6 months. This is a self-limiting condition.
Hepatosplenomegaly.
The developmental changes in globin proteins uniquely impact the presentation of hemoglobinopathies in neonates. For example, anemia due to gamma-hemoglobinopathies resolves spontaneously, whereas beta hemoglobinopathies are clinically inapparent at birth and manifest only after a few months. Anemia from alpha-hemoglobinopathies can occur throughout life (prenatal and postnatal) (see Chapter 9 : Hemoglobinopathies).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here