Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
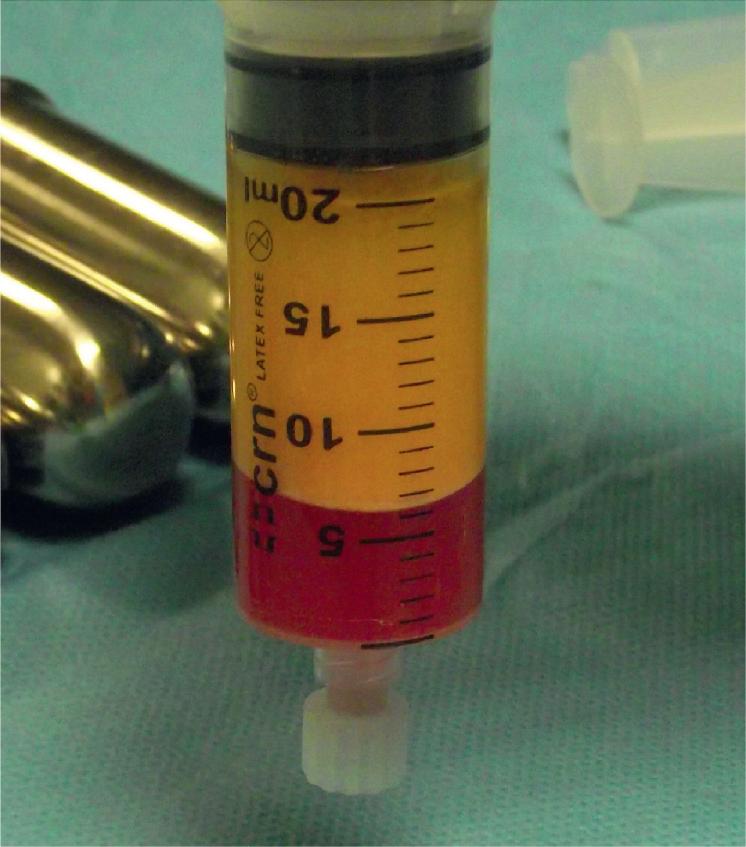
Adipose tissue is a biologic entity composed of various cell types that play a multitude of functions in the body, including insulation, protection and padding, energy storage, and endocrine regulation. Adipose tissue comprises approximately 15% of nonobese female total body mass and 22% of nonobese male total body mass and is distributed all over the body. Liposuction was developed in the 1970s, and it has opened avenues not only to remove excess fat but also to transfer fat from one part of the body to another. Fat grafting was first described in 1893 by Neuber, but it was Coleman who popularized free fat transfer in a minimally traumatic fashion. The so-called Coleman technique has allowed surgeons to obtain a consistently higher graft retention compared with prior techniques. This process produces three layers ( Fig. 14.1 ): the top supernatant layer containing lipid and the lower layer containing blood, tissue fluid, and local anesthetic are both discarded, leaving the middle layer, which contains adipose tissue, adipose-derived stem cells (ASCs), stromal cells, vascular cells, endothelial cells, and mural cells, termed the stromal vascular fraction (SVF). The large number of cells present in adipose tissue highlights what a complex and functional organ it is.
Fat grafting has become one of the most commonly performed procedures by plastic surgeons because of its unique versatility in soft tissue remodeling and its potential in reconstructive and aesthetic surgery. A recent survey by our group showed that approximately 80% of plastic surgeons have used fat grafting in their practice and that last year, a total of 80,000 cosmetic procedures and 30,000 reconstructive procedures involved fat grafting. Over the past decade, fat grafting has been increasingly used to improve facial aesthetics or to correct craniofacial contour abnormalities, ranging from traumatic to congenital deformities, in both the pediatric and adult populations.
Fat graft can be defined as particles composed of adipocytes and interconnecting mesenchyme, including connective tissues, fragments of vessels, and ASCs. Particles do not occur in native fat but, rather, are formed during the harvesting procedure of fat grafting. Fat particles are composed of three sections during healing: the surviving outer layer (1–300 μm), in which all adipocytes survive; the regenerating middle layer (600–1200 μm), where adipocytes undergo necrosis but are replaced by proliferating stem cells; and a necrotizing central core, characterized by oil cysts, necrosis, and fibrosis ( Fig. 14.2 ). ,
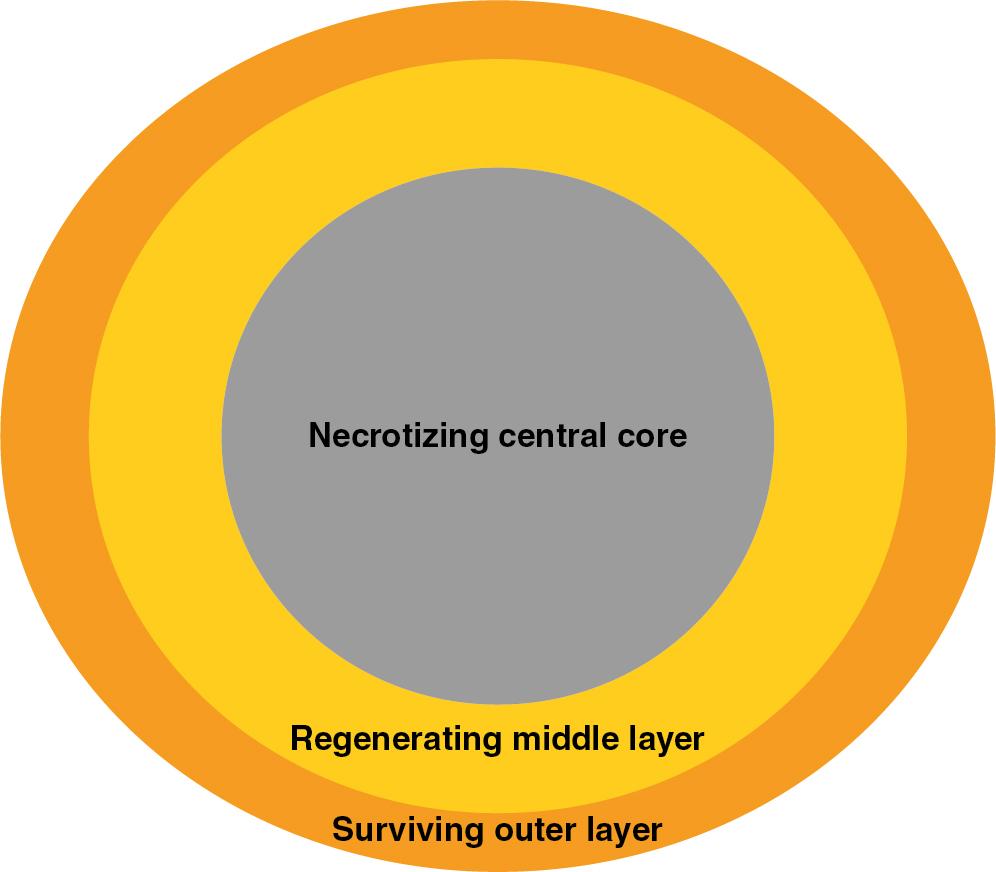
The initial survival of a fat particle relies on simple diffusion of nutrients until blood supply is established. Therefore the larger the diameter of the fat particle, the greater is the necrotizing central core due to diffusional limitations. Fat particle size therefore has a significant influence on the amount of grafted material survival. However, Del Vecchio and Rohrich believe that smaller fat particles may lack stromal components, such a blood vessels, fibroblasts, and connective tissue, for structural support of proliferating stem cells and adipocytes. Therefore one might argue that the ideal fat particle size must be large enough to contain adequate mesenchyme but not too thick to preclude diffusion of nutrients.
Over the past decades, techniques have evolved to produce smaller fat particles. Trepsat described, in 2009, the “microfat” grafting technique, where fat is harvested with a 2 mm cannula with 1 mm side holes. Subsequently, Tonnard et al. described, in 2013, the “nanofat” grafting technique, where fat is harvested with a 3 mm cannula with 1 mm side holes, rinsed with saline, mechanically emulsified by shifting the fat between two 10 cc syringes connected to each other by a female-to-female Luer-Lok connector, and filtered by using a nylon cloth. Nanofat was shown to lack adipocytes, while maintaining a rich supply of ASCs comparable with macro- and microfat in terms of stem cell proliferation and differentiation.
The mechanism by which fat survives after being grafted remains unclear, and various theories have been brought forward. The cell survival theory, introduced by Peer in the early 1950s, hypothesizes that fat graft survival is determined by the successful transfer of viable adipocytes and the development of a vascular network from the recipient site. Furthermore, initial nutrition and survival are through plasmatic imbibition, which requires the graft to be within 2 mm from the edge of vascularized tissues. Therefore fat-grafted cells receiving early and adequate blood supply survive, whereas other cells degenerate and are gradually removed. This theory highlights the importance of atraumatic processing and injection techniques to enable the transfer of viable adipocytes. More recently, the host replacement theory, introduced by Suga et al., hypothesizes that ischemic conditions lead to early death of grafted adipocytes, which, in turn, leads to the activation and regeneration of ASCs promoting the replacement of adipocytes.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here