Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The neuroradiological illustrations were generously provided by Blaine Hart, MD, Department of Radiology (Neuroradiology), University of New Mexico Health Sciences Center.
Increased intracranial pressure (ICP) and cerebral edema are life-threatening complications of shifts in water between cells and tissue that are final common pathways of injury in many neurological disorders. Separation of brain fluids from blood is maintained by a complex series of interfaces between the blood and brain tissues with the major one referred to as the neurovascular unit (NVU). The cerebrospinal fluid (CSF) is continuously formed mainly at the choroid plexus and absorbed at the arachnoid granulations. The interstitial fluid (ISF) bathes the brain cells delivering nutrients and removing waste. Early investigators realizing that the brain lacked a true lymphatic drainage system recognized that the ISF functioned as the lymphatic system and that the CSF and ISF were a continuous fluid. In 1925, Cushing and Weed named this the “third circulation” elevating it to the level of blood and lymph fluid. In 1885, Ehrlich injected blue dye into the bloodstream of mice. The dye stained all of the animals’ organs blue—except their brains. In a follow-up experiment in 1913, one of Ehrlich’s students injected the same dye directly into the brains of mice. This time, the brains turned blue, whereas the other organs did not. From these early studies the concept of a blood–brain barrier (BBB) emerged. It is now well established that at all the interfaces between the blood and brain tissues there are specialized proteins that form tight junctions. In addition to the tight junctions, the NVU has carrier molecules and electrolyte pumps to preserve the fluid balance, provide nutrients, and remove waste materials from metabolism ( ).
Cellular membranes preserve the compartmental structure with water in extracellular and intracellular spaces. When shifts in water from one compartment to another occur under pathological conditions, swelling in the various compartments leads to increased ICP. If the increased water is blocked from exiting the ventricles, hydrocephalus results with transependymal flow of water into the periventricular white matter, resulting in interstitial edema. Loss of energy stores results in cell swelling due to failure of the membrane pumps, which is called cytotoxic edema. Damage to blood vessels leads to leakage of fluid, which expands extracellular space with intact cell membranes, leading to vasogenic edema ( ).
Hypoxia/ischemia and brain trauma initiate a series of molecular events that ultimately lead to cell death. Several molecules play key roles in the injury cascade: aquaporin forms pores in membranes that facilitate passive water movement; hypoxia inducible factor-1α (HIF-1α) is another key molecule that plays a key role in brain injury and repair by activating a cassette of inflammatory and repair-promoting genes ( ). Cytokines, proteases, and free radicals amplify the tissue damage. Advances in magnetic resonance imaging (MRI) have improved the diagnosis of CSF disorders and brain edema. Although we understand the underlying molecular processes involved in edema formation and have better ways of observing its evolution, treatment of brain edema remains a major challenge.
Brain edema represents a serious, often life-threatening consequence of many common brain disorders, including stroke, trauma, tumors, and infection. Early anatomists realized that the bony skull provided a rigid case that prevented expansion of the contents inside the skull and that such an expansion causes increases in ICP. Herniation of brain tissues at several sites occurs when there is an increase in any of the three main brain compartments: brain tissue, blood, or CSF. Brain tumors and space-occupying infections damage cells because the mass distorts the surrounding tissues by compressing vital regions of the brain. Cell injury that occurs in cerebral ischemia, hypoglycemia, and some metabolic disorders causes tissue damage via cell swelling or breakdown of the BBB. It is important to appreciate the physiology of brain fluids as a basis for understanding the pathological changes encountered in clinical practice.
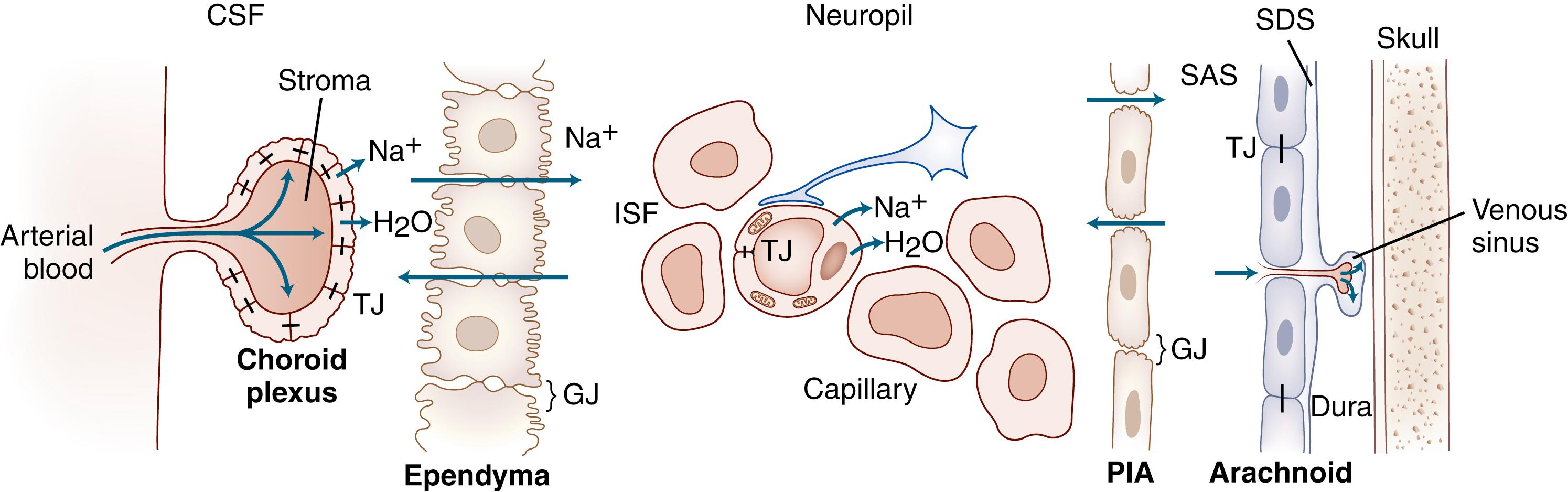
The human nervous system has evolved mechanisms to provide a stable microenvironment for the normal functioning of neurons and other cells. The electrolyte and protein contents of the brain fluids are normally kept within a constant range, which differs greatly from the systemic circulation of blood and lymph. The key to maintaining this privileged environment is a series of interfaces at each of the sites of potential brain and blood interaction. Interfaces formed by endothelial cells, choroid plexuses, ependymal cells, and arachnoid have tight-junction proteins that restrict the transport of nonlipid soluble substances and large protein molecules. In the major site formed by the endothelial cells, other components are important, including astrocytes, pericytes, and the basal lamina. Energy is expended at these interfaces to preserve this balance, and functions that are unique to the brain have evolved to provide for a constant delivery of oxygen and glucose to brain cells as well as the removal of metabolic products.
CSF fills the cerebral ventricles and subarachnoid spaces around the brain and spinal cord, serving along with the fluid between the cells, ISF, as a lymph-like fluid for brain tissue. ISF circulates between cells, draining into the CSF in the ventricle and subarachnoid space. Water moves into the extracellular space along osmotic gradients created at the capillary abluminal surface by the exchange of three sodium molecules for two molecules of potassium through the action of the sodium/potassium-triphosphatase (Na + /K + -ATPase) pump. Once within the ventricles, CSF/ISF circulates through the foramina of Magendie and Luschka to return to the systemic circulation at the sagittal sinus by way of one-way valves at the arachnoid granulations.
Examination of the CSF by lumbar puncture (LP) can provide unique information, aiding diagnosis and patient management. Increased ICP can only be determined by measurements made during removal of CSF; this information is critical in the diagnosis of raised CSF pressure in idiopathic intracranial hypertension (IIH). Studies of cells and proteins in the CSF provide information about infection and inflammation. Cancer cells can be detected and antibodies to infectious agents identified. When the BBB is disrupted, increased blood-derived proteins, mainly albumin that is produced in the liver, move into the CSF. Albumin levels in the blood are in the range of 3–5 g/dL, and in the CSF they are normally 15–60 mg/dL. CSF is critical in diagnosis of brain infection, such as meningitis, and in selection of appropriate treatment. Detection of cells in the CSF aids in the diagnosis of neuroinflammation. Detection of proteins in the CSF is important in the diagnosis of multiple sclerosis (MS): there are increased levels of myelin basic protein along with immunoglobulin (Ig)G endogenous production, which is expressed as an IgG index that is formed by dividing CSF albumin into IgG. When it is elevated, it suggests the IgG is formed in the brain rather than transported into the CSF across a damaged BBB. Patients with Alzheimer disease have low levels of amyloid-β 1-42 (Aβ 1-42 ), and elevated levels of phosphorylated tau. The ratio of Aβ 1-42 /Aβ 1-40 is more accurate in identifying Alzheimer’s disease (AD) patients ( ). Thus, LP to obtain CSF is one of the most cost-effective procedures in daily clinical practice, and when done correctly, it can provide critical diagnostic information that is only available from CSF.
The recognition that the total volume of fluid and tissue contained within the skull of an adult is constant is called the Monro–Kellie doctrine , named after two early anatomists. Changes in volume of blood, CSF, or brain compartments produce compensatory changes in the others, with a resultant increase in CSF pressure. When CSF outflow pathways are blocked, enlargement of the ventricles or hydrocephalus follows, resulting in a buildup of pressure in the ventricles that forces the CSF to move transependymally into the periventricular white matter ( ). Masses enlarge the tissue space and compress CSF and blood spaces. When the compensatory mechanisms are overwhelmed, ICP increases and herniation of brain tissue occurs. Disruption of the blood vessels leads to vasogenic edema that moves through the more compliant extracellular space of the white matter. HIF-1α is another novel molecule that plays a key role in brain injury and repair matter. Finally, an increase in blood volume, as seen in hypercapnia and hypoxia, increases the ICP ( Table 88.1 ).
| Site of Increased Intracranial Pressure | Diseases |
|---|---|
| Increased tissue volume | Tumor, abscess |
| Increased blood volume | Hypercapnia, hypoxia, venous sinus occlusion |
| Cytotoxic edema | Ischemia, trauma, toxins, metabolic diseases |
| Vasogenic edema | Infections, brain tumors, hyperosmolar states, inflammation |
| Interstitial edema | Hydrocephalus with transependymal flow |
The large surface area of capillary endothelial cells forms the major interface between the blood and brain. Other, less-extensive, interface surfaces include choroid plexuses and arachnoid granulations ( Table 88.2 ). At each of the BBB interfaces, high-resistance junctions between cells, which make the surface into an epithelial-like structure, restrict transport. The epithelial sheets impede nonlipid-soluble substances, charged substances, or large molecules, whereas lipid-soluble substances, such as anesthetic gases and narcotics, pass easily through the cells. Water has an anomalous structure that allows it to pass rapidly through endothelial cells but with slight restrictions ( ).
| Interface | Tight-Junction Location | Functional Aspects |
|---|---|---|
| Blood–CSF | Choroid plexus cell | Active secretion of CSF via ATPase and carbonic anhydrase |
| CSF–blood | Arachnoid membrane | Arachnoid granulations absorb CSF by one-way valve mechanism |
| Blood–brain | Capillary endothelial cell | Active transport of ISF via ATPase; increased mitochondria and glucose transporters in capillary endothelial cells |
ISF surrounds brain cells. It is formed by capillaries via an active transport mechanism. It is similar in composition to CSF and circulates. This lymph-like ISF fluid is formed by cerebral blood vessels, which have electrolyte pumps that make fluid in a fashion similar to that of the epithelial cells. Flowing around cells, ISF brings nutrients such as glucose and oxygen to neurons and astrocytes and removes the products of metabolism. ISF is absorbed either into the blood via terminal capillaries and venules or into CSF for eventual absorption through the arachnoid granulations ( Fig. 88.1 ). CSF from the subarachnoid space moves rapidly into the brain along paravascular routes surrounding penetrating cerebral arteries, exchanging with ISF and facilitating the clearance of interstitial solutes, which may be driven by arterial pulsation ( ). Measurements of movement of ISF made with MRI indicate that inspiration facilitates the flow of ISF by its effect on the veins. Studies in mice have shown an influence of arterial pulse pressure on the movement of ISF into and out of the brain, but these studies need to be replicated in higher mammals.
Brain extracellular space comprises 15%–20% of the total brain volume. Complex carbohydrates are found in the extracellular space, including hyaluronic acid, chondroitin sulfate, and heparan sulfate. Hyaluronic acid forms large water domains. These large extracellular matrix glycoproteins impede cell movement. After an injury, astrocytes secrete an extracellular molecule, hyaluron, which impedes movement of fluids in the extracellular space, slowing tissue repair. Treatment with hyaluronidase reduces hyaluron and improves regrowth of injured fibers ( ).
Proteases are secreted during development, angiogenesis, and neurogenesis to clear a path for the growing cells, similar to the secretion of proteases by spreading cancer cells ( ). Rather than a unitary endothelial BBB, transport between blood and brain is modulated by neurons, astrocytes, pericytes, and endothelial cells, forming an NVU. On the abluminal surface of the endothelial cells is a basal lamina composed of type IV collagen, fibronectin, heparan sulfate, laminin, and entactin. Entactin connects type IV collagen and laminin to add a structural element to the capillary. Fibronectin from the cells joins the basal lamina to the endothelium. Basal lamina provides structure through type IV collagen, charge barriers by heparan sulfate, and binding sites on the laminin and fibronectin molecules. Pericytes are embedded in the basal lamina; they are a combination of smooth muscle and macrophage. Pericytes are important in preserving the BBB. Loss of pericytes occurs in a number of neurodegenerative diseases ( ). Astrocyte foot processes form a layer that surrounds the basal lamina. Glia limitans is found at the pial surface and at the interface between astrocytes and blood vessels ( ; Fig. 88.2 ).
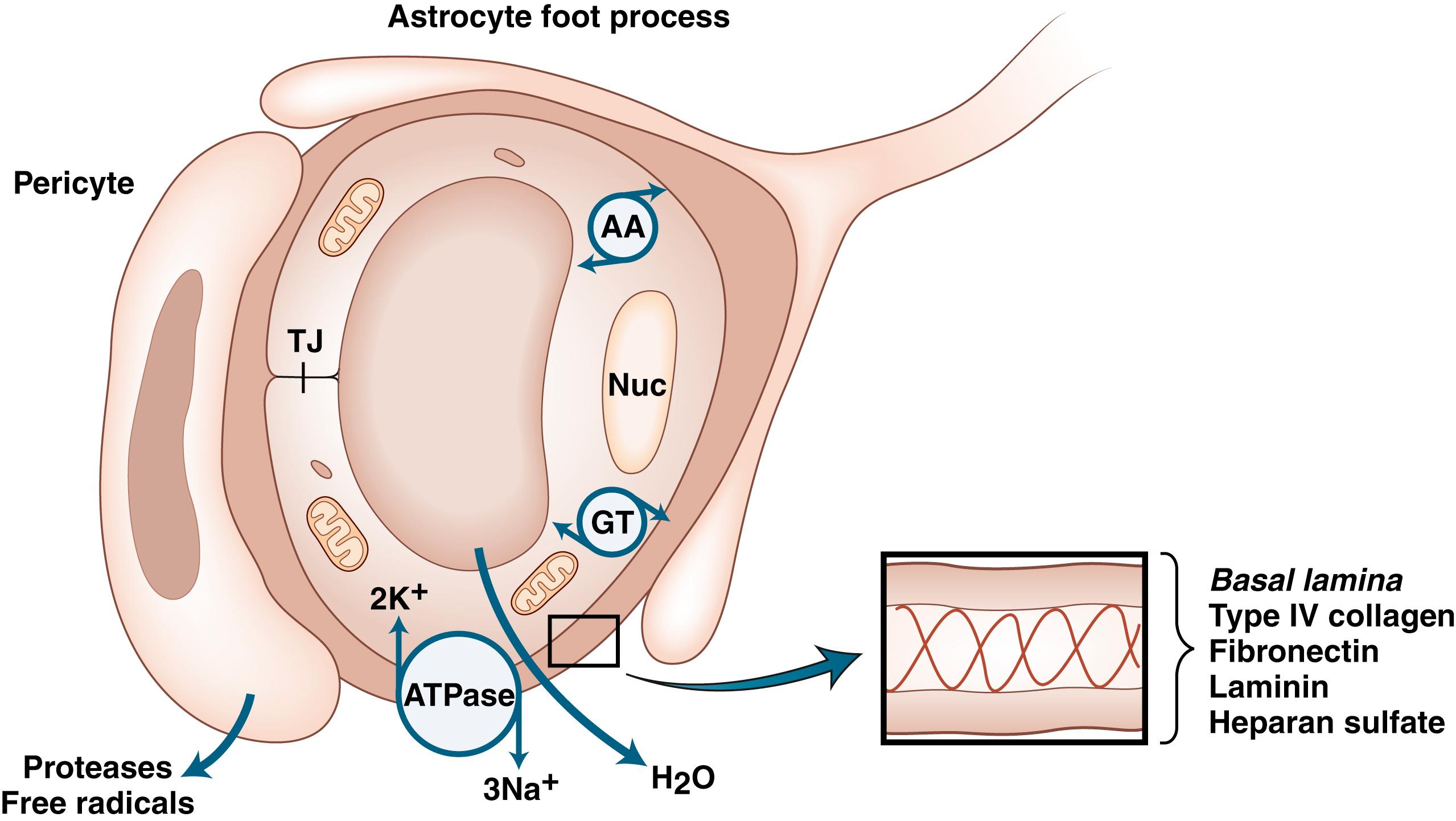
Cerebral blood vessels have very low permeability and high electrical resistance, making them more similar to epithelial cells than systemic capillaries, which are passive structures with low electrical resistance and fenestrations that permit passage of large protein molecules. In addition, cerebral blood vessels have highly selective molecular transport properties. During development, cerebral blood vessels acquire the characteristics that distinguish them from systemic capillaries. Astrocytes are critical in this differentiation process, which involves interactions between blood vessels and astrocytes. The critical nature of the astrocytes in this process was shown in transplantation studies involving chicken and quail cells, which can be separated histologically. Quail brain grafts from 3-day-old quails transplanted into the coelomic cavity of chick embryos become vascularized by chick endothelial cells and form a competent BBB. On the other hand, when avascular embryonic quail coelomic grafts are transplanted into embryonic chick brain, chick endothelial cells form leaky capillaries and venules ( ). Astrocytes are critical in the differentiation process ( ).
At the interface between the systemic circulation and brain cells there are specialized proteins that form the poorly permeable vessels. Tight-junction proteins have been isolated and cloned, permitting immunocytochemical studies of their location in the endothelial cells. Zona occludins tether the tight-junction proteins to actin within the endothelial cells; occludin and claudin form the actual tight junctions within the endothelial clefts. Occludin attaches to the zona occludins, while claudins attach to occludin and protrude into the clefts between cells. The extracellular tails of claudins from adjacent cells self-assemble to form the tight junctions that are “zip-locked” together ( ). During an ischemic injury, the tight junction proteins are degraded, contributing to the disruption of the BBB ( ).
Tight junctions between the endothelial cells create the unique membrane properties of the cerebral capillaries by greatly increasing electrical resistance, blocking transport of nonlipid-soluble substances ( Box 88.1 ).
Tight junctions create high electrical resistance
Adenosine triphosphatase pumps on abluminal surfaces form interstitial fluid
Increased numbers of mitochondria for high-energy needs
Glucose transporters and amino acid carriers
Basal lamina contributes to the barrier
Pericytes act as perivascular macrophages
Astrocytes maintain the tight junctions
Brain tissue has a very high demand for glucose and essential amino acids, which can be met by specialized molecules that transport glucose and amino acids across the BBB. Glucose transporters are densely distributed in the capillaries. At low levels of blood glucose, the carrier proteins function at full capacity to meet metabolic needs, but at higher levels of blood glucose, the carriers are saturated, and transport is dominated by diffusion rather than active transport ( ). High concentrations of one isoform, GLUT1, are found on cerebral blood vessels. GLUT3 is found on neurons and GLUT5 in microglia. GLUT2 is found predominantly in the liver, intestine, kidney, and pancreas. Amino acid transporters carry essential amino acids into the brain. Competition for the amino acid transporters can lead to a deficiency state; serotonin uptake is decreased in patients with phenylketonuria, which competes for the transporter.
Steady-state levels of brain electrolytes are preserved by transport mechanisms at the BBB. Potassium is maintained at a constant level in the CSF and brain by the BBB. This prevents fluctuations of electrolyte levels in the blood from influencing brain levels. Calcium is similarly regulated. Glutamate, which is an excitotoxin, is excluded from the brain. Highly lipid-soluble gases such as carbon dioxide and oxygen are rapidly exchanged across the capillary. Anesthetic gases are effective because they readily cross the BBB and enter the brain.
The presence of the BBB creates a major impediment for the transport of drugs into the brain. For example, penicillin is restricted from entry into the brain; high doses are needed to achieve therapeutic brain levels. Newer generations of antibiotics, such as the cephalosporins, penetrate more readily, making them better agents for treatment of brain infections. Chemotherapy of brain tumors has been hampered by the poor lipid solubility of most agents; to overcome this impediment, chemotherapeutic agents can be injected intrathecally or into catheters implanted into the ventricles, with injection bulbs buried beneath the scalp. Drugs of addiction are often modified to allow them to more readily cross the BBB. For example, heroin, which is derived from morphine, has increased lipid solubility, which enhances its transport into the brain. Similarly, other addictive substances, such as nicotine and alcohol, are highly lipid soluble and easily transported into brain.
Different rates for equilibration of various substances between blood and brain can cause paradoxical clinical situations. For example, to compensate for a metabolic acidosis, bicarbonate levels fall in both the blood and the brain. Metabolic acidosis is balanced by a respiratory alkalosis due to lowering of carbon dioxide by hyperventilation, which compensates for the acidosis; carbon dioxide is reduced in both the blood and CSF compartments, since it readily crosses the BBB, while bicarbonate is much more slowly exchanged between the two compartments. This adjustment results in a stable, albeit pathological, situation. However, when the metabolic acidosis is corrected by intravenous infusion of bicarbonate, there is a rapid adjustment of Pco 2 as the hyperventilation stops and CO 2 builds up. Bicarbonate adjusts very slowly because of the limited transport across the BBB, and the CO 2 entering the brain causes a further fall in brain pH. This dangerous situation continues until the bicarbonate levels in the brain rise. Although treatment is necessary to correct the metabolic acidosis, patients may become worse due to brain acidosis if treatment is too rapid ( ).
Production of brain fluids comes from multiple sources including the choroid plexuses within the ventricles, the electrolyte pumps on the abluminal surface of the cerebral capillaries, and metabolism. The main source is the choroid plexuses, which form an important interface between CSF and blood. Choroid plexuses protrude into the cerebral ventricles; they are covered with a specialized type of ependymal cell that has tight junctions on the apical surface.
Choroid plexus capillaries are fenestrated. Substances from the blood can cross into the stroma next to the ependymal cells. They are blocked from entering the CSF by tight junctions that form at the apical surface of the ependymal cells. Choroid plexus ependymal cells are enriched with mitochondria, Golgi complexes, and endoplasmic reticulum—suggesting a high rate of metabolic activity—and are covered with microvilli that increase their surface area.
In humans, the volume of CSF in the ventricles and around the spinal cord is approximately 140 mL, with a rate of CSF production of 0.35 mL/min or about 500 mL/day, which explains why obstruction of CSF leads rapidly to life-threatening hydrocephalus. CSF production occurs at both choroidal and extrachoroidal sites, and estimates of the proportion of CSF from each site vary, depending on the species and the method of measurement. Removal of the choroid plexus in nonhuman primates only reduces CSF production by 40%, leaving 60% presumably from extrachoroidal production ( ).
Higher levels of sodium, chloride, and magnesium and lower levels of potassium, calcium, bicarbonate, and glucose are found in CSF than are expected from a plasma ultrafiltrate, which suggests that the CSF is actively secreted. An ATPase pump on the apical surface of the choroidal cells secretes three sodium ions in exchange for two potassium ions; osmotic water follows the increased sodium gradient. Carbonic anhydrase converts carbon dioxide and water into bicarbonate, which is removed along with chloride to balance the sodium charge. Production of CSF continues even when the ICP is high. Only acetazolamide, which inhibits carbonic anhydrase, can be used for the long-term reduction in CSF production. Experimentally, hypothermia, hypocarbia, hypoxia, and hyperosmolality have been shown to reduce production, but these are not practical to use for other than short periods. Osmotic agents such as mannitol and glycerol increase serum osmolality, lowering CSF production temporarily by about 50%. Agents that interfere with Na + /K + -ATPase reduce CSF production. Digitalis has an effect on the rate of CSF production, but ouabain, which is a more effective agent experimentally, is too toxic for use in patients. Recently, hypertonic saline has been shown to reduce CSF pressure; some of this effect may be due to a reduction in CSF production, but the mechanism of action remains to be clarified.
Capillaries, which have Na + /K + -ATPase on the abluminal surface, are a source of extrachoroidal ISF production. Gray matter has a dense neuropil that impedes the flow of water, whereas white matter, being more regularly arranged, is a conduit for normal bulk flow of ISF as well as a route for movement of edema under pathological conditions. Normally the flow of ISF in the white matter is toward the ventricle, where it mixes with the CSF from the choroid plexus to be eventually drained across the arachnoid granulations that protrude into the sagittal sinus.
Water molecules have a magnetic moment that allows them to be aligned in a magnetic field. Such a field is created in a magnetic resonance scanner. Because brain tissue is 80% water, and water dipoles can be aligned by manipulating the magnetic fields, they can be made to resonate and the resonance signals from water protons can be detected by MRI; since water is the most abundant source of protons in the brain, water protons dominate the signals.
MRI can detect water diffusion by the use of appropriate pulse sequences. The complex diffusion signals are obtained mainly from intracellular water, with some contribution from extracellular water. Water diffusion between cells in the extracellular space occurs normally. When there is cellular swelling and the extracellular space is compressed, the diffusion of water slows, and the apparent diffusion coefficient (ADC) shows a loss of signal, which appears black on the image. The diffusion-weighted image (DWI) has a bright signal. Because the DWI may show T2 shine-through that will be misinterpreted as restricted diffusion, both a darkened ADC and a bright DWI should be seen in the region of the infarct. In cerebral ischemia, the DWI is abnormal within minutes after the onset of the ischemia, making this an excellent diagnostic test for the presence of acute cerebral ischemia.
Diffusion tensor imaging (DTI) reveals the patterns of white-matter tracts in three dimensions. Taking advantage of the directional flow of water protons along white matter, diffusion is measured in three planes, and the separate pathways for water movement between the fibers are traced. In patients with white-matter pathology, such as in vascular cognitive impairment and MS, injury patterns in the white matter can be revealed by DTI ( ).
Contrast agents are important in determining injury to the BBB. Iodine-containing contrast agents are used in computed tomography (CT) scanning because they are radiopaque. When injected intravenously, contrast agents show the site of injury to the blood vessels by the appearance of the contrast agent on the scan. Iodine-containing contrast agents can cause anaphylactic reactions, however, particularly in individuals with allergy to shellfish. Contrast agents used in MRI studies are safer and more sensitive, making them the agents of choice. Gadolinium-containing compounds are used in MRI because they produce a paramagnetic effect. When they leak from the vessels into tissue, they cause a rapid relaxation of the protons that can be seen on T1-weighted images as a hyperintensity, compared to the pre-contrast scan. There is some retention of gadolinium in the brain, but the significance of this finding is uncertain. However, it has led to more cautious use of gadolinium.
The terminology used to describe various types of central nervous system (CNS) infections is anatomically based ( Table 88.3 ). An infection limited to the subarachnoid space, with inflammation of the meninges, is called meningitis . Meningeal signs of headache, stiff neck, and photophobia are present without focal findings that would indicate spread into the parenchyma. When the infection spreads contiguously from the subarachnoid space through the pial surface or along Virchow-Robin spaces, crossing gap junctions, the brain parenchyma is infected, and the term meningoencephalitis is used. In addition to meningeal signs, there are focal findings and possibly impaired consciousness and seizures. An infection in the brain tissue that is most likely to spread via blood begins as a loose collection of invading cells referred to as a cerebritis ; walling off of the infected brain tissue leads to an abscess . Finally, the term encephalitis is used to describe a more diffuse brain infection in both the gray and white matter, which is usually indicative of a viral infection. Occasionally the infection spreads in a potential space beneath the dura but outside the arachnoid; subdural empyema describes a life-threatening collection of pus over the brain surface that has often spread from an infected sinus through the venous plexus of the ethmoid or sphenoid sinuses into the subdural space. The presence of a subdural empyema should be suspected in a patient with sinus infection, fever, seizures, focal findings, and altered consciousness. Diagnosis of meningitis can be done by examination of CSF for signs of infection such as increased white blood cells or protein. Infections that invade the brain are best diagnosed with MRI, which can readily demonstrate a meningoencephalitis, cerebritis, abscess, or encephalitis. Use of contrast agents increases the potential of reaching a correct diagnosis based on site of infection. Subdural empyema is the most difficult condition to diagnose because it may only be a thin layer of pus on the surface of the brain and be obscured by the skull. Diagnosis can be missed on LP or CT, and MRI is more sensitive.
| Infection | Symptoms | Site of Inflammation |
|---|---|---|
| Meningitis | Fever, stiffness, photophobia, headache | Cells confined to subarachnoid space (SAS) |
| Meningoencephalitis | Meningeal symptoms with focal findings | SAS and brain inflammation |
| Encephalitis | Headache, seizures, altered mental state | Multiple sites of cellular response in brain tissue |
| Cerebritis/abscess | Fever, seizures, focal findings | Cerebritis, early collection of inflammatory cells around vessels; abscess is the walled-off stage |
| Subdural empyema | Fever, seizures, coma | Diffuse collection of pus over the surface of the brain between the dura and arachnoid |
Lining the cerebral ventricles (other than over the choroid plexus) is a layer of ciliated ependymal cells connected by gap junctions. Pial cells lining the surface of the brain, which form the limiting glial membrane, the glial limitans, also have gap junctions. Fluid, electrolytes, and large protein molecules move through the gap junctions, allowing exchange between the CSF and ISF. Intrathecal administration of antibiotics and chemotherapeutic agents has been used to bypass the BBB.
Blood vessels penetrate the brain from the surface. As they enter the brain, they are invested with pia mater. The space between the penetrating blood vessels and the brain, prior to the point where only brain tissue surrounds the vessels, is called the Virchow-Robin space . After injection of substances intrathecally, the large proteins in the CSF space penetrate into the brain from the surface via the Virchow-Robin spaces. These perivascular routes may be involved in the spread of infection into the brain from the subarachnoid space in meningitis.
Arachnoid granulations (pacchionian granulations) are the major sites for the drainage of CSF into the blood. They protrude through the dura into the superior sagittal sinus and act as one-way valves. As CSF pressure increases, more fluid is absorbed. When CSF pressure falls below a threshold value, the absorption of CSF ceases ( Fig. 88.3 ). In this way, CSF pressure is maintained at a constant level, with the rate of CSF production as one determining factor.
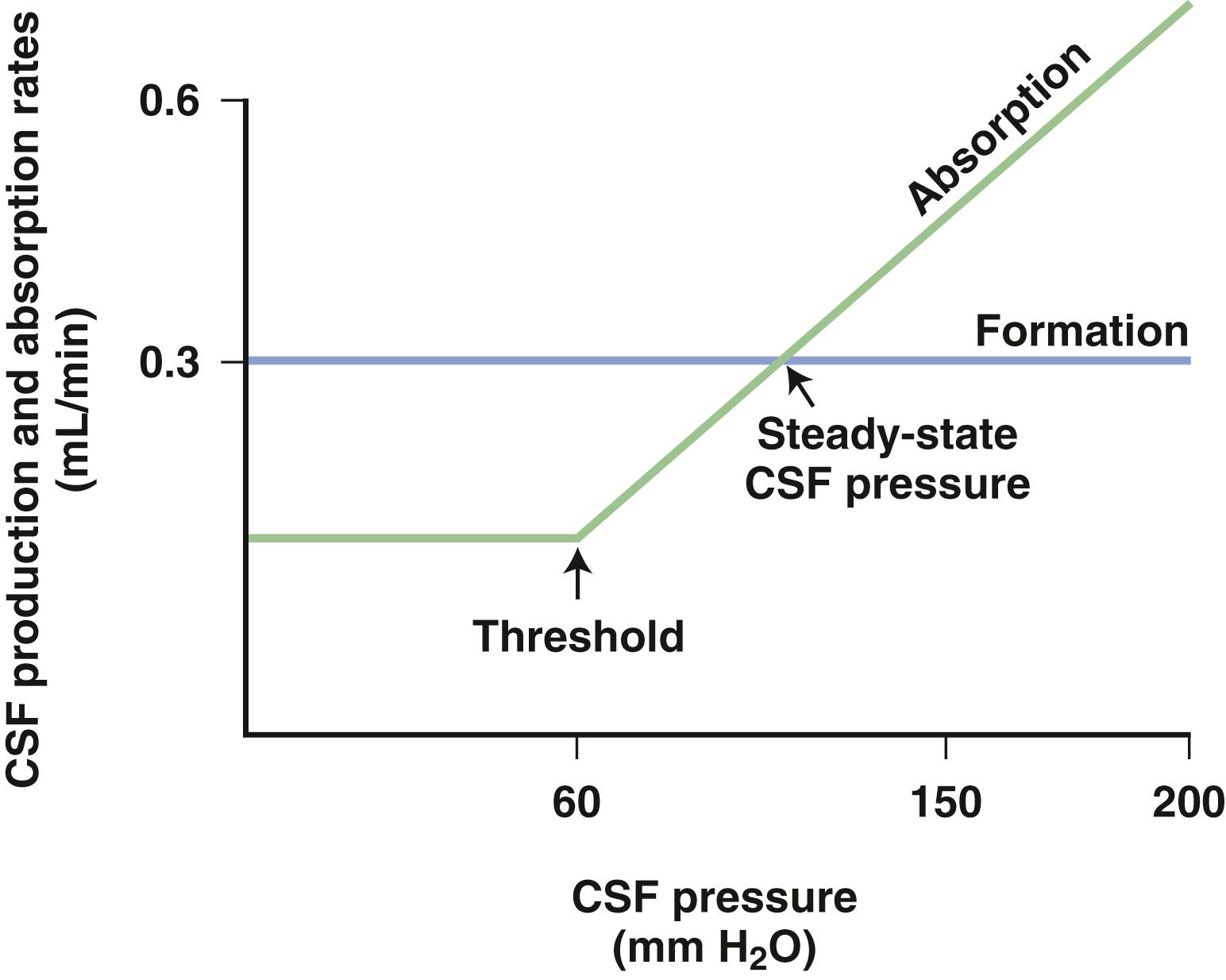
Although channels are seen in the arachnoid granulations, actual valves are absent. Tissue appears to collapse around the channel as the pressure falls, and the channels enlarge as pressure rises. Resistance to outflow across the arachnoid granulations leads to CSF pressure elevation. Substances can clog outflow channels and increase resistance to CSF absorption. Blood cells are trapped in the arachnoid villi, and subarachnoid hemorrhage causes a transient increase in CSF pressure and can occasionally lead to hydrocephalus. Similarly, white blood cells and increased protein from meningitis can block the arachnoid granulations and increase CSF pressure.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here