Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cardiomyopathy is a cardiac muscle disease process that leads to clinical myocardial dysfunction. The disease process results in morphologic changes in the heart that are typically classified as (1) dilated cardiomyopathy, (2) ischemic cardiomyopathy, (3) hypertrophic cardiomyopathy, (4) restrictive cardiomyopathy, and (5) arrhythmogenic right ventricular (RV) dysplasia. Clinical manifestations, predictors of survival and functional outcome, and therapeutic objectives focus on the clinical and pathophysiologic condition termed heart failure .
Heart failure is a clinical syndrome that represents a complication or common final pathway of many heart diseases in which defective cardiac filling (diastolic heart failure) or impaired contraction (systolic heart failure) or emptying results in the heart's inability to pump a sufficient amount of blood to support tissue metabolism, or to be able to do so only with elevated filling pressures. It is commonly characterized by secondary organ abnormalities in the skeletal muscles (fatigue), lungs (dyspnea on exertion and sometimes at rest), and kidneys (salt and fluid retention).
William Withering drew attention to the use of foxglove, Digitalis purpurea, in his classic treatise An Account of the Foxglove and Some of Its Medical Uses: With Practical Remarks on Dropsy and Other Diseases in l785. He began using an old family remedy for dropsy—swelling of the limbs—and determined that the active ingredient of the herbal concoction was derived from foxglove leaves. After 10 years’ experience, he published use of digitalis in treating dropsy, anasarca, and hydrops pectoris. Withering advised digitalis primarily as a diuretic, assuming this was its principal action. He also recognized a powerful action on the heart, quieting and slowing its rate. Cushny, writing about digitalis in 1925, noted that much literature was published regarding its actions until 1810 ; thereafter, clinical use of digitalis remained unchanged for 100 years.
William Heberden, writing in 1802 about diagnosis and treatment of dropsy, stated “A dropsy is very rarely an original distemper, but is generally a symptom of some other, which is too often incurable; and hence arises its extreme danger.” He recommended removing “stagnating water” by purging and diuretic salts. He knew of digitalis and used it in his practice.
Bertin made an important distinction in 1833 between hypertrophy and dilatation of the heart. He noted that dilatation does not constantly accompany thickening (hypertrophy) of the myocardium. He proposed classifying hypertrophy using “simple” to describe a heart whose walls were thickened without dilatation of chambers, “eccentric” to describe increased wall thickness with dilatation of chambers, and “concentric” to describe increased wall thickness and diminished cavity size.
In 1870, J. Milner Fothergill won the Hastings Gold Medal of the British Medical Association for an essay about digitalis. His text on heart disease and treatment provides a glimpse of heart failure treatment in 1872. He stated, “The treatment of [cardiac] dilatation taxes all the powers that can be brought to bear on it, and the success which attends our efforts is very limited indeed.” He recommended “rest, nutritious and easily-digestible diet, … and a residence in a hospital ward for a few weeks.” In addition to rest and abstaining from all exertion, he recommended treatment with digitalis.
When William Osler published The Principles and Practice of Medicine in 1892, treatment of heart failure included “complete bed rest, a carefully regulated diet, and use of aromatic spirits of ammonia, sulphuric ether, and stimulants.” Digitalis was used more selectively. “Morphia” (morphine) was used. Even “strychnia” (strychnine) was given. Cautious use of nitroglycerine was also recommended.
Understanding of heart failure advanced in the 20th century. Paul D. White used the term congestive heart failure to refer to insufficiency of the myocardium in his classic text Heart Disease, published in 1931. Mechanisms and causes of heart failure became more refined. Treatment consisted of:
Rest
Digitalis
Diuretic drug therapy
Other drugs, including cathartics and hypnotics
Other measures, including venesection
Diuretics that were useful at that time were theobromine, mercury, certain salts, and parathormone.
By the I950s, rapid advances were being made in treating diseases of the circulation. Heart failure treatment included not only rest but intermittent exercise to prevent phlebothrombosis and enhance efficacy of diuretic therapy. Dietary sodium restriction was understood. Digitalis was available as powdered leaf and in several purified compounds and was the mainstay of heart failure treatment. Mercurial diuretics were the most effective. These drugs were given parenterally and acted by diminishing reabsorption of chloride by the renal tubules. Their action was enhanced by intravenous administration of aminophylline. Chlorothiazide became available for oral use and was thought to be an effective diuretic. Oxygen therapy was found to be useful. Other measures were still in use, such as sedation with morphine, venesection, thoracentesis, puncture of legs, and radioiodine to induce hypothyroidism.
Loop diuretics were introduced in the late 1960s. For the first time, efficacious diuretic agents were available. Ethacrynic acid, the first of the loop diuretics, is a phenoxyacetic acid derivative; furosemide is a sulfonamide derivative. These drugs selectively inhibit sodium chloride reabsorption in the thick ascending limb of the loop of Henle. Other sulfonamide derivatives were subsequently introduced. These diuretics have become the basis for treating heart failure.
Cardiac remodeling is the central feature of the failing heart. One of the principal mechanisms by which the heart compensates for increased load is ventricular hypertrophy. The basic cellular feature of cardiac remodeling is myocyte hypertrophy, which includes both an increase in myocyte size and presence of additional sarcomeres, whose qualitative aspects differ according to the inciting pathologic conditions. Early stages of myocardial hypertrophy are characterized by increases in number and size of mitochondria in cardiac myocytes and in number of myofibrils. The myocardial cell, therefore, is larger and longer.
Transition from compensatory hypertrophy to heart failure is related to alterations in cell organization and changes in coronary blood flow to the increased cell mass of the hypertrophied ventricle. Myocardial capillary density and coronary reserve are reduced, resulting in myocardial ischemia that is most pronounced in the subendocardium. Ischemic myocyte injury associated with replacement fibrosis impairs systolic and diastolic function and accelerates heart failure. At a more advanced stage of hypertrophy, subtle changes of cellular organization and contour occur. Long-standing hypertrophy disrupts both cellular organization and Z-band architecture. Late stages of hypertrophy are characterized by loss of contractile elements with breakdown of Z bands, loss of normal parallel arrangement of sarcomeres, deposition of fibrous tissue, and tortuosity of T tubules.
When the remodeling process leads to segmental or global ventricular dilatation, the increased curvature results in increased wall tension, as predicted by the Laplace relationship. Increased wall tension induces increased myocardial oxygen consumption, impairs subendocardial blood flow, and decreases arrhythmia thresholds. Increasing degree of remodeling has been correlated with worsening prognosis.
Growth factors present in the embryonic heart but dormant in the normal adult heart are reactivated by myocyte hypertrophy. In the embryonic heart, growth factors provide the stimulus for normal cell division and heart growth. Withdrawal of growth factors inhibits the cell cycle and favors repair in the normal adult heart. The fetal gene repertoire responds to myocyte hypertrophy with induction of β-myosin heavy chain (MHC), atrial natriuretic factor, and repression of α-MHC, among others. Loss of α-MHC content contributes directly to reduced contractility in heart failure. Excitation-contraction coupling is compromised by alterations in the phosphorylation status of troponin-I; and defects in calcium ATPase and calcium release channels impair contractility and promote β-adrenergic desensitization.
The environment of continuous growth signals promoted by the fetal gene profile produces cell dysfunction and eventually cell death, likely related to apoptotic mechanisms. Alterations in the extracellular matrix of the myocardium include replacement fibrosis.
Heart failure begins after an event produces an initial decline in pumping capacity of the heart. Once the injurious event is initiated, three major forces can contribute to chronic heart failure: intrinsic myocardial damage, abnormal load on one or both ventricles, and extrinsic forces.
The major causes of intrinsic myocardial damage include myocardial infarction secondary to ischemic heart disease, inherited conditions that affect the contractile apparatus, infiltrative myocardial diseases, autoantibodies against myocardial antigens, autoimmune injury, infection, metabolic abnormalities (e.g., hyperthyroidism), and toxic agents (e.g., alcohol, certain chemotherapeutic agents). Abnormal loading conditions may result from chronic pressure overload (e.g., chronic hypertension, aortic stenosis) or volume overload (e.g., mitral regurgitation, aortic regurgitation, intracardiac shunts, extracardiac fistulae between arterial and venous circulations). Extrinsic forces include constrictive pericarditis, severe anemia, and substrate-inducing chronic tachycardia.
These conditions initially evoke a variety of compensatory mechanisms that, in the short term, restore cardiovascular function to a normal homeostatic range in which the patient is asymptomatic. Such compensation is termed adaptive . Over time, however, sustained activation of these compensatory systems becomes maladaptive and can lead to secondary damage of ventricular myocardium, worsening ventricular remodeling, and eventually cardiac decompensation and death. Changes in configuration of the ventricular chamber that are also detrimental to cardiac function include dilatation, change in shape (increased sphericity), thinning of the wall, and inflow valve regurgitation.
A common early manifestation of heart failure is excessive salt and water retention resulting from abnormalities of renal blood flow attending reduction of cardiac output and vasoconstriction. Sympathetic nervous system up-regulation increases cardiac afterload and heart rate and decreases renal perfusion. Reduced renal perfusion alerts receptors in renal arterioles, activating the renin-angiotensin-aldosterone system. Renal and tissue renin stimulates production of angiotensin I. Angiotensin converting enzyme (ACE) catalyzes conversion of angiotensin I to angiotensin II. Angiotensin II and aldosterone blood levels are increased, causing vasoconstriction and retention of salt and water. Angiotensin II induces vasoconstriction of efferent arterioles, increasing glomerular filtration pressure despite reduced renal blood flow. This increase in sympathetic activity is initially compensatory for reduction in renal blood flow, but over time becomes maladaptive, accelerating cardiac remodeling.
As cardiac output and tissue perfusion are reduced, complex neurohormonal responses are evoked for maintaining arterial perfusion ( Fig. 20-1 ). This compensatory (adaptive) mechanism is initially beneficial in maintaining tissue perfusion as cardiac output declines, but the process actually increases hemodynamic burden and oxygen requirements of the heart, eventually becoming detrimental (maladaptive). Circulating levels of norepinephrine may be markedly increased, and there is increased activity of adrenergic neurons that cause vasoconstriction and increase afterload of the failing ventricle. Over time, density of adrenergic receptors and concentration of norepinephrine in the myocardium are reduced. These changes are accompanied by reduced activity of adenylate cyclase, which lowers intracellular concentrations of cyclic adenosine monophosphate and reduces activation of protein kinase, affecting phosphorylation of Ca ++ channels and decreasing transsarcolemmal Ca ++ entry, thereby depressing cellular uptake of Ca ++ . The result is depression of the myocardial force-velocity relationship and the length–active tension curve, which reduces the myocardial contractile state.
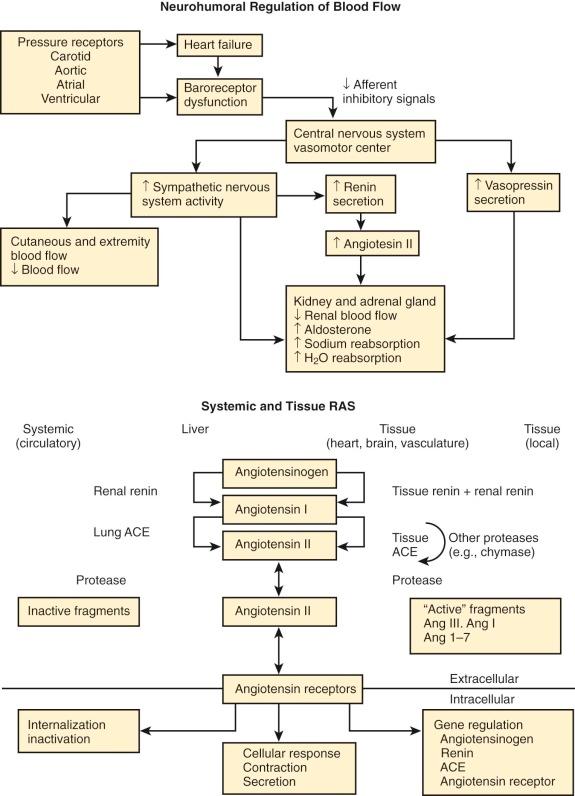
Cardiac output is maintained by increased end-diastolic fiber length and elevation of ventricular end-diastolic volume (Frank-Starling mechanism). Initial improvement of contractility associated with increased norepinephrine is eventually attenuated or prevented by norepinephrine depletion or down-regulation of myocardial norepinephrine receptors. Eventually, myocardial damage is progressive, ventricular failure intensifies, and death ensues.
Heart failure may progress as a result of overexpression of compensatory biologically active molecules that exert toxic effects on the myocardium. A variety of molecules have been implicated as sufficiently toxic to contribute to heart failure, including norepinephrine, angiotensin II, endothelin, aldosterone, and tumor necrosis factor. These molecules are of neuroendocrine origin, but may also be produced by a variety of cell types within the heart, including the myocyte itself.
Heart failure can be an acute decompensation or a chronic progressive disease. It is usually associated with decline in cardiac output. The consequence of elevated left ventricular (LV) preload and pulmonary capillary pressure is dyspnea. Cardiac remodeling is generally accepted as a determinant of the clinical course of heart failure. An approach to classification has been proposed that reflects the progressive nature of the clinical syndrome.
Evolution of the disease is identified by four stages of heart failure ( Box 20-1 ). This classification complements the New York Heart Association (NYHA) functional classification, which gauges severity of symptoms primarily in stages B and C. The classification suggests that patients with heart failure are expected to advance from one stage to the next unless progression of the disease is slowed or stopped by treatment.
High risk for developing heart failure, but with no structural disorder of the heart (hypertension, coronary artery disease, diabetes mellitus, alcohol abuse, rheumatic fever, personal history, family history of cardiomyopathy)
Structural disorder of the heart with no signs or symptoms of heart failure (left ventricular hypertrophy, dilatation or hypocontractility, valvar disease, myocardial infarction)
Past or current symptoms of heart failure associated with underlying structural heart disease (dyspnea or fatigue or asymptomatic patient treated for prior symptomatic heart failure)
Advanced disease requiring continuous or frequent hospitalization or specialized treatment strategies such as mechanical circulatory support, continuous inotropic infusions, cardiac transplantation, or hospice care
Heart failure is estimated to affect more than 5 million people in the United States alone and accounts for 1 to 3 million hospitalizations per year, or about 5% to 10% of all hospital admissions. The number of individuals with heart failure is increasing, with 400,000 to 700,000 new cases diagnosed each year, and the frequency is expected to increase two to three times during the next decade. About 3% of the adult population is treated for heart failure. Occurrence of heart failure increases with age; 6% to 10% of people older than age 65 are affected, and it is the most common cardiac diagnosis in this patient population.
Heart failure results in nearly 300,000 deaths per year in the United States, 60% sudden. Sudden death may be completely unexpected (one third of patients), a consequence of rapid deterioration (one third of patients), or a result of progression of chronic heart failure (one third of patients). About one third of patients die within 1 year of initial heart failure diagnosis, with nearly 80% dead within 6 years.
Cost of treating patients with heart failure is enormous, estimated at more than $40 billion per year, or more than 5% of the healthcare budget. Of that total, about $20 billion goes directly for medical costs, $500 million of which is spent on drugs for treating heart failure.
When a specific treatable medical disorder is responsible for heart failure, specific treatment of the underlying condition is a critical component of initial therapy (see Section VII ).
Standard guidelines for evaluating and treating heart failure have been published. A fundamental goal is to slow or reverse ventricular remodeling. Intense medical therapy, which has evolved over the last 20 years, demonstrably improves survival. ACE inhibitors, new-generation β-blockers, and aldosterone improve survival, attributed in part to their reverse remodeling effects.
Current treatment of heart failure, based on various pathophysiologic theories, recognizes that no single model explains all aspects of the heart failure syndrome and therefore attempts to use all clinical models to develop effective therapeutic strategies. Most patients with symptomatic LV dysfunction should be routinely managed with a combination of four categories of drugs: a diuretic, an ACE inhibitor, a β-adrenergic blocker, and digitalis. Diuretics and spironolactone are used to treat congestion and fluid retention related to cardiorenal mechanisms. Short-term intravenous inotropic support, intravenous vasodilators, or both, are used to treat cardiocirculatory mechanisms. Long-term inotropic support is achieved using digoxin. Intermediate and long-term strategies are directed at neurohormonal mechanisms using carvedilol, a β 1 -, β 2 -blocker with antioxidant properties, and ACE inhibitors. When ACE inhibitors are not tolerated, angiotensin receptor blockers may be effective.
Worsening of LV systolic function is frequently accompanied by impaired electromechanical coupling, which further diminishes systolic function. About 20% to 30% of patients with symptomatic heart failure have a prolonged P-R interval or an intraventricular conduction disorder characterized by wide QRS (left bundle branch block) and a discoordinate contraction pattern. Prolonged P-R interval may result in early contraction of the atria and mitral regurgitation during the diastolic phase, reducing ventricular filling. Proper timing of the P wave relative to mitral valve closure by cardiac pacing may provide more appropriate ventricular filling time.
Prolonged QRS and delay of conduction may result in contraction of the base of the heart well in advance of the LV lateral wall and apex. This may cause paradoxical septal motion and mitral valve regurgitation. Biventricular pacing (also termed cardiac resynchronization therapy ) allows simultaneous electrical stimulation of the RV and LV using an implantable pacing system. A discoordinate contraction pattern of the ventricles may be resynchronized by pacing the lateral wall of the LV synchronous with pacing the apex of the RV. Optimal aortic pulse pressure occurs when the peak of left atrial pressure coincides with start of the LV contraction as a result of optimal preload mechanisms. In addition to optimizing left atrial–to-LV mechanical timing, favorable response to both LV and biventricular stimulation may result from improved synchrony of RV and LV contraction.
In patients with advanced heart failure, the potential for fatal ventricular arrhythmias has prompted consideration of implantable cardioverter-defibrillator (ICD) therapy over chronic anti-arrhythmia drugs.
The effect of various therapeutic interventions has been evaluated in a number of clinical trials in patients with LV systolic dysfunction. Both ACE inhibitors and β-blockers, acting individually or synergistically, reduce morbidity and mortality in heart failure patients treated with diuretics and digoxin. Agents such as ACE inhibitors that inhibit neurohormonal activation relieve symptoms, reduce hospitalizations, and prolong survival. An increasing dose of ACE inhibitor or adding spironolactone to an ACE inhibitor may further improve prognosis. The optimal dose of β-blockers is unknown. Digoxin continues to be a mainstay of heart failure treatment and has been shown to decrease heart failure–related hospitalizations, but there is no evidence that it decreases mortality. It is not known whether digoxin is effective when added to a β-blocker.
Shortening of the P-R interval correlates with improved functional class and increased oxygen consumption. Resynchronization therapy with biventricular pacing has been shown to improve symptoms in patients with moderate to advanced heart failure. Several randomized multicenter trials have examined its effects on functional status, quality of life, and hemodynamics in patients with dilated cardiomyopathy. These studies have demonstrated clinical improvement correlated with narrowing of the paced QRS complex, decrease in interventricular conduction delay, and a trend toward increase in duration of ventricular filling. Clinical benefits appear to be maintained over 12 months in patients with both sinus rhythm and atrial fibrillation. Biventricular pacing incorporating antitachycardia options or an ICD appear to be the future direction of pacing therapy.
Large prospective, randomized multicenter trials have identified patient populations for whom ICD therapy provides survival benefit even in the absence of prior cardiac arrest or sustained ventricular tachycardia.
The 2008 American College of Cardiology (ACC)/American Heart Association (AHA) Practice Guidelines recommend the following indications for cardiac resynchronization therapy (CRT) in patients with severe systolic heart failure.
For patients who have LV ejection fraction (LVEF) of 35% or less, a QRS duration of 0.12 seconds or more, and sinus rhythm, CRT with or without an ICD is indicated for treatment of NYHA functional class III or ambulatory class IV heart failure symptoms with optimal recommended medical therapy. (Level of Evidence: A)
For patients who have LVEF of 35% or less, a QRS duration of 0.12 seconds or more, and atrial fibrillation, CRT with or without an ICD is reasonable for the treatment of NYHA functional class III or ambulatory class IV heart failure symptoms on optimal recommended medical therapy. (Level of Evidence: B)
For patients with LVEF of 35% or less, with NYHA functional class III or ambulatory class IV symptoms, who are receiving optimal recommended medical therapy and have frequent dependence on ventricular pacing, CRT is reasonable. (Level of Evidence: C)
For patients with LVEF of 35% or less, with NYHA functional class I or II symptoms, who are receiving optimal recommended medical therapy and are undergoing implantation of a permanent pacemaker or ICD with anticipated frequent ventricular pacing, CRT may be considered. (Level of Evidence: C)
CRT is not indicated for asymptomatic patients with reduced LVEF in the absence of other indications for pacing. (Level of Evidence: B)
CRT is not indicated for patients whose functional status and life expectancy are limited predominantly by chronic noncardiac conditions. (Level of Evidence: C)
The 2008 ACC/AHA Practice Guidelines indicate the following indications for ICD therapy in patients with severe systolic heart failure.
ICD therapy is indicated in patients with:
LVEF less than 35% due to prior myocardial infarction (MI) who are at least 40 days post-MI and are in NYHA functional class II or III (Level of Evidence: A)
Nonischemic dilated cardiomyopathy who have an LVEF of 35% or less and are in NYHA functional class II or III (Level of Evidence: B)
LV dysfunction due to prior MI who are at least 40 days post-MI, have an LVEF less than 30%, and are in NYHA functional class I (Level of Evidence: A)
Nonsustained ventricular tachycardia (VT) due to prior MI, LVEF less than 40%, and inducible ventricular fibrillation (VF) or sustained VT at electrophysiological study (Level of Evidence: B)
ICD implantation is reasonable for patients:
With unexplained syncope, significant LV dysfunction, and nonischemic dilated cardiomyopathy (Level of Evidence: C)
Who are nonhospitalized and awaiting transplantation (Level of Evidence: C)
With cardiac sarcoidosis, giant cell myocarditis, or Chagas disease (Level of Evidence: C)
ICD therapy may be considered in patients with:
Nonischemic heart disease who have an LVEF of 35% or less and are in NYHA functional class I (Level of Evidence: C)
A familial cardiomyopathy associated with sudden death (Level of Evidence: C)
LV noncompaction (Level of Evidence: C)
ICD therapy is not indicated for patients:
Who do not have a reasonable expectation of survival with an acceptable functional status for at least 1 year, even if they meet ICD implantation criteria specified in the Class I, IIa, and IIb recommendations above (Level of Evidence: C)
With drug-refractory heart failure who are not candidates for cardiac transplantation or CRT (Level of Evidence: C)
Dilated cardiomyopathy is a cardiac muscle disease characterized by enlargement of one or both ventricles and impairment of at least systolic function. It may be considered the final outcome pathway of a variety of causes of myocardial insult. These include coronary artery disease (see Chapter 10 ), infectious etiologies (most commonly viral), toxic substances (e.g., alcohol), certain medications, familial cardiomyopathy, inherited disorders, pregnancy, long-standing supraventricular tachycardias, vitamin and mineral deficiencies, and other rare causes (see Section VII ). In most patients, the causative factor is never identified ( idiopathic cardiomyopathy ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here