Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
|
|
|
|
|
|
|
|
Surgical fusion of the cervical spine is a common procedure with a myriad of indications. Historically, allograft and autograft bone have been the mainstays of cervical fusion; however, in more recent years, surgeons have begun using a class of bone graft substitutes known as biologics. The biologics include demineralized bone matrix (DBM), ceramics, allografts containing mesenchymal stem cells, and growth factors. Each of these biologics has different characteristics and different degrees of osteoconductivity, osteoinductivity, and ostegenicity. This chapter reviews the key basic science principles, preclinical studies, and clinical studies that guide the surgeon in the appropriate use of biologics in the cervical spine.
For successful spinal fusion to occur, sufficient bone graft, adequate vascularity to the fusion bed, and mechanical stability of the fusion levels are required. Without these components, the rate of nonunion is markedly increased. A key component of the fusion cascade begins at the cellular level. Progenitor cells, which respond to the local environment to produce bone, are the ultimate throughput allowing fusion to occur. These cells can be provided by cellular grafting to the fusion site, as well as through vascularity to the fusion bed.
A sufficient vascular supply to the fusion bed is required to complete the various phases of fusion biology. Adequate vascularity allows a hematoma, rich with various activated growth factors, to accumulate at the fusion bed. The factors ultimately attract progenitor cell migration into the fusion bed. These cells can then differentiate into osteoblasts.
The oxygen tension in the fusion bed also depends on blood supply. Investigators have hypothesized that an abundant blood supply leads to a higher oxygen tension level, which favors osteoblastic differentiation rather than chondrogenic differentiation. Following differentiation, the osteoblasts produce osteoid matrix, which calcifies into bone. The newly formed bone ultimately undergoes remodeling according to Wolff’s law, such that reorganization is based on the imposed load.
Mechanical stability is the final requirement for fusion. In bony fracture healing, Perren proposed the interfragmentary strain theory, which states that the amount of strain in the fracture gap determines the type of tissue formed during healing. Strain greater than 100% would lead to nonunion, 10% to 100% would lead to initial fibrous union, and 2% to 10% would lead to cartilage formation and endochondral ossification. Strains less than 2% would lead to direct bone healing.
Furthermore, once tissue forms, the fracture gap could stiffen, which would lower strain and possibly change the tissue characteristics in the gap. Similarly, in fusion healing, a rigid structure is needed to promote the formation of bony tissue rather than a fibrous union. Instrumentation in the spine helps promote bone formation by reducing the “fracture” gap strain between the functional spinal units. These findings suggest that a certain amount of rigidity in fixation may facilitate spinal fusion.
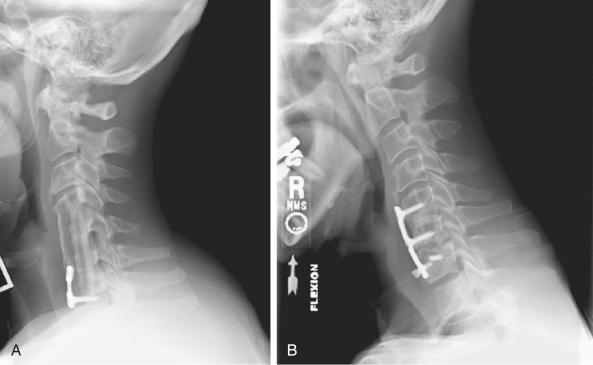
One of the most common cervical procedures performed is anterior cervical diskectomy and fusion (ACDF). ACDF has been highly successful in the treatment of radiculopathy, myelopathy, and myeloradiculopathy secondary to disk herniation and cervical spondylosis; however, the risk of nonunion following ACDF remains a real concern in certain patient populations. Fusion rates in the literature vary because no standard radiographic criteria exist for determining osseous fusion. Furthermore, successful fusion depends on the number of levels, graft type, and use of instrumentation. The fusion rates for single-level ACDF are much higher than for multilevel fusion. More specifically, for one-, two-, and three-level fusions, the fusion rates are 82% to 100%, 73% to 80%, and 70%, respectively. The use of allograft or autograft bone may also play a factor in the fusion rates, but in single-level fusions, this difference appears to be minimal. Instrumentation plays a critical role in fusion, particularly in multilevel constructs, to stabilize the motion segment and prevent graft compression and graft expulsion ( Fig. 48-1 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here