Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Birth injuries are those sustained during the birth process, which includes labor and delivery. They may be avoidable, or they may be unavoidable and occur despite skilled and competent obstetric care, as in an especially hard or prolonged labor or with an abnormal presentation. Fetal injuries related to amniocentesis and intrauterine transfusions and neonatal injuries after resuscitation procedures are not considered birth injuries. However, injuries related to the use of intrapartum monitoring of the fetal heart rate and collection of fetal scalp blood for acid-base assessment are included. Factors predisposing the infant to birth injury include macrosomia, prematurity, cephalopelvic disproportion, dystocia, prolonged labor, abnormal presentation, and certain operative deliveries, particularly vacuum extraction. The fetus may also sustain injury, including death, if the mother is involved in a motor vehicle collision. Fetal deaths may occur from maternal cardiovascular instability, uterine rupture, placental abruption, hemorrhage, and direct injury to the fetus. Although usually protected by maternal soft tissues, the uterus, and amniotic fluid, the fetus may be subjected to the same acceleration-deceleration forces as the mother. This may result in full-thickness bowel injury and fulminant disseminated intravascular coagulation. Thus, a thorough physical examination of the infant is critical after a maternal motor vehicle collision to identify any internal injury that may have occurred.
The significance of birth injuries may be assessed by review of mortality data. In 1981, birth injuries ranked sixth among major causes of neonatal death, resulting in 23.8 deaths per 100,000 live births. During the ensuing decade, because of refinements in obstetric techniques and the increased use of cesarean deliveries over difficult vaginal deliveries, a dramatic decline occurred in birth injuries as a cause of neonatal death. Statistics for 1993 revealed a reduction to 3.7 deaths per 100,000 live births; because of the emergence of other conditions, birth injuries ranked 11th among major causes of neonatal death. The most recent figures available (for 2013-2014) identify only 10 leading causes of neonatal and postneonatal death, with no mention of birth injuries.
Despite a reduction in related mortality rates, birth injuries still represent an important source of neonatal morbidity and neonatal intensive care unit admissions. Of particular concern are severe intracranial injuries after operative vaginal delivery (vacuum-assisted and forceps delivery) and failed attempts at operative vaginal delivery.
The clinician should consider the broad spectrum of birth injuries in the differential diagnosis of neonatal clinical disorders. Although many injuries are mild and self-limited, others are serious and potentially lethal. This chapter describes conditions that can be managed by observation only, as well as those that require more aggressive intervention. In addition to assuring timely institution of therapy when indicated, recognition and documentation before discharge from the hospital will help avoid inappropriate suspicion of inflicted injury (child abuse) at a later date.
Erythema and abrasions frequently occur when dystocia has occurred during labor as a result of cephalopelvic disproportion or when forceps have been used during delivery. Injuries caused by dystocia occur over the presenting part; forceps injury occurs at the site of application of the instrument. Forceps injury frequently has a linear configuration across both sides of the face, outlining the position of the forceps. The affected areas should be kept clean to minimize the risk for secondary infection. These lesions usually resolve spontaneously within several days with no specific therapy.
Occasionally petechiae are present on the head, neck, upper portion of the chest, and lower portion of the back at birth after a difficult delivery; they are observed more frequently after breech and precipitous deliveries and tight nuchal cord.
Petechiae are probably caused by a sudden increase in intrathoracic and venous pressures during passage of the chest through the birth canal. An infant born with the cord tightly wound around the neck may have petechiae only above the neck.
Petechiae may be a manifestation of an underlying hemorrhagic disorder. The birth history, early appearance of the petechiae, and absence of bleeding from other sites help to differentiate petechiae caused by increased tissue pressure or trauma from petechiae caused by hemorrhagic disorders (see Chapter 79 ). The localized distribution of the petechiae, absence of subsequent crops of new lesions, and a normal platelet count exclude neonatal thrombocytopenia. The platelet count also may be low because of infection or disseminated intravascular coagulation. Infections may be clinically distinguished from traumatic petechiae by the presence of other signs and symptoms. Disseminated intravascular coagulation usually is associated with excessive and persistent bleeding from a variety of sites. Petechiae usually are distributed over the entire body when associated with systemic disease.
If the petechiae are caused by trauma, neither corticosteroids nor heparin should be used. No specific treatment is necessary.
Traumatic petechiae usually fade within 2 or 3 days.
Ecchymoses may occur after traumatic or breech deliveries. The incidence is increased in premature infants, especially after a rapid labor and poorly controlled delivery. When extensive, ecchymoses may reflect blood loss severe enough to cause anemia and, rarely, shock. The reabsorption of blood from an ecchymotic area may result in significant hyperbilirubinemia ( Fig. 29.1 ).
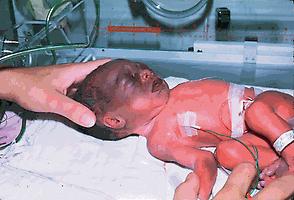
No local or systemic therapy is necessary. The rise in serum bilirubin that follows severe bruising may be decreased by the use of phototherapy (see Chapter 91 ). Ecchymoses rarely result in significant anemia.
The ecchymoses usually resolve spontaneously within 1 week.
Subcutaneous fat necrosis is a rare form of panniculitis seen mostly in term or post-term infants characterized by well-circumscribed, indurated lesions of the skin and underlying tissue (see Chapter 87 ).
Although subcutaneous fat necrosis can occur without any obvious cause, it is most commonly seen in association with perinatal asphyxia. Other etiologic factors that have been implicated include cold exposure, localized skin trauma, obstetric trauma, preeclampsia, gestational diabetes, maternal or fetal risk of thrombosis, maternal cocaine use, hypothermia, prostaglandin E administration, brown fat deficiency, meconium aspiration, sepsis, and intrapartum calcium channel blocker administration. It can also occur as a complication of therapeutic hypothermia for perinatal asphyxia or in newborns undergoing surgical procedures. Many affected infants are large and have been delivered by forceps or after a prolonged, difficult labor involving vigorous fetal manipulation. The distribution of the lesions usually is related to the site of trauma, which explains the frequent involvement of shoulders and buttocks. One suggested mechanism of pathogenesis proposes that diminished in utero circulation and mechanical pressure during labor and delivery result in vascular compromise to specific areas, which eventually causes localized fat necrosis. In maternal cocaine use during pregnancy, it has been postulated that cocaine may decrease placental perfusion with subsequent hypoxemia and alteration of the maternal and fetal pituitary-adrenal axes.
Histopathologic studies reveal initial endothelial swelling and perivascular inflammation in the subcutaneous tissues. This is followed by necrosis of fat and a dense granulomatous inflammatory infiltrate containing foreign body–type giant cells with needle-shaped crystals resembling cholesterol.
Necrotic areas usually appear between 6 and 10 days of age but may be noted as early as the second day or as late as the sixth week. They occur on the cheeks, neck, back, shoulders, arms, buttocks, thighs, and feet, with relative sparing of the chest and abdomen. The lesions vary in size from 1-10 cm; rarely, they may be more extensive. They are irregularly shaped, hard, plaquelike, and nonpitting ( Fig. 29.2 ). The overlying skin may be colorless, red, or purple with no local tenderness. The affected areas may be slightly elevated above the adjacent skin; small lesions may be easily movable in all directions. Fever may be seen in a small subset of infants with subcutaneous fat necrosis.

This condition may be associated with hypoglycemia, hypertriglyceridemia, hypercalcemia, nephrocalcinosis, anemia, and thrombocytopenia. Marked symptomatic hypercalcemia may develop in infants with subcutaneous fat necrosis at 3-4 weeks of age; this has been characterized by vomiting, weight loss, anorexia, fever, somnolence, and irritability, with serum calcium levels as high as 17.3 mg/dL. The treatment includes intravenous hydration, calcium-wasting diuretics such as furosemide, potassium citrate to inhibit renal stone formation, and corticosteroids. Successful short-term treatment with bisphosphonates (e.g., pamidronate or etidronate) has been reported to control hypercalcemia. Investigators have suggested extra renal production of 1,25-dihydroxyvitamin D by the granulomatous cells of fat necrosis as a possible mechanism for the hypercalcemia.
The differential diagnosis includes lipogranulomatosis and sclerema neonatorum, which carry a potentially grave prognosis, and nodular nonsuppurative panniculitis, which is usually associated with fever, hepatosplenomegaly, and tender skin nodules.
Most of these lesions require only observation. Surgical management is not indicated, with the exception of extensive ulcerated lesions.
The lesions slowly soften after 6-8 weeks and completely regress within several months. Occasionally minimal residual atrophy, with or without small calcified areas, is observed. Affected infants should be followed closely during the first 6 weeks for potential development of hypercalcemia. It is important to treat this complication without delay to prevent central nervous system (CNS) and renal sequelae.
Accidental lacerations may be inflicted with a scalpel during cesarean section. They usually occur on the scalp, buttocks, and thighs, but they may occur on any part of the body. If the wound is superficial, the edges may be held in apposition with butterfly adhesive strips. Deeper, more freely bleeding wounds should be sutured with the finest material available. Rarely, the amount of blood loss and depth of wound require suturing in the delivery room. After repair, the wound should be left uncovered unless it is in an area of potential soiling, such as the perineal area. Healing is usually rapid, and complications are rarely seen.
Caput succedaneum, a frequently observed lesion, is characterized by a vaguely demarcated area of edema over that portion of the scalp that was the presenting part during a vertex delivery.
Serum or blood or both accumulate above the periosteum in the presenting part during labor. This extravasation results from the higher pressure of the uterus or vaginal wall on those areas of the fetal head that border the caput. Thus, in a left occiput transverse presentation, the caput succedaneum occurs over the upper and posterior aspect of the right parietal bone; in a right-sided presentation, it occurs over the corresponding area of the left parietal bone.
The soft swelling is usually a few millimeters thick and may be associated with overlying petechiae, purpura, or ecchymoses. Because of the location external to the periosteum, a caput succedaneum may extend across the midline of the skull and across suture lines. After an especially difficult labor, an extensive caput may obscure various sutures and fontanelles.
Occasionally, a caput succedaneum may be difficult to distinguish from a cephalhematoma, particularly when the latter occurs bilaterally. Careful palpation usually indicates whether the bleeding is external to the periosteum (caput) or beneath the periosteum (cephalhematoma). Iatrogenic encephalocele is an infrequent complication of vacuum extraction delivery and may present like a caput succedaneum initially. Imaging should be considered in every child with a large caput succedaneum that does not diminish in 48-72 hours or with enlargement of the swelling more than 24 hours after delivery, especially when there are neurologic deficits and hemodynamic instability.
Usually no specific treatment is indicated. Rarely, a hemorrhagic caput may result in shock and require blood transfusion.
A caput succedaneum usually resolves within several days.
Cephalhematoma is an infrequently seen subperiosteal collection of blood overlying a cranial bone. The incidence is 0.4%-2.5% of live births, with a higher frequency in infants born to primiparous mothers.
A cephalhematoma is caused during labor or delivery by a rupture of diploic blood vessels that traverse from skull to periosteum. Repeated buffeting of the fetal skull against the maternal pelvis during a prolonged or difficult labor and mechanical trauma caused by use of forceps and vacuum suction devices in delivery have been implicated. Petrikovsky and associates described seven infants in whom cephalhematoma or caput succedaneum was identified prenatally before onset of labor. Occurrence of premature rupture of membranes in five of the pregnancies suggests an etiology of fetal head compression by the uterine wall, resulting from oligohydramnios subsequent to the ruptured membranes.
The bleeding is sharply limited by periosteal attachments to the surface of one cranial bone; there is no extension across suture lines. The bleeding usually occurs over one or both parietal bones. Less often, it involves the occipital bones and, very rarely, the frontal bones. The overlying scalp is not discolored. Because subperiosteal bleeding is slow, the swelling may not be apparent for several hours or days after birth. The swelling is often larger on the second or third day, when sharply demarcated boundaries are palpable. The cephalhematoma may feel fluctuant and often is bordered by a slightly elevated ridge of organizing tissue that gives the false sensation of a central bony depression. It may be associated with an underlying linear, nondepressed skull fracture in a small percentage of infants.
Radiographic manifestations vary with the age of the cephalhematoma. During the first 2 weeks, bloody fluid results in a shadow of water density. At the end of the second week, bone begins to form under the elevated pericranium at the margins of the hematoma; the entire lesion is progressively overlaid with a complete shell of bone.
Cephalhematoma must be distinguished from other birth complications such as subgaleal hematoma, caput succedaneum, vacuum caput, leptomeningeal cyst, or congenital anomalies such as meningoceles. It may be differentiated from caput succedaneum by (1) its sharp periosteal limitations to one bone, (2) the absence of overlying discoloration, (3) the later initial appearance of the swelling, and (4) the longer time before resolution. Cranial meningocele is differentiated from cephalhematoma by pulsations, an increase in pressure during crying, and the demonstration of a bony defect on a radiograph. An occipital cephalhematoma may be confused initially with an occipital meningocele and with cranium bifidum because all occupy the midline position.
No therapy is indicated for the uncomplicated cephalhematoma, as more than 80% resolve by gradual hemolysis and resorption in 3-4 weeks. When the hematoma does not resolve spontaneously, it may get organized, and calcification may be seen. It may still get absorbed slowly and often disappears over 3-6 months. Persistent calcification that is not resolved by time may be an indication for surgical excision. Rarely, a massive cephalhematoma may result in blood loss severe enough to require transfusion. Significant hyperkalemia and hyperbilirubinemia may result from resolving hematoma, necessitating appropriate treatment. The most common associated complications are skull fracture and intracranial hemorrhage. Linear fractures do not require specific therapy, but radiographs should be taken at 4-6 weeks to ensure closure and exclude formation of leptomeningeal cysts; depressed fractures require immediate neurosurgical consultation. Routine incision or aspiration of a cephalhematoma is contraindicated because of the risk for introducing infection. Rarely, bacterial infections of cephalhematomas occur, usually in association with septicemia and meningitis. Focal infection should be suspected when a sudden enlargement of a static cephalhematoma occurs during the course of a systemic infection, with a relapse of meningitis or sepsis after treatment with antibiotics, or with the development of local signs of infection over the cephalhematoma ( Fig. 29.3 ). Diagnostic aspiration may be indicated. If a local infection is present, surgical drainage and specific antibiotic therapy should be instituted. Osteomyelitis of the underlying skull may be a rare concurrent problem. The diagnosis may be suggested by periosteal elevation and overlying soft tissue swelling on skull radiographs. Additional rare complications that may accompany an infected cephalhematoma and osteomyelitis include venous sinus thrombosis and cerebellar hemorrhage. Magnetic resonance imaging (MRI) may be used to detect these two intracranial complications, whereas computed tomography (CT) is the best imaging modality to identify the permeative bone erosion and destruction of osteomyelitis.

Most cephalhematomas are resorbed within 2 weeks to 3 months, depending on their size. In a few patients, calcium is deposited ( Fig. 29.4 ), causing a bony swelling that may persist for several months and, rarely, up to several years. Radiographic findings persist after the disappearance of clinical signs. The outer table remains thickened as a flat, irregular hyperostosis for several months. Widening of the space between the new shell of bone and the inner table may persist for years; the space originally occupied by the hematoma usually develops into normal diploic bone, but cystlike defects may persist at the sites of the hematoma for months or years. Rarely, a neonatal cephalhematoma may persist into adult life as a symptomless mass, the cephalhematoma deformans of Schüller.

Subgaleal hemorrhage is a collection of blood in the soft tissue space between the galea aponeurotica and the periosteum of the skull ( Fig. 29.5 ). The incidence is about 4 per 10,000 noninstrumented deliveries, with higher incidence after instrumental deliveries. Ng and colleagues have reported an incidence of 64 per 10,000 deliveries when vacuum extraction is performed.

The most common predisposing factor is difficult operative vaginal delivery, particularly midforceps delivery and vacuum extraction. The risk for subgaleal hemorrhage may be reduced by use of softer silicone vacuum cups instead of the original rigid metallic ones. The major risk factors include coagulopathies, prematurity, macrosomia, fetal dystocia, precipitous labor, intrapartum hypoxia, male sex, cephalopelvic disproportion, prolonged labor, and nulliparity.
When vacuum is used, the mechanism of injury is thought to be the vacuum traction pulling the scalp away from stationary bony calvarium, thus avulsing open the subgaleal space and causing the bridging vessels to tear and bleed into the subgaleal space. The loose connective tissue of the subgaleal space is extremely expansive and extends over the entire area of the scalp. The space can accommodate the entire neonatal blood volume (250 mL or more in a term baby), leading to hypovolemic shock, disseminated intravascular coagulation, and multiorgan failure, resulting in death in 25% of the cases.
Early manifestations may be limited to pallor, hypotonia, and diffuse swelling of the scalp. The development of a fluctuating mass straddling cranial sutures, fontanelles, or both is highly suggestive of the diagnosis. Because blood accumulates beneath the aponeurotic layer, ecchymotic discoloration of the scalp is a later finding. This is often associated with pitting edema and progressive posterior spread toward the neck and lateral spread around the ears, frequently displacing the ears anteriorly ( Fig. 29.6 ). Periorbital swelling and ecchymosis also are commonly observed. Eventually, hypovolemic shock, multiorgan failure, and signs of cerebral irritation develop. Massive lesions can cause extracranial cerebral compression, which may lead to rapid neurologic decompensation. The clinician should be aware of occasional “silent presentation,” in which a fluctuant mass is not apparent initially despite serial clinical examinations. Subgaleal hemorrhage should be considered in infants who show signs of hypoperfusion and falling hematocrit after attempted or successful vacuum delivery even in the absence of a detectable fluctuant mass. Close monitoring is particularly important in those infants who are considered stable enough to allow admission to the normal newborn nursery.

Standard radiographs of the skull may identify possible associated fractures. Computed tomography scanning may demonstrate abundant epicranial blood, parieto-occipital bone dehiscence, bone fragmentation, and posterior cerebral interhemispheric densities compatible with subarachnoid hemorrhage.
In contrast with cephalhematoma, subgaleal hemorrhage is characterized by its more diffuse distribution, more rapid course, significant anemia, signs of central nervous system (CNS) trauma (e.g., hypotonia, lethargy, seizures), and frequent lethal outcome.
Prompt restoration of blood volume with fresh frozen plasma or blood is essential. In the presence of continued deterioration, neurosurgery may be considered as a last resort. A bicoronal incision allows for exposure of the subgaleal space. Bipolar cauterization of any bleeding points can then be accomplished, and a drain can be left in the subgaleal space.
Although nearly 25% of infants with subgaleal hemorrhage die, long-term prognosis for survivors is generally good. More experience with aggressive and timely neurosurgical intervention may help to improve outcomes.
Fracture of the neonatal skull is uncommon, because the bones of the skull are less mineralized at birth and thus more compressible. In addition, the separation of the bones by membranous sutures usually permits enough alteration in the contour of the head to allow its passage through the birth canal without injury.
Skull fractures usually follow a forceps delivery or a prolonged, difficult labor with repeated forceful contact of the fetal skull against the maternal symphysis pubis, sacral promontory, fifth lumbar vertebrae, or ischial spine. They have also been described after vacuum-assisted vaginal delivery. Most of the fractures are linear. Depressed fractures are associated with forceps application. However, they may occur spontaneously after cesarean section or vaginal delivery without forceps. Factors that also have been implicated include pressure on the fetal skull by a maternal bony prominence (e.g., sacral promontory) or uterine fibroid, a fetal hand or foot, or the body part of a twin. Occipital bone fractures usually occur in breech deliveries as a consequence of traction on the hyperextended spine of the infant when the head is fixed in the maternal pelvis.
Linear fractures over the convexity of the skull frequently are accompanied by soft tissue changes and cephalhematoma. Usually, the infant's behavior is normal unless there is an associated concussion or hemorrhage into the subdural or subarachnoid space. Fractures at the base of the skull with separation of the basal and squamous portions of the occipital bone almost always result in severe hemorrhage caused by disruption of the underlying venous sinuses. The infant may then exhibit shock, neurologic abnormalities, and drainage of bloody cerebrospinal fluid from the ears or nose.
Depressed fractures are visible, palpable indentations in the smooth contour of the skull, similar to dents in a ping-pong ball ( Fig. 29.7 ). The infant may be entirely free of symptoms unless there is an associated intracranial injury.

The diagnosis of a simple linear or fissure fracture is seldom made without radiographs in which fractures appear as lines and strips of decreased density. Depressed fractures appear as lines of increased density. On some views, they are manifested by an inward buckling of bone with or without an actual break in continuity. Either type of fracture may be seen on only one view. CT imaging is the optimal diagnostic modality if a skull fracture and possible underlying injury are suspected.
Occasionally, the fragments of a linear fracture may be widely separated and may simulate an open suture. Conversely, parietal foramina, the interparietal fontanelle, mendosal sutures, and innominate synchondroses may be mistaken for fractures. In addition, normal vascular grooves, “ripple lines” that represent soft tissue folds of the scalp, and lacunar skull may be mistaken for fractures.
Uncomplicated linear fractures over the convexity of the skull usually do not require treatment. Fractures at the base of the skull often necessitate blood replacement for severe hemorrhage and shock in addition to other supportive measures. If cerebrospinal fluid rhinorrhea or otorrhea is present, antimicrobial coverage is indicated to prevent secondary infection of the meninges.
Small (<2 cm) “ping-pong” fractures may be observed without surgical treatment. Loeser and associates reported three infants with depressed skull fractures in whom spontaneous elevation of the fractures occurred within 1 day to 3 ½ months of age. Follow-up at 1-2 ½ years revealed normal neurologic development in all three.
Several nonsurgical methods have been described for elevation of depressed skull fractures in certain infants:
A thumb is placed on opposite margins of the depression, and gentle, firm pressure is exerted toward the middle. After several minutes of continuous pressure, the area of depression gradually disappears.
A hand breast pump is applied to the depressed area. Petroleum jelly placed on the pump edges ensures a tighter seal, and gentle suction for several minutes results in elevation of the depressed bone.
A vacuum extractor is placed over the depression and a negative pressure of 0.2-0.5 kg/cm 2 is maintained for about 4 minutes.
Because these methods are technically easier and less traumatic, they may be preferable to surgical intervention in a symptom-free infant with an isolated lesion.
Comminuted or large fractures associated with neurologic signs or symptoms should be treated by immediate surgical elevation of the indented segment to prevent underlying cortical injury from pressure. Other indications for surgical elevation include manifestations of cerebrospinal fluid beneath the galea and failure to elevate the fracture by nonsurgical manipulation.
Simple linear fractures usually heal within several months without sequelae.
Basal fractures carry a poor prognosis. When separation of the basal and squamous portions of the occipital bone occurs, the outcome is almost always fatal; surviving infants have an extremely high incidence of neurologic sequelae.
The prognosis for a depressed fracture is usually good when treatment is early and adequate. When therapy is delayed, especially with a large depression, death may occur from pressure on vital areas of the brain. Because the natural history of depressed skull fractures in neonates has not been clearly elucidated, the outcome is uncertain for infants with smaller lesions managed either by simple observation or by surgery after significant delays.
See Chapter 53 .
Facial nerve palsy in the neonate may follow birth injury or rarely may result from agenesis of the facial nerve nucleus. The latter condition occasionally is hereditary but usually is sporadic.
Traumatic facial nerve palsy most often follows compression of the peripheral portion of the nerve, either near the stylomastoid foramen through which it emerges or where the nerve traverses the ramus of the mandible. The neonate is vulnerable to these injuries because of the superficial course of the extracranial facial nerves. The nerve may be compressed by forceps, especially when the fetal head has been grasped obliquely. The condition also occurs after spontaneous deliveries in which prolonged pressure was applied by the maternal sacral promontory. Less frequently injury is sustained in utero, often in association with a mandibular deformity, by the persistent position of the fetal foot against the superior ramus of the mandible. An extremely rare cause is the pressure of a uterine tumor on the nerve.
This condition may occur rarely with simultaneous ipsilateral brachial plexus palsy, most likely secondary to compressive forces during delivery. Contributing factors include prolonged second stage of labor and midforceps delivery.
Traumatic facial nerve palsy may follow a contralateral injury to the CNS such as a temporal bone fracture or hemorrhage, tissue destruction, or both to structures within the posterior fossa. This CNS injury is less frequent than peripheral nerve injury.
Paralysis is usually apparent on the first or second day but may be present at birth. It usually does not increase in severity unless considerable edema occurs over the area of nerve trauma. The type and distribution of paralysis are different in central facial paralysis compared with peripheral paralysis.
Central paralysis is a spastic paralysis limited to the lower half or two-thirds of the contralateral side of the face. The paralyzed side is smooth and full and often appears swollen. The nasolabial fold is obliterated, and the corner of the mouth droops. When the infant cries, the mouth is drawn to the normal side, the wrinkles are deeper on the normal side, and movement of the forehead and eyelid is unaffected. Usually other manifestations of intracranial injury appear, most often a sixth cranial nerve palsy.
Peripheral paralysis is flaccid and, when complete, involves the entire side of the face. When the infant is at rest, the only sign may be a persistently open eyelid on the affected side, caused by paralysis of the orbicular muscle of the eye. With crying, the findings are the same as in a central facial nerve injury, with the addition of a smooth forehead on the involved side. Because the tongue is not involved, feeding is not affected.
A small branch of the nerve may be injured, with involvement of only one group of facial muscles. Paralysis is then limited to the forehead, eyelid, or mouth. Peripheral paralysis caused by nerve injury distal to the geniculate ganglion may be accompanied by a hematotympanum on the same side.
Central and peripheral facial nerve palsies must be distinguished from nuclear agenesis (Möbius syndrome). The latter frequently results in bilateral facial nerve palsy; the face is expressionless and immobile, suggesting muscle fibrosis. Other cranial nerve palsies and deformities of the ear, palate, tongue, mandible, and other bones may be associated with Möbius syndrome. Congenital absence or hypoplasia of the depressor muscle of the angle of the mouth also may simulate congenital facial palsy and has been associated with an increased incidence of other congenital anomalies.
No specific therapy is indicated for most facial palsies. If the paralysis is peripheral and complete, initial treatment should be directed at protecting the cornea with an eye pad and instilling artificial tears every 4 hours. The functional state of the nerve should be followed closely. Falco and colleagues proposed the following comprehensive approach :
Distinguish developmental from acquired lesions on the basis of the birth history and a detailed physical examination. Patients thought to have developmental palsy should be examined with radiologic and electrodiagnostic studies and brainstem-evoked response as appropriate.
Because of the expected 90% likelihood of complete spontaneous recovery, patients should be observed for 1 year before surgical intervention is considered. If recovery is suggested by physical examination or serial electromyography, observation without surgery may be delayed until the second birthday. Infants who require surgery are best treated with decompression or neuroplasty or both.
Most facial palsies resolve spontaneously within several days; total recovery may require several weeks or months. Electrodiagnostic testing is beneficial in predicting recovery; repeatedly normal nerve excitability indicates a good prognosis, but decreased or absent excitability early in the course suggests a poor outlook. The subsequent appearance of muscle fibrillation potentials indicates nerve degeneration. The prognosis in surgically treated infants worsens with increasing age at treatment.
Facial bone fractures may occur during passage through the birth canal, during forceps application and delivery, and during obstetric manipulation (most often the Mauriceau maneuver for delivery of the fetal head in a breech presentation). Manipulation may result in mandibular fractures and mandibular joint damage but is rarely severe enough to cause separation of the symphysis of the mandible. Fracture of the nose may result in early respiratory distress and feeding difficulties. The most frequent nasal injury is dislocation of the cartilaginous part of the septum from the vomerine groove and columella. This may result from intrauterine factors such as a uterine tumor or persistent pressure on the nose by fetal small parts or during delivery from pressure on the nose by the symphysis pubis, sacral promontory, or perineum. The presence of nasal septal dislocation may be differentiated from the more common normal variant of a misshapen nose by a simple compression test in which the tip of the nose is compressed ( Fig. 29.8 ). In the presence of septal dislocation, the nostrils collapse and the deviated septum becomes more apparent; in the normal nose, no nasal deviation occurs with compression.

Infants who sustain nasal trauma during the birth process may demonstrate stridor and cyanosis, even in the absence of septal dislocation. Miller and coworkers noted high nasal resistance in three such infants, only one of whom was found to have septal dislocation. The authors postulated the presence of edema and narrowed nasal passages from compression forces on the midface during delivery. The problem may be exaggerated by repeated nasal suctioning or transnasal bronchoscopy. These procedures and oral feeding should be avoided until the infant re-establishes normal nasal ventilation. Pulse oximetry measurements are useful in monitoring these infants.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here