Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Microparticle (MP) analysis has become popular in the last decade because of mounting evidence of their active role in multiple cellular processes. Alterations in total MP numbers or changes in the relative abundance of MP populations have been correlated with multiple diseases. It is still unknown whether these MP alterations are markers of disease or whether MPs play an active role in the pathology, progression, or modulation of these diseases. Because of a high variety of protocols and methods available to analyze MPs, there is an urgent need to standardize these techniques to facilitate large-scale multicenter studies to understand the role of MPs in disease and their potential use for clinical diagnosis and prognosis.
MPs are a heterogeneous group of bioactive small vesicles (100–1000 nm) that can be found in blood and body fluids following activation, necrosis, or apoptosis of virtually any eukaryotic cell. It is thought that they play an important role in intercellular communication and participate in the maintenance of homeostasis under physiological conditions.
Healthy individuals have small amounts of circulating MPs, the majority of which are derived from platelets and erythrocytes. Marked elevations of total number of MPs or relative increases of specific MP populations have been associated with a variety of disorders, including deep venous thrombosis and pulmonary embolism, congestive heart failure (CHF), sepsis, heparin-induced thrombocytopenia (HIT), thrombotic thrombocytopenic purpura (TTP), paroxysmal thrombocytopenic purpura, and preeclampsia.
MPs are composed of a phospholipid bilayer, which carries several bioactive molecules, including transmembrane proteins, receptors, counterreceptors, and adhesion molecules, as well as cytoplasmic products such as nucleic acids, chemokines, cytokines, enzymes, growth factors, and signaling proteins. The membrane shows externalization of phosphatidylserine (PS) that provides a platform for the assembly of coagulation factors. The precise content of lipids and proteins depends on their cellular origin and the type of stimulus involved in their formation.
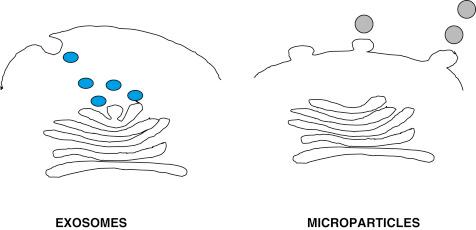
Living cells are capable of releasing different types of membrane vesicles that can be classified based on their size, sedimentation, and secretion mechanisms into exosomes, MPs, and apoptotic vesicles. The term MP encompasses a heterogeneous group of poorly characterized vesicles ranging in size from 100 to 1000 nm that are released by budding of the plasma membrane (ectocytosis). Based on their size, these MPs have been divided into microvesicles (100–1000 nm), ectosomes (50–200 nm), membrane particles (50–80 nm), and exosome-like vesicles (20–50 nm) ( Fig. 162.1 ; Table 162.1 ). These large vesicles should be distinguished from exosomes (<100 nm) that are membrane vesicles that are stored intracellularly in multivesicular compartments and are secreted following the fusion of internal compartments with the cell plasma membrane ( Fig. 162.1 ). Dying or apoptotic cells are also able to secrete membrane vesicles with different features to those from living cells.
| Feature | Exosomes | Microparticles | Apoptotic Vesicles |
|---|---|---|---|
| Size | 50–100 nm | 100–1000 nm | 50–500 nm |
| Density in sucrose | 1.13–1.19 g/mL | 1.04–1.23 g/mL | 1.16–1.28 g/mL |
| Sedimentation | 100,000 g | 20 000 g or greater | 1200, 10,000 or 100,000 g |
| Origin | Multivesicular, internal compartments | Plasma membrane | Cellular fragments |
| Release | Constitutive and/or cellular activation | Cellular activation and early apoptosis | Terminal apoptosis |
| Appearance on electron microscopy | Cup-shaped | Irregular shape, heterogeneous | Heterogeneous |
| Main protein markers | Tetraspanins (CD9, CD63), Alix, flotillin, TSG101 | Integrins, selectins, other antigens of parental cell | Histones |
| Annexin V binding capacity | No or low | High | High |
It is thought that MPs form when the asymmetric distribution of plasma membrane lipids is lost. As highlighted by Hugel et al., under resting conditions, phospholipids are asymmetrically distributed in the plasma membrane, with PS almost exclusively located in the inner membrane layer, and phosphatidylcholine and sphingomyelin located on the external one. Increased concentrations of cytosolic calcium, like those seen in response to cellular activation, may lead to lipid redistribution and surface exposure of PS, with membrane blebbing and subsequent MP shedding. Once exposed, PS promotes blood coagulation by serving as a scaffold for the assembly of the prothrombinase and thrombinase complexes. Of interest, some investigators have shown that platelet-derived MP surfaces have 50- to 100-fold higher procoagulant activity when compared with the normal platelet surface. The physiological importance of MP shedding is exemplified in a rare disorder known as Scott syndrome, which is characterized by moderate to severe bleeding episodes, usually provoked hemorrhages due to an enzymatic deficit that leads to decreased surface exposure of PS and decreased MP formation.
Circulating vesicles originating from apoptotic cells are thought to be produced by less-controlled mechanisms, possibly secondary to loss of membrane integrity or mechanical destruction. Although these apoptotic vesicles display PS on their surface, in contrast to the MPs, they show no or weak procoagulant activity. Surface exposure of PS also serves as a signal for the clearance of senescent cells by the reticuloendothelial system.
MPs play an important role in homeostasis and intercellular communication through several mechanisms including
Transfer of surface receptors,
Transfer of mRNA,
Release of proteins or active lipids, and
Induction of adaptive immune response ( Table 162.2 ).
| Biological Effect | Proposed Mechanisms |
|---|---|
| Procoagulant | Surface exposure of phosphatidylserine |
| Expression of Tissue Factor (mostly monocyte-derived MPs) | |
| Transfer of GPIIb/IIIa | |
| Presence of membrane functional effectors (integrins, P-selectin, VWF) | |
| Anticoagulant (in vitro) | Proteolytic inactivation of FVa by activated protein C |
| Fibrinolytic | Expression of u-PA and u-PAR |
| Proteolytic | Expression of matrix metalloproteinases |
| Vascular | Membrane expression of thromboxane A2 |
| Impairment of endothelium-dependent relaxation through eNOS downregulation | |
| Proinflammatory | Release of proinflammatory endothelial cytokines (IL-6 or MCP-1) |
| Induction of expression of ICAM-1, VCAM-1 and e-selectin | |
| Serve as a substrate for production of lysophosphatidic acid | |
| Antiinflammatory | Secretion of TGFβ, potent inhibitor of macrophage activation |
| Expression of Annexin A1, endogenous antiinflammatory protein | |
| Immunity | Expression of major histocompatibility complex molecules |
| Display of autoantigens, such as RNA and DNA, which may act as potent autoadjuvants, inducing B cell tolerance | |
| Expression of Fas-L leading to apoptosis, leading to immune evasion |
Abnormal levels of circulating MPs have been associated with a variety of disorders ( Table 162.3 ).
| Disease | Source of Microparticles |
|---|---|
| Thrombotic Disorders | |
| Venous thromboembolism | Platelet, endothelial cell |
| Thrombotic thrombocytopenic purpura | Platelet, endothelial cell |
| Antiphospholipid syndrome | Platelet, endothelial cell |
| Heparin-induced thrombocytopenia | Platelet |
| Sickle cell disease | Platelet, red blood cell, endothelial cell, monocyte |
| Paroxysmal nocturnal hemoglobinuria | Platelet |
| Bleeding Disorders | |
| Scott syndrome | Platelet |
| Castaman syndrome | Platelet |
| Cardio- and Cerebrovascular Diseases | |
| Acute coronary syndrome | Platelet, endothelial cell |
| Acute ischemic stroke | Endothelial cell |
| Arteriosclerosis obliterans | Platelet |
| Hypertension | Monocyte, platelet, endothelial cell |
| Hyperlipidemia | Endothelial cell |
| Atherosclerosis | Monocyte, platelet, endothelial cell |
| Congestive heart failure | Endothelial cell |
| Diabetes | Platelet, monocytes, endothelial cells |
| Infectious Diseases | |
| Escherichia coli hemolytic uremic syndrome | Platelet, leukocytes |
| HIV infection | Lymphocyte |
| Prion diseases | Platelet |
| Malaria | Platelet |
| Hepatitis C | Lymphocyte |
| Inflammatory Disorders | |
| Preeclampsia | Leukocytes |
| Sepsis | Platelet, endothelial cells, leukocytes |
| Sepsis-induced immunosuppression | Platelet |
| Other | |
| End-stage renal disease | Endothelial cell |
| Organ transplantation | Endothelial cell |
| Rheumatoid arthritis | Platelet |
| Immunosuppression | Platelet |
| Polycystic ovarian syndrome | Platelet |
Venous thromboembolism (VTE) is a multifactorial disease with a high incidence. Few studies have demonstrated increased levels of endothelial-derived MP (EMP) and platelet MP (PMP) in patients with VTE. A retrospective case–control study conducted by Bucciarelli et al. has demonstrated an association between high plasmatic levels of total MP and the risk of a first VTE. The study showed that compared with individuals with low levels of MP (<10th percentile), individuals with high levels (>90th percentile) had a fivefold increased risk of having had a previous VTE. The investigators concluded that elevated levels of MPs are associated with increased risk of VTE, which is independent of other known risk factors for VTE. A prospective study conducted by Rectenwald et al. suggested that combined detection of PMP, D-dimer, and P-selectin correlates with the diagnosis of deep venous thrombosis with a sensitivity of 73% and a specificity of 81%. Although the underlying mechanisms are still unknown, it is likely that VTE develops as a result of a complex interaction between MPs, endothelial cells, platelets, and inflammatory cells.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here